Abstract
Background
Previous findings indicate important effects of stress, disrupted sleep, and dysregulated circadian rhythms on the progression of cancer. Our understanding of these relationships, however, has been limited by the absence of data from 24-h continuous physiologic monitoring of hormonal and immune activity with simultaneous collection of objective sleep data. The present study successfully employed such an intensive protocol among women with advanced breast cancer.
Purpose
In this paper we describe our protocol in detail. We provide methodological and practical suggestions for assessing sleep and circadian rhythms in the medically ill. Finally, we offer psychological and physical safety guidelines for future studies that aim to utilize similar protocols.
Results
We enrolled 123 women with advanced breast cancer; 18 dropped out, 105 participated (6 provided incomplete data, and 99 completed the protocol). The protocol included: (1) psychological/medical screenings and assessments; (2) a 2-week at-home sleep study, including wrist actigraphy and two nights of polysomnography (PSG); and (3) 28 h in-hospital collection of measures of hormones and immune function, in conjunction with one night of PSG for assessing sleep stages, respiration, and leg movement. Based on the implementation challenges we faced, protocol adjustments were made to address feasibility limitations and to accommodate participant preferences and medical/physiological needs.
Conclusions
By combining and modifying the gold standard protocols for sleep and circadian assessments, researchers can compassionately optimize participant enrollment and protocol compliance, and minimize attrition.
Introduction
Sleep disruption is common among individuals with cancer.1,2 In addition to reducing quality of life, sleep disruption is associated with shorter cancer survival in numerous studies, suggesting it may have medical implications.3–9 Sleep debt, the accumulation of insufficient sleep, can be directly detrimental to the functioning of physiological systems critical to cancer defense, such as natural killer cell activity. 10 Cytokines, in turn, affect CNS function, affecting mood and sleep. 11 Sleep loss also disrupts numerous modulators of immune function necessary for disease recovery, 9 including the sympathetic nervous system, hypothalamic-pituitary-adrenal (HPA) axis hormones, and cytokines. 12 Dysregulation of the autonomic nervous system, as measured by respiratory sinus arrhythmia, mediates the relationship between poor sleep and shorter survival in women with metastatic breast cancer. 13 Other studies have linked HPA dysregulation to cancer progression, showing flatter diurnal cortisol slopes (a potential biomarker for disrupted feedback inhibition of the HPA-axis) 14 are predictive of shorter survival with cancer.4,15,16 We have also observed that lower NK cell number is associated with the flatter diurnal cortisol patterns that predict survival time in breast cancer. 17
The relationship between disrupted sleep and cancer may also be mediated by the desynchronization of the circadian timing system from rest-activity cycles. Support for this comes from numerous studies linking varied forms of circadian disruption exposure to breast cancer risk. 18 For example, two large-scale studies found increased risk of breast cancer after extended nighttime shift work.19,20
Despite the indirect evidence for the potential involvement of the circadian system, few studies have examined the 24-h circadian rhythms of the endocrine and immune systems of cancer patients in concert with subjective and objective measures of sleep/wake activity. 21 ; In general, the extant studies have imputed circadian function through examination of gross motor activity patterns or the daytime slope of cortisol. For example, one study found potential circadian abnormalities, as imputed by abnormal actigraphy data and diurnal cortisol patterns, predicted tumor progression, its conclusions suggesting that either circadian dysfunction may drive changes in biomarkers favoring tumor growth, or the alternative—that tumors may lead to endocrine/circadian abnormalities. 22 The use of both activity patterns and daytime cortisol slope are indirect measures of circadian function as they measure variables that are influenced by the circadian system, but whose underlying circadian component can be masked by behavioral and environmental variables, making them inherently inadequate markers. 23
Controlled studies using rigorous methodological approaches are needed to examine how objective sleep and circadian functions are related to biomarkers of disease progression and survival. To fill these gaps in the literature and improve on existing methodological limitations, we conducted an observational investigation examining relationships among sleep quality, circadian cycles, and disease progression in women with advanced breast cancer (ABC). We systematically studied the full 24-h sleep and activity cycle, including objective and subjective measures of sleep and measures of immune and hormonal patterns. Here we describe the methodology necessary for recruitment, retention, and balancing ethical and practical concerns.
Methods and design
Overview of study design
This was an observational study examining the relationships among sleep quality, circadian cycles, and disease progression (clinicaltrials.gov registration #NCT00519168). All study procedures were approved by the Stanford University Institutional Review Board and conformed to the principles outlined in the Declaration of Helsinki. After meeting the inclusion/exclusion criteria, participants’ data were collected for two consecutive weeks at their homes and during a 28-h hospital visit. We then tracked women over the course of their survival, assessing for disease progression and collecting long-term survival data.
Aims
Our study sought to address the following aims: (1) to study 24-h diurnal rhythms of HPA axis hormones, immune measures, and melatonin in women diagnosed with advanced breast cancer; (2) to describe sleep disruption in women diagnosed with advanced breast cancer and to examine psychosocial, endocrine, and immune factors that may be associated with sleep disruption; (3) to study the relationship between sleep disruption and survival time among patients with advanced breast cancer (ABC); and (4) as an exploratory aim, to investigate whether sleep disruption mediates the relation of psychosocial factors to health outcomes. Our a priori hypothesis was that women diagnosed with ABC will have sleep disruption that causes reduced amplitude and disrupted phase of 24-h diurnal rhythms of endocrine and immune function, ultimately decreasing survival time.
Recruitment of participants
Study recruitment
Recruitment was done through various sources such as the Stanford Cancer Center, referring oncologists in the surrounding communities, participants from previous studies, study flyers located at various clinics throughout the San Francisco bay area and advertisements on the Internet. For the last 2 years of the study, most of the participants were recruited through the Dr Susan Love Research Foundation’s Army of Women® (AOW), whose initiative in part is to connect researchers with men and women willing to participate in breast cancer research studies. Due to our specific screening criteria and the cancer-related health limitations of our population, meeting recruitment goals had become a challenge, even with AOW’s collaboration. As a result, we expanded our catchment area from individuals who lived near Stanford CA to include participants living in all time zones within the United States; the protocol refinements necessary for this adaptation are described below. Participants were paid up to $800 for study completion. This remuneration was to reimburse them for any cost related to study participation, such as taking time off from work.
Screening procedures, consent, and enrollment
Initial phone screen (approximately 20 min). To minimize time burden, we conducted an initial intake phone screen. Study staff informed potential participants that they were required by Stanford’s Institutional Review Board to read from a telephone script to ensure that everyone received the same information. Women were asked about how and where they had learned about the study and basic contact information was collected. Of the women diagnosed with ABC, 67% (412 out of the 545 initially contacted) gave permission to have their screening data and contact information saved for data analysis and for being used to contact them in regards to potential participation in future research studies.
