Abstract
Introduction:
Although people with mental health disorders are more likely to die by suicide, individuals experiencing suicidality are frequently excluded from clinical trials of mental health treatment due to safety and liability concerns. This approach limits the generalizability of trial results and opportunities for intervention. This descriptive study aimed to report outcomes and lessons learned for a suicide risk management protocol implemented for participants reporting suicidal ideation in a comparative effectiveness clinical trial that enrolled patients screening positive for posttraumatic stress disorder or bipolar disorder. Specifically, we examined the proportion of trial participants reporting suicidal ideation, their chosen risk management plan, suicide attempts, and death by suicide. Also, because few studies have examined whether the survey modality of suicide screening impacts endorsement rates, we compared suicide ideation endorsement, patient demographics, and chosen risk management plans across phone and web survey modalities.
Methods:
Descriptive statistics were used to report the proportion of participants in the comparative effectiveness trial who reported suicidal ideation and activated the suicide risk management protocol, as well as the chosen risk management plans for those with active suicidal ideation. Chi-square tests of independence and Fisher’s exact tests were used to test for differences in demographics, screening question responses, and chosen risk management plans, respectively, between web versus phone survey modalities among those that activated the suicide risk management protocol.
Results:
Of the 1004 participants in the trial, 72% endorsed current suicidal ideation or previous suicidal behavior at baseline and activated the study’s suicide risk management protocol. There were two suicide attempts in the sample (0.28%), and one of which resulted in death (0.14%). There were no statistically significant differences in SRMP activation between phone and web-based survey modalities. Among participants who activated the suicide risk management protocol and endorsed active suicidal ideation, selection of risk management plans did not vary by survey modality. Participants most frequently opted to visit their community health center (42%) or to call the National Suicide Prevention Lifeline (32%) as their chosen risk management plan.
Discussion:
We developed and implemented the suicide risk management protocol for a multisite clinical trial enrolling patients with complex mental health conditions. Although a higher proportion of participants activated the SRMP compared to previous trials, rates of suicide attempts and suicide deaths were low. Our findings indicated no differences in positive screening rates among trial participants and no differences in safety plan selection by survey modality among participants entering the SRMP. This suggests that similar protocols may be used to screen for and manage suicidality in clinical trials, and protocols can be administered via phone and web-based surveys.
Keywords
Introduction
In the United States, suicide rates increased 36% from 2000 to 2021. 1 Suicide risk is substantially (up to 10–30 times) higher than the general population among individuals with mental health disorders such as posttraumatic stress disorder (PTSD), 2 depression, 3 and bipolar disorder. 4 Including individuals at risk for suicide in clinical trials for these conditions is key to optimizing the generalizability of research. Participating in these trials may also offer individuals an opportunity to receive evidence-based mental health care.
Suicidality refers to an elevated risk of suicide and encompasses a range of suicidal ideation and behaviors. Suicidal ideation may include passive suicidal ideation, or thoughts related to wanting to be dead, without intent or plan. Active suicidal ideation may include varying degrees of intent, planning, and securing of methods.5,6 Death ideation refers to thoughts related to death and dying without a specific focus on suicide, but is potentially a precursor to suicidal ideation and subsequent behaviors. 7 Screening for suicidality with a standardized instrument or protocol can uniformly identify individuals at risk for suicide. Once identified, individuals at risk for suicide should have a more comprehensive evaluation to confirm suicide risk, estimate the imminence of risk, and formulate treatment recommendations, including a risk management plan connecting the participant to appropriate care. 8
To maximize the generalizability of clinical trials of mental health interventions, individuals at elevated risk for suicide should not automatically be excluded. In an examination of randomized clinical trials (RCTs) focusing on online depression interventions, more than 70% of these trials excluded individuals screening positive for suicidality. 9 Similarly, Iltis and colleagues observed high and increasing exclusion rates of individuals with suicidality in RCTs of pharmacological treatments for depression. In drug trials from 1991 to 2000, 72% of studies excluded individuals with suicidality, and from 2000 to 2013, 86% of the drug trials excluded individuals with suicidality. 10 Exclusionary practices of individuals with suicidality in pharmacological RCTs have also been identified in recent antipsychotic clinical trials. 11 A 2020 meta-analysis of 48 RCTs of clinical interventions for PTSD, spanning publications from 1980 to 2019, revealed similar trends, with 65% of studies excluding individuals with suicidal ideation. 12 Reasons for exclusionary practices include lack of familiarity and proficiency among clinical trialists in implementing protocols to monitor and manage suicide risk.
