Abstract
Objectives:
Cancer patients routinely exhibit dysfunctional circadian organization. Indeed, a dysfunctional circadian organization is a hallmark of advanced cancer. A cohort of advanced cancer patients undergoing chemotherapy was recruited to investigate whether manipulating exposure to blue light could restore or ameliorate their circadian organization.
Methods:
Thirty advanced metastatic cancer patients participated in a randomized crossover trial to evaluate whether blue light-blocking night-simulating eyeglasses could ameliorate a disrupted circadian organization better than sham eyeglasses. Circadian organization was evaluated by actigraphy and patients’ self-reports of sleep, fatigue, and quality of life. Kruskal–Wallis tests compared patients’ outcomes in circadian organization with a cohort of non-cancer, disease-free individuals with normal sleep as a negative control, and with advanced cancer patients with disrupted circadian organization as a positive control. Quality-of-life outcomes of the patients were compared with population-based controls (negative controls) and with cohorts of advanced cancer patients (positive controls).
Results:
Actigraphy measurements, self-reported sleep, fatigue levels, and quality-of-life outcomes of trial participants were similar to those of negative controls with a normal circadian organization, in spite of the trial patients’ concurrent chemotherapy. Night-simulating glasses did not improve circadian organization. The 24-h correlation of day-to-day rhythms of rest and activity was 0.455 for the experimental eyeglasses and 0.476 for the sham eyeglasses (
Conclusion:
The circadian organization of patients in this study unexpectedly resembled that of healthy controls and was better than comparison populations with disrupted circadian organization. The study clinic implements chronomodulated chemotherapy and a systematic, supportive integrative treatment protocol. Results suggest a need for further research on interventions for circadian rhythm. Although the study intervention did not benefit the participants, this work highlights the value of supporting circadian time structure in advanced cancer patients.
Keywords
Background
Life emerges from modular, interacting systems that range in complexity from an enzyme to food webs. Circadian molecular clocks directly or indirectly synchronize these interacting systems at all levels of biological complexity.1,2 Circadian clocks regulate clinically important biological functions such as heart rate, serum glucose levels, pain, renal function, immune function, and other physiological or psychological variables that affect an individual’s clinical status.3,4 Mammals have a small number of photoreceptor cells in their eyes called photosensitive retinal ganglion cells. These cells use the photopigment melanopsin, which is sensitive to blue light wavelengths, to entrain mammalian physiology to the solar day by suppressing the synthesis of melatonin during daylight hours.5–7 The regulatory effects of melatonin are critical to maintaining circadian function, which suggests that manipulation of light wavelengths reaching the eyes may have clinical impact. Reducing evening screen time, maintaining complete darkness in the bedroom, or dimming home lighting in the evening are suggested as ways to reduce blue light exposure, but more systematic study of this area is needed.
Circadian dysfunction is a hallmark of advanced cancer. A healthy circadian organization (CO) exhibits consistent day-to-day rhythms of sleep and activity. Cancer patients who have a functional CO maximize their body’s capacity to repair cellular damage caused by conventional treatments to collateral tissues. Advanced cancer patients with intact CO experience less insomnia, daytime fatigue, sleepiness, and depression.8–13 A functional CO has been identified as an independent prognostic variable in several clinical trials. Multiple studies show that disrupted CO predicts a significantly shorter than expected survival in breast, lung, ovarian, kidney, and colorectal cancers.10,12,14–17 Consequently, CO is emerging as a new target in cancer therapy, since therapies that improve CO may improve survival.13,17–20 Because of the degree of disruption of CO in advanced cancer patients, it is questionable whether lifestyle suggestions like reducing evening screen time have the potential to remediate their circadian dysfunction.
This report describes an unexpected finding from a trial designed to investigate whether a noninvasive intervention for environmentally induced circadian rhythm disorganization caused by blue light wavelengths can affect CO in advanced cancer. The study design could help to justify trials of lifestyle-based interventions. However, the trial cohort of advanced cancer patients did not report the signs and symptoms of a disrupted CO either before or during their use of the intervention. Actigraphy data confirmed that these patients had a functional CO at baseline and during the trial intervention periods. To our knowledge, this is the first report of an advanced cancer patient cohort receiving chemotherapy that showed CO characteristics indistinguishable from a healthy population.
Methods
Trial design for night-simulating eyeglasses
This pilot clinical trial had a single-blind, sham-controlled, randomized crossover design. It investigated whether night-simulating eyeglasses that block blue light wavelengths could restore the circadian time structure in patients with advanced metastatic cancer receiving chemotherapy.
The trial took place at the Block Center for Integrative Cancer Treatment in Skokie, Illinois, a comprehensive integrative medicine center. In addition to outpatient chemotherapy, the Center provides individualized supportive treatment that addresses patient outcomes and quality of life (QOL) by enhancing performance status and ameliorating symptoms and side effects.21–25 The Center’s registered dietitians supervise patients in implementing a prescriptive plant-based diet. 22 A physical activity program is designed and individualized for each patient’s clinical needs.26,27 Credentialed staff assist patients with the psychosocial challenges of cancer and its treatment. Interventions include sleep hygiene, daytime light exposure, providing information on optimizing circadian rhythms, cognitive-behavioral therapy for insomnia, mindfulness, and progressive muscle relaxation for stress, sleep problems, and fatigue.28–30 Previous research, however, did not exhibit any effects of behavioral therapies on CO in patients with advanced cancer. 31 Nutritional therapies are implemented based on laboratory testing for fatigue- and cancer-promoting metabolic states such as inflammation and insulinemia.31,32 Test results of genetic profiling are addressed clinically and nutritionally. 22 Cancer chemotherapy is routinely administered using chronomodulated regimens where indicated. 33 This includes optimal drug timing incorporating a sinusoidal curve of drug administration rates. 34
The inclusion criteria were advanced cancer patients at least 18 years old; histologically confirmed solid tumors other than brain, esophageal, or pancreatic cancer; life expectancy of at least 6 months as estimated by the patient’s treating oncologist; eligible to receive at least three cycles of chemotherapy or hormonal therapy, or daily oral therapy, with any cycle length permissible; Eastern Cooperative Oncology Group (ECOG) Performance Status of 0, 1, or 2; and residence within driving distance of the Center. Any number of previous systemic therapies or prior participation in a clinical trial were permissible. The exclusion criteria were an uncontrolled concomitant malignancy, pregnancy, or lactation; less than 30 days from major surgery or radiation therapy; uncontrolled or symptomatic central nervous system (CNS) metastases; symptomatic spinal cord compression; uncontrolled psychiatric disorders that would prevent participation in the study; serious comorbidity that would interfere with the patient’s capacity to participate in the study; concurrent melatonin supplementation; and blindness or shift work.
