Abstract
Introduction
To produce robust evidence RCTs need to be rigorously conducted as poorly performed studies introduce bias and can mislead clinicians and policy makers. Poor allocation concealment has the largest single impact on bias in RCTs than other methodological aspects. Envelopes are frequently used as a method of allocation concealment and can be associated with increased risk of bias. This paper aims to review envelope use in RCTs published in 2017–2018 and create a guide as a reference for researchers when planning and publishing RCTs when using envelopes as an allocation concealment method.
Methods
RCTs that used envelopes as a form of allocation concealment that were published in BMJ, JAMA, NEJM and The Lancet in 2017 and 2018 were identified and methodological data on their envelope use extracted and authors were contacted to ascertain reasons for using envelopes in their research.
Results
338 RCTs were identified that were published in 2017 and 2018. 8% (n = 29) of the RCTs published used envelopes as an allocation concealment method. 24.1% (n = 7) of studies reported envelope studies robustly with all required methodological information stated to enable an assessment of quality. Budget was the most frequent reason given for envelope use (41.7%).
Discussion
Only 24% of published RCTs, that used envelopes, contained robust methodological information to enable the reader to judge whether the randomisation and allocation concealment method was adequate.
Conclusion
RCTs are not reporting envelope use well. RCTs using envelopes should be designed and reported clearly ensuring all necessary methodological information is included.
Keywords
Introduction
Randomised Controlled Trials (RCTs) are considered to be the gold standard in assessing the effectiveness of interventions. To produce robust evidence RCTs need to be rigorously conducted as poorly performed studies introduce bias and can mislead clinicians and policy makers. Probably the single design element associated with biased findings in trials is poor or absent allocation concealment.1,2
Allocation concealment
Allocation concealment is defined as the method used to conceal the randomisation sequence from all study personnel until after the patient has been recruited into the study. This stops the randomisation sequence being subverted and the study having a high risk of bias. It has been shown that having an inadequate allocation concealment method can exaggerate the effect size by 41%.1,2 There are multiple ways that the randomisation sequence can be concealed, such as web-based or telephone systems. Traditionally, before web and telephone systems were available envelopes were used. The use of sealed envelopes as a method still lingers on as a concealment method for a significant proportion of RCTs. For instance, Yelland et al found in 2015 that 9% of RCTs employed sealed envelopes as a method of concealment. 3
Advantages and disadvantages of envelope use as a method of allocation concealment
There are significant disadvantages to using envelopes for allocation concealment. They can be opened in advance for example, 4 trans illumination can determine the allocation5–8 such methods allow subversion of the randomisation. On the other hand they are relatively cheap and logistically practical in remote areas that are internet or telephone free or in emergency medicine situations.
In this paper we aim to describe the types of trials that continue to use envelopes and the quality of the envelope concealment used and to provide advice on their safer usage.
Methods
RCTs published in BMJ, JAMA, NEJM and The Lancet in 2017 and 2018 were identified. Two reviewers extracted data from each paper on the randomisation and allocation concealment methods. Those RCTs that used envelopes to conceal the randomisation sequence were identified.
Envelope concealment
We used the approach described by Doig and Simpson
9
to define high quality envelope concealment. There are three areas that were assessed as follows:
If the person who created the envelope was stated
Best practice for the use of envelopes in RCTs would be that a randomisation sequence would be generated and personal not involved in the RCT would create the envelopes for the RCT.
2. Whether the envelopes had an additional security measure.
Envelopes should have an additional security measure rather than just being closed and be opaque and sequentially numbered. This order can then be checked and anomalies will be identified if the randomisation sequence has been violated. Other additional security measures include the person who has created the envelope signing the back of the envelope when sealed so it is obvious if it has been tampered with. Inserting foil and or carbon paper into the envelope prevents trans-illumination and the carbon paper allows an additional audit trail as the participants name and date of recruitment can be written on the envelope at the point of recruitment before the envelope has been opened and the carbon paper prints this information to the allocation insert. The envelopes should be kept securely and not with the research team who are responsible for recruiting participants into the study.
