Abstract
Objectives:
Mid-regional pro-adrenomedullin (MR-proADM) and monocyte CD169 (CD169) are valuable prognostic indicators of severe COVID-19.
Methods:
We assessed the predictive ability of a single measurement of MR-proADM and CD169 at emergency department (ED) admission to forecast in-hospital and 60-day mortality in adult COVID-19 patients. We analyzed clinical and laboratory data, with in-hospital mortality as the primary endpoint and 60-day mortality as the secondary endpoint. We examined associations with clinical and laboratory variables through univariate and multivariate analyses.
Results:
Data from 382 patients over 14 months were analyzed. Significant predictors of in-hospital mortality included age ⩾ 70 years (hazard ratio [HR] 8.1; 95% confidence interval [CI] 2.2-29.5), CD169 ratio ⩾ 20 (HR: 2.4; 95%CI: 1.6-5.6), MR-proADM ⩾ 1.1 mmol/L (HR: 5.1; 95%CI: 1.7-15.6), the need for invasive mechanical ventilation (HR: 6.8; 95%CI: 2.4-19.1), and active cancer (HR: 5.2; 95%CI: 1.8-15.2). For 60-day mortality, only elevated MR-proADM levels showed predictive value (HR: 6.7; 95%CI: 1.7-25.0), while high serologic titer was protective (HR: 0.4; 95%CI: 0.1-0.9).
Conclusion:
A single MR-proADM and CD169 measurement upon ED admission has prognostic value for in-hospital mortality, with MR-proADM also predicting 60-day mortality.
Introduction
The SARS-CoV-2 pandemic has resulted in over 600 million reported cases globally, periodically overwhelming healthcare systems. Therefore, early patient assessment and appropriate management are essential to ensure safety and optimize hospital operations. 1 Effective tools to identify patients with poor prognoses are critical for optimizing resource allocation. 2 Numerous predictors have been evaluated to develop prognostic models for COVID-19, aiding triage and early assessment of disease severity. 3 The variability of inflammatory and immune responses to SARS-CoV-2 means that no single marker can reliably indicate disease phase, severity, or progression. During the host response to COVID-19, a range of biomarkers is produced at different stages, which may be useful for diagnostic and prognostic purposes. 4
Among these, type I interferons (IFN-Is) are key players in the innate immune defense against viruses, significantly upregulating CD169 (also known as Siglec-1) on the surface of monocytes—normally undetectable in non-infected individuals. This CD169 expression has been proposed as a promising marker for viral infections. 5 Studies conducted during the SARS-CoV-2 pandemic confirmed CD169 overexpression in COVID-19 patients, with sensitivity and specificity rates of 97% and 80%, respectively, even in the early stages of the disease.1,5 -8 Moreover, a strong IFN-I response in the early phase of SARS-CoV-2 infection can restrict viral replication, and CD169 expression levels have correlated with viral load and disease severity.
Another emerging blood biomarker, mid-regional pro-adrenomedullin (MR-proADM), has been linked to disease severity and mortality in COVID-19.1,9,10 As a specific indicator of endothelial dysfunction, MR-proADM has demonstrated value in predicting the risk of progression of infectious diseases in the emergency department (ED). 11 Identifying the most effective mortality predictors has become paramount in the pandemic context. This study examines whether CD169 and MR-proADM can serve as reliable prognostic biomarkers for emergency department decisions related to in-hospital and 60-day mortality in COVID-19 patients.
Materials and Methods
Study design
We conducted a prospective cohort study using the internal database of COVID-19 cases from the “Friuli Occidentale” health authority’s 800-bed hospitals in northeastern Italy, covering the period from October 30, 2021, to December 31, 2022. The Institutional Review Board exempted this study from review. All patients provided consent for processing their personal data for care and research purposes. The study adhered to international and national regulations in accordance with the Declaration of Helsinki. The clinical data entry staff was not involved in patient management or treatment decisions.
Patients included were over 18 years of age hospitalized for confirmed SARS-CoV-2 infection by real-time polymerase chain reaction (RT-PCR) or rapid antigen testing via nasal swab. Exclusion criteria included pregnancy, transplant recipient status, do-not-resuscitate (DNR) orders, and a life expectancy of less than 1 year due to comorbidities. Prior therapies (eg, steroids, antiviral drugs, monoclonal antibodies, antibiotics, anticoagulants) were recorded but did not constitute exclusion criteria. Raw data were collected at Santa Maria Degli Angeli Hospital in Pordenone, Italy. Derived data supporting the findings of this study are available upon request from the corresponding author.
