Abstract
Introduction:
Human immunodeficiency virus (HIV) infection is a risk factor for the occurrence of a large of Mycobacterium tuberculosis (Mtb) antigen load in the body. The antigens cocktail namely early secretory antigenic target protein 6-kDa (ESAT-6), Culture filtrate protein 10 kDa (CFP-10), and Mycobacterium tuberculosis protein 64 (MPT-64) are secreted by Mtb during replication, hence, their concentration increase in patients with active Tuberculosis (TB). This increased levels facilitates their entry into the systemic circulation, followed by secretion by the glomerulus into the urine. The aim of this study was to determine the positivity rate of the urinary Mtb antigens cocktail between TB patients with and without HIV infection.
Methods:
This is an observational descriptive comparative study conducted with a cross-sectional design. Random urine samples were collected from patients diagnosed with active TB in Dr. Hasan Sadikin Bandung Hospital in 2021. The subjects were divided into 2 groups, TB-HIV group and TB without HIV group. The samples were tested using the quantitative immunochromatography method.
Result:
Sixty active TB patients consisting of TB patients with HIV infection (n = 30) and TB patients without HIV infection (n = 30). The positivity in the urinary Mtb antigens cocktail was 93.3% for TB-HIV group and 100% for TB without HIV group (P = .492). The median concentration of urinary Mtb antigens cocktail in TB patients without HIV infection was higher than that of TB patients with HIV infection (137.73 ng/mL vs 96.69 ng/mL, respectively; P = .001).
Conclusion:
There was no significant difference in the positivity rate, meanwhile, there was a significant difference in concentration of the urinary Mtb antigens cocktail between active TB patients with and without HIV infection. Interestingly, this urinary Mtb antigens cocktail can be found in both groups without being affected by the patient’s immune condition, thus becoming a test to assist diagnose active TB.
Introduction
World Health Organization (WHO) stated that Indonesia is one of the countries with the most cases of TB and HIV in the world. 1 Human immunodeficiency virus infection is a risk factor for the occurrence of a large Mtb bacterial load or higher bacillary burden and antigen load in the body. 1 Therefore TB is a prevalent opportunistic disease and causes the death of ±40% to 50% of people living with HIV (PLHIV). 2 This high mortality is especially found in acid-fast bacilli (AFB) negative pulmonary and extra-pulmonary TB-HIV which is most likely due to delays in diagnosis and therapy. 2 The death of TB patients can be prevented by establishing a diagnosis and early administration of therapy, therefore WHO recommends increased access to early and accurate. TB diagnoses are conducted using the WHO-recommended rapid diagnostic (WRD), diagnostic examination of molecular techniques to detect Mtb. 3 Certain problems occur when the facilities and infrastructure are not available to meet the WHO recommendations in the laboratory. Therefore, other strategies are needed to detect Mtb bacteria with accompanying benefits in the form of a more affordable examination both in terms of cost, security, practicality, and safety. 4
The test currently being developed to diagnose TB is antigen identification. Further, several research groups reported that a combination of multiple antigens could improve the diagnosis of TB,5 -8 The antigens cocktail namely ESAT-6, CFP-10, and MPT-64 are protein secreted by Mtb that plays an important role in virulence factors for the spread of Mtb between cells to the extra pulmonary. The antigens are encoded by the Regions of Differences (RD) 1 to 3 gene. 9 This antigens are secreted during replication, hence, its levels can increase in patients with active TB. 10 The increased levels makes it easier for the antigens to enter the systemic circulation which will then be filtered by the kidneys into the urine. The antigens cocktail can be freely filtered by the glomerulus because it has a molecular weight of less than 67 kDa. In addition, it is not reabsorbed in the proximal tubule but excreted directly into the urine. 6 Noninvasive body fluid such as urine is the ideal clinical specimen because it is excreted in large quantities and the collecting process does not require invasive methods, and facilitating utilization in low resource settings. There were studies that can detect Mtb lipoarabinomannan (LAM) antigens in the urine of pulmonary TB patients co-infected with HIV, but not in TB patient without HIV infection. 11 However, there is still no study evaluating the performance of a Mtb antigens cocktail ESAT-6, CFP-10, and MPT-64 from urine samples of TB-HIV patients. Therefore, the aim of this study was to determine the positivity rate of the urinary Mtb antigens cocktail between TB patients with and without HIV infection.