Data collection
Patients were instructed on the use of actigraphs, maintenance of a sleep diary, urine collection procedure, and completion of questionnaires. They underwent home-based actigraphy for 7 consecutive days immediately before the start of their next three chemotherapy cycles (baseline, first treatment, and second treatment periods). On their last day of actigraphy measurement, patients collected all overnight and first morning urine and answered all study questionnaires. The questionnaires, actigraph, sleep diary recordings, and urine collection were brought to the patient’s chemotherapy visit the next day and were collected by study staff. Washouts between intervention periods depended on the length of each patient’s chemotherapy cycle and varied from 1 to 3 weeks. Urine sample volume was noted and 25 mL aliquots of urine were collected and frozen at −20°C until shipment for analysis of 6-sulfatoxymelatonin at Tulane University.
Patients did not wear study eyeglasses during the baseline period. During the first study treatment period, patients received either night-simulating glasses or blue-light-transmitting (sham) glasses. The order of treatments was assigned by the study coordinator based on a computer-generated randomization table of even and odd numbers, constructed and held by a research assistant. Patients were instructed to begin wearing glasses at local sunset time and to remove them upon going to bed. Each patient was provided with a night-light containing a small light bulb that did not emit blue light wavelengths for use when getting out of bed at night. During the second treatment period, patients were crossed over to the alternative type of eyeglasses. At each study visit, patients were asked about adverse events they attributed to the study glasses. Patients, laboratory staff, staff who processed actigraphy data and patient-reported outcomes, and the statistician were blinded to the order of treatments until the statistical analysis of trial data was complete.
All patients recruited to the study were already participants in the Center’s integrative cancer treatment program and all signed written informed consent forms for the study. Recruitment occurred from January 2011 to June 2014. The study was approved by the Office for the Protection of Research Subjects of the University of Illinois at Chicago (#2010-0372). The trial concluded after all 30 patients completed study procedures.
Actigraphic measurements of rest–activity cycles
A watch-like actigraph, worn on the non-dominant wrist, continuously measures and records a patient’s movement patterns (Ambulatory Monitoring, Inc. (AMI), Ardsley, NY, USA). Internal motion sensors (accelerometers) capture data on patients’ movements, measured as the number of accelerations (movements or activity counts) per minute. These data are transferred to a computer for analysis and used to produce a report containing parameters from a cosinor analysis. The analysis, which describes the patient’s daily activity rhythm, was performed by AMI for this study. The actigraph also quantitates the patient’s sleep experience into duration, wake after sleep onset, and several other measurements. The primary outcome variable was 24-h autocorrelation of actigraphy scores (24-h rest–activity rhythm), which describes the consistency of day-to-day sleep–wake rhythms. Intradaily stability and relative amplitude were calculated, as was the dichotomy index (DI). The DI is the percentage of activity counts per minute measured by the actigraph when a patient is in bed that are lower than the median activity count per minute measured when the patient is out of bed.
Melatonin analysis
Urinary melatonin was analyzed at the Laboratory of Chrono-Neuroendocrine Oncology, Tulane University (New Orleans, LA, USA); 6-sulfatoxymelatonin and creatinine were measured using radioimmunoassay. Total excretion was calculated by multiplying the concentration of 6-sulfatoxymelatonin by the total overnight urine volume. The total amount of 6-sulfatoxymelatonin was expressed in total units and in units per unit of creatinine excretion, which is stable throughout the 24-h period. This normalizes melatonin values according to the differing kidney function and urinary outputs among patients. However, a number of urine samples were lost due to laboratory equipment failure and could not be analyzed.
Study night-simulating eyeglasses
Study eyeglasses were obtained through Lighting Innovations, LLC (University Heights, OH, USA) along with matching sham eyeglasses. The night-simulating eyeglasses are amber plastic, blue wavelength-blocking eyeglasses that can fit over regular eyeglasses. They block all wavelengths of light less than 530 nm from reaching the eyes but allow wearers to function normally in electric light. The night-simulating glasses are considered as sunglasses by the US Food and Drug Administration and do not require an Investigational Device Exception. Sham glasses are similar in construction, yellow-colored, and transmit all visible light wavelengths. Indoor use of blue-blocking eyeglasses in the evening may promote normal CO by preventing the suppression of melatonin synthesis and the consequent disruption of sleep caused by blue light from electric light fixtures and electronic device screens.
Study instruments measuring patient self-reported outcomes
The Pittsburgh Sleep Quality Index (PSQI) 35 is a validated 19-item questionnaire that measures self-reported sleep quality, sleep efficiency, sleep latency, and napping. Scores range from 0 to 21; higher scores indicate worse sleep quality. Cancer patients with scores of 8 or greater are considered symptomatic.
The European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ-C30) 36 assesses cancer patients’ capacity to function. The 30-item questionnaire has a global health status subscale, 5 functional domains, and 9 symptom scores. The functional domains are physical, role, emotional, social, and cognitive function. The symptom scores measure fatigue, nausea and/or vomiting, appetite, pain, dyspnea, insomnia, constipation, diarrhea, and financial concerns. This tool has normative data from population-based surveys, and differences in patient responses can be translated into clinical significance.
Patients with symptom scores above the 75th percentile of the symptom scales reported in EORTC population-based surveys were defined as being outside the normal range or symptomatic. 37 For fatigue, pain, dyspnea, and insomnia, all scores greater than 34 are considered out of normal range. For nausea and/or vomiting, appetite loss, constipation, diarrhea, and financial difficulties, scores greater than 0 are considered out of normal range. Out of normal range for functional domains was defined as scores below the 25th percentile in data reported in population-based surveys.
The Piper Fatigue Scale (PFS) measures four dimensions of fatigue: behavioral, sensory, cognitive, and affective. Total fatigue score is bounded between 0 and 10; higher scores reflect more fatigue. Total scores of 0 to 3.99 represent mild fatigue, 4 to 6.99 moderate fatigue, and 7.0 or higher severe fatigue. 38
The Ferrans and Powers Quality of Life Index–Version 3 (QLI) measures patients’ level of satisfaction with their well-being. 39 It consists of two parts: patients’ descriptions of their satisfaction with 33 aspects of life, and their rating of the importance of each aspect in their own lives. Four subscale scores, including health and functioning, social and economic, psychological and spiritual, and family, plus one overall quality-of-life score, are calculated. The range of scores is 0 to 30, and higher scores indicate better quality of life. The five scores have previously been shown to be correlated with actigraphic variables in cancer outpatients. 40 The correlation between the items making up the EORTC QLQ-C30 and QLI 41 is minimal or nonexistent. This shows that these tools measure unrelated aspects of quality of life.
Statistical analysis
Prior research indicates that night-simulating eyeglasses are effective in restoring a disrupted circadian rhythm in some clinical populations. 4 We projected from the literature and our prior experience with patients that we could expect a 20% improvement in one or more of the chosen endpoints in this pilot study. Lower effect sizes, we believe, would not be clinically meaningful. The sample size was projected to be 30 patients for a pilot crossover study design.
Statistical analysis of the data and a confirmation of no crossover effects followed the methods of Wellek and Blettner. 42 The Wilcoxon Rank Sum test was used to compare the distributions when patients used the night-simulating glasses versus the control.
