Abstract
Background and Objectives
The present study is done to evaluate the “Role of Simvastatin in Enhancing Bone Regeneration in Posterior Maxilla.”
Method
Sample size was determined to be 30 patients. Patients who required extractions at contralateral sites in the posterior maxilla were included in the study. Simvastatin graft was placed in one socket called the study socket and secured with gel-foam, and the control socket on the opposite side was allowed to heal without any intervention. Routine hematological investigations were done for the patient before extractions. Informed consent was taken. Radiographic follow-up was planned after extractions post-operatively 1 week, 4 weeks, and 12 weeks. Parameters used for comparison were pain, swelling, and bone density measurements.
Results
Scores for bone density were assessed radiographically, which were significantly higher on the study site than the control site. No significant difference was noticed between pain and swelling scores between study and control sockets.
Conclusion
Simvastatin graft use as a socket preservation material turned out to be on the positive side, as its application as a graft produced significant results in enhanced bone regeneration in the posterior maxilla, with extra added benefits of cost-effectiveness and complete eradication of donor-site morbidity.
Introduction
The posterior maxilla has been reported as the least predictable area for implant survival. 1 Bone in the posterior maxilla has been known to be D3 type in the majority of the cases. D3 bone has less cortical bone and more cancellous bone, creating challenges for implant rehabilitation or misbalancing the “pivotal role of stability” toward rehabilitation failure.
Traditionally, various attempts have been made to enhance the bone density of the posterior maxilla from D3 quality to D2 bone, like bone compaction, osseodensification, and bone grafts. With a vast variety of bone grafts available in the global market, and the quest to achieve the replacement of lost bone, this study aims to follow in the same footsteps by finding an ideal bone graft with osteoinductive potential.
Mundy et al. 2 reported in their in vitro study that they examined more than 30,000 compounds and their effects on the expression of the BMP-2 gene (bone morphogenetic protein-2).3, 4 Bone morphogenetic proteins are important regulators of osteogenic differentiation, and BMP-2 is the most potent of the osteoinductive factors. This dates back to the study of Yasko et al., 5 where he clearly showed the effect of BMP-2 on the lineage of multipotent stem cells, leading to the differentiation of osteoblast cells, leading to bone regeneration. Topical application of bone morphogenetic protein in the recombinant forms proved to be an effective alternative, but expensive, with an added disadvantage of an immune reaction.6–8 Mundy et al., through their study, found statins as the sole product that had a positive effect on increased expression of the BMP-2 gene.
Statins are a group of cholesterol-lowering drug that inhibits 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA), which is a rate-limiting enzyme in the cholesterol biosynthesis pathway. These lipid-lowering agents are considered a boon in restraining atherosclerotic events. In addition to their cholesterol-lowering activity, statins have pleiotropic effects such as vasodilative, antithrombotic, antioxidant, anti-inflammatory, and anti-proliferative effects.9–11
Various studies have been performed in the past, investigating the role of statins and have concluded that simvastatin, with its pleiotropic actions, helps in osteoblastic differentiation and angiogenesis. A few pioneer works include various in vivo studies showing the bone regenerative capacity of simvastatin.12–15
In vitro investigations have explored the mechanisms of statin effects on the mevalonate pathway, as well as osteoblast and osteoclast function. In vivo animal studies, it was confirmed that systemic statins could reduce inflammation-induced bone loss, and various cross-sectional and observational human clinical trials showed promising results in increasing bone regeneration.
Understanding the anabolism mechanism of simvastatin requires a thorough interpretation of the cholesterol biosynthesis pathway. A recent review of the literature suggests that the action of simvastatin can be categorized into three major mechanisms: promotion of osteogenesis, suppression of osteoblast apoptosis, and inhibition of osteoclastogenesis.16, 17
Stimulation of BMP-2, because of its osteogenic potential, has been the basic requirement of any bone graft material. Statins have been proven to stimulate BMP-2, which helps in bone regeneration.
This study is another attempt to validate the anabolic action of simvastatin intraorally in an extracted socket through its cumulative action on osteoblasts. In a desperate need to reverse the predictable failure of implant survival in the posterior maxilla, an attempt was made to preserve the sockets after extraction.