MR-proADM measurements
Blood samples collected in tubes containing EDTA K3 were centrifuged at 2000 × g for 7 minutes. Aliquots of 1 mL of plasma were then frozen, stored at −20°C, and tested within 48 hours of collection. The MR-MR-proADM concentration was determined using a commercial fluorescence-based immunoassay with a time-resolved amplified cryptate emission (TRACE) assay (KRYPTOR®, Brahms Thermo Fisher Scientific Inc.). According to the manufacturer, the detection limit was 0.05 nmol/L.
Flow cytometry procedures
A 10 μL EDTA whole blood sample was lysed with 500 μL of VersaFix lysis solution (Beckman Coulter, Hialeah, FL, USA) and stained with 0.5 mL of CD45KO and 10 μL IOTest Myeloid Activation Antibody Cocktail (Beckman Coulter), containing 3 markers: anti-CD169-PE (clone 7-239), anti-CD64-PB (clone 22), and anti-HLA-DR-APC (clone Immu357). After a 15-minute incubation at room temperature in the dark, the samples were analyzed on a three-laser, 10-color Navios EX flow cytometer (Beckman Coulter) according to a compensation-free protocol and evaluated with Kaluza software version 2.1.1 (Beckman Coulter). For data analysis, leukocytes were gated using side scatter (SSC) versus cluster of differentiation (CD45) positive, CD64 expression as lymphocytes (low SSC, CD64−), monocytes (intermediate SSC, CD64+) and neutrophils (high SSC). The median fluorescence intensity (MFI) of CD64 expression relative to the neutrophil-to-lymphocyte ratio (nCD64) and the MFI of CD169 expression relative to the monocyte-to-lymphocyte ratio (CD169) were calculated, with a cutoff value of 3.51 for the CD169 ratio and 4.59 for nCD64, respectively.
Study variables
On admission, we collected the following data: demographic data, signs and symptoms (eg, fever), immunization status, comorbidities (eg, obesity, COPD, arterial hypertension, diabetes mellitus, cardiovascular disease, cerebrovascular disease, chronic renal failure, liver disease, cancer, cognitive impairment, and connective tissue disease), vital signs, blood gas analysis, blood chemistry values, inflammatory biomarkers (C-reactive protein and procalcitonin, MR-pro-ADM, CD64, and CD169), coagulation tests, administered treatments (eg, remdesivir, monoclonal antibodies), serologic titers, oxygen therapy or ventilation (invasive and non-invasive) and prognostic scores (SOFA, ROX, aPNea, 4C, NEWS2). 12 The outcomes of hospitalization and survival at 60 days were also recorded in the database. Patients underwent diagnostic work-up and were treated in COVID-19-dedicated units according to interim WHO guidelines and shared hospital protocols.
Statistical analysis
The variables are given as medians (interquartile ranges) or frequencies (%). Univariate hazard ratios (HRs) were calculated using a Cox model in which the grouping variable was introduced as an integer predictor. The P-values for the hazard ratios were calculated using the log-rank or Walt test under Cox proportional hazard regression when the row variable was categorical or continuous, respectively. An alpha error ⩽ .05 (P-value) was considered statistically significant. Multivariable Cox proportional hazards survival analysis was performed to identify variables significantly associated with in-hospital and 60-day mortality. To test the assumption that Cox regression is inherently time-invariant, we verified that the coefficient of the corresponding set of scaled Schoenfeld residuals with time was zero. In addition, the linearity assumption was confirmed by plotting the Martingale residuals against the continuous covariates. When hazards were found not to be time-invariant, we estimated the slope of the covariates over time by testing Aalen’s additive regression.
Kaplan–Meier survival analysis was performed to compare survival times between variables significantly correlated with in-hospital mortality and 60-day mortality. A log-rank test was conducted to determine whether there were statistically significant differences in the survival distribution among different subgroups. Statistical analysis was performed using the R environment (version 4.1.2, R Foundation for Statistical Computing, Vienna, Austria).