In a 2009 survey of 125 research ethics committee members from several countries, 65% of respondents suggested that participant suicidality might be exacerbated or “reinforced” by the inclusion of questions pertaining to suicidal thoughts or feelings. 13 Safety and ethical considerations of conducting research with individuals at increased risk for suicide are salient concerns and frequently cited as factors preventing inclusion of participants experiencing suicidal ideation in research. 14 However, research indicates that suicide screening and assessment are associated with reductions in suicidal ideation,15–18 which calls into question the ethics of excluding such individuals from clinical trials. Prioritizing participants’ safety while producing research findings generalizable to populations at risk for suicide is challenging but possible.19,20
Establishing effective suicide risk management protocols (SRMPs) in multisite clinical trials poses additional logistical challenges, requiring considerable planning to establish streamlined assessment, data monitoring and reporting processes, and local knowledge of clinical resources available in the study participant’s location. 21 Common data collection methods include staff administered questions and self-report surveys, including written, web, phone, email, and text surveys.8,22 Past findings indicated suicidal ideation and previous suicide attempts are endorsed less frequently in phone administered interviews vs in-person interview or web-based survey. 23 As such, it is necessary to consider the impact of survey modality on potential underreporting bias inaccuracy of the suicide risk data collected when designing SRMPs.
The objective of this article is to describe the SRMP used in a large, multisite clinical trial enrolling patients screening positive for PTSD and/or bipolar disorder. In this post hoc descriptive analysis, we examine the prevalence of positive suicidality screening in the baseline survey of the trial and compare these proportions across web and phone survey modalities. We also share rates of suicide attempts and death by suicide among trial participants over the 12-month follow-up period. Among participants screening positive for suicidality, we compare demographic and clinical factors, and risk management plans chosen between participants who self-selected phone or web survey modalities. In alignment with previous findings, 23 we hypothesized that the frequency of positive suicidality screening would statistically significantly differ by survey modality, with a greater frequency of positive screening among participants responding via web versus phone. We also hypothesized that risk management plan selection would statistically significantly differ by survey modality, with more participants responding via phone opting to call the National Suicide Prevention Lifeline.
Methods
Participants
The current analyses involve data originally collected during the Study to Promote Innovation in Rural Integrated Telepsychiatry (SPIRIT) trial, a pragmatic comparative effectiveness trial that compared two telehealth approaches to managing PTSD and bipolar disorder in primary care. The trial was registered at ClinicalTrials.gov (NCT02738944). Individuals were invited to participate in SPIRIT if they were receiving primary care at one of 12 participating Federally Qualified Health Centers in three U.S. states (Washington, Michigan, and Arkansas). Individuals were eligible if they screened positive for PTSD via the 6-item Posttraumatic Stress Disorder Checklist (PCL-6 ≥ 14) 24 and/or for bipolar disorder via the Composite International Diagnostic Interview (positive stem question responses and score ≥ 8) 25 during a primary care wellness visit. Patients were ineligible for inclusion if they were currently prescribed psychotropic medication by a mental health specialist, lacked capacity to provide informed consent, or did not speak English or Spanish. There was no exclusion criterion related to suicidal ideation.
Participants completed a baseline survey via phone or web, depending on personal preference. All procedures and materials were approved by the University of Arkansas for Medical Sciences, University of Michigan, and University of Washington Institutional Review Boards (IRBs). For additional details, see papers describing the trial protocol 26 and primary outcomes. 27
Measures
Demographic measures
Demographic information was self-reported via baseline survey, including date of birth, gender identity, race, ethnicity, employment, education, health insurance status, and ZIP code (to assess rurality). Annual household income and reported number of household members were used to assess financial status using 2016 Federal Poverty Level data. 28 Rurality was assessed using the Rural-Urban Commuting Area Categorization D. 29
Other clinical survey measures
Other measures collected include the 20-item PTSD Checklist-5 (PCL-5) 30 to assess past-month PTSD symptom severity and the Internal State Scale (ISS) 31 to monitor mood state. PCL-5 response options range from 0 (not at all) to 4 (extremely) and are summed to produce a total score of 0–80. Higher scores indicate greater severity of PTSD symptoms. The ISS is a 15-item instrument to identify and track mood state in patients with bipolar disorder. The instrument’s activation subscale is used to identify hypo(mania), mixed state, euthymia, and depression mood states.
Death and suicidal ideation and behavior measures
Hopkins symptom checklist depression scale
Death ideation and suicidal ideation were assessed in the baseline survey using two items from the 20-item Hopkins Symptom Checklist Depression Scale (SCL-20), 32 which asks participants how much they were distressed by various feelings within the past two weeks. Participants responded to questions about “Thoughts of death or dying” (death ideation) and “Thoughts of ending your life” (suicidal ideation), indicating responses from 0 (not at all) to 4 (extremely). Any response other than “Not at all” to either question activated the SRMP.
Borderline personality disorder screener
The 15-item self-reported borderline personality disorder screener from the Structured Clinical Interview for DSM-IV (SCID-II) 33 was included in the baseline survey. One Borderline Personality Disorder screening item, “Have you tried to hurt or kill yourself or threatened to do so?” was used to assess previous suicidal behaviors. Response options included “Yes” or “No,” and any response other than “No” activated the SRMP.