Methods
Study population
This is a preliminary study conducted with a cross-sectional design was performed from June 2021 until September 2021 at Dr. Hasan Sadikin General Hospital in West Java Province, Indonesia. Patients visiting the outpatient clinic or admitted to the TB ward were screened for eligibility. The inclusion criteria were: (1) adult (>18 years old), (2) suspected to have active pulmonary (according to the International Standards of Tuberculosis Care) 12 or extrapulmonary TB who had been diagnosed by clinicians, (3) accompanied with or without HIV infection, (4) the CD4 examination is only performed on subjects with HIV. The exclusion criteria were: (1) urinary tract infections diagnosed with dipstick and culture testing; (2) abnormal kidney function tests or any history of renal disease. Study participants were placed into 2 groups: (a) an active pulmonary TB or extrapulmonary TB-HIV group, (b) an active pulmonary TB or extrapulmonary TB without HIV group. The number of samples is determined based on the “rule of thumb” principle, and is taken consecutively until the number of samples of research subjects required in a different test is 30 in each group. 13
Specimen collection
Expectorate sputum was obtained from participants from the pulmonary TB patient. Specific clinical specimens were obtained from extrapulmonary according to the hospital standard operating procedure for AFB direct smear. On the same day, each included participant was also required to submit a random urine specimen with a clean-catch collection technique. 14
Laboratory testing for TB and HIV
Sputum and nonsputum specimens were assessed for AFB with the Ziehl Neelsen (ZN) technique (ST Reagensia Company, Jakarta, Indonesia) according to the standard protocol. 15 Anti-HIV examination according to The Regulation of the Minister of Health of the Republic of Indonesia algorithm No. 15 of 2015. 16
The urine specimen was collected in a sterile container and directly centrifuged at 1000g for 15 minutes to separate the low molecular weight analytes, and then kept at −80°C for further examination of the Mtb antigens cocktail (ESAT6, CFP10, and MPT64). The cocktail was detected using the semiquantitative immunochromatography test (ICT) method (Jei Daniel Biotech Corp., Taiwan), in which 100 µL of sample buffer was placed into the specimen collection box with 100 µL of urine sediment and mixed well by dropper for 30 to 60 seconds then left to stand for 30 minutes. Four (60 µL) drops were applied to the “S” region of the testing card, and the result was read after 30 minutes using an automatic reader (JD Reader) to determine the Mtb antigen cocktail concentration of the clinical specimen. Quality control and instrument assurance were performed according to the manufacturer’s recommendations.
Data analysis
The data were analyzed using the Statistical Package for the Social Science (SPSS) software version 25.0 for Windows. The urinary Mtb antigens cocktail Positivity rate was analyzed using the Fisher Exact, and the significance of differences in median urinary Mtb antigens cocktail concentration among groups was evaluated using the Mann-Whitney test. We considered tests with P < .05 to be significant. The study was approved by the Health Research Ethics Committee of Dr. Hasan Sadikin General Hospital Bandung through the issuance of ethical approval no. LB.02.01/X.6.5/195/2021.
Result
A total of 110 participants were screened in this study, and their positivity in the urinary Mtb antigens cocktail was 93.3% for the TB-HIV group and 100% for TB without HIV group (P = .492; Figure 1).

Study flowchart.
The proportion of males among participants was dominant (73.3%), there are 60% of TB-HIV patients who have negative smear results. The characteristics of the subjects are shown in Table 1.
Characteristics of subjects.