Results
During the study period, 393 patients were enrolled. Of these, 11 were excluded due to incomplete data or missing informed consent, resulting in a final study population of 382 patients (Figure 1).

Recruitment flowchart. The initial population consisted of 393 unselected adult patients with COVID-19. Three hundred and eighty-two patients were included in the study after the exclusion of 11 patients due to missing data or lack of consent. Of these patients, 50 (13%) died during hospitalization. A further 48 patients (12%) died during the 60-day follow-up period.
Table 1 summarizes the study population’s characteristics (Table 1). The median age was 69 years (IQR: 53-81), with 194 male patients (51%). Hypertension was the most common comorbidity (169; 44%), followed by obesity (62; 16%) and heart disease (61; 16%). The primary type of respiratory support was high-flow nasal cannula (149; 39%), followed by noninvasive ventilation (NIV) in 101 patients (26%). Seventeen patients (4%) required invasive mechanical ventilation. Remdesivir was administered to 84 patients (20%), and 30 patients (8%) received monoclonal antibodies. A SOFA score ⩾ 2 was observed in 26% of patients, while a ROX index ⩾ 30 was noted in 29%. Fifty-two patients (13%) were immediately admitted to the ICU.
Characteristics of the study population categorical variables are given as absolute values and percentages (parentheses); continuous values are given as medians and interquartile ranges (square brackets).

Abbreviations: A-a, alveolar-arterial difference; aPTT, activated partial thromboplastin clotting time; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; Hb, hemoglobin; HCO3, bicarbonate; ICU, intensive care unit; INR, international normalized ratio; LDH, lactate dehydrogenase; LOS, length of stay; MAP, mean arterial pressure; MR-proADM, pro-adrenomedullin; NIV, noninvasive ventilation; PaCO2, arterial carbon dioxide pressure; PaO2, arterial oxygen pressure; SpO2, peripheral capillary oxygen saturation; WBC, white blood cells.
The in-hospital mortality rate was 13% (50 patients), while the 60-day mortality rate stood at 12% (48 patients). Multivariate Cox survival analysis (Figure 2) showed the following variables to be significantly associated with in-hospital mortality: age ⩾ 70 years (HR: 8.06; 95%CI: 2.20-29.52; P = .002), high CD169 ratio (>20) (HR: 2.42; 95%CI: 1.06-5.56; P = .036), elevated MR-proADM levels (⩾1.1 nmol/L; HR: 5.14; 95%CI: 1.70-15.62; P = .004), the need for orotracheal intubation and invasive mechanical ventilation (HR: 6.75; 95%CI: 2.38-19.11; P < 0.001), and oncologic comorbidity (HR: 5.20; 95%CI: 1.78-15.22; P = .003). Conversely, alkalosis was associated with lower in-hospital mortality (HR: 0.18; 95%CI: 0.06-0.58; P = .004) (Figure 3). Aalen’s test (χ² = 20.87; P = .004) indicated an exponential increase in mortality with increasing age and MR-proADM levels (Figure 4).

Cox regression multivariable analysis of predictive in-hospital mortality variables. Agecat = age (Young: <70 years; Old: ⩾70 years); cd169cat = CD169 (Low: <20 ratio; High: ⩾20 ratio); proADMcat = MR-proADM (Low: <1.0 nmol/L; High: ⩾1 nmol/L); IOT = orotracheal intubation and need for invasive mechanical ventilation (0: no; 1: yes); pHcat = pH (Acid: <7.30; alkalosis: >7.45); and tumore = active cancer (0: no; 1: yes). AIC 250.6; P-value = 4.88 × 10−9; concordance index = 0.84.

Kaplan–Meier curve for pH (P-value = .0004) for in-hospital mortality. In yellow, patients with pH > 7.45; in red, patients with pH in the normal range (7.35-7.45); in blue, patients with pH < 7.35. Survival rate (%) on the y-axis, time (days) on the x-axis.
