Abstract
Introduction:
Poroid neoplasms (PN) are a heterogeneous group of tumors deriving from sweat glands and folliculo-sebaceous units. Their histological classification and clinical features are challenging. Our aim was to report clinicopathological features of poroid neoplasms.
Methods:
It is a retrospective study including all cases of poroid neoplasms registered at our Pathology laboratory of Niamey National Hospital (February 2020-February 2024).
Results:
We registered 13 cases of benign poroid neoplasms: 10 classic poromas (CP) (76.9%), 2 poroid hidradenomas (PH) (15.4%) and 1 dermal duct tumor (DDT) (7.7%). Nine cases (69.2%) had preoperative clinical diagnosis of malignancy. The mean age was 41.1 years (range of 12-70 years) with a slight female predominance. Only 4/13 cases (30.8%) had classical palmoplantar locations. The tumors mean size was 3.7 cm (range of 0.4-8 cm). Clear cells were present in 7 cases (53.8%), apocrine ductal differentiation (mixed or pure) in 6 cases (46.2%), keratin horns in 2 cases (15.4%), squamous eddies in 6 cases (46.2%), melanin pigments in 1 case (7.7%) and sebaceous differentiation in 2 cases (15.4%).
Conclusions:
Unlike what is classically reported, our study shows that apocrine ductal differentiation, younger age and non-palmoplantar locations are common in poroid neoplasms.
Introduction
Poroid neoplasms (PN) are a group of heterogeneous tumors with sweat glands and folliculo-sebaceous differentiation representing 10% of primary sweat gland tumors.1 -3 Since the first description by Goldman et al. 4 in 1956, it is now well established that this group of neoplasms include 4 main benign entities according to their architecture, location in the dermis and connexion with the epidermis: classic poroma (CP), formally known as eccrine poroma, that is located in the dermis with broad epidermal connexion, hidroacanthoma simplex (HS) restricted to the epidermis, dermal duct tumor (DDT) that is intradermal with numerous small lobules, and poroid hidradenoma (PH) which is also intradermal but with larger cystic nodules.3,5 All these neoplasms show a mixture of small, round, basophilic poroid cells, and larger eosinophilic squamoid cuticular cells, often associated with ductal structures that have eccrine or apocrine differentiation.1,3 -7 Also, these 4 benign lesions could coexist in a same lesion.1,7 It is now well established by many studies that PN derive from the basal keratinocytes of the sweat duct ridge and the lower acrosyringium.3,8 Malignant transformation of PN into porocarcinoma has been reported in the literature.9,10 Rarely PN can present with sebaceous11,12 or follicular differentiation 13 along with clear cell changes, keratinization 1 or melanin pigments.14,15 In earlier studies, PN have been reported with classical palmoplantar locations in elderly patients,4,6 however subsequent studies challenged these reports, with non-palmoplantar locations and occurrence in younger patients.3,16 -19 Also, apocrine differentiation has been reported in later reports and is not so unusual as believed.7,19 -21
We reported herein, to the best of our knowledge the first series about clinical and histopathological features of PN in our subsaharan African country.
Methods
As the study was a retrospective report with de-identified, anonymous data, ethical approval, and consent to participate were not required due to local/national guidelines (DAMTE/HNN, Ny-Nig/01/24).
This is a retrospective study on PN registered at our newly operative Pathology laboratory from February 2020 to February 2024 (4 years). Clinical data were retrieved from the pathology request forms as well as from phone calls of patients’ surgeons.
The histological diagnosis has been made on formalin-fixed, paraffin-embedded surgically resected specimens, and stained by hematoxylin-eosin (HE).
Additional serial sections were performed on all retrieved archival paraffin-embedded specimens and re-assessed by two pathologists (BE and IB) in search for some eventually initially missed histological features: epidermal connexion, folliculo-sebaceous differentiation, clear cell changes, horn cysts, squamous eddies, necrosis en masse, eccrine, or apocrine ductal differentiation.