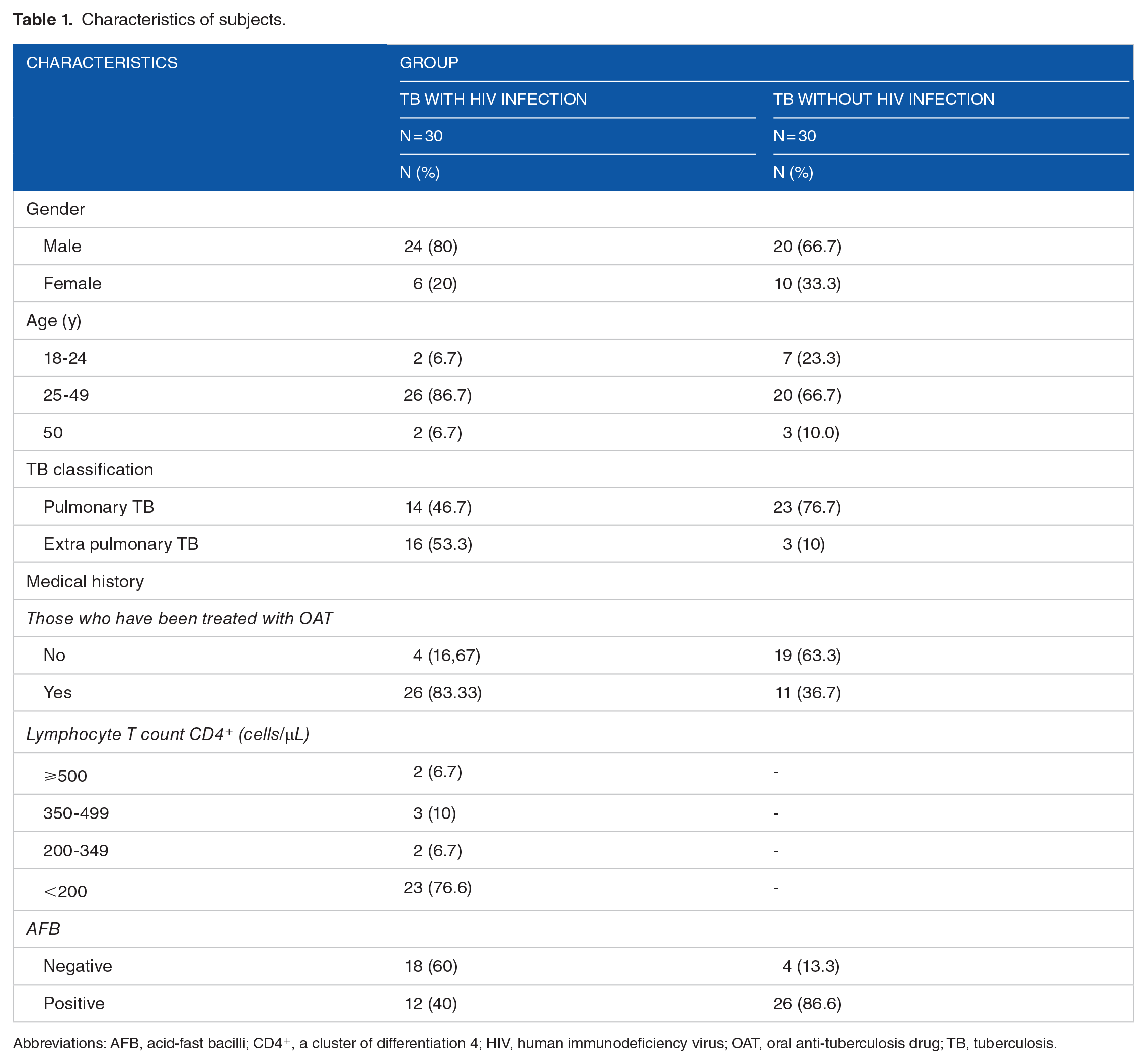
Abbreviations: AFB, acid-fast bacilli; CD4+, a cluster of differentiation 4; HIV, human immunodeficiency virus; OAT, oral anti-tuberculosis drug; TB, tuberculosis.
The urinary Mtb antigen cocktail was successfully detected in the TB-HIV and TB without HIV groups. A higher median urinary Mtb cocktail antigen concentration was found in the TB without HIV group compared to the other groups (P = .001; Figure 2).

Urinary Mtb antigens cocktail levels in patients with TB-HIV and TB without HIV.
A lower median urinary Mtb antigens cocktail concentrations were found in the TB-HIV group with extrapulmonary infection compared to the other groups (P = .004; Table 2).