In person/phone consenting and in depth screening (approximately 2–4 h). Once the initial screening criteria were met, the potential participant was scheduled to come to Stanford for a more detailed screening and consent signing. Individuals who lived too far from Stanford to conveniently attend an in-person screening were screened by phone. Prior to their screening interview, potential participants were mailed or e-mailed a copy of the study consent form that included study contact information so that they could review the details of the study at their leisure and contact the team should they have any questions. In order to use the potential participant’s time more efficiently, forms and questionnaires (e.g. cancer diagnosis and treatment) were included such that they could be completed before the screening visit. The study investigator or a research associate reviewed the study consent form and protocol in detail with potential participants and answered any questions they had. More in-depth screening was done by administering the Mood and Substance Use Disorders modules from the Structured Clinical Interview for DSM-IV-TR Axis I Disorders (SCID - I) and collecting information regarding their cancer diagnosis and treatment. Once determined eligible, screened individuals indicating willingness to enroll were then scheduled for participation.
Rationale and List of Study Inclusion Criteria
Participants were postmenopausal non-smokers diagnosed with ABC. Participation also required scoring a Karnofsky rating of at least 70% (physical ability measure for medically ill patients) 24 and ability and consent to use a heparinized forearm catheter for blood sample collection. Women had to be willing to travel to Stanford, California for a 28-h hospital stay and agreed to abstain from travelling two or more time zones away from their local time zone for the 2 weeks prior to and during participation in the study. If women were not living within Pacific time zone, they were instructed to maintain their local time zone schedule during their three-night visit to California.
Postmenopausal. As most women diagnosed with ABC are likely to be postmenopausal due to age or cancer treatment-induced menopause, we enrolled only postmenopausal women, thus eliminating normal hormonal variation and sleep changes related to the menstrual cycle and menopause.
Non-smoker. Only non-smokers were enrolled as the study was conducted in a hospital in which smoking is not allowed inside the hospital. As participants were asked not leave their hospital room during the 28-h in-hospital sleep and circadian assessment (necessary to control for the effects of light exposure on outcomes (see below), acute nicotine withdrawal might be physiologically and psychologically stressful and could affect the outcome of the psychological, sleep, endocrine, and circadian measures.
Rationale and List of Study Exclusion Criteria
Exclusion criteria were designed to minimize potential risk to participants and control for factors that could influence outcomes. Women were excluded if they had any concurrent medical condition likely to influence short term survival; had active cancers (other than breast cancer, basal cell or squamous cell carcinomas of the skin, or in situ cancer of the cervix) within the past 10 years; utilized corticosteroids, glucocorticoids, or benzodiazepines (these medications affect the HPA measures) or melatonin within the week preceding and during their in-hospital study; had a history of major psychiatric illness that required hospitalization in the preceding year because of possible effects on the sleep measures, given that psychiatrically fragile populations often experience insomnia; or had current substance/alcohol abuse/dependence (these affect the HPA measures). To reduce the risk of lymphedema as a result of catheterization for blood draws, women were also excluded if they had bilateral lymph nodes removed. In addition, women who engaged in shift work (e.g. working 4pm–midnight or 10pm–6am) during 3 months prior to the study were also not included due to potential pre-existing circadian dysregulation associated with shift work schedules.
Scheduling Considerations
Several factors were considered when scheduling participants for study participation. To minimize the effects of chemotherapy on the immune measures during a 28-h hospital study, the participants on chemotherapy were scheduled a few days before the start of the next chemotherapy cycle. Women diagnosed with influenza and those who were taking antibiotics were rescheduled to minimize the impact of the illness and medication on the immune measures and sleep. For participants who did not live locally, we considered flight times, transportation, and hotel stays that would minimize the impact of travel on their sleep and circadian rhythms. Participants paid for their own flight, but the hotel and other transportation costs were covered by the study budget. Weekend schedules were made available for participants who worked and were not able to take time off during the work day.
Study Population
One hundred and five women diagnosed with ABC participated in the study (Figure 1). Six provided partial data (did not participate in the 28 h hospital stay), and 99 completed the study protocol. Participants had documented metastatic or recurrent breast cancer, were between 45 and 75 years old, and were proficient enough in English to complete questionnaires and follow instructions. Participants at minimum had graduated high school or obtained a certificate of high school equivalency and were U. S. Citizens or residents able to receive payment of the study stipend. For the 10 years following participation, individuals’ disease progression and mortality rates were followed by reviewing their medical records. Twenty-seven aged-matched healthy controls were also recruited for this study. The healthy controls underwent the same procedures as the ABC group. CONSORT diagram of study flow for ABC participants.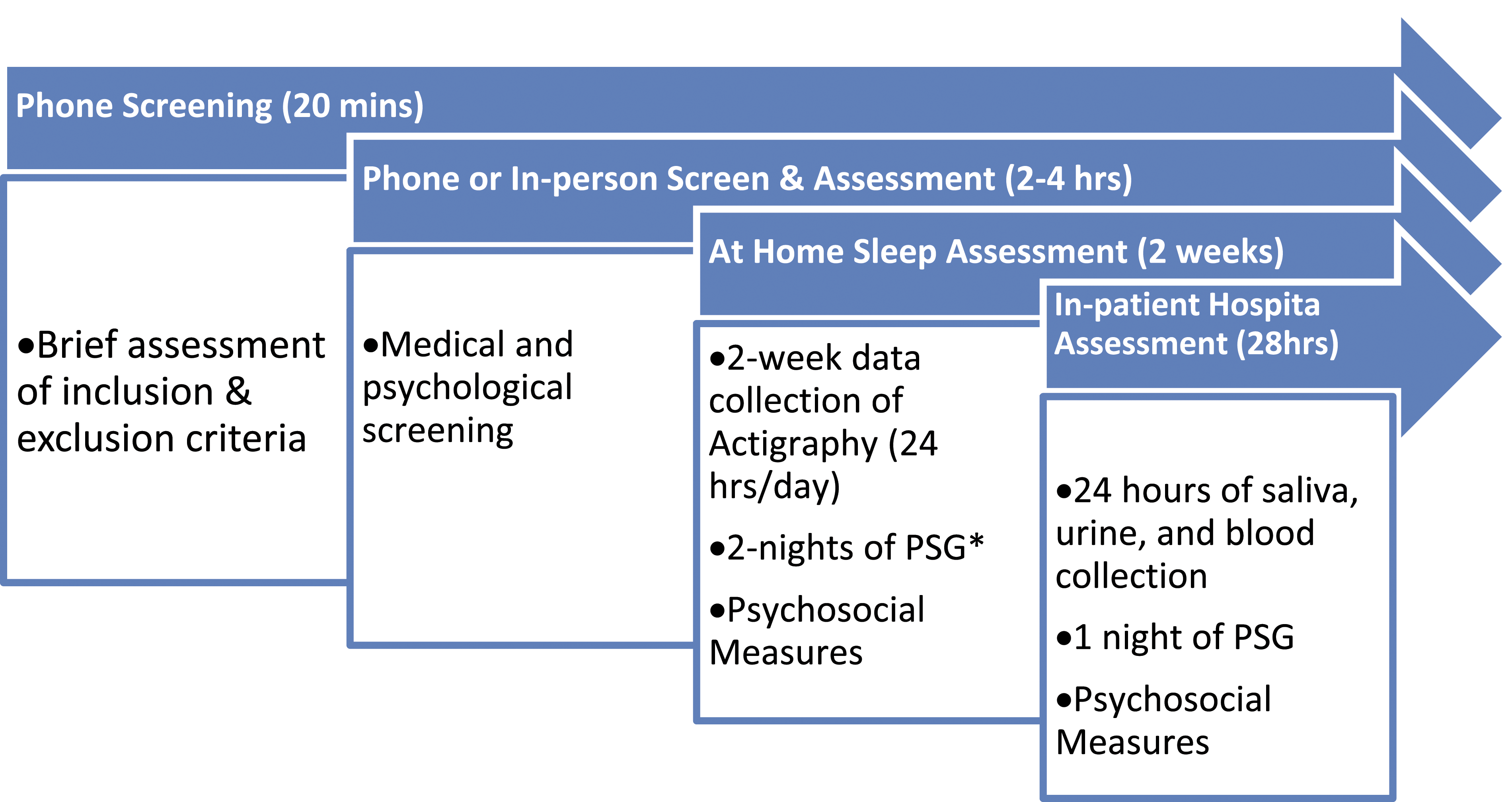