3. If the person who opened the envelope (recruited participants) was stated
The person who created the envelope should not be the same person who recruits participants to prevent the ordering of participants into one treatment arm or another.
We identified these and other quality factors and extracted them from each RCT that used envelopes: the envelope description, whether who created the envelope and who opened the envelope was stated. We emailed each corresponding author of the RCTs that used envelopes as an allocation concealment method and inquired as to why they have chosen envelopes as an allocation concealment method.
Results
A total of 338 RCTs were identified that were published in 2017 and 2018. 7.5% of RCTs published in 2017 used envelopes and 9.5% in 2018. Combined, 8.6% (n = 29) of the RCTs published in 2017 and 2018 used envelopes as an allocation concealment method. We emailed each author of the RCTs using envelopes and received responses from 12 (41%) of them. Figure 1 shows the flow of studies.
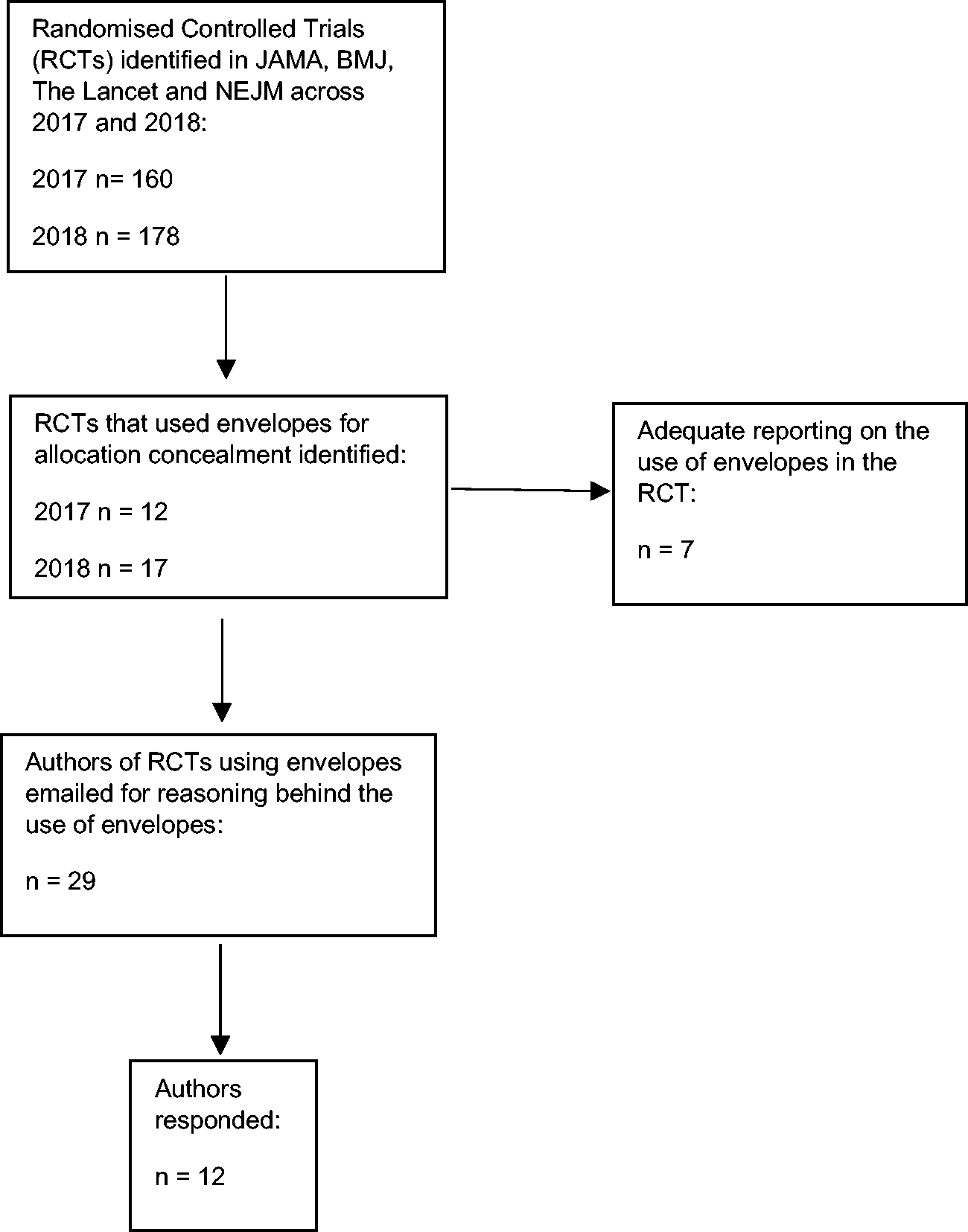
Flow of trials through study.
