Abstract
Background
Clinical trials involving pharmacologic or behavioral treatments often assess depression and suicidal ideation for purposes of screening, baseline assessment of potential moderators or mediators of treatment, or as a study outcome, even if the primary condition under study is not a mental health disorder. Suicide risk management in the context of clinical research poses significant clinical, ethical, and practical challenges, and the literature provides little guidance with respect to outcomes of suicide risk management protocols (SRMPs) or suicide risk assessment instruments deployed in the clinical research setting.
Methods
We report our experience using a novel SRMP in the Program for Lung Cancer Screening and Tobacco Cessation (PLUTO) trial through in-person and remote interactions.
Results
An SRMP was developed for non-clinical research staff to assess and respond to participants who express suicidal ideation. Between September 2016 and April 2021, the SRMP was used 61 times for 59 individuals. The SRMP was activated by explicit probing of suicidal ideation in 46 of 61 uses (75%). Subject risk was categorized as high-risk in 6 of 61 SRMP uses (10%).
Conclusion
Our findings demonstrate a useful tool for the management of suicidal ideation and behavior in a clinical trial. Suicidal ideation may be endorsed by only a small number of study participants, however participant safety dictates the need to develop and implement a practical SRMP. These findings may be of relevance to researchers collecting patient reported outcomes remotely. Researchers should consider available resources for SRMPs during design and start-up phases of research.
Introduction
Approximately 700,000 people die by suicide worldwide annually. 1 An even larger number of people have thoughts of suicide. In 2017, approximately 4.3% of U.S. adults experienced suicidal ideation. Alarmingly, the rate of suicide increased 35.2% between 2000 and 2020. 2 Depression—a major risk factor for suicide—is common among the general population, affecting approximately 8% of U.S. adults3,4 Historically, researchers may have been reluctant to include patients with depression, suicidal ideation and other mental health diagnoses in clinical trials, citing patient safety. 5 Suicide risk management in the context of a clinical trial poses varying practical and ethical challenges to researchers. However, exclusion of patients with suicidal ideation and behavior from clinical trials undermines generalizability and limits the scope of empirical evidence to inform treatment decisions in this marginalized population.
Participant disclosure of suicidal ideation and behavior in non-psychiatric research
In clinical trials outside the psychiatric field, suicidal ideation and behavior among research participants is often identified through depression assessments or prospective monitoring of treatment-emergent adverse events. Even if depression severity is not a study outcome, it is often monitored using standardized instruments as a potential moderator or mediator of treatment effect. With a growing emphasis on the importance of capturing patient reported outcomes (PROs) to better inform analyses of treatment effectiveness, the scope of information captured during data collection may reveal information about a participant that necessitates intervention.
Several widely used standardized measures of depression, such as the Beck Depression Inventory and Patient Health Questionaire-9 (PHQ-9), explicitly probe suicidal ideation and behavior.6,7 Other assessments may reveal factors associated with increased risk for suicidal behavior, such as hopelessness or impulsivity, or prompt a participant to disclose suicidal ideation spontaneously. 8 Studies evaluating pharmacologic or behavioral interventions for conditions such as tobacco use, known to be associated with depression, often monitor mood during treatment and require adequate protections for participant safety to be in place.