Materials and Methods
This study was approved by the institute, and all participants signed an informed consent agreement, following the Declaration of Helsinki on medical protocol and ethics, and the Regional Ethical Review Board of our institute approved the study. The present study was carried out on patients reporting to the Department of Oral and Maxillofacial Surgery. Patients who required extractions at contralateral sites of the posterior maxilla were included in the study. Informed consent was taken after a thorough explanation of the procedure. Regular hematological investigations and pre-operative radiovisiography (RVG) were done before the extractions. Inclusion criteria were patients in the age group of 18-0 years, who were willing to participate in the study and were undergoing extractions at contralateral sites in the posterior maxilla, whereas patients with periapical infection, metabolic bone disorders, radiation, and chemotherapy to the head and neck region, and medically compromised patients were excluded. Extractions were performed under local anesthesia with 2% lignocaine with 1:80,000 adrenaline (Figure 1). The study socket site was packed with simvastatin crushed tablet 10 mg (Figures 2 and 3). Gel-foam was placed over the powdered tablet to avoid extrusion of the graft, and a figure-of-eight suture was placed. The control socket site was allowed to heal in its natural way without any graft (Figure 4). Post-extraction instructions were given to the patient, and antibiotics and analgesics were prescribed for 3 days. Radiographic follow-up was done post-extraction, at 1 week, 4 weeks, and 12 weeks (Figure 5). Evaluation criteria included post-operative pain assessment using a visual analog scale, ease of operator’s comfort, inflammation assessment on the 1st, 7th, and 14th days, presence or absence of infection on the 1st, 2nd, and 7th days, and bone density measurement and analysis using post-operative RVG, using Sophix RVG software.
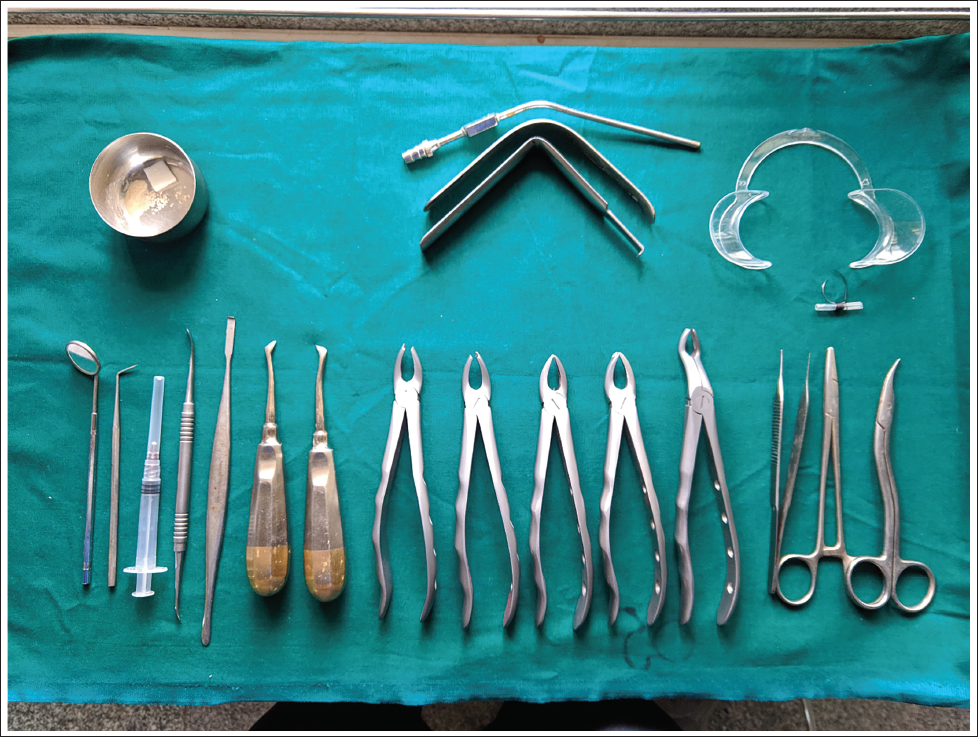
Surgical Armamentarium.
