Abstract
Background
The surgical repair and reconstruction of defects in the craniomaxillofacial region can be exigent because of complex anatomy, sensitivity of the involved systems, and uniqueness of each defect. The need to reconstruct the defect in the most precise way for the surgical procedure is of crucial importance to surgeons for enhancing patient outcomes and well-being. Autogenous grafts are considered the gold standard for reconstruction but are associated with an unpredictable resorption and donor site morbidity. The introduction of 3-dimensional printing in the biomedical field has led to the utilization of patient-specific implants (PSIs) in the surgical repair of maxillofacial defects, which occur congenitally, posttraumatically, or postsurgically. PSI fit quickly with greater accuracy with the added benefit of shorter rehabilitation time.
Material and Method
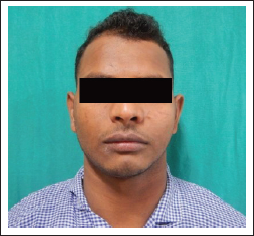
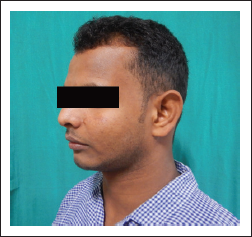
A 21-year-old male with a history of left zygomatico–orbital–maxillary complex fracture presented complaining of secondary deformity as a result of facial trauma. He underwent zygomatic osteotomy and repositioning followed by PSI reconstruction of the left zygoma, left orbital floor with titanium implants. A hemicoronal, subcilliary, and maxillary vestibular approaches were used.
Result
Good esthetic results with no postoperative complications are obtained.
Conclusion
The use of PSI for the reconstruction of oral and craniomaxillofacial defects should be considered an accurate alternative to noncustom-made implants. Automation allows for the application of safe, time-effective procedures not requiring specialized, and software-specific knowledge. The major drawback of PSI is its high cost, but many advantages of using PSI outweigh this disadvantage.
Introduction
Oral and maxillofacial defects are both functionally and esthetically debilitating for the patient and the prominent position of the face makes it more vulnerable to trauma, accidents, and war injuries. Similarly, various benign and malignant maxillofacial pathologies add to the etiology of such defects. The reconstruction of these hard and soft tissue defects is always challenging for surgeons. 1 Different autogenous and alloplastic materials are used by the reconstructive surgeons but accurate replicating the details of the lost tissues precisely is seldom achieved. The last decade has seen many options of 3-dimensional (3D) correction of these defects with various alloplastic materials such as silastic sheets and blocks, Polymethylmethacrylate (PMMA), or polyether ether ketone implants. Medical grade titanium alloy also has recently been used to fabricate 3D customized implants for the reconstruction of various facial bony defects. 2 This paper presents one case of midface reconstruction with the help of a customized 3D titanium alloy.
Case Report
Case 1
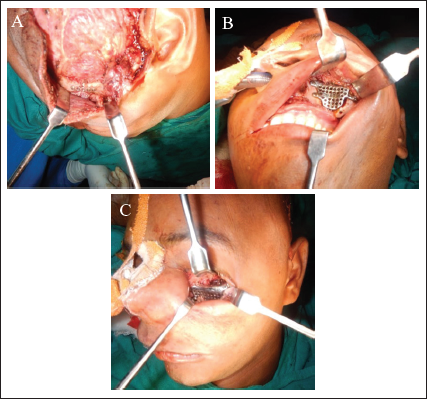
A 21-year-old male reported to the outpatient department who had a history of left zygomaticomaxillary complex fracture 3 years back. He presented with a chief complaint of secondary deformity as a result of facial trauma (Figure 1). He underwent zygomatic osteotomy and repositioning followed patient-specific implant (PSI) reconstruction of the left zygoma, left orbital floor with titanium implants under general anesthesia.
A hemicoronal, subcilliary, and maxillary vestibular approaches were used for the exposure of the defects (Figure 3). Computed tomography (CT) was used for the acquisition of patient data. The scan extended from cranial vertex to midface. Spiral CT data sets with 0.6 mm slice thickness and 08 gantry tilt were acquired. CT data in Digital Imaging and Communications in Medicine format were sent to the manufacturer for fabrication of the stereolithographic model (Figure 2).
The bony defect on the stereolithographic model was reconstructed using modeling wax. The contours were individually shaped by the surgeons themselves. Morphometric measurements of the uninvolved side were duplicated on the defect side. Extension wings were created on the wax model to aid as fixation elements and the waxed-up model was sent to the engineering lab for implant fabrication.
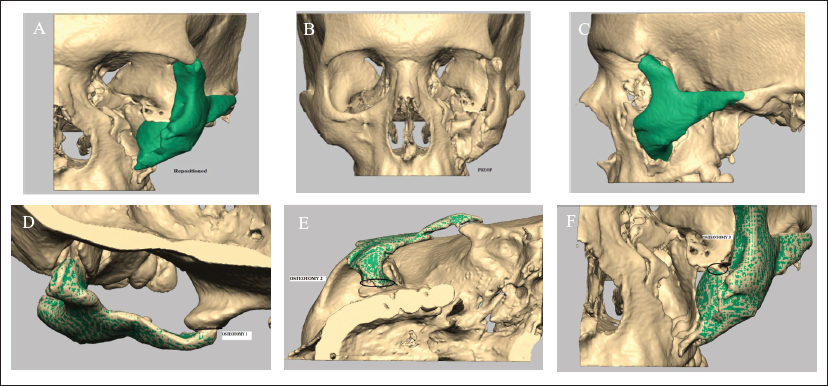
The wax model was scanned using a visible blue light scanner. The digital data of wax reconstruction obtained in the stereolithography (STL) were transferred to the control data of computer numerical control (CNC) milling machine. The titanium implant was fabricated through this milling process. The final thickness of the implant was fixed at 0.6 mm. Holes were drilled over the surface of the implant to aid in the drainage and to prevent any extradural collection. Holes were also drilled into the extension wings to facilitate screw fixation.