Table 1 shows the description from the paper of each envelope trial with the necessary quality factors and trial setting. 24.1% (n = 7) of studies that use envelopes for allocation concealment, reported envelope use robustly with all required methodological information stated to enable an assessment of quality. 44.8% (n = 13) reported who created the envelope, 44.8% (n = 13) reported who opened the envelope and 62.1% (n = 18) reported the envelope description adequately.
Quality factors and setting reported in included RCTs.

aTwo types of randomisation – SNOSE and drawing cards at random in different centres.
Table 2 shows the author stated reasons for envelope use, it was found that the most frequent reason given was budget (41.7%).
Author stated reasons for envelope use.
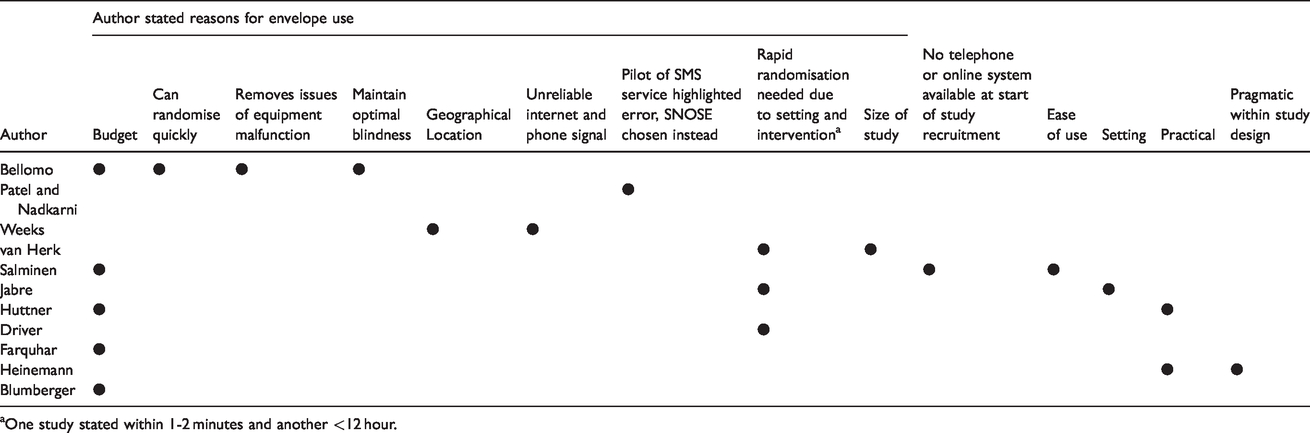
aOne study stated within 1-2 minutes and another <12 hour.
Discussion
Envelope use is similar to 2015 where Yelland found, in a similar group of journals, that 9% of RCTs used envelopes as a form of allocation concealment [2]. Reasons for envelope use were all appropriate for the trial design and setting that the research was being conducted.