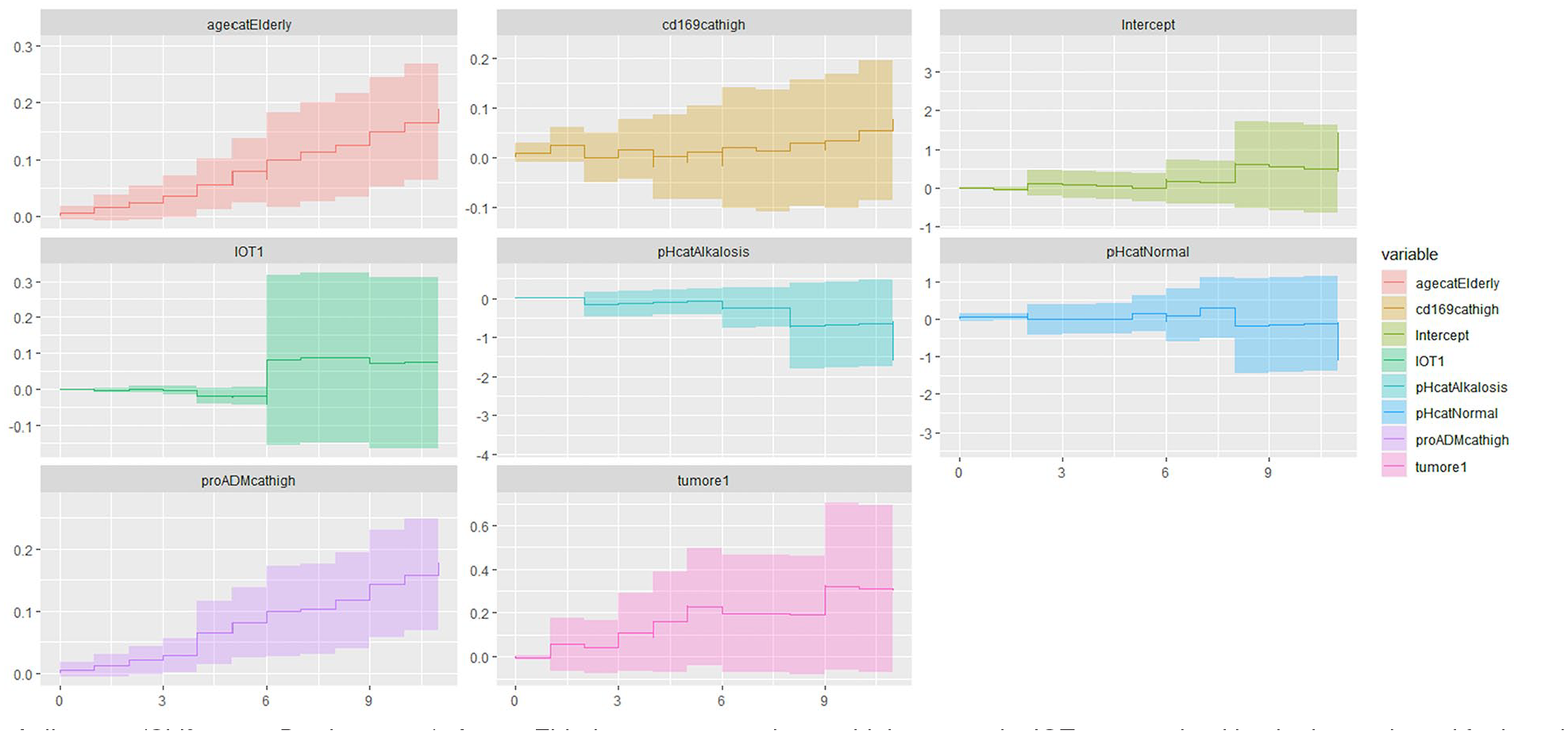
Aailen test (Chi2 20.87; P-value = .004). AgecatElderly: ⩾70 years; cd169cathigh: ⩾20 ratio; IOT1: orotracheal intubation and need for invasive mechanical ventilation; pHcatAlkalosis: pH < 7.45; pHcatNormal: pH 7.35 to 7.45; MR-proADMcathigh: ⩾1.1 mol/L; tumore 1: active cancer. Age andMR-proADM level showed an exponential increase in mortality over amount.
The multivariate Cox survival analysis for 60-day mortality (Figure 5) identified high pro-adrenomedullin levels (>1.1 nmol/L; HR: 6.56; 95%CI: 1.72-25.02; P = .006) as the only statistically significant predictor. Additionally, a high SARS-CoV-2 serum antibody titer (⩾5 U/mL) provided a protective effect (HR: 0.35; 95%CI: 0.14-0.87; P = .024).