Suicide risk management protocols in clinical trials
Suicide risk management in the context of a clinical trial poses varying practical and ethical challenges to researchers.9,10 One common misconception is that asking a person about suicidal thoughts or behaviors will induce suicidality, and a recent meta-analysis evaluating iatrogenic risks of evaluating suicidality in prospective research studies has demonstrated that this is not true. 11 The challenges of retaining participants who express suicidal ideation can be mitigated by using a study-specific suicide risk management protocol (SRMP), a component of the study protocol that provides systematic guidance to research staff for managing participants who disclose suicidal ideation and behavior. The primary goal of the protocol is to standardize the steps necessary to identify participants who need escalated levels of care. Such decision aids are especially important for non-clinical research staff (e.g., research coordinators or research assistants) without formal mental health training who would otherwise be left to rely on personal judgment to determine a participant’s level of risk. An SRMP should facilitate—but not replace—comprehensive suicide risk assessment and intervention facilitated by a provider with specialty mental health training.12,13
The literature describing the features and outcomes of SRMPs deployed in non-psychiatric clinical research is limited.13–16 In a 2010 review of National Institute of Mental Health-funded studies, Vannoy et al. proposed a framework for development of an SRMP. Core components include clear decision rules for entry and exit of the protocol, risk assessment instrumentation or questions to gauge intensity of risk, specific actions for staff responses, contingency instructions, and documentation guidance. 13 Guidelines by Schatten et al. echo Vannoy and colleagues’ framework and also offer considerations for a range of study samples, data collection methodologies, and study designs. 17 A scoping review by Stevens et al. additionally offers a recommendation for providing educational resources to research participants as an additional consideration. 18
Suicide risk assessment in clinical research
In many non-psychiatric research settings (e.g., studies not evaluating psychotropic agents, or where suicide prevention is not a study outcome) it may not be operationally feasible to have a mental health professional available to provide urgent, comprehensive suicide risk assessment for each participant who expresses suicidal ideation. Yet, researchers have an obligation to minimize risk to participants by eliminating immediate dangers.10,19 Thus, short, easily-scored suicide risk assessment instruments are ideally suited for use as part of an algorithmic approach to identifying participants who pose an imminent danger to themselves and warrant emergency intervention (e.g., a participant who discloses intent to die by suicide and who has access to lethal means during a telephone data collection assessment). In clinical practice, a prevailing consensus holds that suicide risk assessment instruments should not be used as an absolute determination of risk but can be valuable when used in combination with other assessment strategies.20–23 Several instruments to quantify suicide risk have been proposed, but the heterogeneity of suicidal ideation and behaviors—and the rarity of completed suicide relative to the prevalence of suicidal ideation and behaviors—limits the reliability and validity of standardized measures.21,24 Additionally, translations from the English language and cross-cultural validation of published instruments are lacking.25,26 Despite the available literature describing the utility of suicide risk assessments instruments for a range of clinical applications, how well these instruments perform in the context of a non-psychiatric clinical trial as a risk-mitigation tool remains largely unreported.
Aim
The aim of this article is to describe the suicide risk management algorithm used to standardize non-clinician research staff response to participants who disclosed suicidal ideation in a smoking cessation trial. We report the outcomes of the protocol and describe challenges encountered monitoring depression using the PHQ-9. Finally, we describe common conceptual and methodological issues for the development and implementation of SRMPs and highlight critical knowledge gaps suitable for future investigation.
Methods
PLUTO trial
This research was conducted as part of the Program on Lung Cancer Screening and Tobacco Cessation (PLUTO), a study which employed a sequential, multiple assignment, randomized trial (SMART) design to evaluate different year-long permutations of evidence-based tobacco cessation interventions (i.e., nicotine replacement therapy, tobacco longitudinal care, pharmacist-administered medication therapy management) in patients aged 55–79 eligible for lung cancer screening by low dose computed tomography. 27 PLUTO was funded by the National Cancer Institute (R01CA196873) and was approved by the University of Minnesota, Minneapolis VA Health System, and Allina Health Institutional Review Boards (NCT02597491). All participants provided written informed consent. This study was considered greater than minimal risk. In addition to internal protocol adherence and safety monitoring by our research team, an independent Data and Safety Monitoring Board (DSMB) provided further oversight.
Cohort characteristics
Patients were included in this report if they activated our SRMP either during PLUTO trial eligibility screening or during their participation in the study. Eligibility screening and data collection occurred between September 2016 and April 2021. Current daily smokers between the ages of 55–79 who were participating in or eligible (on the basis of their age and smoking history) for lung cancer screening using low-dose computed tomography were invited to participate in the PLUTO trial. Patients with unstable mental health disorders were not eligible for participation, defined by hospitalization for mental health reasons within 3 months of screening, suicidal ideation within 2 weeks of screening, or report of significant changes to medications for mental health indications within 3 months of screening (e.g., recent initiation of an antipsychotic). Patients with stable depression, anxiety, and other mental health disorders were eligible to participate.