Procedure
Suicide risk management protocol (SRMP)
During the SPIRIT trial, the SRMP was activated by endorsement of one of two SCL-20 and/or one borderline personality disorder baseline survey questions. A complete description of the SRMP is available in Supplementary File 1. See Figure 1 for a flowchart of the SRMP. Briefly, participants who endorsed death ideation, suicidal ideation, or past suicidal behavior on at least 1 of the 3 screening items in the baseline survey or answered, ‘Don’t Know “Refused” or who did not answer activated the first SRMP suicidal ideation question: “Do you have thoughts of ending your life today or in the near future?” Participants responding “Yes,”‘Don’t know,’ or “Refused” to this question were immediately randomized to a treatment arm so the suicidality information could be sent through automatically generated emails to the relevant provider team. Participants responding “No” received contact information for the National Suicide Prevention Lifeline before continuing with the baseline survey. Participants responding with anything other than “No” continued through the remaining steps of the SRMP. By the end of the protocol, participants selected from four risk management plans: calling the National Suicide Prevention Lifeline, contacting their local crisis center, visiting their community health center during normal hours, or visiting their nearest emergency room. In cases where no risk management plan was selected during a phone survey, administrators relayed that they were not trained professionals and were required to have their supervisor call emergency responders (e.g. local sheriff) for a wellness check. Risk management plan options were reoffered. If no plan was selected, the study team (researchers, clinicians, survey supervisors) was notified by automatic email, and a participant wellness check was requested. In cases where no risk management plan was selected during a web-based survey, the study team was immediately notified via email and communicated with the participant’s clinic, instructing them to call local emergency responders to conduct a wellness check for the participant. Participants could activate the SRMP multiple times, in which case they were asked to reconfirm their previous responses (see Supplementary File 1).
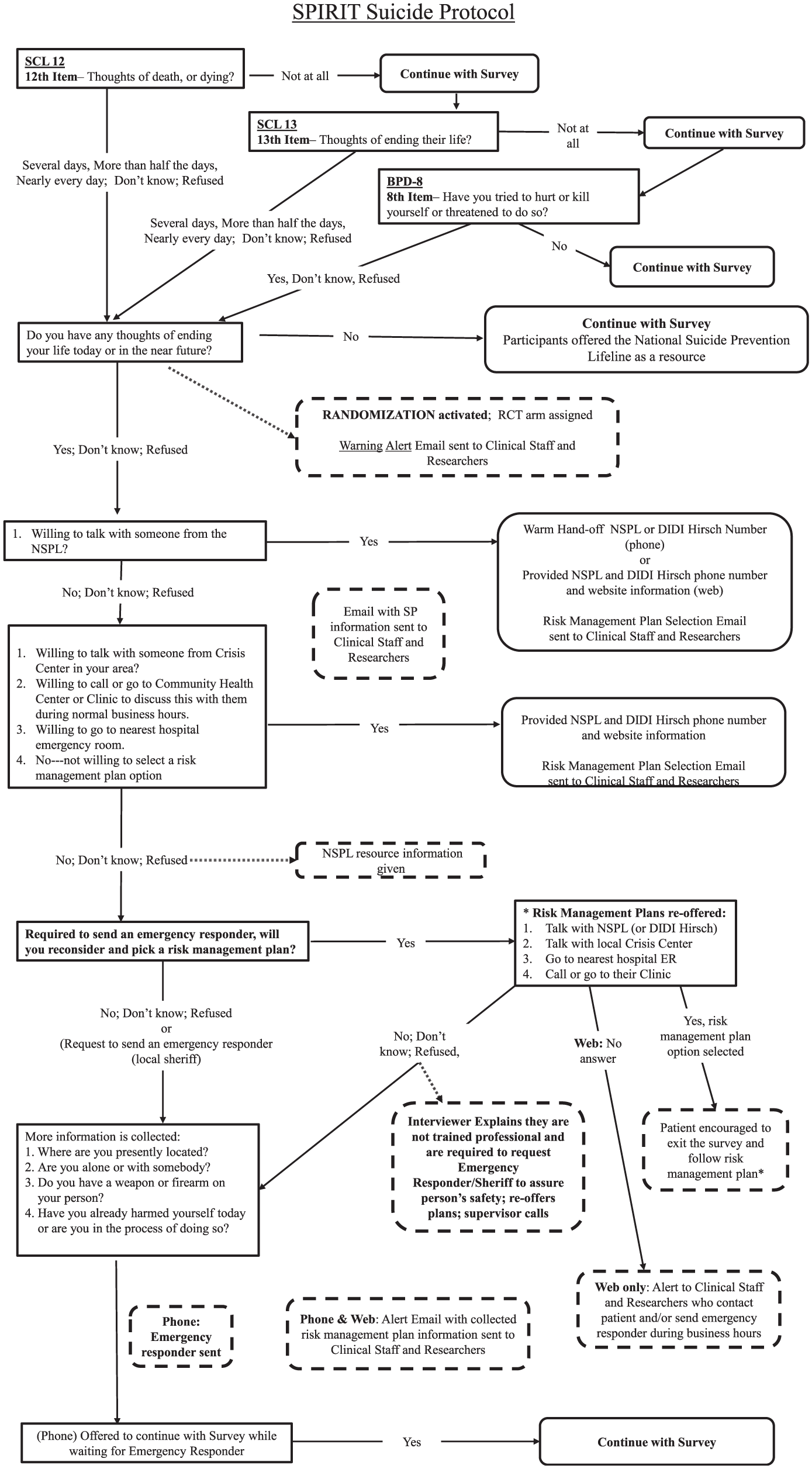
SPIRIT suicide risk management protocol.