Cox regression multivariable analysis of predictive 60-day mortality variables. AIC 139.5, concordance index 0.72; P-value = .007. MR-proADMcat = MR-proADM (Low: <1.1 nmol/L; High: ⩾1.1 nmol/L); pHcat = pH (Acid: <7.35; alkalosis: >7.45); and serology = anti-SarsCoV2 antibody titer (low_titre: <5 U/mL; high_titre: ⩾5 U/mL).
Kaplan–Meier curves (Figure 6A and B; Figure 7A and B) demonstrated that elevated MR-proADM levels negatively impacted prognosis, even in vaccinated patients (P < .001 for in-hospital mortality; P = .06 for 60-day mortality), although vaccination itself was protective compared to non-vaccinated patients.

Kaplan–Meier curves for in-hospital mortality according to the MR-proADM level (A) and further subdivided by SARS-CoV-2 vaccination status (B). (A) In-hospital mortality according to MR-proADM values, P < .0001. (B) In-hospital mortality according to MR-proADM values and SARS-CoV-2 vaccination status, P < .0001: in red, vaccinated patients with low MR-proADM levels; in blue, unvaccinated patients with low MR-proADM levels; in green, unvaccinated patients with high MR-proADM levels; in purple, vaccinated patients with high MR-proADM levels. Survival rate (%) is shown on the y-axis, and time (days) is shown on the x-axis.
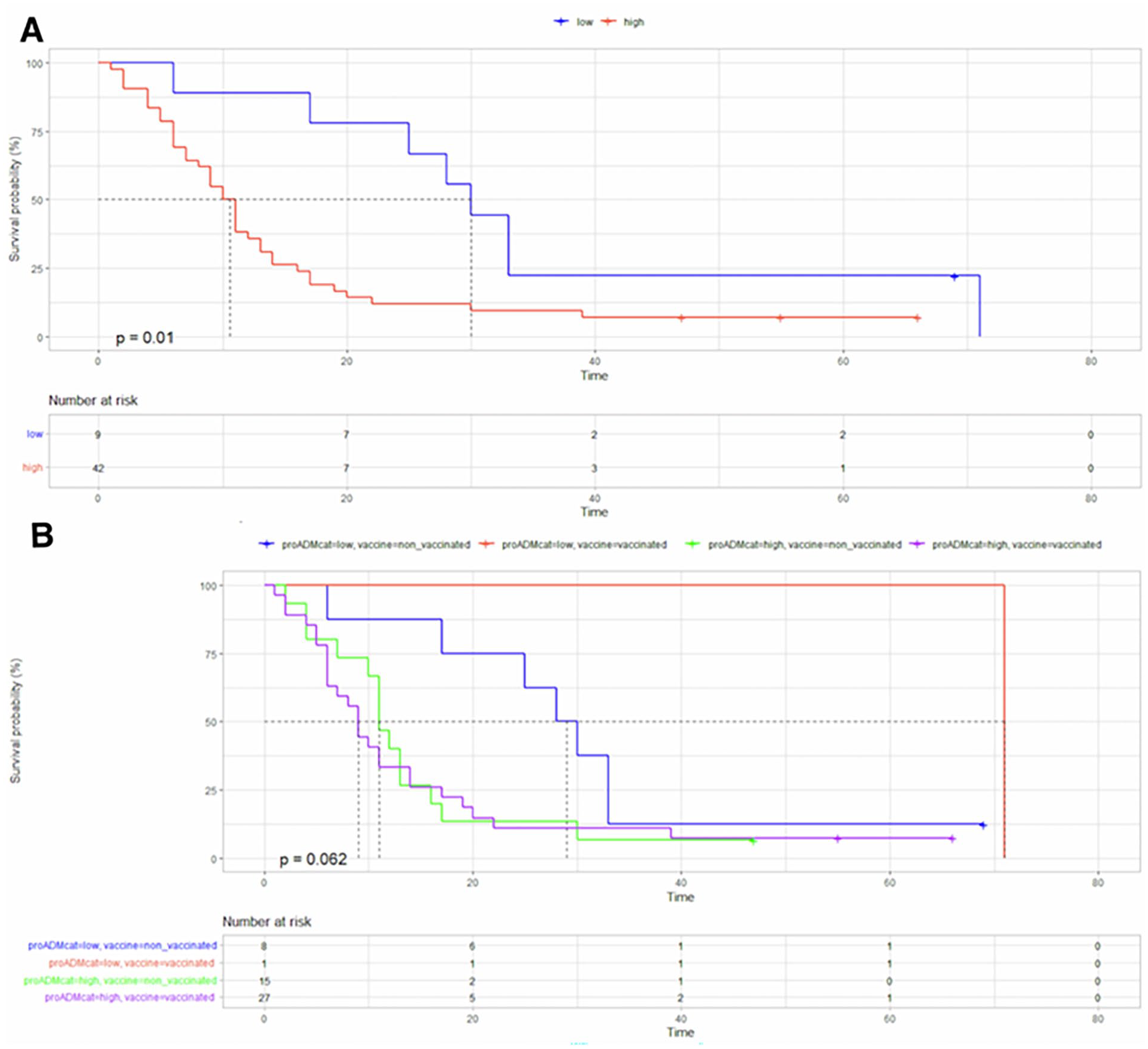
Kaplan-Meier curves for 60-day mortality according to the MR-proADM level (A) and further subdivided by SARS-CoV-2 vaccination status (B). (A) 60-day mortality according to MR-proADM values, p value = 0.01. (B) 60-day mortality according to MR-proADM values and SARS-CoV-2 vaccination status, p value = 0.062: in red, vaccinated patients with low MR-proADM levels; in blue, unvaccinated patients with low MR-proADM levels; in green, unvaccinated patients with high MR-proADM levels; in purple, vaccinated patients with high MR-proADM levels. Survival rate (%) is shown on the y-axis, and time (days) is shown on the x-axis.
Discussion