A series of secondary outcome analyses of trial data was instigated following the failure of the night-simulating eyeglasses to improve participants’ 24-h rest–activity rhythm. These follow-up analyses were not part of the original study protocol. They should therefore be regarded as exploratory and hypothesis-generating. A number of comparisons were made between study participants and outside cohorts in order to evaluate their clinical status (Figure 1). Comparisons were made with cancer-bearing (positive controls) and non-cancer populations (negative controls).

Comparison of trial participants with outside cohorts (ad hoc analysis). The figure describes the ad hoc analysis evaluating the underlying causes of why this trial failed. The chart identifies the cohorts used in the analysis. Participants’ results were compared to positive controls (cancer cohorts with a disrupted CO) and negative controls. Positive and negative controls were found for actigraphy, PSQI, EORTC QLQ-C30, and QLI. Piper Fatigue Scale had no positive control. Finally, a comparison was made with a hospital patient cohort of recurrent breast and colon cancer patients as a sound comparator cohort for a positive control using the EORTC QLQ-C30.
Several populations were used for comparisons of actigraphy and patient-reported outcomes. The PSQI has test norms associated with symptom-free populations, and the PFS has defined symptom ranges. QLI and EORTC QLQ-C30 both have population-based data to describe the range of values found in the general population. The data on population-based norms served as negative controls. 43 An estimate of actigraphic normal daily rest–activity rhythm derived from a convenience control group with no known disease or sleep problems, ages 20–50, provided by AMI, was used as an additional negative control.
An advanced cancer patient comparison group with a disrupted daily rest–activity rhythm acted as a positive control. It comprised 84 advanced stage (IIIb and IV) non-small-cell lung cancer patients who participated in a clinical trial evaluating the benefits of adjuvant melatonin. 44 These patients were untreated or had only one previous chemotherapy regimen. Their baseline values before melatonin administration were used for comparison. Actigraphy data and data from the PSQI, EORTC QLQ-C30, and QLI were derived from this cohort. Finally, cohorts of North American recurrent colorectal and breast cancer patients who responded to the EORTC QLQ-C30 41 at an initial hospital visit for oncology consultation made up an additional positive control. The demographics of these colorectal and breast cancer cohorts resemble those of the patients participating in the trial.
These evaluations were performed for the purpose of making meaningful clinical comparisons between the trial participants and groups of patients with and without a disrupted CO. Comparisons were not designed to make cause and effect conclusions. Rather, they simply assessed the clinical status of the CO and patient-reported outcomes of the trial participants relative to the positive and negative controls to detect whether the results observed in the trial suggested normal or disrupted CO. The subsequent ad hoc analyses used the
For each set of comparisons, the 19 QOL scales from the EORTC QLQ-C30 and QLI were assumed to be independent. Consequently, they were used to test 19 null hypotheses. The multiple comparisons problem was accounted for by a factor of 19 using the Bonferroni correction. Statistical significance was assessed using the Mann–Whitney test with a significance threshold of
Finally, to check for the safety of the integrative program, survival times of trial participants were calculated and compared with a hospital population of advanced cancer patients, a national cancer database, and the advanced cancer patient comparison group.
Results
Participants
The CONSORT diagram (Figure 2) describes the recruitment, participation, and reasons for study discontinuation. Chief reasons for not meeting inclusion criteria were insufficient number of remaining treatment cycles and travel distance too far from the trial center. Of the 69 eligible Block Center advanced cancer patients, 43.5% were recruited to the study. Of the 24 patients who refused to participate, 8 wished to take melatonin supplements, 7 were too busy, and the remainder declined for personal reasons. Participants were predominantly female (73.3%) with a median age of 55 years (Table 1). Most had metastatic disease at the time of recruitment, and 17 had previously undergone chemotherapy. Colon and breast were the most frequent cancer sites. Participating patients had received a median of three prior cycles in their current treatment regimen (interquartile range (IQR) = 2–7; minimum 1, maximum 23). These regimens are routinely given with chronomodulation at the study clinic. In addition, four participants reported taking sleep supplements and four took sleep medications that were permitted in the exclusion criteria. No adverse events were reported due to study glasses.
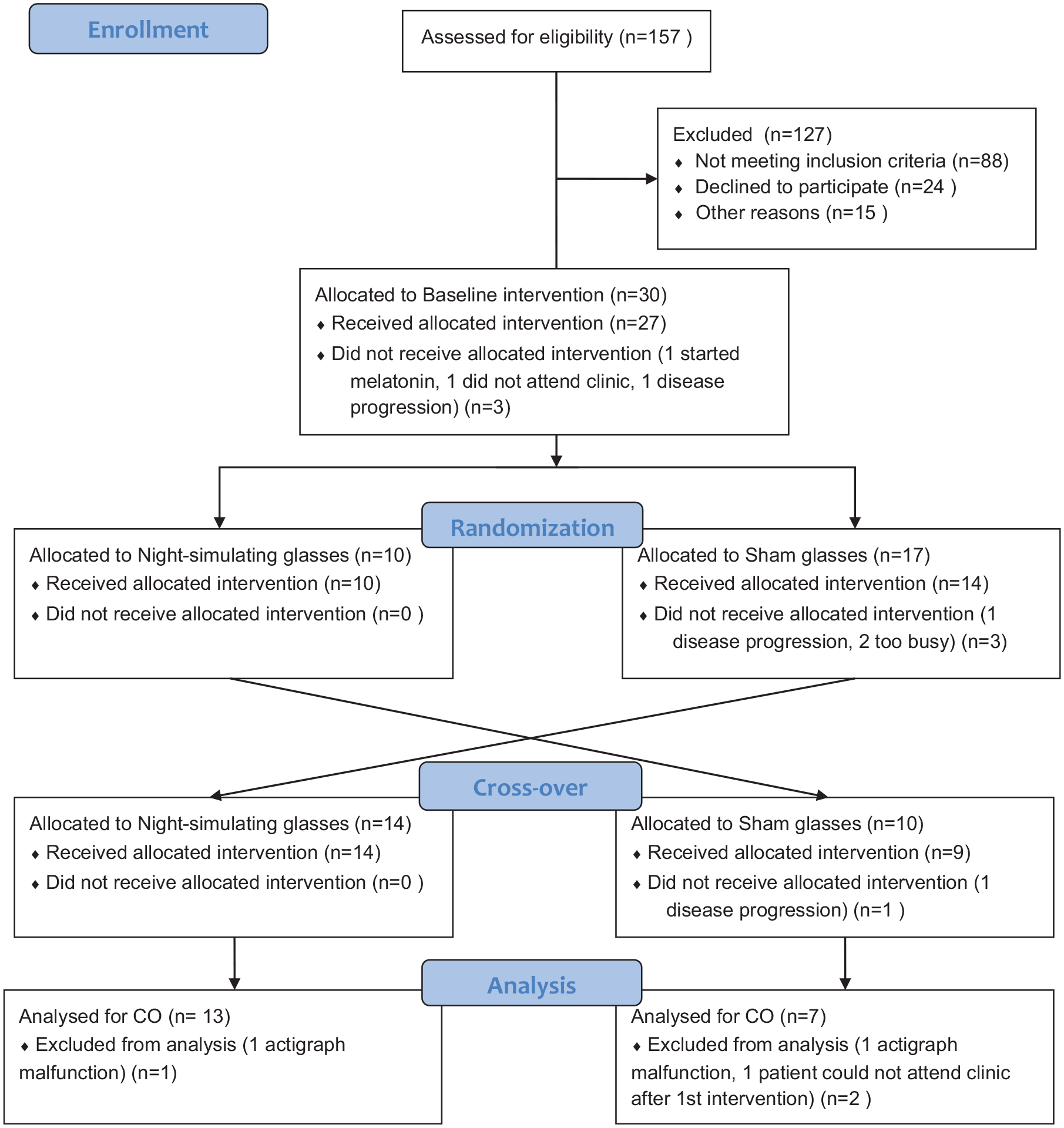
Study flowchart describing the participant recruitment process and participant retention.