Data analysis
Descriptive statistics of responses to death ideation and suicidality screening questions among SPIRIT participants (n = 1004) were totaled using baseline survey data. We also reported rates of suicide attempts and deaths by suicide. Chi-square tests of independence were used to determine if responses to the three screening questions and activation of the SRMP statistically significantly differed by survey modality. Among participants activating the SRMP, t-tests, chi-square tests of independence, or Fisher’s exact tests (when n < 5) were used to determine if self-selection to web or phone survey modality statistically significantly differed by demographic characteristics. A Fisher’s exact test was used to examine if chosen risk management plans differed by survey modality for those with active suicide ideation that continued through the SRMP. Analyses were completed in RStudio Version 4.3.2. 34
Results
Demographic and clinical characteristics
Table 1 presents descriptive statistics. The average age was 39.4 years, and most participants were female (70.3%, n = 703). Most of the sample identified as white (70.3%, n = 699), followed by Black or African American (11.9%, n = 118) and “Another race” (7.3%, n = 73). Most (71.8%, n = 721) participants in the trial endorsed suicidality on one or more screening questions at baseline and activated the study’s SRMP. Among participants activating the SRMP, choice of survey modality statistically significantly differed by age, gender, race, education, employment, poverty level, rurality, depression, and mood state (see Table 1).
Baseline participant characteristics.
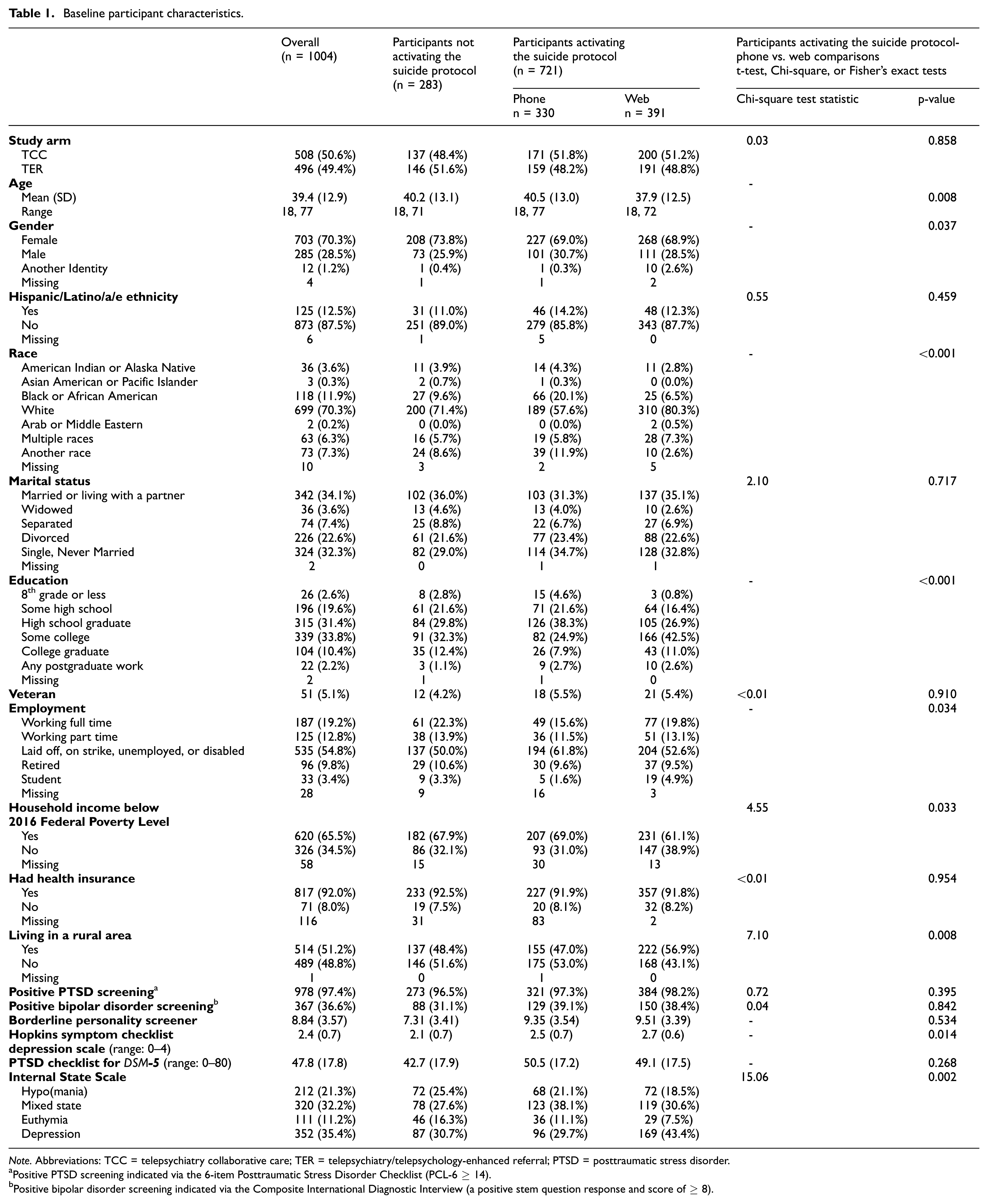
Note. Abbreviations: TCC = telepsychiatry collaborative care; TER = telepsychiatry/telepsychology-enhanced referral; PTSD = posttraumatic stress disorder.