Recruitment and study procedures
Potential participants were identified in partnership with the lung cancer screening programs at all study sites. Interested patients completed a brief set of questions to determine eligibility. Eligibility screening was facilitated by a non-clinician research assistant in person or by telephone. Patients completed an in-person or telephone enrollment encounter to provide informed consent and data collection. In-person assessments occurred in clinical settings (e.g., radiology clinic or primary care clinic). All participants received 1 year of intervention in parallel with 18 months of telephone data collection. In this longitudinal study, depression, anxiety, and alcohol use were monitored as potential moderators of treatment effect. The rationale, study design, outcome measures, and recruitment strategy was described in greater detail in a separate publication. 27
Data management
Study data were collected and managed using REDCap (Research Electronic Data Capture) electronic data capture tools hosted at the University of Minnesota. The data collection and counseling teams had separate longitudinal REDCap environments with configurations that allowed for specific fields (e.g., participant name, phone number) to be shared between environments. The SRMP was nested within each REDCap project as a repeating form (to allow multiple SRMP uses for the same participant) and utilized filters and branching logic to (1) auto-prompt use, (2) provide specific triage points, and (3) offer follow-up instructions specific at each triage point.
Analysis
Descriptive statistics were used to summarize the characteristics (e.g., age, sex, participating site, research setting at time of activation) of patients who activated the SRMP and to summarize the indication for use and risk classifications. Medical records were reviewed for subsequent psychiatric adverse events following use of the SRMP. Comparisons between SRMP activation rates pre- and post-revisions to the PHQ-9 suicide item were made using a chi-squared test. p-values <0.05 were considered statistically significant. Analyses were completed using SAS Version 9.4.
Results
Suicide risk protocol development
Approach for the design of a suicide risk management protocol.

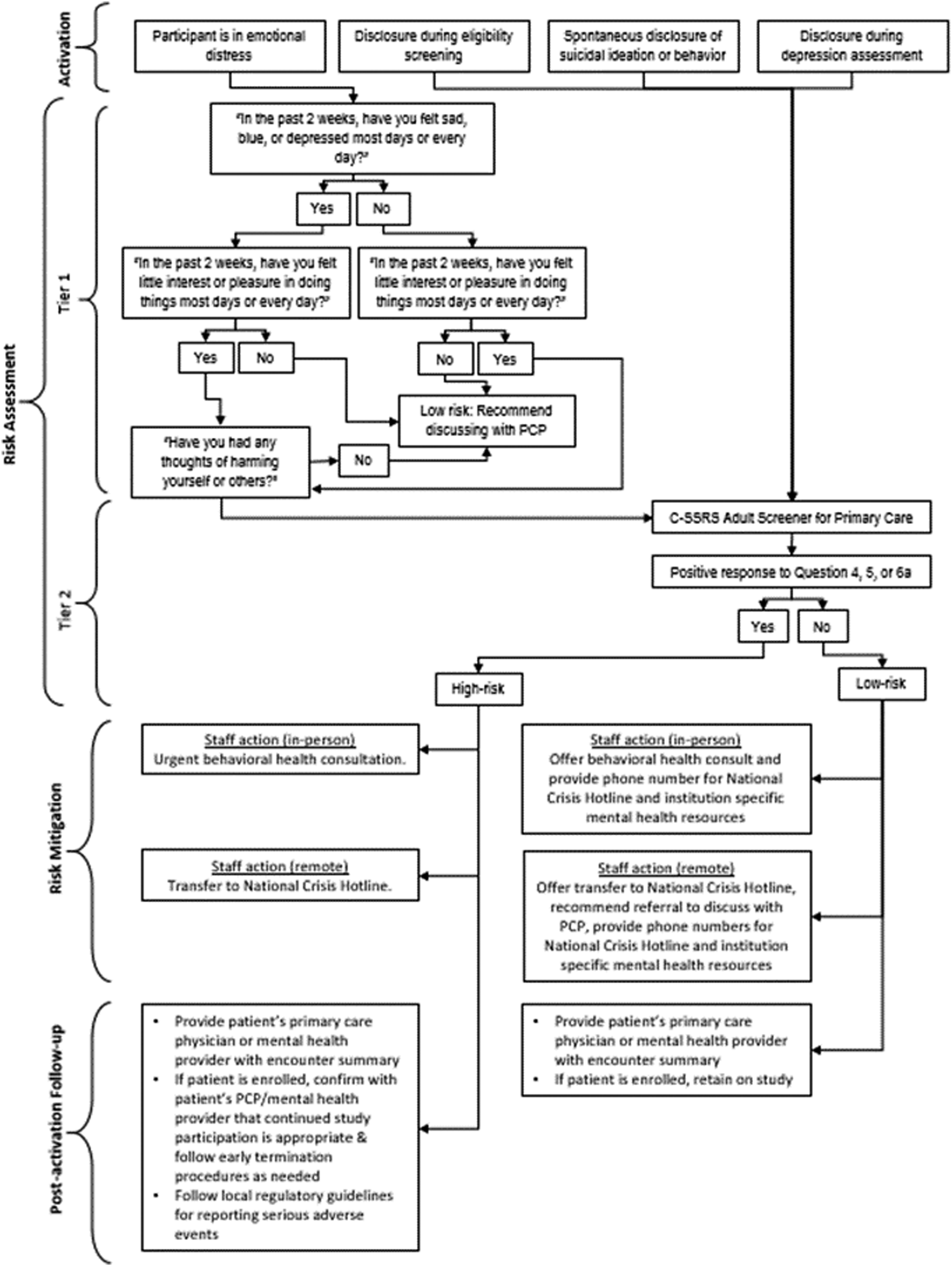
PLUTO trial suicide risk management protocol.
Activation was prompted by clear decision rules that accounted for the multiple scenarios in which staff might encounter suicidal ideation and behavior during implementation of our study protocol. The activation scenario informed entry into the risk assessment phase, which included two tiers. In some circumstances depression or suicidal ideation and behavior was suspected by our research staff but was not explicitly endorsed; in that case, staff could initiate Tier 1 of the risk assessment phase, which used a modification of the Patient Health Questionnaire-2 to assess depression and thoughts of self-harm. 28 When suicidal ideation and behavior was directly endorsed, staff entered the risk assessment phase at Tier 2, which utilized the Columbia-Suicide Severity Rating Scale (C-SSRS) Adult Screener for Primary Care to assign risk strata and subsequent triage points. 29 This concise measure includes six questions and can be administered by non-clinicians remotely or in-person. 30