Clinical and demographic characteristics of trial participants (
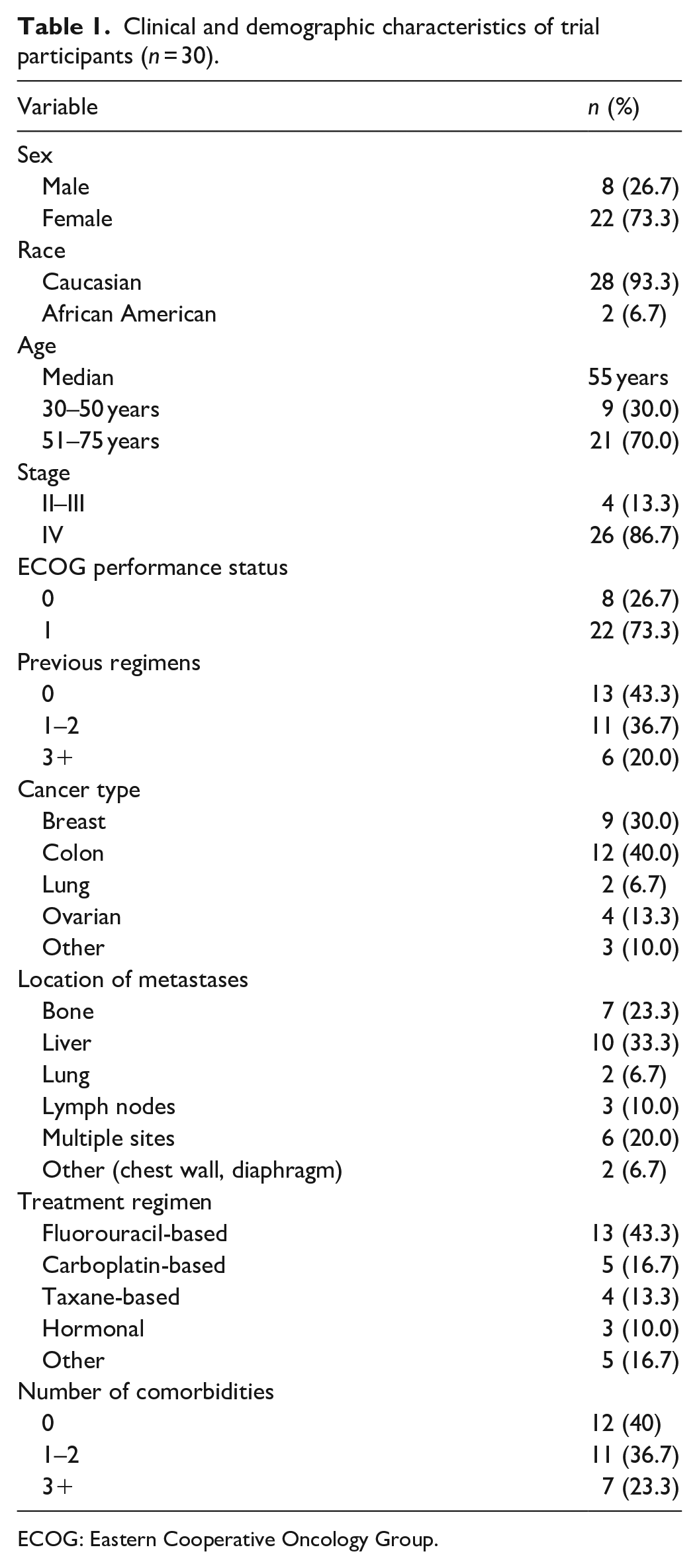
ECOG: Eastern Cooperative Oncology Group.
Of the 30 participants, 7 dropped out or were withdrawn from the study. Of these, six left the study before completing the baseline period. Three were withdrawn due to disease progression (two before completing the baseline), two reported that they were too busy to complete the baseline intervention, one did not attend the clinic, and one began melatonin supplementation (Figure 2). In addition to the drop-outs and withdrawals, actigraphic data from three participants were not analyzed: in two instances, actigraphs malfunctioned and one participant forgot baseline actigraphy. Participants who did not drop out completed study assessments successfully at rates of 87% to 100% (Supplemental File 1, Table 1).
Effects of study eyeglasses on trial outcomes
No evidence of effects of the study eyeglasses on actigraphic variables, melatonin levels, or patient-reported study outcomes was found. No evidence of carryover effects was found, indicating that the crossover study design was conceptualized and conducted correctly. (Supplemental File 1: Supplemental Tables 2–4; study data are available in Supplemental File 2; study questionnaires are available in Supplemental File 3).
The secondary analyses were subsequently carried out to determine whether the CO and patient-reported outcomes of the Block advanced cancer patients were too disrupted to remediate; whether they were essentially intact, which would suggest a ceiling effect; or whether their CO was typical of advanced cancer patients but the glasses were simply ineffective.
Secondary analysis of reasons for failure of eyeglasses to improve outcomes: Was CO disrupted in trial population?
The first step in the secondary analysis was to determine whether the Block advanced cancer patients’ 24-h rest–activity rhythm was disrupted. Their actigraphy data were compared to two groups: a convenience sample of healthy individuals without sleep problems, whose actigraphy indicated normal rest–activity rhythms, and the advanced cancer patient comparison group from the melatonin trial. Actigraphy verified that the latter population had disrupted CO.
No statistically significant difference was found in any shared actigraphy parameter between the Block advanced cancer patients and the healthy control sample (Table 2). A large statistically significant difference in 24-h correlation exists, however, between the Block patients and the advanced cancer patient comparison group (Table 3). These results demonstrate normal CO among the Block patients. In addition, the median DI (Table 2) of the Block patients was compared with data on two groups of metastatic colorectal cancer patients. 45 Abnormal DI values (<97.5%) indicate disrupted CO. No Block patients had abnormal DI values in the first treatment period, 15% had abnormal values in the second treatment period, and 5% had abnormal values in the third treatment period. In contrast, the percentages of abnormal DI in the two comparison groups of colorectal cancer patients were 54.9% and 44.4%. 45
Circadian parameters compared between a healthy control sample and trial participants.