It was disappointing that only 24% of the published RCTs in this sample contained robust methodological information to enable the reader to judge the RCT as adequate and low risk of bias when assessing the randomisation and allocation concealment methods. This therefore means that 76% of RCTs cannot enter into systematic reviews with a low risk of bias, there will be a higher level of uncertainty of the validity of the systematic review. All resources used to perform this research is wasted as the published report is not clear.
Many envelopes were not described as having any additional security measures. They were simply stated as being ‘sealed’, they may have been sequentially numbered however this was not stated so it cannot be assumed. Only one study (Boden – see Appendix 1 for a list of all included studies) stated that they used very secure envelopes with the addition of foil to wrap the allocation cards within the envelopes. Foil prevents the trans-illumination of the envelopes and further protects an RCT from subversion.1,9 It has been found that RCTs employing the use of envelopes without additional security measures are associated with an exaggerated effect size. 10 Results from insecure envelopes will be treated with caution by policy makers – thus highlighting the important of using secure envelopes and ensuring that if secure envelopes were used the details are reported comprehensively to enable policy makers to have confidence in the reported results. The use of foil with sequentially numbered sealed opaque envelopes in the opinion of the authors of this paper should be the gold standard way to set up an envelope for use within an RCT.
One point of interest is there are two RCTs in this sample that describe the same methodology from the large scale RCT but are reporting different results. Interestingly one study was deemed methodologically robust (Patel) and one was not (Nadkarni) as they did not report who created the envelope. This small omission has resulted in a study not being classed as having robust methodology.
There are a variety of valid reasons why envelopes are used within RCTs. We would urge researchers to carefully consider their research budget and assess whether they should allocate additional funds to cover the cost of a more robust and secure randomisation and allocation concealment method if their research design allows. Envelopes are inexpensive but if not executed and published robustly the entire research cannot contribute meaningfully to the evidence base.