Study protocols for 2 weeks of at home data collection
Objective and subjective measures of sleep
Two-Week at-Home Sleep Study. The at-home sleep study was conducted over 14 consecutive days and it was scheduled to take place within a 1-month period prior to the 28-h in-hospital assessment. The 1-month proximity of these two assessments was important since the sleep/wake time for the 28-h in-hospital stay was determined by the average sleep/wake time obtained from the 2-week at-home sleep study. During this 2-week period, participants were asked to wear an actigraph (Actiwatch-L, Philips/Respironics), a small (16 g) wrist-worn device that records arm movement and is useful for determining sleep/wake patterns.25,26 We requested that participants wear the actigraph 24 h a day and to complete a daily sleep log. For the participants’ convenience, the 2-week data collection kit was mailed to them and upon its receipt, a staff member called to review the contents of the kit and the protocol in detail.
Two nights of ambulatory at home polysomnography (PSG). During the 2-week at-home sleep study, in addition to actigraphy, there were two nights of at-home polysomnography. Surface electrodes (Grass Instruments Co, Braintree MA) were taped to standard (International 10-20 System) locations on the face (bilateral electro-oculogram, EOG: above and below the outer canthi; electromyogram, EMG: chin) and to the scalp (electroencephalogram, EEG: C3, C4, Fz, Oz) via Quik-Cap (Neuroscan, Charlotte NC). Signals were collected by a Siesta system (Compumedics, El Paso TX) onto compact flash memory cards. To increase participants’ comfort, we assigned a sleep technologist to arrive about 2–3 h before each participant’s usual bedtime. Participants were given instructions on how to remove the electrodes the following morning. The next evening, the technician returned to reattach the electrodes for the second night of recording. For the participants who did not live locally, the protocol was conducted at a hotel near their 28-h hospital study, beginning either two nights before or one night after completion of the 28-h hospital stay. For any post-hospital PSG measurement, a one-night break was added to address any sleep rebound issues resulting from possibly disrupted sleep at the hospital. Participants from a different time zone were asked to keep their sleep schedule on their local time.
Subjective measures of sleep. A sleep log was completed within 30 min of waking each day. Using this log, participants recorded the time at which they went to bed and arose, how long it took them to fall asleep, how long they remained awake in bed before getting out of bed, and how many times they awoke at night. If the proximity of the 2-week at home protocol was more than 4 weeks prior to their in-hospital stay, they were asked to retake this questionnaire 1-week before their in-hospital stay. A nap log was completed just after the nap or before bedtime and allowed participants to record the timing and duration of any daytime naps. While the actigraphs were supposed to be worn continuously during the 2-week at-home data collection, if participants needed to remove their actigraph, they were asked to record the timing and duration in an actigraph log.
Psychosocial measures
ACASI, Audio Computer Assisted Self-Interview (ACASI). In an effort to obtain reliable self-report and increase participant comfort in disclosing sensitive information, we used audio computer-assisted self-interview (ACASI) instead of paper-and-pencil questionnaires. We employed Questionnaire Development System (QDSTM, Nova Research Company, Bethesda, MD) to implement ACASI. Our investigators’ team has successfully used ACASI in previous studies with similar samples. 27 We found that ACASI ensures participant confidentiality, assists those with low literacy, and minimizes staff support required for data entry. In addition, studies suggest that ACASI is effective at reducing response bias, minimize the requirement that respondents have to be literate and able to follow skip patterns (allow the ACASI to ask only those questions that pertain to the answers previously selected). 28 Also, ACASI results in reduction of data entry errors by project staff and can be programmed to include automatic range checking and internal consistency checks as the participant is entering her information.
Psychosocial measures were administered via ACASI during the 2-week at-home sleep study visit. In order to use the participants’ time efficiently, the psychosocial measures were administered during the second night of the at home PSG, while the sleep technician downloaded the data from the previous night and set up for the second night of PSG recording. The questionnaires took approximately 1 hour to complete. For list of these measures please see Appendix A.
Protocols for the 28 hour stay
This section of the study protocol was completed at the Stanford Hospital’s General Clinical Research Center (GCRC) or at the Stanford Sleep Medicine Center. It lasted for approximately 28 consecutive hours (24 h of blood draws). Participants were asked to arrive approximately 5 h after their typical wake time. The timing of all procedures that occurred during the 28-h stay were customized to each individual participant based on the sleep/wake data obtained from the 2-week at-home actigraphy data collection. Using these actigraphy data, the timings of sleep onset and offset were estimated for each of the 14 days, from which an average sleep onset and offset were calculated. The midpoint of the onset and offset of sleep was then calculated and used as the basis for the timing of all events during the 28-h stay. Notably, women were scheduled to sleep for 8 h in darkness, starting 4 h before the calculated midpoint of their at-home sleep. This procedure helps to align the sampling protocol with the approximate time of the circadian clock 29 and ensures that participants are sleeping during their habitual window of sleep.