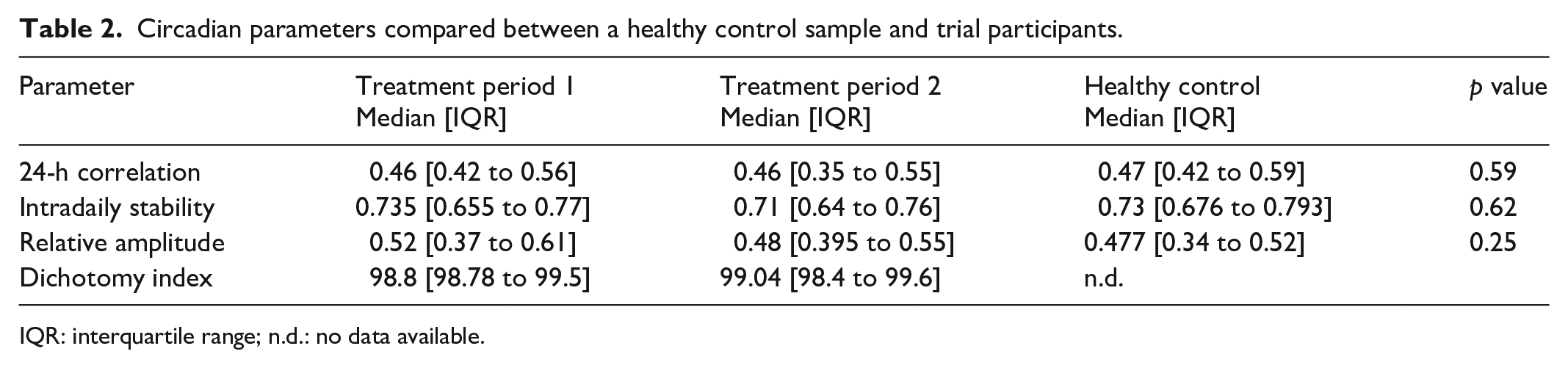
IQR: interquartile range; n.d.: no data available.
Circadian parameter 24-h correlation for Block advanced cancer patients and advanced cancer patient comparison group.
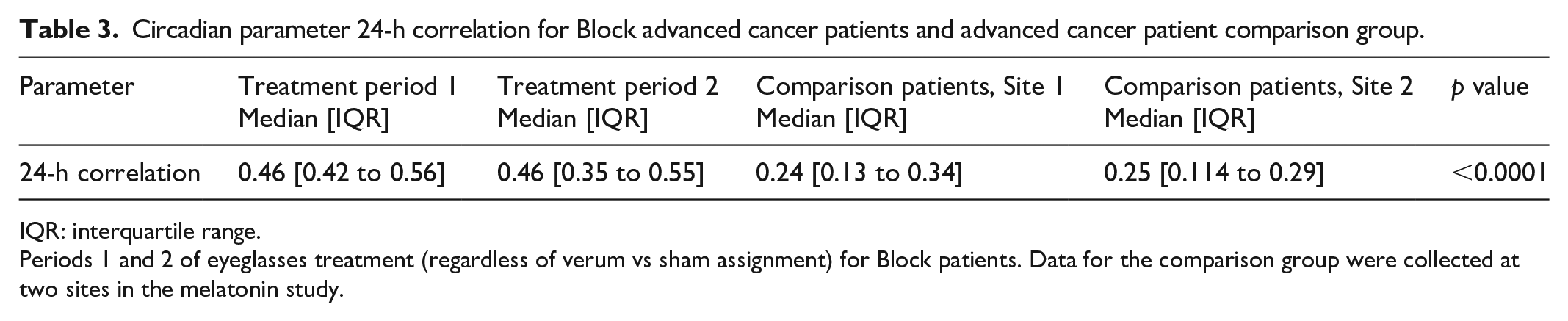
IQR: interquartile range.
Periods 1 and 2 of eyeglasses treatment (regardless of verum vs sham assignment) for Block patients. Data for the comparison group were collected at two sites in the melatonin study.
To illustrate the differences in 24-h rest–activity rhythm, mean daily activity counts derived from actigraphy of the Block advanced cancer patients were graphed alongside the healthy controls without sleep problems and the advanced cancer patient comparison group (Figure 3). Actigraphy counts were compared using Wilcoxon and Kruskal–Wallis tests. The three groups were found to differ significantly (
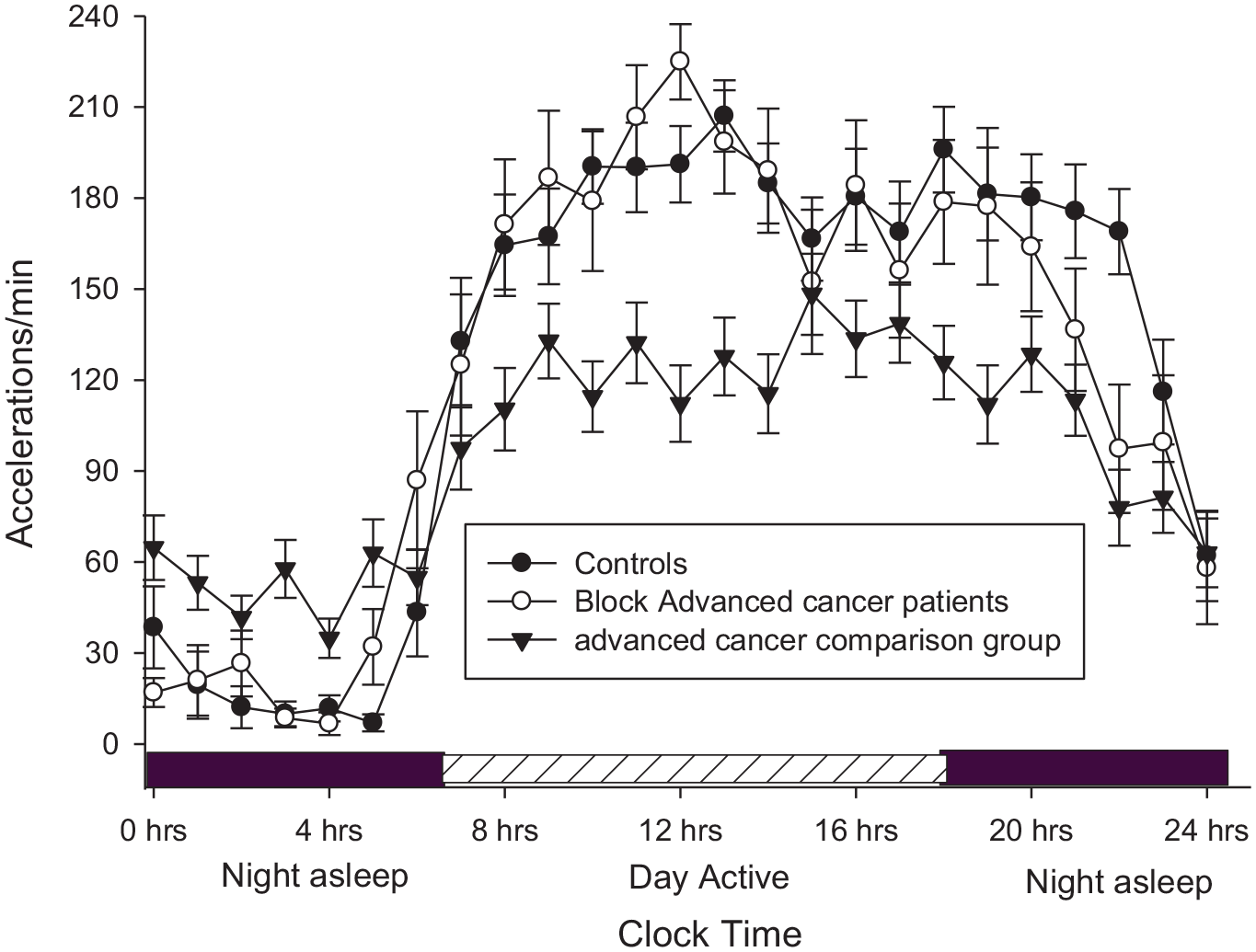
The 24-h activity counts (mean accelerations/minute ± standard error) of Block advanced cancer patients, the advanced cancer patient comparison group, and controls without sleep problems. The three groups differ significantly (Wilcoxon,
A progressive disruption of CO with increasing numbers of chemotherapy cycles was recently reported for breast cancer patients in treatment. 46 However, 24-h rest–activity values were not correlated with numbers of previous chemotherapy treatments in the Block patients (Table 4).