The risk mitigation phase utilized responses from the C-SSRS to further stratify patients in Tier 2 into low risk or high risk categories to determine immediate allocation of resources. 29 In this population of older adults – some of whom were veterans – and a research setting which involved in-person and remote assessments, our SRMP incorporated multiple types of responses. Some practical issues contributed to staff response, such as proximity to the patient, resources available at the study site, and level of risk. Based on these factors, responses options ranged from providing resources for support, to referral for assessment, immediate evaluation by a mental health professional, phone transfer to a crisis hotline (e.g., the National Suicide Prevention Lifeline), admission to the emergency department, or contacting 911 to coordinate a welfare visit. 31
Research staff were required to report on the use of the SRMP within the same day of use to the lead study coordinator and site principal investigator. All uses of the SRMP were documented in both the patient’s electronic medical record (including for patients not enrolled in the study) and study records as an adverse event. The adverse event was considered a serious adverse event (SAE) if the patient was classified as high risk by the SRMP.
Development of the SRMP was an iterative process; some elements were added to the protocol after initial experience. For example, decision rules for cross-communication with the participant’s medical team and rules for subject disposition (e.g., early termination) were built into the protocol after initial experiences highlighted their necessity, and trial and error provided clarity regarding appropriate avenues for communication. The participant’s mental health or primary care provider was notified with documentation (via the electronic medical record) of the encounter if the patient had an established provider within the study site’s medical system. For patients classified high risk, consultation with the participant’s physician or mental health care provider was sought to determine if continued trial participation was appropriate.
These protocol elements precipitated a change in our informed consent to ensure transparency in the confidentiality clauses and to promote trust between the participant and research team. Participants were informed during the informed consent process that cross-communication between the research team and their medical provider might occur. The following was added to the informed consent document: “In the event that you disclose or endorse any thoughts to harm yourself to the research staff, we may provide you with referrals for treatment, contact your primary care physician or mental health care provider, or work with you on a plan that may include getting you to a hospital for safety.”