A part of this study has been presented as an E – poster at the 61st International Academy of Pathology (IAP), Thailand division, Annual meeting 2022, 2nd to 4th November 2022, virtual meeting.
Results
Clinical features
The Table 1 summarizes the clinical features of our series of PN. The mean age was 41.1 years (range of 12-70 years) with a slight female predominance (sex ratio = 1.16). The most frequent tumor’s locations were the scalp (3 cases, 23.1%; Figure 1a), the sole (3 cases, 23.1%; Figure 1b), and the thoracic wall (2 cases, 15.4%). Only 4/13 cases (30.8%) had classical palmoplantar locations as the majority of patients had tumors located in hair-bearing parts of the body. The mean tumor size was 3.7 cm (range of 0.4-8 cm). All lesions were grossly well-circumscribed, with ulcerations in 4 cases (30.8%). PN appeared grossly as solid (7/13 cases, 53.8%) or solid-cystic lesions (6/13 cases, 46.2%) with whitish or pink color and mucinous content (Figure 1c). The preoperative clinical diagnosis was suggestive of malignancy in 9/13 cases (69.2%).
Clinical features of our series of poroid neoplasms.


(a) Clinical image showing a well-defined nodule with smooth surface in the scalp (case 1), (b) Clinical image showing a well-circumscribed nodular lesion in the sole (case 13), and (c) Resected surgical specimen after formalin-fixation. The tumor is whitish, solid-cystic with mucinous compact content (case 1).
All patients had complete surgical resections of their tumors, and none had reported recurrence at the time of this report.
Histopathological features
Histopathological features of our patients were summarized in Table 2.
Histopathological findings in our cases of poroid neoplasms.

: present; −: absent.
After serial additional sections, the final diagnoses of the 13 patients were: 10 classic poromas (CP) (76.9%) (Figure 2a), 2 poroid hidradenomas (PH) (15.4%) and 1 dermal duct tumor (DDT) (7.7%). All cases were made of a variable mixture of poroid (uniform small cuboidal cells with round nuclei) and cuticular cells (larger cells with abundant eosinophilic cytoplasm), without cellular atypias, mitoses, or stromal infiltration. The tumors’ stroma was fibro-hyaline and inflammatory in all cases, sometimes loose and myxoid. A broad epidermal connexion was observed in all cases of CP, whereas absent in cases of PH (cases 6 and 9) and DDT (case 12). Clear cells were observed in 7 cases (53.8%; Figure 2b). Focal keratin horns were present in 2 patients (15.4%; Figure 2c, arrow), while focal poroid cells maturation as squamous eddies were found in 6 cases (46.2%; Figure 2d, arrow). Ductal structures were generally dilated and cystic with eosinophilic amorphous contents, with pure eccrine differentiation in 7/13 cases (53.8%; Figure 3a, arrow), mixed eccrine/apocrine differentiation in 5/13 patients (38.5%) and 1 pure apocrine differentiation (bulging apical cytoplasm with decapitation secretions; Figure 3b, arrow). Sebaceous differentiation (Figure 3c) and melanin pigments (Figure 3d) were present respectively in 2 and 1 patients. Despite serial additional cuts, follicular or necrosis en masse, have not been found in all the 13 cases of the current series. Also, we have not registered malignant PN such as porocarcinoma.

(a) Histological view of an eccrine poroma showing a dermo-epidermic tumor with predominantly poroid cells, broad-epidermal connexion, and large anastomosing trabeculae disposed in a hyaline and inflammatory stroma (hematoxylin-eosin × 100) (case 2), (b) Histological image showing large areas of clear cell changes with abundant clear cytoplasm (hematoxylin-eosin × 200) (case 2), (c) Horn cyst consisting of focal concentric lamellar keratin within a microcystic space (hematoxylin-eosin × 200) (case 1), and (d) A squamous eddy is seen in the image as focal maturation of poroid cells (red arrow) (hematoxylin-eosin × 100) (case 5).