COVID-19 displays distinctive virus-host interactions, immune activation, and endothelial inflammation, which have been extensively investigated since the pandemic’s onset for diagnostic and prognostic insights. Beyond traditional biomarkers, newer indicators such as MR-proADM and CD169 have emerged as promising prognostic tools.6,13,14 However, data validating a single biomarker for both diagnosis and outcome prediction are still limited. Our findings demonstrate that MR-proADM and CD169 are reliable and scalable markers of disease severity, with survival analyses indicating a significant prognostic role for these markers in predicting in-hospital mortality in a broad adult COVID-19 patient cohort.
Severe COVID-19 often presents as a multisystemic disease, with endothelial damage as a key feature. Vascular endothelial alterations, prothrombotic states, and cytokine overexpression contribute to disease progression toward ARDS, multiorgan involvement, and increased mortality. MR-proADM is a well-established marker of endothelial dysfunction in sepsis and pneumonia, with elevated levels predictive of severe disease and poor outcomes.15 -17 Numerous studies have attempted to link MR-proADM levels to mortality risk or clinical deterioration in SARS-CoV-2 patients.
Our analysis of a single MR-proADM measurement at ED presentation provides significant prognostic value. de Montmollin et al. 13 demonstrated that prognostic accuracy for in-hospital and 60-day mortality is consistent regardless of the timing within the first day post-admission. Thus, even a single MR-proADM determination is predictive and may reduce costs and resource usage. The optimal cutoff value for MR-proADM identified in our cohort aligns with findings from other studies,11,18 indicating that an MR-proADM level ⩾1.1 nmol/L has strong prognostic performance in COVID-19 patients presenting to the ED. In most studies, MR-proADM levels above 1 to 2 nmol/L have been linked to an elevated risk of death and disease progression (requiring NIV or invasive mechanical ventilation), with satisfactory sensitivity and specificity.14,19
In an emergency setting, MR-proADM can aid clinicians in making decisions to escalate care for high-risk patients while enabling safe, lower-complexity management for low-risk patients, either in less intensive wards or outpatient settings.