Urinary Mtb antigens cocktail levels in patients with pulmonary and extra-pulmonary TB.

Abbreviations: HIV, human immunodeficiency virus; TB, tuberculosis.
Mann-Whitney test, statistically significant if P < .05.
In both groups, median urinary Mtb antigen cocktail levels between patients who did not receive TB therapy and those who received TB therapy had a significant difference (P < .05; Figure 3).

Urinary Mtb antigens cocktail levels in patients with TB-HIV and TB without HIV versus OAT therapy.
Discussion
This study shows that the Mtb antigen cocktail (ESAT-6, CFP-10, and MPT-64) can be found in the urine of TB patients with and without HIV. Numerous studies suggest that ESAT-6 and CFP-10 have been linked to the cell lysis of both macrophages and pneumocytes. They also suggested that ESAT-6 could induce pore formation on the macrophage and dendritic cell membranes, resulting in the spreading of intracellular bacteria independently of bacterial load.10,17
This finding contrasts with studies using the LAM antigen recommended by WHO, which can only be used in TB-HIV. 18 The differences in the results of this studies with LAM antigen may be due because some evidence in human studies and animal challenge models underscores the importance of T cells in controlling Mtb infection. T cells trigger cellular Immunity whereas humoral immunity depends on B cells. However, the role of B-cells and antibody depletion is another ongoing debate in TB. This is due to the lack of information regarding optimal antibody concentrations needed for protection and the heterogeneity of humoral immunity against TB. The humoral immune response to TB is heterogeneous, mainly because of differential antigen expression at different stages of infection. Therefore it is still doubtful whether these antibodies bind to ESAT6, CFP 10, and MPT 64 antigens in TB patients without HIV. Therefore, they are freely filtered by the glomerulus and then excreted in the urine in both TB patients with and without HIV.17,19
Microscopic investigation of AFB is still the most commonly used method for tuberculosis diagnosis. Although the microscopic investigation is the fastest diagnostic method, the performance of sensitivity is relatively low. 20 In our study, only 40% of the TB-HIV group had positive smear results, meanwhile, the urinary Mtb antigen cocktail test result in this group was 93.3%. Our previous study mentioned the added value of urinary Mtb antigen cocktail, we found the sensitivity of AFB staining was 45.4% to 90% when combined with urinary Mtb antigen cocktail assay. 21 In accordance with the WHO statement that TB patients with co-infection with HIV are often associated with smear-negative disease cases, the urinary Mtb antigens cocktail assay can be used to improve TB diagnosis in the TB-HIV group.
The median concentration levels of the urinary Mtb antigens cocktail in TB without HIV group was higher (137.73 ng/mL) than in TB-HIV group (96.69 ng/mL) (P = .001). Some aspects can contribute to the concentration levels of the urinary Mtb antigens cocktail. First, the TB without HIV group was dominated by pulmonary TB patients with positive smear results (86.6%). This higher concentration indicates, that in active pulmonary TB, ESAT-6, CFP-10, and MPT-64 antigens are secreted by Mtb replication and metabolism10,17 Second, the TB-HIV group was dominated by extrapulmonary TB patients with negative smear results up to 60% (Table 1). Pulmonary tuberculosis is the main predilection site for TB because Mtb is an obligate aerobic bacterium that prefers areas with higher oxygen levels. Consequently, they are more commonly found in the lungs, especially in the upper lobe. When the host immunity fails like in HIV patients, the mycobacteria will spread to areas other than the lungs.22,23 Extrapulmonary TB is usually paucibacillary form of TB, therefore found less antigens cocktail is detected in the urine (Table 2).10,24 Third, another suggestion regarding the significant difference in levels of the urinary Mtb antigens cocktail between active TB patients with and without HIV infection, it could be that the cooperation between T and B lymphocytes in patients with TB and HIV, could be affected by the decrease of Th2 cells. Unfortunately in this study, a decrease in the number/function of Th2 was not identified.