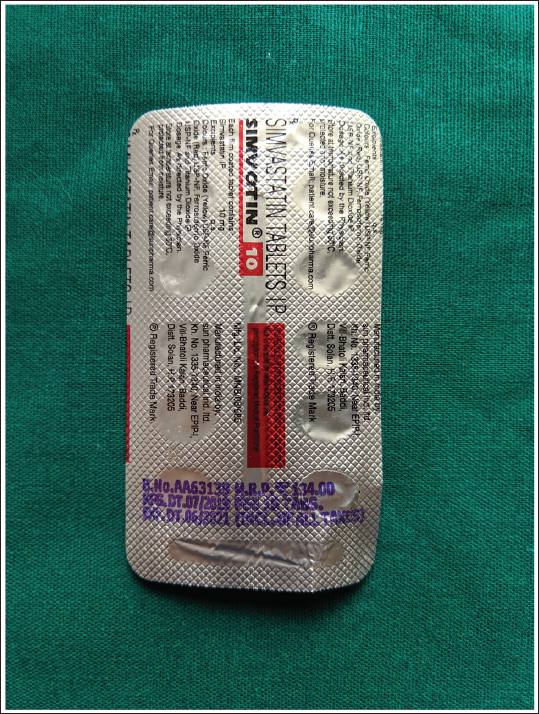
Simvastatin 10 mg Tablet.
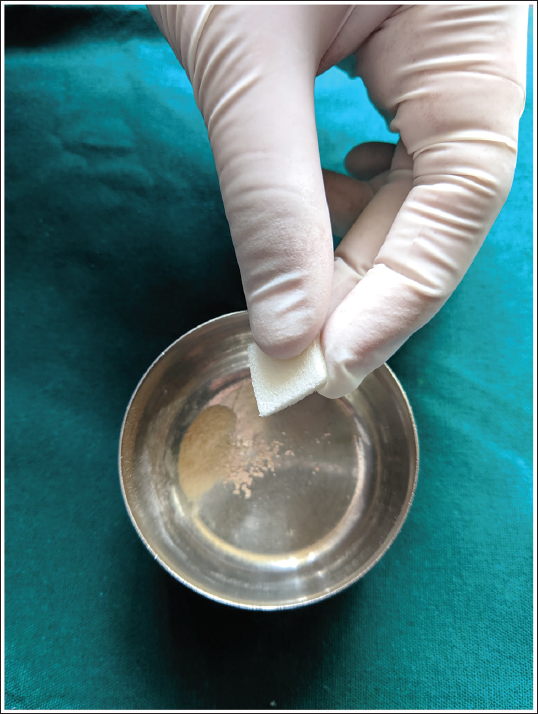
Crushed Simvastatin Tablet Using Mortar and Pestle.
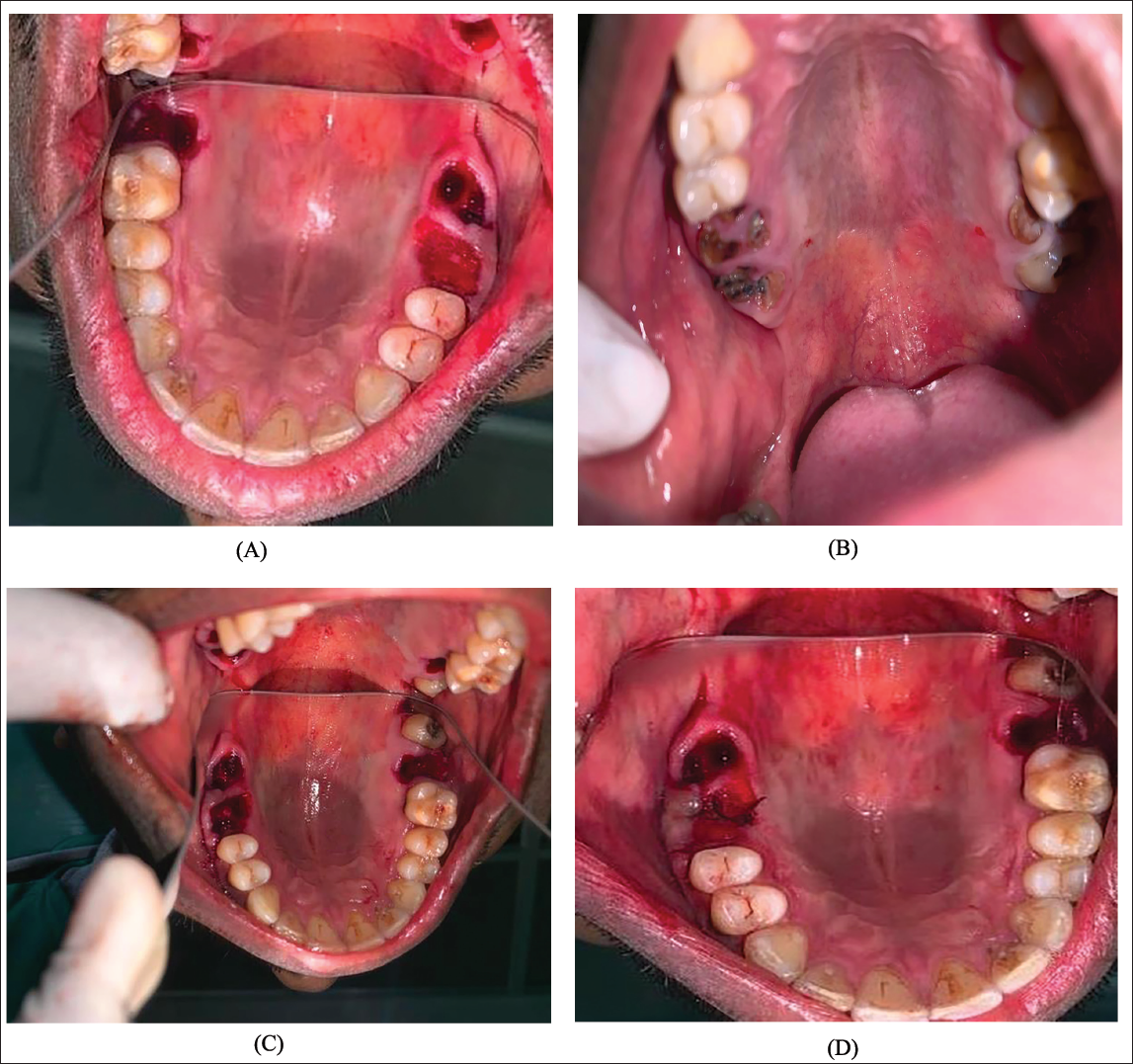
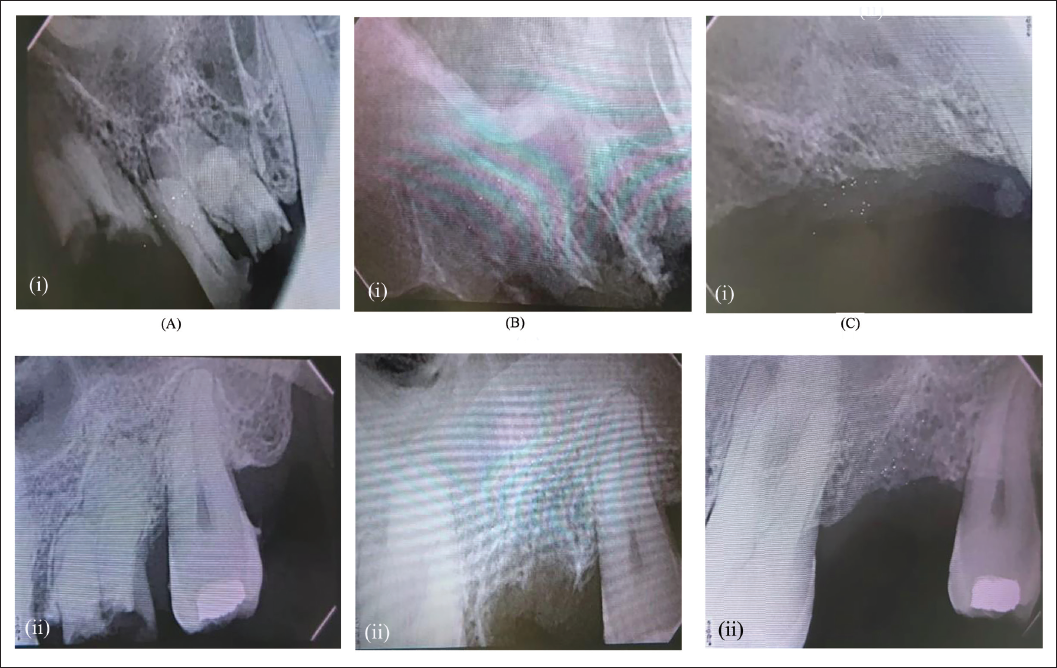
Results
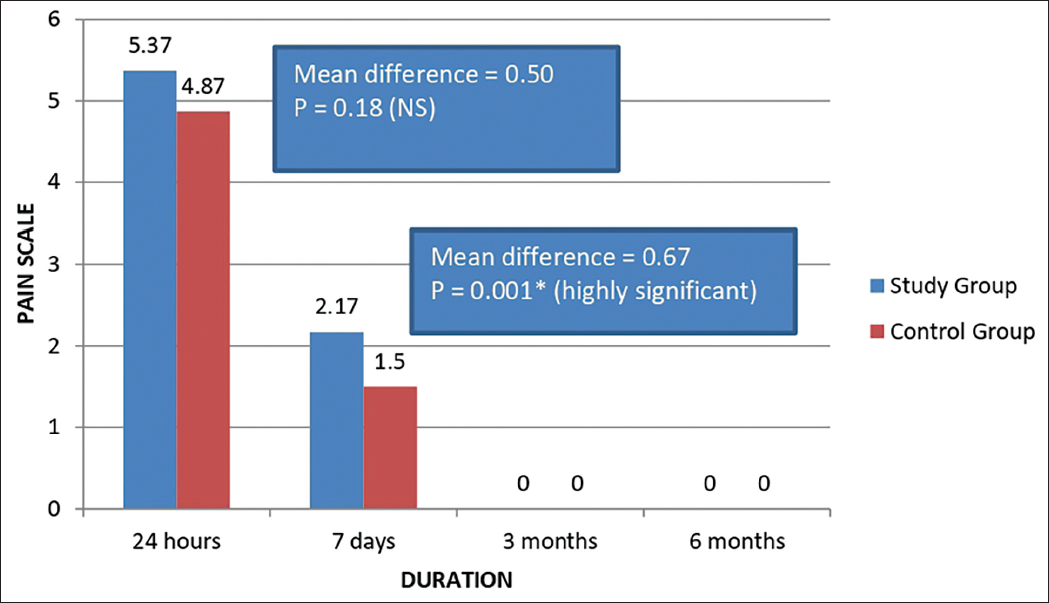
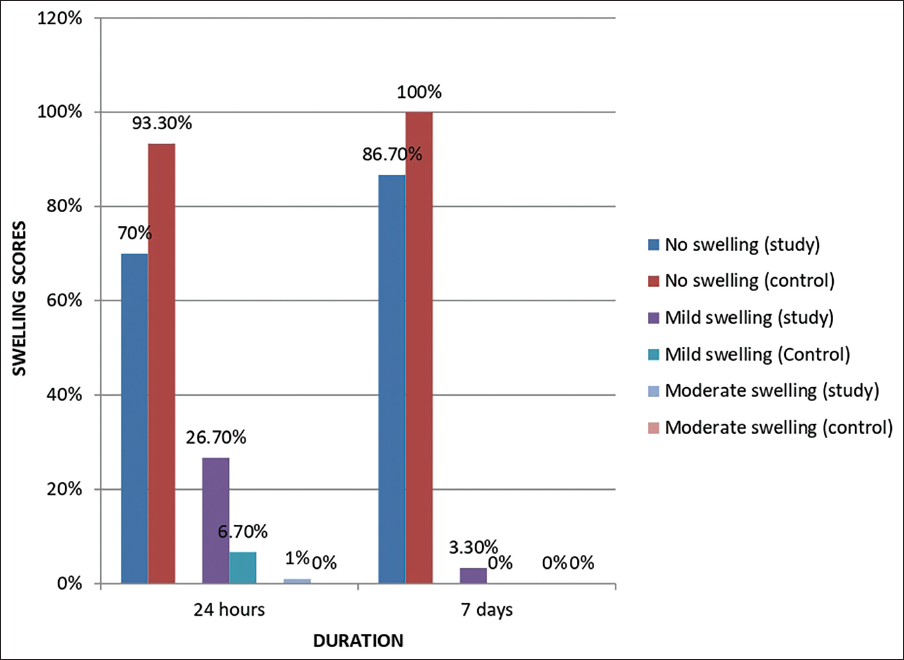
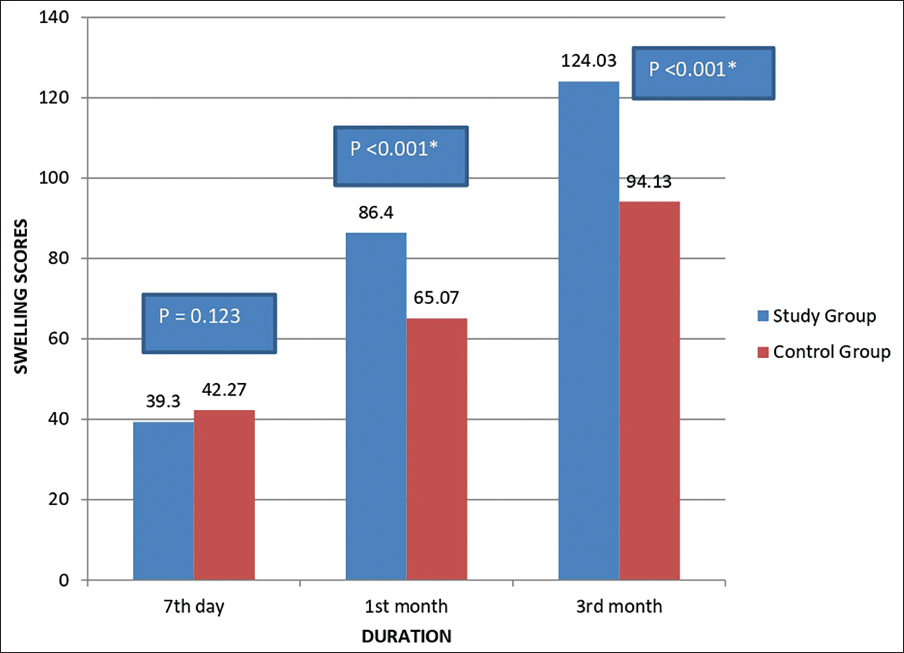
Thirty patients volunteered to participate in the study, where a simvastatin tablet was crushed and placed into the posterior maxillary contralateral extraction sockets and secured with gel foam. The percentage distribution of gender in our study was 60% male and 40% female, and 46.6% belonged to the 21-30 years age group. Mann–Whitney U test was used to compare mean pain scores between the study and control groups in relation to the duration of assessment, and no significant result was obtained (Figure 6). Evaluation of facial swelling post-operatively after a day revealed higher values at the study site compared to the control site (Figure 7). Scores of facial swelling were significantly reduced after a week. The intensity of swelling in the study and control groups among the study population after 24 h of treatment showed an insignificant difference using the chi-square test. Similarly, an insignificant P value was calculated after a week of comparing the study and control sites for the assessment of swelling using the chi-square test. The results showed a marked increase in mean gray-level histographic values of the study socket compared with the control socket, with a drastic increase in gray-level histographic values for the study socket at the end of 12 weeks, producing a significant P value < .001 (Figure 8).
Showing the Mean Pain Scores Between Study and Control Groups with Respect to the Duration of Assessment.
Intensity of Swelling Between Study and Control Groups in Relation to the Duration of Assessment.
Comparison of Mean Gray-scale Value Between Study and Control Groups (Mann–Whitney U Test).