The postoperative outcome has been satisfactory, and the patient has been on regular follow-up.
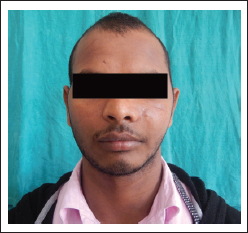
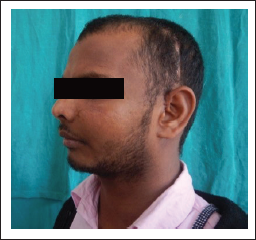
Discussion
There are many challenges unique to the bony reconstruction of the maxillofacial skeleton, including anatomic diversity, complex movement of the mandible, saliva contamination, and dental rehabilitation. Internal fixation in maxillofacial surgery gained popularity after the introduction of antibiotics in the 1940s. Luhr 3 was the first to study rigid internal fixation for maxillofacial surgery, and he also introduced compression plates as well as self-threading screws to the specialty in the 1960s. Before the development of the custom implants, mandibular reconstruction used to be carried out using rigid fixation plates and locking screws designed to fit the “average” mandible, usually at the inferior border and away from relevant anatomic structures, such as inferior alveolar neurovascular bundle and teeth. The titanium reconstruction plates are flat and require bending intraoperatively while the patient remains under general anesthesia after adequate exposure is achieved and sometimes after resection has taken place. Some prefabricated plates have a built-in angle to simulate the in-plane bend at the angle of the mandible. Despite this shaping, a mandibular reconstruction plate to fit a particular patient’s surgical defect is time-consuming and also weakens the integrity of the plate.
A major advancement to PSIs is the rapid prototyping of stereolithic models to scale, first described in oral and maxillofacial surgery by Brix and Lambrecht4, 5 in 1987. The printed models can be used to manually bend reconstruction plates fitted for a particular defect before the day of surgery, a concept is commonly known as “Prebending.” This technique allows for the accurate adaptation of the reconstruction plate to the patient’s anatomy without the patient being under anesthesia with an open wound. As the prices of desktop 3D printers and resins decreased over time, it has become feasible and practical for individual institutions to fabricate stereolithic models on their own using in-house computer aided design (CAD) software. Improved accuracy of prebent plates compared with the conventional method of intraoperative bending for mandibular reconstruction was demonstrated in a study of 42 patients in 2015. 6 The drawback of plate weakness that arises with bending, however, still remains, though at lesser values because of more direct and improved application.
PSIs with titanium alloy have revolutionized reconstructive surgeries as well as their outcomes. It is now being used in many medical specialties such as orthopedics, neurosurgery, and craniomaxillofacial surgery. Its planning and manufacturing require acquiring technical data from the CT scan in a Digital Imaging and Communications in Medicine file format and then the file is converted into a STL file that depicts the dimensions of the implant. This STL file is converted into scalable link interface file format that designs the implants into multiple layered (30 microns approx.) structures. This scalable link interface format file is then sent to the 3D printer that builds the implant by the process of additive (layer-wise) manufacturing. Once out of the 3D printer, the implant is removed from the platform, and it is polished, wiped, and sterilized before packaging and final delivery. 7 The surgeon is also intricately involved in the designing and approval of the implant.
In the case discussed here, it was nearly impossible to reconstruct the 3D architecture of the zygoma with an autogenous bone graft. Zygoma has a tetrapodal structure that supports the eye-globe and gives anteroposterior projection or prominence to the face. Its flattening or loss can severely affect the overall facial esthetics and also the ophthalmic functions. Most of the time the patient carries the burden of a disfigured face and unesthetic appearance for life which also affects the social life.
Titanium is a biocompatible inert material that shows the tendency of osseointegration with the underlying bone and the implants are being used for internal maxillomandibular fixations also as dental implants since the last few decades. Its biocompatibility is also proven and remains the material of choice for maxillofacial internal fixation as well as reconstruction. Therefore, titanium 3D PSIs hold a promising future for such patients. 8
Patient-specific custom implants made with autogenous and adipose-derived stem cells in custom bioreactors have already proven efficacy and superiority to traditional implants in large animal studies. Bhumiratana et al 9 demonstrated that anatomically correct bone grafts from adipose-derived stem cells were grown and implanted in Yucatan mini pigs to reconstruct the ramus-condyle unit (Figure 4). The custom stem cell grafts were tested against both no graft and scaffold only and were found to have superior characteristics in terms of strength, volume, and shape. The stem cell grafts were also found to benefit from antiresorptive properties.
PSIs are oral and maxillofacial surgery’s answer to the personalized medicine. Although this technology has been in use for many years in some areas such as total joint replacement of temporomandibular joint (TMJ); it is relatively new in other areas, such as reconstruction and orthognathic surgery. 8 Advances in computer aided design and computer aided manufacturing (CAD/CAM) technology with decreasing costs will continue to allow this field to evolve in order to improve accuracy, efficiency, and overall outcome.
Footnotes
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
Acknowledgment
The authors are thankful to the Department of Dentistry, All India Institute of Medical Sciences (AIIMS), Patna and Department of Oral & Maxillofacial Surgery, Sardar Patel Post Graduate Institute of Dental and Medical Sciences, Lucknow for allowing them to conduct this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