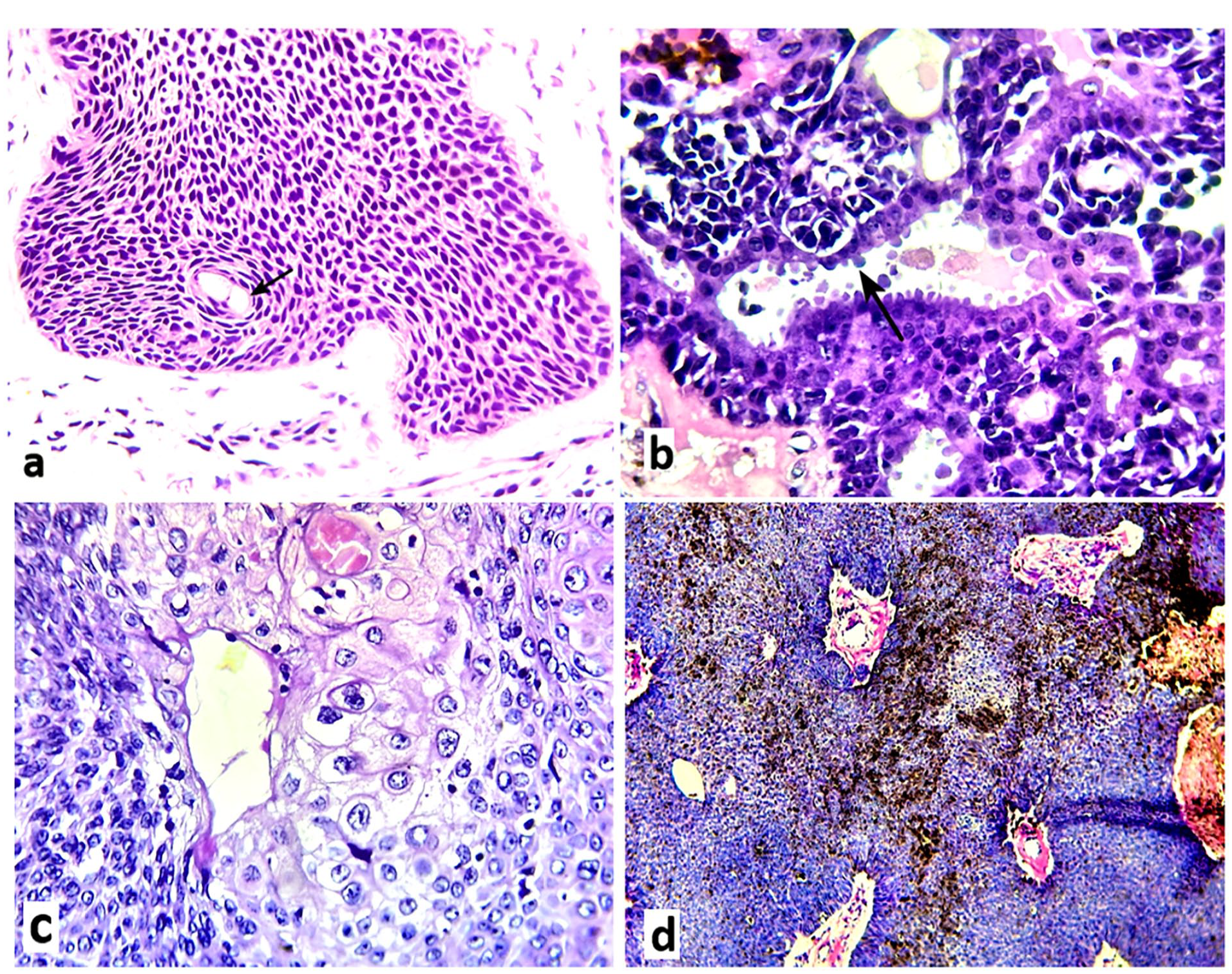
(a) Eccrine ductal differentiation shown as a small duct lined by flat cells with conspicuous eosinophilic cytoplasm (black arrow) (hematoxylin-eosin × 200) (case 5). (b) Apocrine ductal differentiation seen as a cystic duct lined by epithelial cells with bulging apical eosinophilic cytoplasm with decapitation secretion (black arrow) (hematoxylin-eosin × 400) (case 9). (c) Sebaceous differentiation consisting of large cells with abundant clear granular cytoplasm with central rounded nuclei (hematoxylin-eosin × 400) (case 9). (d) a case of CP showing large areas with melanin pigments (hematoxylin-eosin × 100) (case 11).