Discussion
Prerequisites for bone regeneration constitute three crucial components: a morphogenetic signal, a suitable carrier matrix acting like a scaffold for new bone formation, and the ability of host cells to differentiate into bone-forming cells. 9 Statins are efficacious lipid-lowering drugs widely used in the suppression of the risk of cardiovascular diseases. Statins include naturally occurring lovastatin, chemically modified simvastatin and pravastatin, and the synthetically derived atorvastatin, fluvastatin, and cerivastatin. Statins inhibit HMG-CoA reductase, a rate-limiting enzyme in the synthesis of endogenous cholesterol, and reduce the prenylation of GTPases like RAS and RHO, which serve an important role in the maintenance of cellular integrity, cytoskeleton, and vesicle traffic. Statins are known to have pleiotropic effects on bone metabolism, with their direct impact on BMP-2 messenger ribonucleic acid (mRNA) expression and vascular endothelial growth factor (VEGF) expression in osteoblastic cells. Additionally, statins appear to reduce bone resorption by decreasing the expression of receptor activator of nuclear factor kappa-B ligand (RANKL) and Cathepsin K, and by disrupting the actin ring of osteoclasts, decreasing their activity. Now, the next question that arises is, “Why was simvastatin preferred over the other statins?” Simvastatin has been proven in in vitro and in vivo studies to stimulate osteoblastic differentiation and mineralization in nontransformed osteoblastic cells. It increases mRNA expression for BMP-2, alkaline phosphatase, type I collagen, bone sialoprotein, and osteocalcin in nontransformed osteoblastic cells and decreases gene expression for collagenase-1 and collagenase-3. Statin-actuated VEGF assumes a bigger part in angiogenesis and osteogenesis. Simvastatin improves trabecular bone density, promotes, and hastens early consolidation and mineralization. Mundy et al. 2 developed a cell-based screening assay in which the murine BMP-2 gene was transfected into osteoblasts, linked to the firefly luciferase reporter gene, and screened 30,000 compounds. Statins increased luciferase activity in a significant way, and their action on bone is due to the inhibition of the HMG-CoA reductase enzyme. Cultured murine (2T3) and human (MG-63) bone cells exposed to statins also showed enhanced expression of BMP-2 mRNA specifically. 2 Statins inhibit the rate-limiting enzyme HMG-CoA reductase in the cholesterol metabolism pathway. An immediate consequence of this inhibition is the reduced synthesis of mevalonate and downstream isoprenoid precursors, which are farnesyl pyrophosphate (FPP) and geranylgeranyl pyrophosphate (GGPP). During statin-induced osteoblastic differentiation and subsequent osteogenesis, both FPP and GGPP are reduced in their levels. FPP and GGPP hold a special place in the post-translational lipid modification of certain guanosine triphosphate (GTP)-binding proteins like rho. The repercussion of the inhibition of protein prenylation is the increased synthesis of VEGF, which is an anabolic factor for osteogenesis. Another pathway of its mechanism enlightens on “statins-inhibit osteoblast apoptosis.” Statins increase bone regeneration by inhibiting osteoblast apoptosis through transforming growth factor-β (TGF-β). It plays a critical role in bone formation, and Smad proteins are key components of the TGF-β signaling pathway. Smad 3 is regulated by the TGF-β receptor. TGF-β activates type II receptors, resulting in the activation of type I receptors and the phosphorylation of TGF-β type I receptor-like kinase, which activates Smad3, which is essential for bone mass maintenance. Statins’ anti-osteoclastic effect is due to their effect on the osteoprotegerin (OPG)/RANKL/RANK signaling pathway. It has been reported that simvastatin induces estrogen receptor-α (ER-α) in murine bone marrow stromal cells. Statins inhibit osteoclastogenesis by impairing the maturation and the integrity of the osteoclasts cytoskeleton. These effects are a consequence of the inhibition of prenylation of target proteins by prenyl protein transferases through a decrease in their substrates, including FPP and GGPP. 10 Wong and Rabie examined, in their study, the early histological expressions of VEGF, BMP-2, and core binding factor alpha 1 (Cbfa1) in healing bones with or without simvastatin. In their study, they showed that staining with their collagen matrix caused more new bone formation than the collagen matrix alone. They found that VEGF was expressed on day 3 after grafting, BMP-2 was expressed on day 4, and Cbfa1 was expressed on day 5 at the same time as new bone formation. Cbfa1 inhibits the differentiation of osteoclasts. They explained histologically the difference between bone regeneration under two mechanisms, osteoconduction and osteoinduction. 