If a participant was considered unfit for continued participation in the study by their medical provider or the study team, they were withdrawn from the study. Language describing the possibility that a participant might be withdrawn from the study without their consent and the subsequent consequences of early termination of participation (e.g., prorated compensation and continued use of study data already collected) was added to the informed consent.
Interim feedback from our research assistants facilitating the baseline assessments indicated that participants sometimes had difficulty responding to Question 9, the double-barreled suicide item included in the PHQ-9 (“In the past two weeks, have you been having any thoughts that you would be better off dead, or of hurting yourself?”). The two-part sentence structure (“…better off dead…or of hurting yourself?”) presents two different concepts that participants occasionally had trouble reconciling. In this population of older, heavy tobacco users in which comorbid illness and pain were common, we suspected that the PHQ-9 suicide item might an overly sensitive measure of suicidal ideation. At the recommendation of the DSMB, we stopped using the PHQ-9 and began using the PHQ-8 while the trial was ongoing. 32 The PHQ-9 suicide item was replaced with a modified item to assess suicide risk adapted from the Ask Suicide-Screening Questions (ASQ) “In the past two weeks, have you been having thoughts about killing yourself?”), which was selected because of the direct, action-oriented wording. 33
Suicide risk management protocol implementation experience
Experience using the PLUTO Trial suicide risk management protocol.

Five of the 14 patients (35.7%) who activated the protocol during screening accepted suicide prevention resources offered by the study team. All three patients who were assessed as high risk during eligibility screening accepted immediate referrals—one patient accepted an in-person suicide risk assessment facilitated by mental health clinician and two patients accepted phone calls with the National Suicide Prevention Lifeline. None of the 14 patients who expressed suicidal ideation during eligibility screening were enrolled in the study.
The majority (84.2%; n = 32 of 38) of SRMP activations at baseline were the result of suicidal ideation endorsed during the depression assessment (Patient Health Questionnaire-9, while it was still in use). Six of the 15 situations (42%) where suicidal ideation was spontaneously disclosed, or the participant’s mood warranted staff-initiation of the SRMP, occurred during the baseline data collection assessment while the remainder occurred during the 78-weeks intervention and follow-up period. A total of 660 participants completed the PHQ-9 at baseline; 32 of 572 patients (5.6%) asked about suicidal ideation in the original PHQ-9 format and 0 of 88 patients (0%) asked about suicidal ideation in the modified format activated the SRMP (p = .027).
Fourteen of the 55 participants (23%) assessed as low risk accepted mental health resources offered by the study team. Low risk participants who declined resources were encouraged to schedule an appointment with their mental health or primary care provider. Two of the 38 participants (5.3%) who activated the protocol at baseline were assessed as high risk. Both patients accepted immediate referral; one patient was evaluated in person by a mental health clinician and one patient accepted a phone call with the National Suicide Prevention Lifeline. Neither of the patients assessed as high risk by the protocol at baseline required psychiatric hospitalization and both were retained on the study following consultation with the patients’ mental health providers. All 38 participants who disclosed suicidal ideation during baseline assessment were retained on study. A review of adverse events revealed that in the 78 weeks following activation of the SRMP, none of the 38 participants requiring baseline SRMP activation had subsequent psychiatric serious adverse events.
One participant was assessed as high risk during their final routine data collection follow-up telephone call and refused all resources offered. Upon further discussion between the participant and the research assistant facilitating the call, the participant clarified that their suicidal ideation was not a currently ongoing, but a prior issue related to a negative life experience. The research assistant who facilitated the data collection encounter was immediately able to contact the study and site principal investigators and it was determined that based on the context provided by the participant and established care within the health system, a welfare check with the local emergency medical services was not necessary. The team determined that notifying the participant’s primary care physician was sufficient.