Discussion
Over a period of 4 years since the creation of the first operative Pathology laboratory in a public hospital in our country, we have registered 13 cases of poroid neoplasms (PN). The duration of our study is too short to reflect the real epidemiological characteristics of these neoplasms in our context, however some conclusions could be drawn from our current small series.
Poroid neoplasms are rarely reported tumors across the literature, with case reports,11,15,17,22 -25 small series and few large series with limited sample (no more than 400 cases).1,3,9,26,27 From 1988 to 2003 Chen et al. 1 have registered 25 cases in Taiwan, while Betti et al. 16 in Italy, Batistella et al. 3 in France, have respectively reported 101 cases from 1994 to 2012, and 266 cases from 1980 to 2008. To the best of our knowledge, Ito et al. 7 have reported the largest series of PN with 384 cases.
In our series, the mean age was 41.1 years (range of 12-70 years) with a slight female predominance (sex ratio = 1.16). Our patients were younger than what is commonly reported in the literature where the mean age turns around 50 to 60 years, with no established sex predominance in larger series.1,3,7,28 However, cases of PN in children and younger patients were reported.23,29,30
Earlier reports found that PN were classically located in palm and sole,4,28 however additional studies proved the opposite by reporting more frequent locations in hair-bearing areas of the body such as the head and neck regions, the trunk and limbs.1,7,16 Our results are in accordance with these later studies as 9/13 (69.2%) of our patients had tumors located in hair-bearing sites of the body.
Poroid neoplasms are usually solitary lesions but multiple locations (eccrine poromatosis) have been reported.31 -34 Eccrine poromatosis was usually reported in patients with a history of immunosuppression from radiation, chemotherapy or transplantation.32,33 All of our patients had single tumors, and none was immunosuppressed. Tumors were well-circumscribed with ulcerations in 4 cases (30.8%), with a mean size of 3.7 cm (range of 0.4-8 cm). This relatively larger size of the tumors reflects the socio-economic context of our study (low-income settings) where diagnoses are often delayed.
In our series, PN appeared as solid or solid-cystic lesions with whitish or pink color and mucinous content. Often, PN are red, pinkish, or skin-colored elevated lesions on clinical examinations 6 with solid or solid-cystic cut surface. The gross features reported in our series correspond mainly to resected specimens (ex vivo) after formalin fixation, thus quite different from clinical aspects (in vivo) of the tumors. Ulcerations were present in 4 of our cases, a fact that is not unusual according to previous studies.3,27
The majority of cases were clinically suspected of malignancy in our series (9/13 cases), especially in patients with ulcerated or larger lesions. The clinical features of PN lack any specificity even on dermoscopic analysis.26,27 However, a large multicentre observational case study by Marchetti et al. 35 showed that there were 4 dermoscopic features associated with PN: white interlacing areas around vessels, yellow structureless areas, milky-red globules, and poorly visualized vessels. In our series, 12 patients out of 13 (92.3%) have been managed by surgeons, only one patient has been treated by a dermtologist (case 13) and dermoscopic analysis was not performed in all cases.