After analysing the data gained in this research we can see that there is still a two-fold issue with envelope use. Envelopes are not being prepared in a rigorous manner with additional security measures and they are not reported in a transparent robust way ensuring all methodological information is provided. There is unclear information given to ascertain whether the envelope had additional security measures and whether the person creating the envelope is separate to the person who opens the envelope at the point of recruitment.
Future recommendations
Figure 2 shows the recommendation we have to the following when performing research with envelopes as an allocation concealment method.
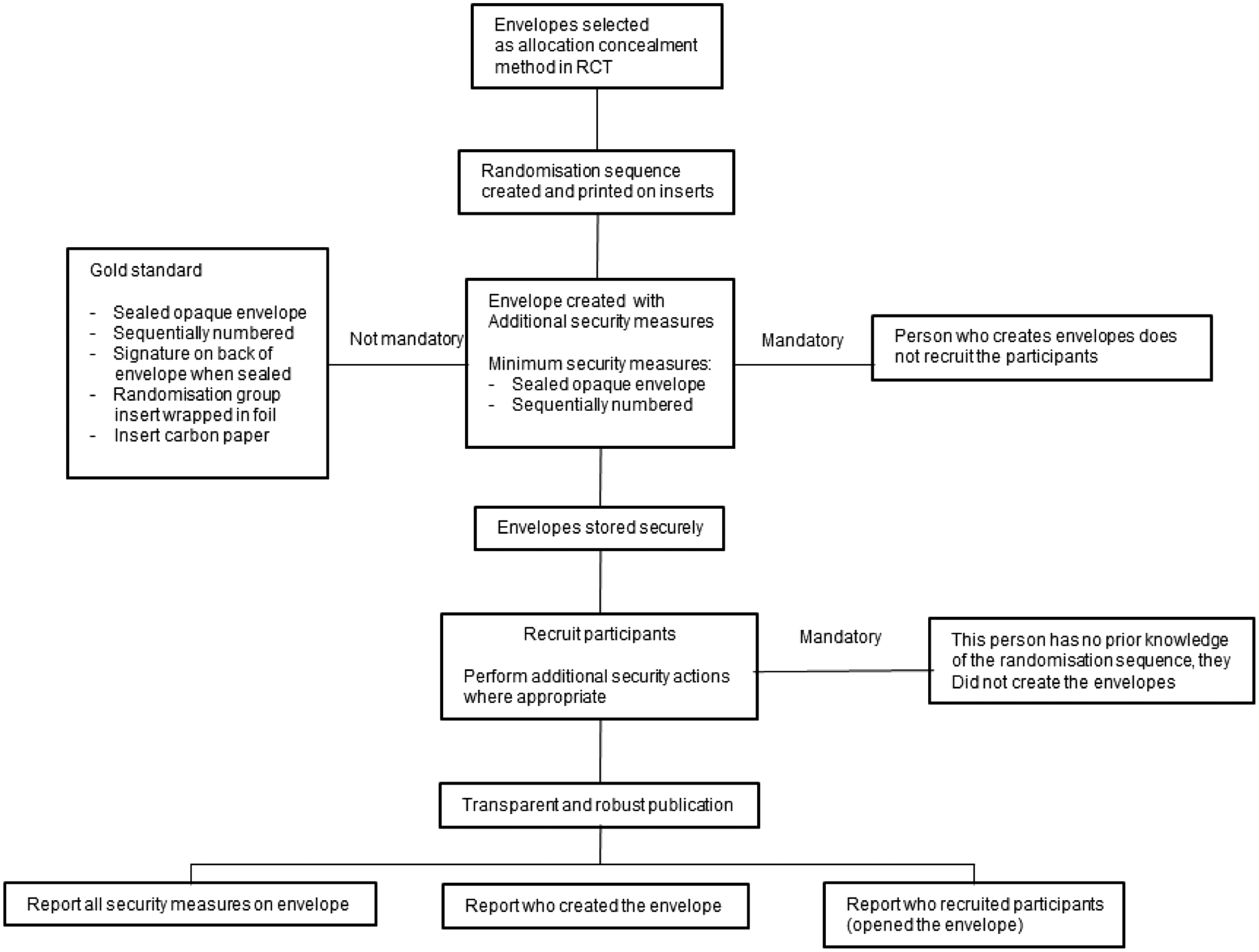
Flow diagram stating methodological steps to creating a robust envelope as an allocation concealment method in an RCT and essential methodological information to be reported in the publication of an RCT.
It is also pertinent to discuss the evolving nature of technology and allocation concealment methods. For rapid randomisation envelopes are a sensible choice, there are however apps being created that can rapidly randomise participants. These are also a relatively inexpensive method of randomisation and allocation concealment and may be used more widely in the future. Even with the use of innovative technology methodology will still need to be published thoroughly for methodological quality judgements to be made.
Moving forward we would urge authors to plan for and create secure envelopes when using envelopes as a form of allocation concealment are the only option for their RCT and to write their research transparently to include all the methodological information stated in Figure 2. Journals should ensure that any RCT published that uses envelopes as a form of allocation concealment should be reported robustly.
Conclusions
Allocation concealment methods are one of the most influential methodological factors on the validity of an RCT. Envelopes can be used as a robust method of allocation concealment. However, they are the most insecure method associated with subverting an RCT. If they are used within a research design they should be created robustly and reported clearly ensuring all necessary methodological information is included.
Supplemental Material
sj-pdf-1-rmm-10.1177_2632084320957204 - Supplemental material for Envelope use and reporting in randomised controlled trials: A guide for researchers
Supplemental material, sj-pdf-1-rmm-10.1177_2632084320957204 for Envelope use and reporting in randomised controlled trials: A guide for researchers by Laura Clark, Alexandra Dean, Alex Mitchell and David J Torgerson in Research Methods in Medicine & Health Sciences
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