To our knowledge, few studies have examined the prognostic potential of CD169 in COVID-19 patients. In hospitalized COVID-19 patients, CD169 correlates with inflammatory and immune status and is associated with respiratory outcomes, with marked overexpression seen in critically ill patients. 13
Monocyte CD169 ratio measurement in the emergency setting has shown high sensitivity for detecting SARS-CoV-2 infection, even in the early stages. It offers several advantages: high sensitivity, easy integration with existing laboratory equipment (flow cytometry), affordable reagents, minimal invasiveness, a turnaround time of under 1 hour, and 24/7 availability.1,7 Our findings indicate that CD169 expression provides valuable risk stratification for in-hospital mortality. However, unlike MR-proADM, CD169 does not demonstrate significant prognostic value for mid-term (60-day) mortality. Minutolo et al. 14 found that CD169 is strongly associated with various clinical and biological parameters, reflecting more than just patient status at admission. Additionally, CD169 modulation is influenced by treatment factors, such as glucocorticoids, which reduce interferon production by inhibiting Toll-like receptors. 20
In our study, additional significant factors influenced in-hospital mortality, including age over 70 years, the need for invasive mechanical ventilation, and oncologic comorbidities. These findings align with existing literature showing that elderly and cancer patients have an inherently higher risk of short-term mortality from SARS-CoV-2 infection, as do those requiring mechanical ventilation upon admission. 21 Acidosis reflects both a severe form of respiratory failure (eg, hypercapnia) and possible metabolic acidosis due to renal failure, both of which have previously been identified as poor prognostic factors. 22
Regarding in-hospital mortality, a high antibody titer appeared to be associated with a more favorable outcome. Recent studies indicate that anti-SARS-CoV-2 IgG protects against symptomatic COVID-19, suggesting that antibody measurement could enhance prognostic assessment in current diagnostic protocols. 23 Although a clear threshold for low titers is not established, we followed Malipiero et al.’s 24 guidelines, considering titers below 5 times the cutoff (5 U/mL) as low. Post-vaccination values in our cohort ranged from 27.55 to 466 U/mL, supporting the notion that a level below 5 U/mL reasonably indicates a low titer. 25
Our study has several limitations. First, recruitment occurred over an extended period, during which multiple COVID-19 variants emerged. Determining the extent to which these variants may have influenced our findings is challenging. Additionally, there is no definitive threshold defining a low antibody titer below 5 U/mL; we adopted the guideline from Malipiero et al., 24 considering titers below 5 times the cutoff as low. Post-vaccination titers in our cohort ranged from 27.55 to 466 U/mL, supporting the notion that a value under 5 U/mL reasonably represents a low titer.
As with all predictive models, ours is fundamentally limited by the variables considered. We cannot exclude the possibility that unaccounted-for variables may have a stronger impact on patient outcomes than the identified predictors, and some pre-existing conditions (eg, cancer, advanced cardiovascular disease, and renal failure) may alter baseline biomarker kinetics. 10 Although the literature supports the use of a single determination, we acknowledge that serial determinations may offer even greater predictive accuracy.
Conclusion
In conclusion, we can affirm that determining MR-proADM and CD169 levels at the time of patient presentation in the ED is indicated. Even though these data are not monitored over time, they still prove helpful regarding the patient's 30-day prognosis. Such measurements could be implemented in any hospital since they are simple, rapid, and easily accessible. When combined with a comprehensive patient assessment, these biomarkers could enable rapid stratification of COVID-19 patients, saving resources and optimizing decision-making processes in the ED and healthcare costs. Finally, the necessity of maintaining a high antibody level, particularly in at-risk patients, becomes even clearer.
Footnotes
Acknowledgements
Not applicable.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Conceptualization, methodology, data collection and analysis, project administration: Sergio Venturini. Data collection: Giovanni Del Fabro, Dina Giordani, Astrid Callegari, Elisa Pontoni, Gian Luca Colussi. Sample analysis: Danilo Villalta, Chiara Pratesi, Paolo Doretto. Methodology and statistical analysis: Daniele Orso, Francesco Cugini. Writing original draft and revisions, review and editing: Sergio Venturini, Ingrid Reffo, Francesco Cugini. Review and final version approval: Laura De Santi, Maurizio Tonizzo, Massimo Crapis.
Ethical Approval
The Hospital Institutional Review Board waived the need for ethics approval for the collection, analysis, and publication of the anonymized data for this non-interventional study.
Informed Consent Statement
Each patient consented to processing their personal data for care and research purposes.
Data Availability Statement
The derived data supporting this study’s findings are available upon request from the corresponding author.