Positive PTSD screening indicated via the 6-item Posttraumatic Stress Disorder Checklist (PCL-6 ≥ 14).
Positive bipolar disorder screening indicated via the Composite International Diagnostic Interview (a positive stem question response and score of ≥ 8).
Screening question responses
Responses to the SCL-20 and borderline personality disorder suicidality screener questions are presented in Table 2, stratified by survey modality. Most participants activating the suicide protocol indicated death ideation by providing a response other than “Not at all” to the SCL-20-based question, “How much in the past two weeks have you been distressed by thoughts of death or dying?” Of the 721 participants who activated the SRMP, 102 (14.1%) expressed active suicidal ideation in the first SRMP question (“Do you have thoughts of ending your life today or in the near future?”) and continued the SRMP to choose a risk management plan.
Participant responses to screening questions.
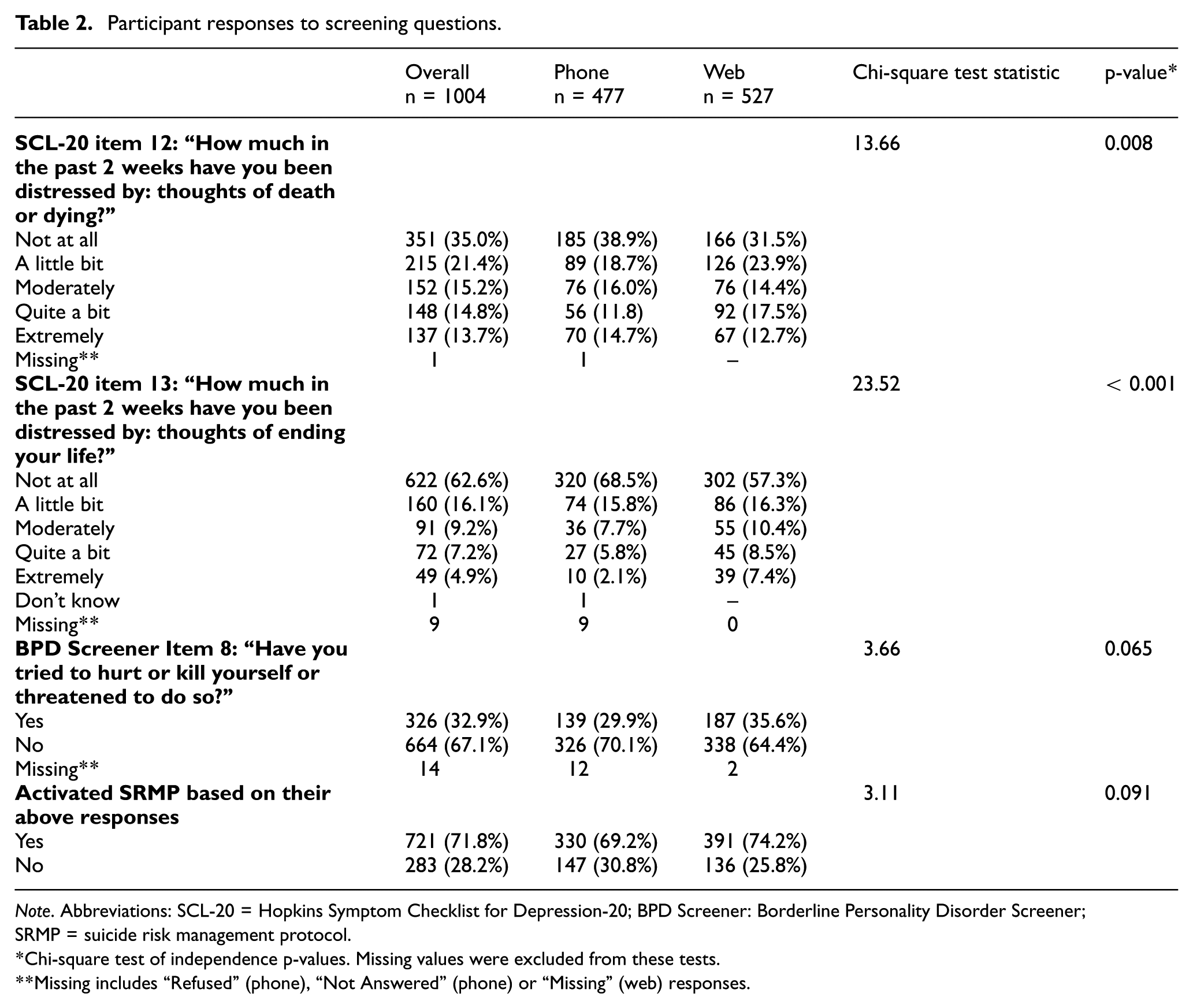
Note. Abbreviations: SCL-20 = Hopkins Symptom Checklist for Depression-20; BPD Screener: Borderline Personality Disorder Screener; SRMP = suicide risk management protocol.