To accurately assess the underlying rhythm of variables such as plasma melatonin and cortisol, certain environmental factors were kept constant during a 24-h window of blood sampling. During their 2-day stay, women were unaware of the timing of the study procedures and isolated from external time cues (including clocks, radios, television, visitors, sunlight), but maintained contact with staff members and were able to listen to recorded music and watch videos. They were allowed to bring in a laptop computer, but the clock and internet access was disabled before they could use it. Environmental temperature was maintained at 23°C ± 2°C. During the scheduled waking hours, participants were allowed to be ambulatory in their room, in which there was an en suite bathroom. During scheduled sleep hours, participants were required to remain in bed in a constant semi-recumbent position; they were asked to use the bed pan if they needed to urinate. Maintenance of posture (upright or seated during scheduled waking hours, semi recumbent during scheduled hours of sleep) was necessary to control for the effects of posture on apparent plasma hormone concentrations.30,31 Indoor light remained dim (<15 lux) and at the same illuminance throughout the study period, except during the scheduled 8 hours of sleep when the rooms were dark (<0.05 lux). No accessory lighting was available during the protocol. Constant low illuminaion was necessary to prevent the acute suppressive effects of light on the production of melatonin and shifting of the timing of the circadian system. 32 At no time during their stay in the hospital, other than what was scheduled, were participants allowed to sleep. During wake time, on an hourly basis, the participants received a small, isocaloric snack and an isovolumetric serving of water. The total nutrition and calories of a normal breakfast, lunch, and dinner were equally distributed in each snack, as calculated by hospital dieticians and based on the Harris-Benedict equation, adjusted for inactivity. 33 The participants were asked to drink all the water and eat all the food whenever possible. Blood samples were collected 1 to 3 times per hour, with the increased rate occurring from 4 hours before bedtime until 2 hours after bedtime (including during sleep). A total of 222 cc of blood were obtained over the sampling protocol. Urine samples were collected every 3 h or ad libitum during the sleep period. Saliva samples were collected before bedtime and in the morning of day 2.
Research nurses were present 24 h a day to carry out the protocol and collect biologic specimens. The PI and study co-investigators provided initial and ongoing in-service to train the nurses for conducting the protocol. The project director and an on-call physician were available during the hospital stay to answer any protocol questions or issues with the participants. Study flow sheets were provided to the nurses detailing each step of the protocol.
Study staff would met with participants upon arrival, come in for checking in during the 24 h stay, and at the end for debriefing and walking the participants to their cars.
In-hospital sleep protocol
28-h In-Hospital Sleep Study. The same sleep recording montage was used in the hospital as was used during the at-home recordings, with the addition of bilateral leg electromyogram (EMG), nasal cannula, thoracic and abdominal pressure bands, body position monitor, and pulse oximetry. For participants’ comfort, the PSG hookup was scheduled to begin 1–2 h before bedtime.
Blood collection procedures
Upon admission, a peripheral saline lock was placed in a forearm or hand vein on the side contralateral to the affected (either from a sentinel lymph node procedures, axillary sampling, or axillary lymph node dissection) arm and a single sample was drawn for measuring hematocrit (HCT). If the participant preferred, their port-a-cath was allowed to be used for this sample only. If HCT was equal or greater than 32, the protocol was continued. If HCT was 30 or 31, the protocol was continued and the HCT was repeated 12 h later. If at that time or at the time of the initial draw, the values were equal or lower than 29, blood draws were stopped for the remainder of the protocol and saliva and urine samples were collected in place of blood when possible.
For the intravenous line, the smallest possible needle was used, starting with an 18 gauge and going to a 20 gauge, if necessary. A 1.5 m intravenous line was attached to a saline bag with an electronic pump set to provide an intravenous microdrip infusion of saline with heparin (500 mL bag of 0.9% sodium chloride with 1000 U heparin – 2 units/mL) at a very slow rate (10 mL/h, TKO) between blood samples. The use of such a heparin anticoagulation procedure, although somewhat uncommonly used by the staff, proved necessary to avoid closing of the line through clot formation. In front of the saline bag was a triple stopcock manifold. A syringe attached to the stopcock closest to the participant was used draw mixed saline/blood. Once whole blood reached the manifold, the next stopcock was used to draw the whole blood sample (2–8.5 cc per draw). Once a sample was drawn, the mixed saline/blood was reinfused and the line was cleared with a saline flush. To minimize sleep disruption during the draws scheduled to occur during sleep, an identical apparatus (stopcock, IV bag) with an additional 1.5 m extension tube was set up in the adjacent room. A light-shielded porthole in the wall allowed connection of the extension tubing between the participant and the adjacent room. The switch between the apparatuses occurred just before lights out (bedtime). Given the minimal amount of heparin, no problems were anticipated, however, as a safety measure the last blood draw was used for measuring partial prothrombin time (PTT). To avoid any possible interference from the heparinized line a different site was used for this draw. The PTT was done to confirm that the heparin was not affecting clotting time. A total of 221.5 cc of whole blood was drawn over the course of the 24 h of blood draws, which is less than half of which is normally drawn when donating blood. We intentionally kept the volume low given risks of lower hematocrit in our participants. PTT was assessed in 68% (N 67) of the participants. Of those 3% (N 2) had marginally higher values in the upper range of normal.
Urine collection procedures
The volume of all urine was recorded and a 10 mL sample saved and frozen at −80°C.
Salivary collection procedures
Saliva was collected at three time points: (1) 9 p.m. on day one, and (2) at wake and (3) 30 min after wake time on day 2. Saliva was collected using untreated cotton Salivettes (Sarstedt, Newton NC). Saliva was spun in a centrifuge for 2 min at 1000 ⨯ g and the resulting supernatant stored at −80°C.
Psychosocial measures during in-hospital procedures
Participants completed a battery of computerized psychosocial measures on an hourly basis during wake time. The computer would prompt the participant on an hourly basis to complete the battery. See Appendix B for psychosocial measures administered during the in-hospital stay.
Primary outcome measures
Home and in-hospital PSG for total of three nights
PSG data were analyzed using Pro Fusion PSG software (Compumedics, El Paso TX) and scored in 30-s epochs after the method of. 34 Sleep scoring yielded both total amount and percentage of stages 1, 2, 3, and four of NREM sleep, REM sleep, and wake time. Other calculated metrics include time available for sleep, total sleep time (TST), wake time after sleep onset (WASO), sleep and REM onset latency, sleep efficiency, and the number and type of arousals throughout the night. The Apnea-Hypopnea Index, number of leg movements resulting in arousals, and time in minutes that participants slept with a blood oxygen content below 90% saturation was also calculated from PSG data collected during the in-hospital stay.