Discussion
Our initial experiences with this SRMP demonstrate that it is fairly common for participants in a tobacco cessation clinical trial to express suicidal ideation. Using a novel, systematic approach, we have identified and responded to participants who disclosed suicidal ideation and behavior. The SRMP was of particular use in the context of this trial because many participant-study interactions were facilitated by research staff who were not clinicians and did not have specialty mental health training. The C-SSRS was an effective tool to gauge the severity of suicidal ideation disclosed to our research team for the purposes of tailoring specific referrals and the intensity of follow-up. Conversely, we found some components of the PHQ-9 to be ambiguous and overly sensitive for this setting. Specifically, participants may have answered Q9 “yes” due to chronic pain rather than having genuine plans for suicide. This limitation of the PHQ-9 has been noted before and evaluation of a PHQ-8 supports its reliability.32,34
We found that participants do endorse suicidal ideation and behavior both spontaneously and over the course of longitudinal data collection that probes suicidal ideation and behavior and while these events were rare, the SRMP was necessary in this population of older smokers. Other studies have reported SRMP activation in 3%–25% of their cohort, consistent with our findings.14,15 When an SRMP risk assessment yielded a low risk classification, a combination of resources were offered—including referrals to the National Hotline, site-specific mental health intake phone number or offer of urgent in-person consult, or referral back to their primary care physician—but these were commonly declined. However, our team had success triaging resources in situations where the SRMP yielded a high risk classification. Suicidal ideation and behavior may be endorsed by only a small number of study participants; however, the importance of patient safety outweighs the administrative burden to develop and implement an SRMP.
Evaluation of iatrogenic risk was not within the scope of this study; however, we did not observe any deaths due to suicide in this study which involved repeated measures of mood and, when indicated, discussions about suicidal ideation and behaviors. This observation is consistent with formal evaluations of the iatrogenic effect of suicide assessment, which have debunked the myth that asking an individual about suicide increases their suicide risk. 11 Further, we found that by embedding this SRMP within the study protocol, we were able to provide a proactive risk management clinical research environment whereby suicide risk could be identified, triaged, and effectively managed. As suicidal ideation within 2 weeks of eligibility screening was an exclusion criterion for this study, our experience using the SRMP highlights the fact that suicide risk can fluctuate throughout the course of a clinical trial and ongoing monitoring of suicide risk is recommended.
Limitations of this report include that some issues faced by our team may be site-specific or specific to an older study population. These data suggest this protocol is an effective strategy for managing disclosure of suicidal ideation in the context of data collection, but the low rate of high risk episodes limits our ability to fully evaluate clinical outcomes from the analysis. Additionally, our small sample size precluded statistical analyses of patient characteristics or SRMP activation outcomes within our cohort. Knowledge gaps regarding the management of participants with suicidal ideation and behavior via SRMPs can be categorized into (1) the organizational and operational features of these protocols and (2) the effectiveness of these protocols in increasing patient safety. We further explore some of the conceptual challenges to highlight opportunities for future investigation.
Timing and setting of identification of suicidal ideation and behavior
SRMPs must account for a variety of situations in which suicidal ideation and behavior can be encountered, including by (1) mail-in survey, (2) electronic survey portal, (3) telephone or other remote data collection, or (4) in-person study visit. When data collection occurs in the absence of research staff, as is generally the case for mail-in or online assessment, there is usually a time lapse between disclosure of suicidal ideation and behavior and identification and potential intervention by research staff. If disclosure of suicidal ideation and behavior occurs during telephone or in-person assessments, immediate intervention by research staff is possible. In addition, SRMPs may need to include procedures for suicidal ideation and behavior identified during recruitment, before the individual has provided informed consent.