In larger reported series, the clinical diagnosis of PN was more often missed, with numerous differential diagnoses such as pyogenic granuloma, nevus, hemangioma, basal cell carcinoma, melanoma, . . .etc.1,3,6
The definitive diagnosis of PN relies on the histopathological analysis. Poroid neoplasms have well-described characteristic morphological features, thus ancillary testing such as immunohistochemistry are not required for the diagnosis in routine histopathological practice.3,6,26 As in the majority of previous studies, our current cases have been diagnosed with the routine standard histopathology techniques (formalin fixation, paraffin embedding, and HE-staining). On immunohistochemical analysis, PN express a variety of cytokeratins (CK) with a phenotype similar to that of the upper acrosyringium keratinocytes.3,8,36 Poroid cells express CK5/8, CK14, CK1/5/10, CK11, CK7, CK8/18, and CK19 while cuticular cells express CK1/5/10/14, CK10/11, CK7, CK6, CK8/18, and CK19; the duct-lining cells are positive for CK1/5/10/14, CK10/11, CK6, CK7,CK8/18, CK19, and CK77.3,36 Also, compared to poromas (benign tumors), porocarcinomas show altered p53, p16, and/or Rb immunostaining. 37 Molecular analysis showed that PN (benign or malignant) are characterised by recurrent gene fusions involving YAP1 (a transcriptional co-activator) with MAML2 or NUTM1.38 -40 Interestingly, these YAP1 rearrangements can be detected by immunohistochemistry or by fluorescence in situ hybridization.38,39
Our histological diagnoses of PN consisted of 10 CP (76.9%), 2 PH (15.4%), and 1 DDT (7.7%). We have not registered malignant tumors with poroid differentiation (especially porocarcinoma). Our results are quite similar to those reported in the literature with DDT being the less commonly found variant among PN.7,41 Apocrine ductal differentiation was observed in 6/13 cases (46.2%); this frequent apocrine differentiation is in contradiction of what is commonly reported in the literature.7,19,20 The seemingly rare apocrine differentiation in previous studies would be likely due to the fact that apocrine ductal differentiation was overlooked on morphological evaluation and classification of PN.7,20,36 In fact apocrine differentiation seems to be relatively common in PN as Ito et al. 7 have reported that a quarter of them show apocrine differentiation. In our series, sebaceous differentiation were observed in 2 cases (15.4%). In fact, sebaceous differentiation was found in 1.3% to 4.8% cases of PN in some reports.7,11
Poroid neoplasms are characterized by an admixture of poroid and cuticular cells in variable proportion as reported in our series, however cases of PN with only poroid cells have been reported. 1 Usually the proportion of poroid cells is predominant in PN, however in 2017 Alegria-Landa et al. 42 have reported 2 cases of poromas mostly composed of cuticular cells and named them cuticular poromas. We found clear cells in 7/13 cases (53.8%), intermingled with other tumor cells, sometimes forming large clusters, a finding that is not uncommon in PN. 1 Also, in 6/13 cases (46.2%), we have found focal maturation of poroid cells termed as squamous eddies. This feature was rarely reported in the literature, 1 likely because of its lack of diagnostic value. However we have observed focally keratin cysts (horn cyst) in 2 cases of our patients, a feature that could suggest the diagnosis of seborrheic keratosis especially in small biopsies when the lesion is connected to the epidermis and ductal structures not observed in the sample. 1
Cases of recurrences (if incomplete resection)6,25 or malignant transformation10,43 of PN have been reported previously. Until now, we have not registered recurrence or malignant transformation in all of our patients. The surgical resection was complete in all cases, and there were no history of trauma or immunosuppression. Trauma and immunosupression have been reported as predisposing factors in patients with PN.6,31,34
Conclusion
Poroid neoplasms are rare benign adnexal tumors derived from sweat glands and folliculo-sebaceous units. Occurrence in younger patients and location in hair-bearing body parts are frequent. The clinical features are often misleading, with the definitive diagnosis relying on histopathological analysis of the resected specimens. Apocrine differentiation and clear cell changes are common in poroid neoplasms.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
BE wrote the article and made substantial contributions to its conception and design. IB, KAOK, ABAB, HSB, AS and HN were involved in drafting the manuscript and its critical revision. All authors read and approved the final version of the manuscript.
Data Availability Statement
All data generated or analyzed during this study are included in this article.