Actigraphy
Actigraphy data were analyzed using Sleepwatch software (v.2.82, Cambridge Neurotechnology, Cambridge UK). The actigraphy and light exposure data, used in conjunction with the sleep self-reports, were used to approximate the timing of both daytime and nighttime sleep. The light exposure data were also used to examine if self-reported sleep disruptions were due to unusual patterns of nocturnal light exposure. Actigraphy is a common way to examine ambulatory patterns of sleep and wakefulness. 25
Self-Report of Sleep
Nap Log. Participants recorded their at-home napping during the 2 weeks of the at-home data collection. This log was used to aid in the analysis of the actigraphy data (see above).
Actigraph Log. This log was used to aid in the analysis of the actigraphy data (see above).
Sleep Log. The information regarding the self-reported timing of sleep further aided in the analysis of actigraphy data (see above). This questionnaire includes the Stanford Sleepiness Scale that was used to examine daily subjective sleepiness.
Endocrine measures
Serum obtained from whole blood was stored at −80°C for later assay of melatonin and cortisol using commercially-available enzyme immunoassay kits.
Flow Cytometry. Whole blood was collected into EDTA and sodium heparin tubes, and maintained at room temperature. Blood from the EDTA tube was used to obtain white blood cell counts on a hematology analyzer (Beckman Coulter, Brea, CA). Blood from the heparin tube was used to enumerate specific leukocyte subtypes using flow cytometry (FACSCalibur, Becton Dickinson, San Jose, CA).
Serum IL-6. Every 4 hours, blood was drawn into serum separator tubes for the measurement of IL-6. Cytokine concentrations were measured in duplicate using commercially available, ultra-sensitive ELISAs.
Secondary outcome Measures
See Appendix A for Psychosocial Measures.
Protocol modifications and adaptations
Revisions were made to the study protocol in order to accommodate the study population, as well as to improve recruitment and retention. The combination of at-home actigraphy and PSG with in-hospital PSG and blood assessment allowed us to best characterize the sleep and circadian systems of the participants.
Actigraph
Actigraphy provides a low burden, non-invasive, objective assessment of sleep and wakefulness patterns and was used to corroborate participant sleep complaints. Using 14 days of collection, rather than just 1 week, reduces random variability and provides greater accuracy of typical sleep.
PSG
The PSG assessments of sleep provide information to characterize the physiologic cause of actigraph-determined and self-reported sleep disruptions (e.g. sleep apnea, change in EEG power spectrum, sleep onset or maintenance difficulties, early morning awakening, periodic limb movements, etc.).
Adaptations to sleep measurement
We considered a 4-day, 3-night stay at the GCRC to obtain data that might be more typical of normal sleep patterns (day 1: disrupted sleep, day 2: sleep debt recovery, day 3: most typical) and to reduce the effect of large inter-night variability in sleep parameters. However, there is controversy in the sleep literature about whether this protocol provides truly typical sleep patterns. Furthermore, we felt that this would restrict generalizability because a substantial number of eligible women might refuse to spend 4 days and three nights in the hospital. Given the possibility that even three nights might not provide typical sleep data, we opted for two nights of in-home sleep monitoring. This provided excellent objective sleep data and allowed us to compare in-home and in-hospital assessments. A reduced PSG montage (i.e. fewest electrodes necessary to obtain valid sleep scoring) was used in the at-home study to minimize disruption of participant sleep. The additional sensors required for assessment of breathing and limb movements were used in the hospital-based PSG assessment. The unusual hospital environment coupled with the additional sensors makes the in-hospital PSG assessment less useful for determination of “normal” sleep patterns, but allowed for determination of limb movements, breathing-related arousals, and to obtain in-hospital sleep measurements concurrent with assessment of 24-h hormone profiles (i.e. direct association between concurrent sleep and endocrine patterns).
Endocrine measures
The assessment of hormones in the blood was used to examine basal circadian function and its relationship to the observed sleep patterns. The use of these three complementary, overlapping techniques are a powerful way to substantiate sleep hormonal characterizations.
Constant routine protocol
A constant routine protocol is designed to remove or hold constant factors that would otherwise obscure underlying, endogenous circadian function. The typical protocol 35 is 40 h long (i.e. 2 days awake, missing one night of sleep) and occurs in constant dim light with participants remaining in bed in a constant posture the entire time. These protocols are often conducted in time isolation, to avoid knowledge of time influencing subjective responses to questionnaires, and food and water are given in hourly aliquots. Our protocol was modified in two ways. First, it was reduced from 40 h of continuous wake to 16 h of continuous wake. Second, it no longer required participants to remain in bed in a constant posture (CP) for their entire 24-h assessment. The use of a 40-h constant routine would have lengthened the protocol by a day and included a night of sleep deprivation, both of which we felt were burdensome to participants. As such we just examined the 24-h endocrine profile in the presence of sleep and selected markers of the circadian system (melatonin, cortisol) that are only minimally affected by sleep.36,37 The constant posture is used to reduce fluid shifts between the blood and interstitial space that occur during shifts between supine and standing.30,31 Such shifts can change the apparent hormone concentrations without reflecting changes in actual hormone production or release. By limiting posture to seated/standing during the daytime and lying during the nighttime, we attempted to minimize the effect of postural shifts on apparent hormone concentrations. Other aspects of the constant routine were maintained and accepted by participants.
Bedpan during sleep time
Participants were instructed to use a call button, placed on the bed rail, to contact the nurses should they need anything during sleep time, they were also given a sleep mask to use as needed in order not to be exposed to light. Before entering the room, nurses would check with the participant to be sure they had their sleep mask on. To minimize postural shifts during sleep, participants were asked to use a bedpan if they needed to urinate upon awakening during sleep time. The bedpans were provided without turning on the lights. Of the 99 women who completed the protocol, 70 (71%) voided during sleep time. Of these, 22 (31%) were not able to use the bed pan and had to get up and use the bathroom. Since some of the initial participants reported trying to not urinate at night in order to avoid using the bed pan, as they were worried about spilling, feeling embarrassed, and trying very hard but unable to urinate or have a bowel movement in a supine position, we shifted the protocol instructions for the nurses to first provide the bedpan and, if unable or unwilling to use, then offer the use of the toilet. Use of the toilet was also done while the participant was wearing a sleep mask guided by the nurse carrying a flashlight for safety.