Correlation of actigraphy variables for trial participants with number of prior chemotherapy cycles at baseline (range = 0–24).

Are patient-reported outcomes outside or within normal ranges?
The self-reported outcomes of the Block advanced cancer patients were compared to the outcomes of the same tests in the advanced cancer patient comparison group as well as to published population-based test norms. Since actigraphic data indicated no differences in patient-reported outcomes between the study periods, data from the baseline period were used for comparisons.
Was sleep quality normal?
Global PSQI scores >8 indicate poor quality sleep for cancer patients, while scores >5 indicate poor quality sleep in a disease-free cohort. Throughout the trial, 75% of Block advanced cancer patients reported a Global PSQI score that was less than 8 and over half reported scores below 5. The advanced cancer comparison group had a median Global PSQI score of 12.50 (Table 5).
Patient-reported outcomes compared between baseline values of Block advanced cancer patients and advanced cancer patient comparison group.

IQR: interquartile range; PSQI: Pittsburgh Sleep Quality Index; n.d.: no data available; QLI: Ferrans and Powers Quality of Life Index–Version 3; Socio/eco: social/economic; Psych/spir: psychological/spiritual.
Were participants fatigued?
The PFS has cut points for mild, moderate, and severe fatigue of 1 to 3, 4 to 6, and 7 to 10, respectively. 47 Three-quarters of the Block advanced cancer patients reported levels of total fatigue ranging from mild to moderate (median = 2.45). The clinical significance of these values is unclear due to a lack of reference data from a normal population, but stages 3 and 4 breast cancer patients in treatment had a mean total fatigue score of 5.76 47 (Table 5).
The EORTC QLQ-C30 tool has a multiple-question fatigue item, for which the normal score is less than 33. Three-quarters of the Block patients reported fatigue levels within normal limits (Table 6). In contrast, the median fatigue level reported by the advanced cancer comparison group was 66.7, a statistically significant difference (
Comparison of EORTC symptoms of Block advanced cancer patients versus advanced cancer patient comparison group.

EORTC: European Organization for Research and Treatment of Cancer; ONR: out of normal range; IQR: interquartile range; QOL: quality of life.
ONR scores provided for each variable indicate cutoff for “normal’ scores.
Was quality of life diminished?
EORTC comparison to advanced cancer patients with known disrupted CO
The Block advanced cancer patients had significantly better scores than the advanced cancer comparison group for all EORTC functional domains except emotional function and social function. The differences were large, and both statistically and clinically significant (Table 6). 48 Pain, dyspnea, and fatigue were also diminished in the Block patients.
EORTC comparison to a population-based survey
We compared the Block advanced cancer patients’ quality of life with a general population-based survey. 43 At least 75% of the Block patients reported normal functioning in the global health, physical, role, emotional, and cognitive subscales throughout the trial, but social functioning was compromised (Table 7). Up to 75% reported healthy, non-cancer levels of fatigue, pain, dyspnea, and insomnia, and up to 50% reported healthy, non-cancer levels for nausea and/or vomiting, loss of appetite, and diarrhea.
EORTC scores of Block trial participants compared to healthy controls and patients with recurrent cancer, showing clinically significant differences in comparison with recurrent cancer populations.

EORTC: European Organization for Research and Treatment of Cancer; IQR: interquartile range; QOL: quality of life; NA: not applicable.
Trial participants’ EORTC scores [median, IQR] are compared to reference values for healthy population-based controls 43 and patients with recurrent breast or colorectal cancer. 41 Clinical significance of differences follows Cocks et al. 48
Comparison to advanced breast and colon patients whose CO is unknown
Most of the Block advanced cancer group were breast or colon cancer patients who either did not respond to definitive therapy and/or who developed metastatic disease. A comparison between the Block patients and the EORTC results from cohorts of recurrent breast and colon patients treated in a North American hospital system showed favorable differences for global health quality of life, physical function, and five symptom items, including insomnia (Table 7). These differences were sufficiently marked to be defined as clinically large (unequivocal clinical relevance).41,48 All other domains also showed clinical differences with the exception of social function.
QLI comparison
The Block advanced cancer patients’ QLI responses showed no significant differences compared to the general non-cancer population for any of the four scales (Table 8). Their level of satisfaction with their health and functioning was indistinguishable from a random survey of the general population. They differed significantly from the advanced cancer comparison group in health and functioning, social and economic, and family scales, but no significant difference was found in psychological and spiritual quality of life. The advanced cancer patient comparison group was markedly less satisfied with their health and functioning than the general population but in the other domains was fairly similar to the general population.
QLI scores compared to reference values from population-based controls and advanced cancer patient comparison group.

QLI: Ferrans and Powers Quality of Life Index–Version 3; IQR: interquartile range; SD: standard deviation.
Block Center trial participants’ QLI scores are compared to reference values from QLI population-based controls (mean, SD) and advanced cancer patient comparison group. QLI population-based control is taken from a database of 339 randomly selected subjects, ages 18 and above.