Chi-square test of independence p-values. Missing values were excluded from these tests.
Missing includes “Refused” (phone), “Not Answered” (phone) or “Missing” (web) responses.
Chi-square tests of independence indicated SCL-20 death ideation question responses were statistically significantly different; 38.9% (n = 185) of participants responded “Not at all” via phone compared to 31.5% (n = 166) via web. This was similar for the SCL-20 suicide ideation question (68.5% of participants (n = 320) responded “Not at all” via phone compared to 57.3% via web (n = 302)). Responses to the borderline personality disorder screening item were not statistically significantly different across survey modalities. Participant activation of the SRMP one or more times was not associated with survey modality (see Table 2).
Risk management plans
Among participants reporting active suicidal ideation and responding by phone, the National Suicide Prevention Lifeline was the most frequently chosen plan (41.1%, n = 23). Web participants with active suicidal ideation most frequently opted to visit their community health center (52.2%, n = 24). Chosen plans were not statistically significantly different between survey modalities. Seven participants (6.9%, n = 7) did not select a risk management plan, either by hanging up the phone (n = 2) or logging out of the web survey before completing the risk management plan questions (n = 5). Nearly half (40.5%, n = 292) of participants activated the SRMP more than once. Most (93%, n = 271) participants re-activating the SRMP indicated they did not have present thoughts of ending their life when asked for a second time and thus did not select an additional risk management plan. See Table 3.
Risk management plans chosen by participants reporting active suicide ideation.
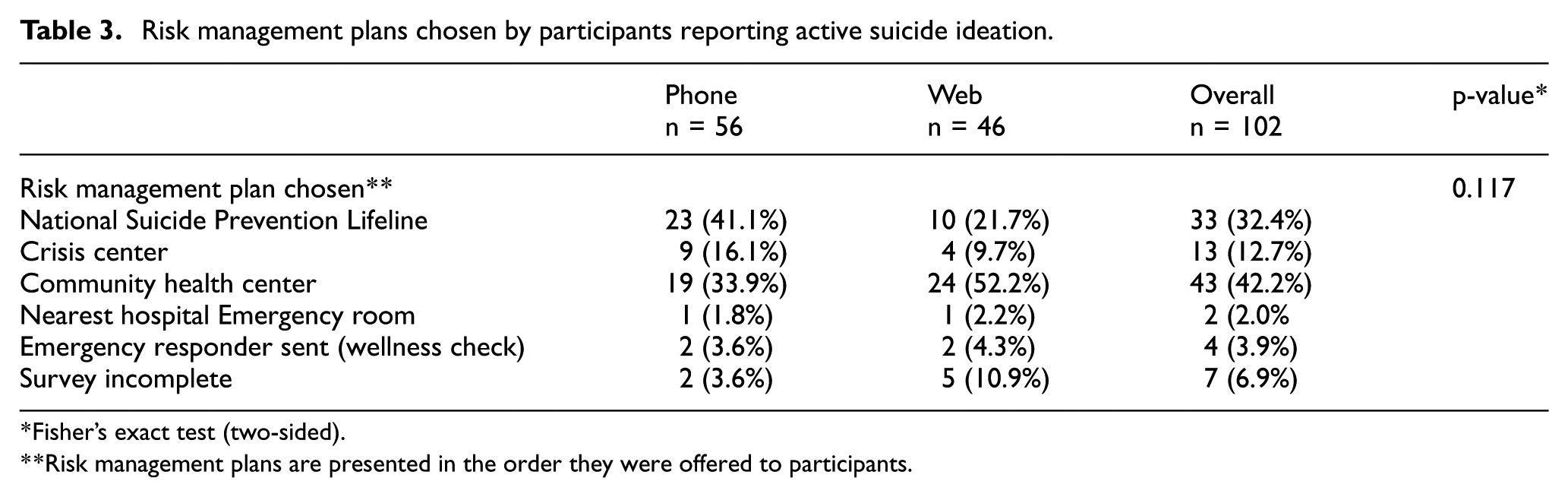
Fisher’s exact test (two-sided).
Risk management plans are presented in the order they were offered to participants.
Suicide-related serious adverse events
During the trial, one participant was hospitalized after attempting suicide and another participant died by suicide. Details on these participants’ responses to the screening and SRMP questions and their associated IRB determinations are available in Supplementary File 2.