Diet
To address participants’ dietary concerns some minor accommodations were made. Participants could adjust the volume of hourly water during the first three snacks, after which time, the volume of water provided was constant. Hot water or herbal tea (non-caffeinated) was available instead of room temperature or cold water, except for the last drink before bedtime. Before their arrival, the participants were given a few choices of sandwiches/snacks to select from, such as peanut butter and jelly or turkey. All ingredients were organic, and they had their choice of bread (e.g. white, wheat, gluten free).
Retention and sustainability strategies
In order to better prepare the participants and improve adherence to the protocol, participants were given as much detail as possible at various time points of the study. The details of the protocol were described in the consent form, but at each phase they would receive detailed instructions to review, followed by a phone or in person meeting with a staff member to go over the details and answer any questions. At all times, a staff member was available by cell phone to address any questions or concerns. The participants received copies of their PSG reports and clinical blood tests done during the 28-h hospital stay. If needed or requested, study physicians would communicate the results (notably those related to sleep disordered breathing) with the participant’s health care team. Study staff kept in in touch with participants via email and phone to provide them with study results as they become available, and the study investigator and his team were available for answering any questions about the results. The overall philosophy of our team was that our participants are part of our research team working towards a common goal, and we attempted to have our participants experience this.
Participant safety
The study inclusion/exclusion criteria were designed keeping in mind the participants’ psychological and physiological safety in addition to study requirements, in particular during the in-hospital protocol. Participants were asked about any difficulty they might experience during their 28-h hospital stay in regards to being confined to their dimly lit room during wake time, as well as the 8 h of required darkness during sleep time. Difficulties such as claustrophobia, previous negative/traumatic experiences related to hospital/medical settings (including receiving cancer diagnosis and/or recurrence/metastasis as well as their experiences during treatment), and past or current history of trauma/abuse. Physiological safety guidelines established by the consulting oncologist excluded women with any combination of bilateral sentinel lymph node procedures, axillary sampling, or axillary lymph node dissection (in order to reduce the risk of lymphedema), average hematocrit values equal or lower than 29, and peripherally-inserted central catheter (PICC) lines or ports as the only available site of blood draws.
Follow-up and study endpoint protocols
As discussed above, over the 10 years following the conclusion of the in-hospital portion of the protocol, participants’ disease progression and mortality rates will be followed by reviewing their medical records.
Study fidelity procedures/Assessment of compliance
Given the complexity of the protocol, various strategies were employed to remind the participants about their schedules and of the details of the protocol. Initially they were provided with a simple table of events for the entire protocol, similar to Figure 2, and then the details of the protocol were reviewed for each time point. The study consent form also contained the protocol details, and it followed the same order of procedures as the table of events so that they could use the consent form as a reference. Once they were scheduled for study participation they were provided with a copy of the calendar containing their schedule, both in paper and electronic format. Study Design. *If not from the San Francisco Bay Area, the two nights of the PSG were done in a hotel when the participant traveled to Stanford for the 28-h hospital stay.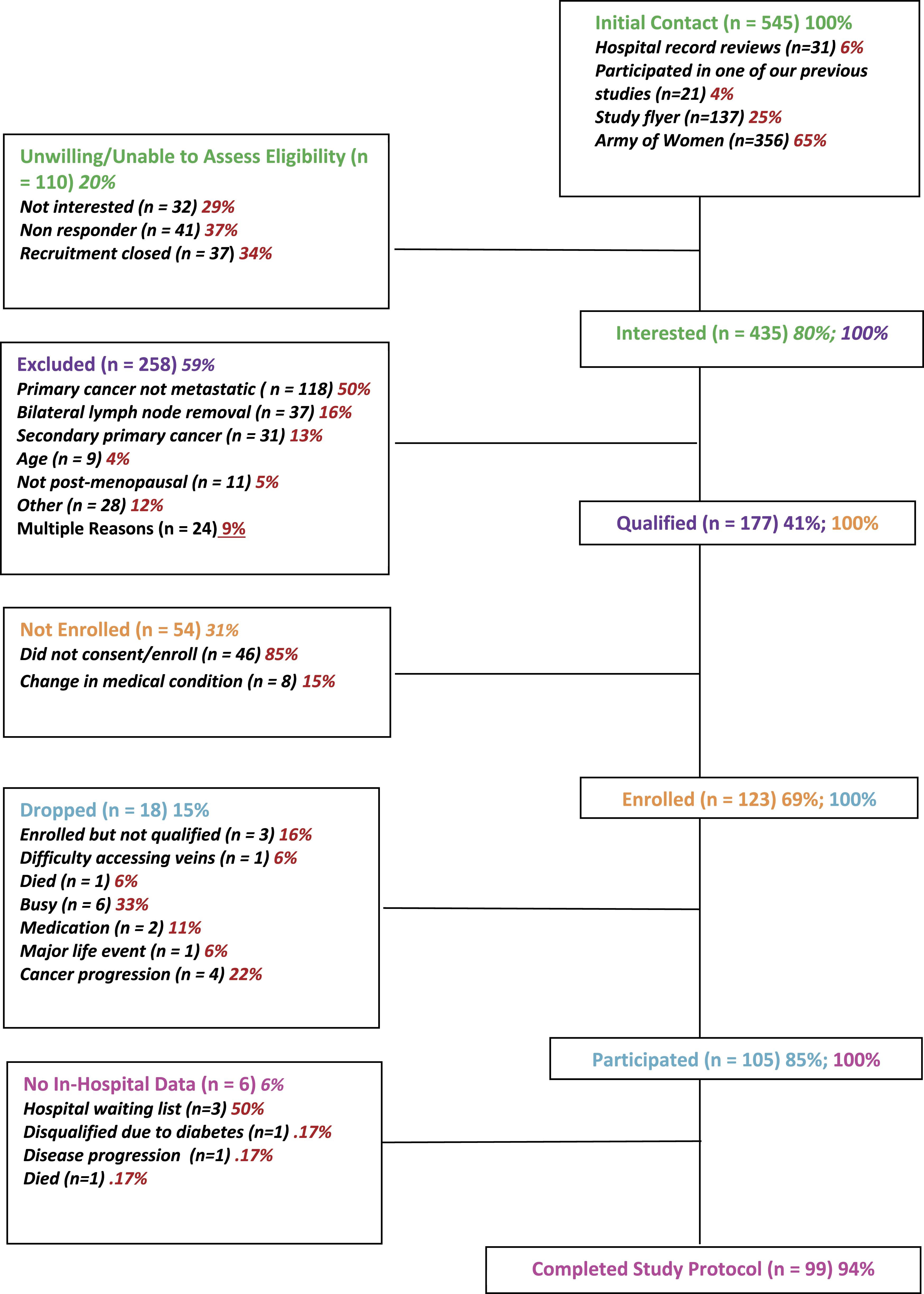
Two week at-home data collection. Once the participant received the at home data collection kit by overnight mail, study staff would call to review the details of the protocol, and there was always someone available to answer any questions they had during the 2 weeks. Once the completed kits were returned, study staff would review the data and contact the participant for any missing information. The study staff would also review the PSG and actigraph recordings.