43
The
Survival
Concerns are sometimes raised about the impact of nutritional or dietary supplement interventions on the efficacy of conventional cancer treatment. The median survival of all Block advanced cancer patients following admission into the study (not from the time of diagnosis or of coming under the care of the study clinic) was 28.8 months. Cox proportional hazard analysis identified two variables correlated with the survival of these patients: chronological age at time of consent (younger patients lived longer) and length of time since diagnosis (the longer the history of a diagnosis, the shorter the survival after trial admission).
For participants with metastatic disease, median survival from date of diagnosis with distant metastases was 32.6 months for breast cancer patients and 29.6 months for colon cancer patients. Median survival of metastatic breast cancer ranges from 18 to 30 months; 49 median survival of colorectal cancer ranges from 13 to 29 months.50,51 In Table 9, 2- and 5-year survival data for Block advanced cancer patients are compared with data from a hospital system and from the National Cancer Institute Surveillance, Epidemiology, and End Results (SEER).52,53 These data suggest favorable 2-year survival (for Block breast cancer patients 87.5%, hospital 69%, SEER 49%; for Block colon cancer patients 58.3%, hospital 42%, SEER 31%).
Comparison of survival times of metastatic breast and colon cancer patients from SEER, a large hospital system, and the Block Center.

SEER: Surveillance, Epidemiology, and End Results.
Survival curves of the SEER, hospital, and Block Center patients were calculated using a Kaplan–Meier nonparametric product-limit estimate.52,53
Figure 4 shows Kaplan–Meier survival curves comparing the advanced cancer patient comparison group with the colon, rectal, and lung cancer patients in the Block population. Hormonal cancer cases such as breast cancer were not included in this analysis since they tend to survive longer than the lung cancer patients in the comparison group. Although the Block population is small for survival comparisons, the statistical analysis indicates significantly longer survival (log-rank test, χ2 = 7.89,
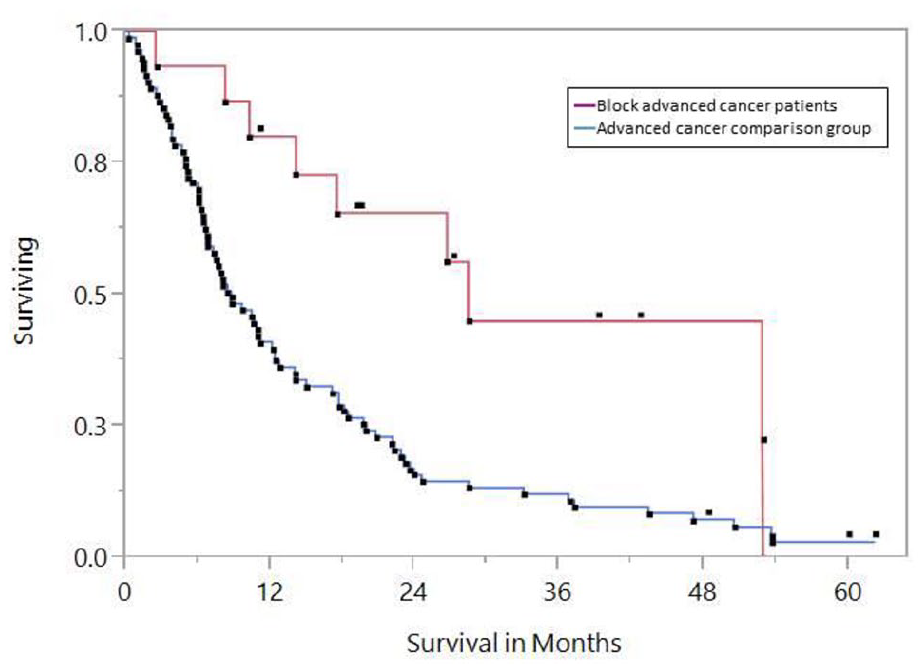
Kaplan–Meier survival curves: the trial participants (Block advanced cancer patients), omitting hormonally driven cancers (e.g. breast, prostate), and the advanced cancer comparison group.
Discussion
A cohort of advanced cancer chemotherapy patients with intact CO is an unexpected finding. This result emerged from a trial that found no clinical benefits of night-simulating glasses on the CO of advanced cancer patients. The lack of carryover effects and the high rate of completion of study assessments by the participants suggest that the study design was feasible and was carried out correctly. The lack of effect of the study eyeglasses raised the question of whether the CO of the Block advanced cancer patients was too disrupted to remediate; whether it was essentially intact, which would suggest a ceiling effect; or whether the CO was typical of advanced cancer patients but the eyeglasses were simply ineffective. Follow-up statistical analyses were conducted to explore these possibilities.
Direct measurements of CO by actigraphy in cancer patients with diagnoses ranging from early stage breast cancer to metastatic colorectal cancer consistently reveal a high prevalence of disrupted CO.11,13,45 Actigraphic measurements of a group of patients with mixed advanced cancers, collected for 3 days before the start of a chemotherapy cycle, recorded a mean DI below 97 and a mean 24-h correlation value of 0.35 (vs 0.455 for the experimental condition in the present trial). 13 The low percentage of Block advanced cancer patients with DIs signaling disrupted CO when compared to a group of metastatic colorectal cancer patients was noted above. 45
The findings of the follow-up statistical analysis suggest that the Block advanced cancer patients had intact CO, implying a ceiling effect. Their actigraphy findings were statistically indistinguishable from the convenience control group composed of healthy adults but were significantly better than those of a cohort of advanced cancer patients with a known disrupted CO. Block patients’ CO was unaffected by the number of prior treatment cycles or regimens, whereas an observational study reported that breast cancer patients’ CO worsened with increasing numbers of chemotherapy cycles. 46 Over 75% of the Block advanced cancer patients had PSQI scores consistent with normal sleep, whereas 75% of the advanced cancer patient comparison group values were consistent with poor sleep. The responses of the advanced cancer comparison patients to the EORTC revealed multiple symptoms and functional capacity that was reduced by a clinically significant amount. The Block patients’ EORTC QLQ-C30 functional and symptom scores were indistinguishable from those derived from general population-based surveys: up to three-quarters reported healthy levels of fatigue, pain, dyspnea, and insomnia. The Block advanced cancer patients’ results for all QLI domain scores were within the normal range, especially the health and functioning domain. Survival data suggest that outcomes in the Block clinic are at least as good as those reported elsewhere, implying safety of the integrative interventions. All these results depict a CO close to normal, which suggests that the patients’ outcomes could not have been improved by the study eyeglasses, implying a ceiling effect.
Although this study cannot determine whether the lifestyle interventions at the study clinic affected the CO of the Block patients, other recent studies have evaluated interventions to improve CO in cancer patients. A randomized study of nightly dosing of 5 mg melatonin in advanced cancer patients reported improvements in sleep and quality of life in cancer patients, but found no effect on CO measured by actigraphy or diurnal cortisol excretion. 54 However, a 2018 randomized study of stages I–III breast cancer chemotherapy patients with insomnia used cognitive-behavioral therapy for insomnia that was modified to include education about circadian disruption as well as increased physical activity and encouragement of daytime bright light exposure. 28 This study reported improvements in circadian parameters in the experimental group. These results suggest that melatonin alone may be insufficient to improve CO in cancer patients and that lifestyle interventions that are similar to those implemented in the Block clinic should receive more attention in research on CO.