Discussion
Among patients screening positive for PTSD and/or bipolar disorder, 72% of participants endorsed or did not respond to suicidal ideation screening questions and activated the SRMP. This rate was higher than is typical of most research studies, which was an estimated 46% in a recent meta-analysis of nearly 100 studies. 35 Our sample demonstrated low rates of suicide, with two attempts (n = 2; 0.28%), one of which resulted in death (0.14%). This finding is clinically meaningful given our sample comprised people screening positive for PTSD and/or bipolar disorder and thus at increased risk of suicidal ideation and dying by suicide. These findings indicate that SRMPs, like the one used in the SPIRIT trial, can be used to monitor and respond to suicidal ideation among clinical trial participants at elevated risk for suicidality.
Frequencies of responses to the SCL-20 death and suicidal ideation screening were statistically significantly different across modality, with more web participants endorsing than phone participants. However, frequencies of responses to the borderline personality disorder screening item, activation of the SRMP, and risk management plan selection did not vary significantly by modality. Our findings that more participants reported ideation via web vs phone are aligned with previous studies which found people are more likely to disclose when answers are perceived as more anonymous.23,36,37 For example, Milton et al. 38 found young men disclosed suicidal ideation more frequently in web-, rather than phone-based surveys. Participants self-selected their preferred modality of research assessments at the start of the study and only one modality was used (unlike the within-person design used by Milton et al.); therefore, our findings are not directly comparable to existing literature. Preferred modality was statistically significantly different across demographics and clinical factors, though SRMP activation and risk management plan selection did not significantly differ by modality.
The prevalence of risk management plans chosen was not statistically significantly different by survey modality. When selecting risk management plans, most participants chose to visit a community health center (42%), followed by the National Suicide Prevention Lifeline (32%). It should be noted that the National Suicide Prevention Lifeline was the first option presented across modalities, and participants completing the survey via phone were transferred to the National Suicide Prevention Lifeline by the interviewer if they chose that option. We are the first (to our knowledge) to compare risk management plans chosen across modalities during a clinical trial SRMP, so additional research is warranted on risk management plan selection.
Lessons learned
Despite successfully assessing suicidality risk and developing risk management plans with large numbers of study participants, one participant died by suicide. Although people who die by suicide are less likely to disclose suicidal ideation than community samples, 35 this represents a potential opportunity to reduce this participant’s risk. One lesson learned is to underscore the importance of providing additional training, scripting tools, and advice to phone survey administrators in the case of a patient who changes their answer to suicide questions, as this person did. We advise not allowing survey software to overwrite previous responses indicating suicide risk but rather store these responses in a new field and flag these cases for further checking. This ensures the history of responses can be accurately determined when reporting serious adverse events to the IRB and maximizes the chance of addressing suicide risk through SRMP implementation.
In addition, rather than using a single question to assess current thoughts of self-harm, a measure assessing the spectrum of suicidality with evidence of psychometric validity, sensitivity, and specificity, such as the Columbia Suicide Severity Rating Scale, 6 should be used. This would provide more information to clinicians to better help their patients. Note that questions about lifetime suicidal ideation should not activate the SMRP because this does not capture current ideation and is less clinically actionable.
When a study participant declines all presented risk management plans, necessitating a wellness check per our safety protocol, the participant should be informed about its purpose in ensuring safety, and inherent risks associated with unplanned interactions with authorities, especially law enforcement. 39 Wellness checks from law enforcement may increase risk of harm to participants, especially among individuals from communities of color.40,41 Vital information about the participant’s level of risk and presence of weapons should be communicated to law enforcement for a safe and effective wellness check (see Supplemental File 1). 42 If available, trained mobile crisis units should be asked to conduct wellness checks instead of law enforcement. Participants should also have an opportunity to reconsider and choose an alternative risk management plan. When a participant selects a risk management plan and exits the survey to follow it, they should be asked if they followed the plan immediately upon survey re-entry. They should not be allowed to continue the survey until they report invoking their chosen plan or another effective solution for mitigating risk. Finally, it is important to clearly document all SRMP changes, including reasons for changes and the dates changes were made. This helps facilitate reporting of serious adverse events to the IRB, contextualizes data at study completion, and most importantly, minimizes participants’ suicide risk.