Twenty 8 hour in-hospital stay. Details of the protocol were mailed or e-mailed to the participants and were reviewed one to 2 weeks before their scheduled hospital stay.
The psychosocial measures administered during these two time points were computerized and were designed to prompt the participant for any missing data before proceeding to the next question. We maintained regular contact with participants via phone and mailings throughout the study.
Study progress - Results
Recruitment results
Data were collected over a 4 year period, with first participant being enrolled in July 2007 and last participant visit being at the end of July 2011. A total of 123 women with ABC were enrolled, 18 dropped before any data was collected. From the 105 women who participated, 99 completed study participation (Figure 1), meeting the recruitment goal. As shown in the CONSORT diagram (Figure 1), out of 177 eligible participants diagnosed with ABC, 123 (69%) enrolled to be in the study, 18 (15%) dropped before any data was collected, partial data was collected for 6 (85%), and 99 (80.5%) completed the study protocol. We also recruited and enrolled a healthy control comparison group of 27 women, exceeding the recruitment goal of 20. As expected, the completion rate was slightly higher for healthy controls (89% vs. 80.5%) than for women diagnosed with ABC, but the difference was not statistically significant (z = 1.03, p = 0.3, test?).
Participant characteristics
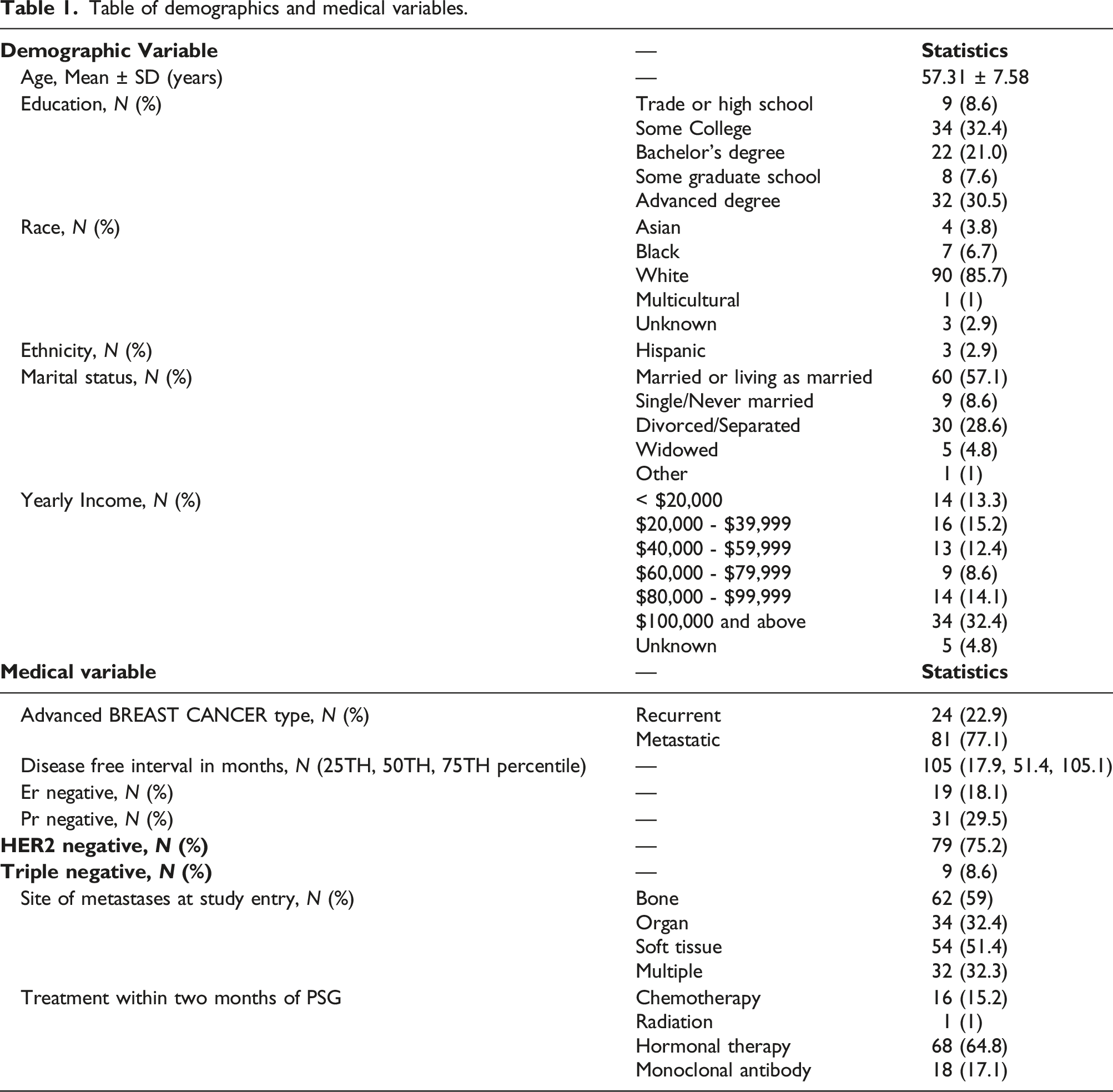
Table of demographics and medical variables.
Adherence and retention
From the 123 women initially enrolled, 18 dropped out or withdrew (14.63% attrition). Accordingly, 105 women participated in the study protocol; six of these women provided incomplete data (no in-hospital stay), leaving 99 who completed the entire study protocol, including both at-home and in-laboratory data.
Among the 18 enrolled participants who did not participate in the study protocol (i.e. dropped out or withdrew), for six, their work and life schedules became too demanding leaving no time for study participation; four had cancer progression and were not able to participate due to poor health; three were disqualified (one was not able to get confirmation from her oncologist that there was no bilateral lymph node removal, one had brain metastasis post-enrollment, one had two primary breast cancer diagnoses and not a recurrence); two were on excluded medications (medications containing benzodiazepines or steroids) and were not able to stop during the period required by the study; one had difficulty with venous access for blood draws; one had a family crisis; and one died before study participation. For the six who participated but did not complete the in-hospital portion of the study protocol, three were not able to be scheduled before the study completion deadline, one was disqualified due to the diagnosis of diabetes, one became too ill due to cancer progression, and one died.