7 Bone defects with collagen matrix alone showed osteoconduction, as a result of growth factors expressed near the end of the bony defect, and bone formation was limited to a thin film of new bone next to the edges of the host bone. Bone defects with statins showed osteoinduction with subsequent 308% more new bone formation. Recently, in vitro and in vivo studies on simvastatin have documented strong evidence in the direction of bone regeneration. Osteoinductive properties of simvastatin need a special mention in the scenario of posterior maxilla D3 bone. D3 bone refers to the type of bone that has a thinner, porous cortical crest, and fine trabecular bone within. Many researchers have investigated its utilization in bone grafting in vitro. This study aims to define its role with strong statistically significant evidence in attaining bone regeneration in the posterior maxilla in order to achieve primary stability in delayed implant rehabilitation. The successful use of simvastatin to promote bone formation in vivo depends on the local concentration. Advantages associated with an ideal local delivery carrier include localization of the graft to the site of application in order to reduce the loading dose, and provision of the 3-D matrix for mesenchymal cell infiltration and a substrate for cell growth and differentiation. Gel foam was used in our study as it is biocompatible, bioresorbable, and adapts easily to the shape of defects. It performed like an ideal carrier that does not inhibit bone growth and resorbs optimally. 11 This study attempts to act in the progress of finding an ideal graft material with cost-effectiveness. The present study used a randomized, split-mouth, single-blinded study to ascertain the efficacy of the local application of simvastatin in bone regeneration after the extraction of the teeth contralaterally in the posterior maxilla. The results showed a marked increase in mean gray-level histographic values of the study socket compared with the control socket, with a drastic increase in gray-level histographic values for the study socket at the end of 12 weeks. Our study was in accordance with the results obtained by Chauhan et al., 12 Hassan et al., 13 and Saifi et al. 14 The posterior maxillary site stands as the most challenging site for the attainment of primary stability in implant rehabilitation. This study holds the position of a pioneering research study in proving the efficacy of simvastatin in bone regeneration in the most challenging site, that is, the posterior maxilla. The posterior maxilla has more cancellous bone than cortical bone, which puts the primary stability of an implant in question. Therefore, the local application of this simvastatin proved beneficial in the accelerated growth of bone, as well as increased bone density without any adverse reactions to the drug.
Conclusion
This study aimed at evaluating the bone regeneration ability of simvastatin, a cholesterol-lowering drug. It is used as a socket preservation graft material in the posterior maxilla and has been proven beneficial in improving and accelerating bone regeneration. Gray-scale values of study sites are significantly higher than the control sites, indicating the bone regeneration ability of simvastatin. Further research in the form of histological and immunohistological studies can be done to probe the role of simvastatin in accelerated bone regeneration and achieving superior results. Based on this study, we can conclude that the use of simvastatin has proved beneficial, simple, and cost-effective, and requires no special armamentarium or infrastructure for preparation. This study stood on the side of proving significant results in establishing the efficacy of simvastatin in bone regeneration in the posterior maxilla, and hence it can be used as a socket preservation graft after extraction.
Footnotes
Abbreviations
BMP-2: Bone morphogenetic protein-2; GTP: Guanosine triphosphate; HMG-CoA: 3-Hydroxy-3-methylglutaryl-coenzyme A; OPG: Osteoprotegerin; RANKL: Receptor activator of nuclear factor kappa-B ligand; RVG: Radiovisiography; TGF-β: Transforming growth factor-β; VEGF: Vascular endothelial growth factor.
Declaration of Conflicting Interests
The authors declared no conflict of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was obtained from the Krishnadevaraya College of Dental Sciences and Hospital.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Necessary informed consent was received and obtained, respectively, before initiating the study from all participants.