The urinary Mtb antigens cocktail test results were positive in 28 TB-HIV patients, 23 patients (76.6%) had CD4+ counts <200 cells/µL. This leads to a state of immunosuppression that makes the patient susceptible to various diseases including TB. CD4+ cells are involved in the main resistance against Mtb by producing IFN-ɤ and IL-12 to activate macrophages which play an important role in controlling and eliminating bacteria. They also strengthen the phagocytic potential of Mtb-infected macrophages by stimulating the formation of phagolysosomes. A decrease in the number of CD4+ will affect the body’s defense mechanism against Mtb due to the restriction of the immune system that works to fight TB infection, culminating in increased levels of Mtb bacteria in the body. 25 There were 2 patients who gave negative urinary Mtb antigens cocktail test results, they had CD4+ counts >500 cells/µL. The 2 patients were extrapulmonary TB patients (lymphadenitis TB) with negative smear results. In addition, these patients had received oral anti-tuberculosis therapy. From these explanations, it is possible that the number of bacteria in the 2 patients has decreased.10,23,26,27
In this study, it was found that TB-HIV patients who did not receive OAT therapy had a median of urinary Mtb antigens cocktail level was 147.73 ng/mL, in contrast to patients who received OAT therapy, which was 86.47 ng/mL (P = .01). It was also found that TB without HIV patients who did not receive OAT therapy had a median of urinary Mtb antigens cocktail level was 140.5 ng/mL, while patients who received TB therapy were 121.99 ng/mL (P = .028). This is similar to previous studies which found that OAT therapy can reduce TB antigen levels.10,18,26
The limitations of this study is the different levels of education of the subjects. This may influence the understanding on how to collect proper sputum and urine, although study staff had taught all subjects. Another limitation of our study is the difficulty of getting active TB subjects with and without HIV infection as well as those who have not received OAT therapy.
Conclusion
There was no significant difference in the positivity rate, meanwhile, there was a significant difference in concentration of the urinary Mtb antigens cocktail between active TB patients with and without HIV infection. Further investigation is needed on diagnostic tests for urinary Mtb antigens cocktail to obtain a cut-off point for establishing TB diagnosis and therapy. Interestingly, this urinary Mtb antigens cocktail can be found in both groups without being affected by the patient’s immune condition, thus becoming a test to assist diagnose active TB in TB-HIV and TB without HIV groups.
Footnotes
Acknowledgements
The authors would like to thank Rudi Wicaksana and Hijriyanti providing the clinical samples. We are extremely grateful to the patients who participated in the study.
Declaration Of Conflicting Interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dewi Kartika Turbawaty is an academic author, a clinical pathologist, and the Head of Clinical Pathology, staff of Microbiology and Biomolecular Department, Faculty of Medicine Padjadjaran University, Dr. Hasan Sadikin General Hospital. Novie Rahmawati Surdjaja is a clinical pathologist. Agnes Rengga Indrati is an academic author, a clinical pathologist, and staff of the Immunoserology Department, Faculty of Medicine Padjadjaran University, Dr. Hasan Sadikin General Hospital. Leni Lismayanti is an academic author, a clinical pathologist, and staff of the Hematology Department, Faculty of Medicine Padjadjaran University, Dr. Hasan Sadikin General Hospital. Verina Logito is an academic author, a clinical pathologist, staff of Immunoserology Department, Faculty of Medicine Padjadjaran University, Dr. Hasan Sadikin General Hospital. The remaining authors declare no relevant financial disclosures or conflicts of interest.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Universitas Padjadjaran through grant-in-aid for HIU for Dewi Kartika Turbawaty.
Author Contributions
Dewi Kartika Turbawaty, Novie Rahmawati Surdjaja, Agnes Rengga Indrati, Leni Lismayanti, and Verina Logito had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Verina Logito contributed to the study design, data analysis, interpretation, and writing the manuscript. Dewi Kartika Turbawaty, Novie Rahmawati Surdjaja, Agnes Rengga Indrati, Leni Lismayanti, and Verina Logito was the primary author and Dewi Kartika Turbawaty was a major contributor in writing the manuscript. All authors read and approved the final manuscript.
Ethics Approval and Consent to Participate
This study was approved by the Ethical Committee of the Dr. Hasan Sadikin General Hospital (LB.02.01/X.6.5/195/2021). Written informed consent was obtained from all participants.