Discussion and Conclusions
Lessons learned
While there are study designs meant to obtain the “perfect” data sample, in clinical populations, these protocols are often excessively burdensome to accomplish or to justify their use. Modifications to the design of research protocols in clinical settings are often made without reference to methodological articles demonstrating the consequences of such decisions. In this study, we have empirically examined these questions and report on practical methods to facilitate further research in this area. Below we recommend seven principles for designing and implementing research on sleep and circadian rhythms in women with advanced breast cancer.
1. Take steps in the design of the study protocol to minimize risk to participants. To become a participant in this study, women agreed to the use of heparin for blood collection. Heparin is used as an anticoagulant to prevent the formation of blood clots blocking IV patency. Physiological safety guidelines established by the consulting oncologist excluded women with any combination of bilateral sentinel lymph node procedures, axillary sampling, or axillary lymph node dissection (in order to reduce the risk of lymphedema); peripherally-inserted central catheter (PICC) lines or ports as the only available site of blood draws; and hematocrit values equal to or lower than 29. Moreover, the maximum amount of blood drawn during the 28-h hospital stay was not to exceed half a pint (<250 cc). Participants were asked about any difficulty they experienced during their 28-h hospital stay with regard to being confined to their dimly lit room during wake time or to 8 h of required darkness during sleep time. Difficulties reported were related to claustrophobia, previous negative/traumatic experiences related to hospital/medical settings (including receiving cancer diagnosis and/or recurrence/metastasis, as well as their experiences during treatment), and past or current history of trauma/abuse. We found that most subjects expressed appreciation for the special care they received and found participation in the hospital stay generally pleasant.
2. Choice of disease status depends on the research question pertaining to sleep. Women diagnosed with advanced breast cancer were the focus of this research because the primary aims pertain to disease progression. This particular population is important to study for several reasons. Many studies have reported sleep difficulties in this population. We were interested in looking at disease progression and survival, thus this outcome is more relevant to this population in comparison to, for example, women diagnosed with primary breast cancer.
3. Design the study to minimize extraneous influences on the assessments. Research nurses who were trained in the study protocol were available 24 h/day to conduct the study procedures in a standardized manner. Room temperature and indoor light were kept constant during the in hospital stay. Food and drink were standardized as well, with minimal variation allowed. Rescheduled women diagnosed with influenza and women who were taking antibiotics to minimize the effects of illness and medications on the measures of sleep and immune function. Women’s in hospital assessments were tailored to their individual sleep-wake cycles as assessed by the 2-week home actigraphy collection. For women who flew to the study location from other time zones, flight times were chosen to reduce the effects of disruption in their sleep schedule.
4. Reduce study burden on participants. To reduce burden, an important aspect of the study design was to limit the duration of the in-hospital stay for the sleep assessment to approximately 28 h so as to include 16 h (versus 40) of continuous wake. Many women diagnosed with breast cancer already have sleep challenges. It would have been a great burden to ask, especially women with advanced breast cancer, to remain awake this long. Also, to reduce participant burden protocol, we did not require that participants remain in bed in a constant posture for their entire 24-h assessment and only requested that they remain in bed keeping a constant posture during the night time (i.e. during the 8 h of sleep time). We addressed possible concerns about physical discomfort and stress of a full bladder by permitting women who had difficulty using the bed pan to get out of bed to use the bathroom, thereby disrupting posture. Regarding diet, protocol augmentations were made to make the strict diet more tolerable. Many of our participants were on healthy diets and drinking plenty of water and having low carb and low sugar meals; therefore, we revised the standard peanut butter and jelly and water protocol so that participants could adjust the volume of water during the first three snacks and be given a few choices of sandwiches/snacks to select from, such as peanut butter and jelly (they could pick strawberry or blueberry) or turkey. All ingredients were organic and they had their choice of bread (e.g. white, wheat, gluten free). Participants were allowed to do mild stretching to better tolerate being relatively immobilized for 24 h. Weekend assessments were conducted for those participants who worked and were not able to take time off.
5. Build in steps to communicate respect and appreciation to the participants. In a complex and demanding research protocol assessing sleep and circadian rhythm, it is particularly important to incorporate practices into the protocol that communicate to the participants that their contributions are valued. One such practice was to debrief participants for their feedback at the end of the study, both to reinforce this point throughout the process and to obtain feedback about the quality of their interactions with various study staff with whom they came into contact. Another practice was for the staff to walk participants to and from their cars during study visits.
6. Consider multiple methods and modalities for evaluating sleep in cancer patients. We included a home study of sleep that included PSG and actigraphy, as well as a hospital-based study to ensure that we captured different aspects of sleep and circadian rhythms, given that neither modality is without limitations.
7. When possible, consider adding benefits for participants. Apart from the participant cash incentive, participants were provided with feedback on their blood test results and vital signs that could be used for potential medical benefit. This was implemented by having the on-call physician to meet with the participant at the end of the hospital stay to review the results of the blood tests, and vital signs. Participants were given a copy of the results of their clinical blood tests (e.g. hematocrit, complete blood count with differential, lipids, hepatic function panel, and creatinine), and the on-call physician would answer their questions. Results from the two nights of at-home polysomnography were given to the participants and a sleep researcher was available to answer any questions they had. In addition, we provided participants who expressed interest in receiving updates on the research findings with journal articles and information to access a Web site with links to the most recent publications resulting from the current study.
Footnotes
Acknowledgements
We thank our research participants for their time, wisdom, and willingness to participate, and the Dr Susan Love Research Foundation’s Love/Avon Army of Women Program for their assistance in recruitment.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Cancer Institute/National Institutes of Health (R01CA118567) and UL1 RR025744 .
Appendix A
(1) Demographics and Medical History Form
(2) Structured Clinical Interview for DSM-IV-TR Axis I Disorders (SCID - I) 38
(3) Childhood Trauma Questionnaire 39
(4) Comprehensive Pain Evaluation
(5) Short-Form McGill Pain Questionnaire 40
(6) Brief Pain Inventory (Short Form) 41
(7) Center for Epidemiological Studies- Depression Scale (CES-D) 42
(8) Positive and Negative Affectivity Scale (PANAS) 43
(9) Perceived Stress Scale (PSS) 44
(10) State Trait Anxiety Inventory 45
(11) Stanford Acute Stress Reaction Questionnaire (SASRQ) 46
(12) PTSD Checklist- Civilian Version (PCL-C) 47
(13) Difficulties in Emotion Regulation Scale (DERS) 48
(14) The Weinberger Adjustment Inventory (WAI-LF) 49
(15) Single-Item Measure of Social Supports (SIMSS) 50
(16) Interpersonal Support Evaluation List (ISEL) 51
(17) Exercise Behaviors 52
(18) Religion/Spirituality Questionnaire 53
(19) Caffeine Consumption Questionnaire 54
(20) Owl and Lark Questionnaire 55