Special populations and settings
SRMPs may need tailoring for research in populations where suicide risk is a known concern (e.g., veterans, patients who are homeless or have mental health disorders, and people who are incarcerated) or in other vulnerable populations (e.g., minors). For example, the 2020 U.S. veteran suicide rate was 57.3% higher than the non-Veteran adult suicide rate. 35 Resources are available to veterans that may not be available to the general population. The Veterans Crisis Line (1-800-273-8255*1 or 988*1) can connect callers with suicide prevention coordinators situated within all major Veterans Affairs Health Care Systems who have outreach protocols in place to connect veterans with resources. 36 In populations where mental health or substance abuse disorders are prevalent, ,for example in homeless populations, SRMPs may need to include referrals to community-based resources (e.g., 1-800-662-HELP, the Treatment Referral Routing Services). Studies that include homeless individuals may benefit from coordinating with local shelters to establish protocols that are feasible, practical, and will safeguard participant welfare while maintaining fidelity to study procedures. 37 Individuals who are incarcerated are known to have a higher rate of suicide than the general non-incarcerated population 38 Best practice standards for suicide prevention in correctional facilities, such as those set by the U.S. National Commission on Correctional Health Care, provide a useful framework for researchers to design an SRMP that is specific to the correctional facility setting. For research involving children and adolescents, guidelines for contacting a parent or legal guardian of a child at increased risk may be necessary.
There are specific settings in which identifying suicide risk for an individual is an important consideration as it may require additional management in an already overtaxed environment. For example, identifying suicide risk in medical settings, especially acute care settings such as an emergency department, or other settings required to manage high volumes of patients such as primary care, are important contexts to consider how to best collaborate with healthcare partners. Research shows that medical providers in emergency medicine and primary care value having access to consulting behavioral health professionals or having these professionals available in a multidisciplinary team-based model of care as well as having a known referral mechanism for mental health treatment as important in effectively management suicide risk.39,40 Consultation with relevant stakeholders and careful consideration of resources for research conducted in these settings are important factors for designing an effective SRMP.
Infrastructure support
Electronic database software with interactive skip logic and auto-prompts can help research staff navigate a SRMP appropriately and efficiently.14,15 Electronic SRMPs can be programmed to accommodate multiple types of participant-staff interaction, risk level cut-off points, and guidance for staff to escalate participants to appropriate levels of care. Multi-site trials have the additional challenge of harmonizing SRMP procedures and documentation across sites, which can be further complicated if Institutional Review Board (IRB) or Data Safety Monitoring Board (DSMB) oversight is not centralized.
Communication between clinical research and clinical practice
Recent studies have highlighted a need for improved communication between clinical research and clinical practice.41,42 If a participant activates an SRMP, establishing communication with the primary care or mental health provider to promote continuity of care is ideal. However, there are practical challenges to this practice, including that the research might occur outside of the provider’s medical system. For community-based research, a participant’s provider may be unknown entirely unless this information is collected during enrollment. Sometimes suicidal ideation and behavior are identified during screening, prior to informed consent. Disclosure of patient health information, including mental health information, is permissible in the event it is necessary to prevent imminent self-harm or harm to others. 43 But what communication is appropriate, or necessary, if a participant is determined to harbor low risk for suicide? Given the paucity of evidence to guide researchers in the development and implementation of SRMPs, collaboration with local IRBs and DSMBs is likely necessary to establish protocols that weigh privacy and confidentiality regulations against necessary steps to minimize risk.
Considerations for staffing
Study design and start-up should involve careful consideration of research staff qualifications and experience. Suicide risk assessment is a significant responsibility and research staff have reported discomfort administering suicide assessment questions. 44 It is critical that the assessments do not exceed the expertise or scope of training of staff. Investigators should provide appropriate training, including crisis de-escalation skills for a smooth hand-off to a provider with specialty mental health training, and opportunities for debriefing after the suicide risk protocols are activated.
Conclusions
Depressive disorders and suicidal ideation are prevalent conditions. Therefore, it is important to include patients with depression in clinical trials that address other medical conditions, like tobacco use, as exclusion will limit generalizability of results. Future studies are needed to provide empirical evidence on the efficacy of SRMPs in non-psychiatric research. Key issues include prevention of poor clinical outcomes and the balance between efficient data collection to reduce participant and research staff burden and appropriate attention to mental health concerns. There is a need to assess the psychometric performance of standardized suicide risk instruments in the context of clinical trials, including patient outcomes. Future studies employing longitudinal designs could provide more information about the degree to which SRMPs are truly effective in preventing suicide.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Cancer Institute at the National Institutes of Health (R01CA196873) as part of the Program for Lung Cancer Screening and Tobacco Cessation (PLUTO Trial).
Trial Registration
ClinicalTrials.gov identifier number: NCT02597491.