Implementation lessons learned include optimizing clinician notification of participant responses to questions about suicidality. Initially, clinicians were emailed after participants chose a risk management plan to minimize clinician burden. However, because some participants closed the web-based survey prior to selecting a risk management plan, no email was sent. Therefore, we modified our SRMP so clinicians were notified when their patient responded yes to active suicidal ideation and again when they selected a risk management plan. Collaborating with participating clinicians to design SRMPs is critical to balance the sharing of time-sensitive clinically relevant patient information and minimizing clinician burden. 43
Strengths, limitations, and future directions
Strengths of this study include a large sample of participants with diverse identities and backgrounds across multiple states in the United States. The SRMP was developed by a multidisciplinary research and clinical team that continuously refined its protocol throughout the study, integrating new insights and best practices to improve participant safety. The study successfully screened and facilitated risk management planning for a large sample of participants with elevated risk of suicidality in a multisite trial. Inclusion of participants with suicidal ideation increases generalizability of trial results but requires planning and resources to manage risk and is a potential source of liability for researchers. Our results are potentially limited in generalizability beyond Federally Qualified Health Center patients who chose to participate in a clinical trial. Other limitations include the use of an active suicide ideation question developed for the study without prior psychometric evaluation and not assessing suicidal intent before initiating risk management planning during the SRMP, both of which would have been addressed using the Columbia Suicide Severity Rating Scale. We also did not assess the cost of developing and implementing the SRMP, which would have helped researchers prepare budgets for future studies. Future directions include qualitative work to explore experiences and preferences of participants who completed research SRMPs, cognitive interviews to determine why people respond “don’t know” or refuse to answer screening questions, and co-designing SRMPs with people with lived/living experience of suicidality to optimize participant experience and safety. Questions about chronicity and fluctuations of suicidal ideation and related behaviors will likely strengthen SRMPs. Future work could translate temporal dynamics of suicidality research to improve risk identification. 44
Conclusion
We presented an SRMP for a multisite pragmatic clinical trial with primary care patients who screened positive for PTSD and/or bipolar disorder. Most participants (72%) endorsed current death ideation, suicidal ideation, or previous suicidal behaviors and activated the SRMP, but subsequent rates of suicide attempts and completion were low. Our findings suggest this protocol is effective for suicide screening and risk management and can be administered via phone and web-based surveys.
Supplemental Material
sj-docx-1-ctj-10.1177_17407745251389222 – Supplemental material for Implementing a suicide risk management protocol as part of a multisite clinical trial: Findings and lessons learned
Supplemental material, sj-docx-1-ctj-10.1177_17407745251389222 for Implementing a suicide risk management protocol as part of a multisite clinical trial: Findings and lessons learned by Erin Chase, Nicole Moreira, Brittany E Blanchard, Julien Rouvere, Lori Ferro, Jared M Bechtel, Danna L Moore, Daniel Vakoch, Keyne C Law, Jürgen Unützer and John C Fortney in Clinical Trials
Supplemental Material
sj-docx-2-ctj-10.1177_17407745251389222 – Supplemental material for Implementing a suicide risk management protocol as part of a multisite clinical trial: Findings and lessons learned
Supplemental material, sj-docx-2-ctj-10.1177_17407745251389222 for Implementing a suicide risk management protocol as part of a multisite clinical trial: Findings and lessons learned by Erin Chase, Nicole Moreira, Brittany E Blanchard, Julien Rouvere, Lori Ferro, Jared M Bechtel, Danna L Moore, Daniel Vakoch, Keyne C Law, Jürgen Unützer and John C Fortney in Clinical Trials
Footnotes
Acknowledgements
We would like to thank the patients and staff of Family Health Centers, Moses Lake Community Health Center, Sea Mar Community Health Centers, Yakima Neighborhood Health Services, Boston Mountain Rural Health Centers, East Arkansas Family Health Center, Lee County Cooperative Clinic, Cherry Health, Family Medical Center, Great Lakes Bay Health Centers, InterCare Community Health Network, and Upper Great Lakes Family Health Center. We would like to thank the staff of the Community Health Plan of Washington, the Community Health Centers of Arkansas, and the Michigan Primary Care Association. We would also like to thank our Policy Advisory Board: Donald Weaver (National Association of Community Health Centers), Ed Larkins (Family Medical Centers of Michigan), Jay Shore (University of Colorado at Denver), LaShannon Spencer and Victoria Miller (Community Health Centers of Arkansas), Nancy Speck (National Association for Rural Mental Health), Sara Coates and Dawne Velianoff (Michigan Primary Care Association), Susan Ward-Jones (East Arkansas Family Health Center), Jay Shore (University of Colorado at Denver) and Kim Arnold (National Alliance on Mental Illness). We would like to thank our Consumer Advisor Board: Bernadette Mcdonald, Florence Fee (No Health without Mental Health), Ingrid Deetz, Kevin Einbinder and Betsy O’Brien (Depression and Bipolar Support Alliance), Kim Arnold, Marquitta Magnini and Shawn McCown (National Alliance on Mental Illness), Jode Freyholtz-London (Wellness in the Woods), Tammy Heral, and Jeremy Mullins. We would like to thank the members of our external Data Safety Monitoring Board: Thomas Belin (University of California, Los Angeles), John Williams (Duke University), and Mark Williams (Mayo Clinic). We would also like to acknowledge the survey team at the Social & Economic Sciences Research Center including Nikolay Ponomarev, PhD, and Rose Krebill-Prather, PhD. Finally, we would like to thank Katherine Comtois, PhD, MPH for providing feedback on this manuscript and sharing her expertise in suicide prevention and care.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Patient Centered Outcomes Research Institute (PCS-1406-19295). Dr. Blanchard was supported by University of Washington’s Institute of Translational Health Sciences KL2 Program through the National Center for Advancing Translational Sciences of the NIH (KL2TR002317) and the National Institute on Drug Abuse Loan Repayment Program (L30DA056956).
Trial registration
The pragmatic trial was preregistered with ClinicalTrials.gov (Identifier: NCT02738944).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