As has been shown by Levi et al. 34 and ourselves, among others, careful and diverse supportive measures paired with cancer therapies given at or near optimal times of day maintain properly timed nighttime sleep and robust daytime activity, signaling healthy circadian time structure. This multidimensional therapeutic strategy may concurrently reduce cancer-associated anxiety, fatigue, and other burdensome disease- or treatment-related symptoms, thereby enhancing patients’ quality of life.
Study limitations
Although this pilot study was correctly implemented, the study group was quite small and heterogeneous. The study design assumed that the trial participants would have disrupted CO similar to that reported in other advanced cancer patients, but the study results unexpectedly indicated intact CO in the study group that was maintained through three treatment cycles. This suggests that disrupted circadian rhythms should be an inclusion criterion in future studies aiming to improve CO, even in advanced cancer populations. A formal sample size calculation was not conducted. The heterogeneous nature of the patient population limits the conclusions that can be drawn from comparisons with the advanced cancer comparison group, which was made up of patients with a single type of cancer and which may differ in several ways from the Block patients. The survival curves showing the Block advanced lung, colon, and rectal cancer patients and the advanced lung cancer comparison group (Figure 4) are meant only as a general comparison of advanced non-hormonal cancers. The small number of lung cancer patients in the Block advanced cancer population did not allow for construction of a reliable survival curve based only on lung cancer patients.
The clinic population is self-selected and therefore subject to potential systematic differences for selection of physically robust subjects relative to some of the comparison populations. 55 Patients who use complementary medicine are observed to use different coping strategies from non-users, such as confrontive coping (e.g. “fighting” and active engagement in treatment plans) rather than evasive coping (e.g. minimizing the importance of symptoms and side effects). 56 However, while this observation may explain patient self-reports of QOL outcomes, it may be less likely to apply to an objective measurement such as actigraphy. The good ECOG status of the patients (all ECOG 0 and 1) may seem unusual, but an ECOG status of 0–2, while not the norm, has certainly been observed in other populations of heavily pretreated cancer patients. 57 The lifestyle interventions at the Block Center may have contributed to the good condition of the patients, but the study was not intended or designed to make conclusions about this possibility. The analysis cannot determine whether participants improved their CO soon after beginning the chronomodulated chemotherapy and systematic supportive integrative treatment protocols,58–60 or whether they simply maintained preexisting good CO throughout chemotherapy regimens. 31
Conclusion
This study identified a cohort of advanced cancer patients who had a non-disrupted CO during chemotherapy treatment accompanied by normal values for patient-reported outcomes. Night-simulating eyeglasses trialed in this group had no benefits, possibly due to a ceiling effect, which implies that advanced cancer patients’ CO should be assessed before admitting them to studies that attempt to improve CO.
While a causal connection cannot be made, these results emerged from a clinic that implemented principles of chronomodulated chemotherapy and comprehensive supportive integrative treatment. Cancer patients who maintain normal CO live longer and better than those whose time structure is damaged. Combined supportive care and optimally timed cancer therapy may be helpful if researched and applied more broadly. Although blocking of blue wavelength light did not benefit the patients in this study, our understanding of the value of supporting time structure among advanced cancer patients undergoing therapy has been advanced by this work.
Supplemental Material
sj-docx-1-smo-10.1177_20503121221100137 – Supplemental material for Advanced cancer patients in a randomized clinical trial of night-simulating eyeglasses observed to have a normal 24-h circadian rhythm during chemotherapy
Supplemental material, sj-docx-1-smo-10.1177_20503121221100137 for Advanced cancer patients in a randomized clinical trial of night-simulating eyeglasses observed to have a normal 24-h circadian rhythm during chemotherapy by Keith I Block, Charlotte Gyllenhaal, James F Grutsch, Penny B Block, Thomas Kazlausky, David Blask, Edward Carome, Justin Reynolds, Dinah Faith Q Huff and William Hrushesky in SAGE Open Medicine
Supplemental Material
sj-xlsx-2-smo-10.1177_20503121221100137 – Supplemental material for Advanced cancer patients in a randomized clinical trial of night-simulating eyeglasses observed to have a normal 24-h circadian rhythm during chemotherapy
Supplemental material, sj-xlsx-2-smo-10.1177_20503121221100137 for Advanced cancer patients in a randomized clinical trial of night-simulating eyeglasses observed to have a normal 24-h circadian rhythm during chemotherapy by Keith I Block, Charlotte Gyllenhaal, James F Grutsch, Penny B Block, Thomas Kazlausky, David Blask, Edward Carome, Justin Reynolds, Dinah Faith Q Huff and William Hrushesky in SAGE Open Medicine
Footnotes
Acknowledgements
Study glasses were obtained through Lighting Innovations, LLC. Actigraphs and analysis of actigraphic data were provided by Ambulatory Monitoring, Inc. We thank Vilnius Kubulins, Michael de la Torre, Sue-Jong Hung, and Tomasz Wilmanski for their assistance with the study. The late Professor Edward Carome of John Carroll University and Lighting Innovations LLC contributed substantial expertise as well as technical support to the study. We gratefully acknowledge his valuable collaboration. We sincerely thank the trial participants for their willingness to take part in the study.
Author contributions
K.I.B. directed medical and integrative treatment of trial participants, assisted in design and conduct of the study, and contributed to writing the manuscript. D.B. and members of his laboratory analyzed urinary melatonin samples. D.F.Q.H. assisted with statistical analysis. E.C. provided study eyeglasses and low-blue light bulbs, background information on these, and logistical assistance. T.K. provided actigraphs and technical assistance as well as data analysis. P.B.B. directed biobehavioral treatment of patients and assisted in assessing patients for inclusion criteria as well as contributing to writing the manuscript. C.G. was the clinical study coordinator and contributed to writing the manuscript. J.R. coordinated the initial organization of the study and was coordinator of laboratory activities at the WJB Dorn Veterans Affairs Medical Center. J.F.G. provided statistical analysis and manuscript writing. W.H. led the design, conduct, and analysis of the study and participated in manuscript writing.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K.I.B. is the owner and Medical Director of the Block Center for Integrative Cancer Treatment. P.B.B. is the Executive Director of the Block Center for Integrative Cancer Treatment. E.C. was the owner of Lighting Innovations, LLC. C.G. is the Research Manager of the Block Center for Integrative Cancer Treatment. All other authors declare no competing interests.
Ethical approval,consent to participate,and protocol availability
The study was approved by the Office for the Protection of Research Subjects of the University of Illinois at Chicago (#2010-0372). All trial participants signed written informed consent documents before beginning the study. The trial protocol is available on request from the study author.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the study was provided by the Institute for Integrative Cancer Research and Education. The Institute for Integrative Cancer Research and Education had no role in study design, data collection, analysis, data interpretation, or writing of the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
