Abstract
Perivascular epithelioïd cell tumor (PEComa) is a mesenchymal neoplasm with epithelioïd or spindled morphology with numerous thin-walled capillaries between tumor cells. They co-express markers of both melanocytic and smooth muscle differentiation. PEComas are rare, presenting in numerous anatomic sites including lung, kidney, liver, genitourinary tract, soft tissue, and skin. Primary cutaneous PEComas are very rare entity, and malignant ones are even more uncommon. Herein, we report the case of a 92-year-old female which was presenting with 7 cm exophytic, ulcerated, hemorrhagic nodular tumor, and rapidly growing for 8 months over the right thigh. On histologic examination, we found a dermal neoplasm formed by an atypical clear cell tumor with numerous branching capillaries between tumor cells. The mitotic count was found 6 mitotic figures/10 HPF. On immunohistochemistry, tumor cells co-expressed smooth muscle and melanocytic markers, CD10, and CD68. Based on these findings, the diagnosis of primary cutaneous malignant perivascular epithelioïd cell tumor (PEComa) was made. The large size (7 cm), the count of mitoses (6 mitotic figures/10 HPF), and the nuclear pleomorphism argued for malignancy. The absence of soft tissue or visceral localization argued for the cutaneous primitive origin. Adjuvant radiotherapy and targeted therapy with mTOR inhibitor (nab-sirolimus) was indicated. To the best of our knowledge, this is only the eighth case of a primary cutaneous malignant PEComa reported in the literature to date.
Keywords
Introduction
Perivascular epithelioïd cell tumors (PEComas) are mesenchymal neoplasms composed of perivascular epithelioïd cells (PECs), distinctive epithelioïd cells that are often closely associated with blood vessels. These epithelioïd cells express both melanocytic and smooth muscle markers. 1 They are rare mesenchymal tumors, presenting in numerous anatomic sites including lung, kidney, liver, genitourinary tract, soft tissue, and skin. Primary cutaneous PEComas are very rare entity, and malignant ones are even more uncommon. This family of tumors includes renal angiomyolipoma, clear cell “sugar” cell tumor of the lung, lymphangioleiomyomatosis of the lung, clear cell myomelanocytic tumor of the falciform ligament, PEComas of somatic soft tissue, of visceral organ, and gynecologic tract. 2 In 2005, Folpe et al 3 proposed classifying PEComas as benign, uncertain malignant potential, or malignant, based on these criteria, tumor size ⩾5 cm, infiltrative growth pattern, high nuclear grade, necrosis, mitotic count up to 1/50 HPF, and aggressive clinical behavior. Therefore, A benign PEComa would have none of thfan aggressive skin malignancy.
Case Presentation
A 92-year-old female with no significant medical history, coming with a 7 cm exophytic, ulcerated, hemorrhagic nodular tumor rapidly growing for 8 months over the right thigh (Figure 1a and b). Magnetic resonance imaging showed budding cutaneous mass, extra-aponeurotic, located at the internal face of the right thigh, contrast enhancing intensely, and heterogeneously after injection of contrast product, it measured 7 cm × 5.5 cm × 5 cm. This was clinically suggestive of melanoma, squamous cell carcinoma, or sarcoma. A biopsy have been performed and sent to us. Histological examination after 10% buffered formalin fixation, impregnation, and block paraffin embedding, showed a dermal, and subcutaneous neoplasm with irregular nests and nodules, numerous branching capillaries were noted between tumor cells (Figure 2). Cells had abundant clear cytoplasm and enlarged polymorphic and vesicular nuclei with prominent nucleoli (Figure 3). The mitotic count was found 6 mitotic figures/10 HPF with atypical mitoses. There was no necrosis or vascular invasion. On immunohistochemistry, tumor cells co-expressed smooth muscle, and melanic markers, they are positive for Melan-A (Figure 4a), and smooth muscle actin (SMA; Figure 4b), they also expressed vimentin, CD10 (Figure 4c), and CD68. They were negative for epithelial markers (CK AE1/AE3, CK 7, CK20, EMA, P63), neuroendocrine markers (synaptophysin, chromogranin A), other muscle markers (Desmin, H-caldesmon), vascular markers (CD34, CD31), other melanin markers (PS100, HMB45), CD45, TFE3, and for myogenin. It should be noted that the immunostaining by CD34 highlighted the numerous branching capillaries between tumor cells (Figure 4d).

(a) Clinical feature: exophytic, ulcerated, hemorrhagic nodular tumor of 7 cm, over the right thigh (arrow). (b) Macroscopically, dermo-hypodermic tumor, multinodular, whitish with hemorrhagic areas (circle).

Tumor section, sheets of clear cells with numerous branching capillaries between tumor cells (hematoxylin and eosin stain ×100).
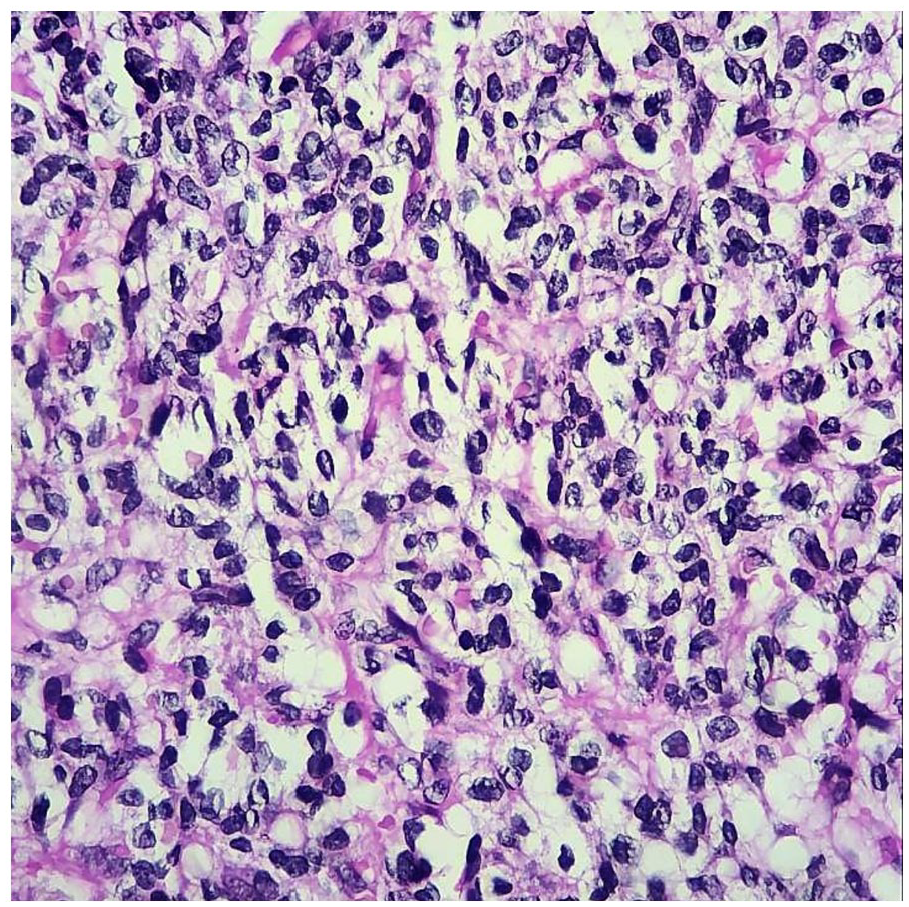
Higher magnification shows there are cellular atypia and atypical mitotic figures (hematoxylin and eosin stain ×400).
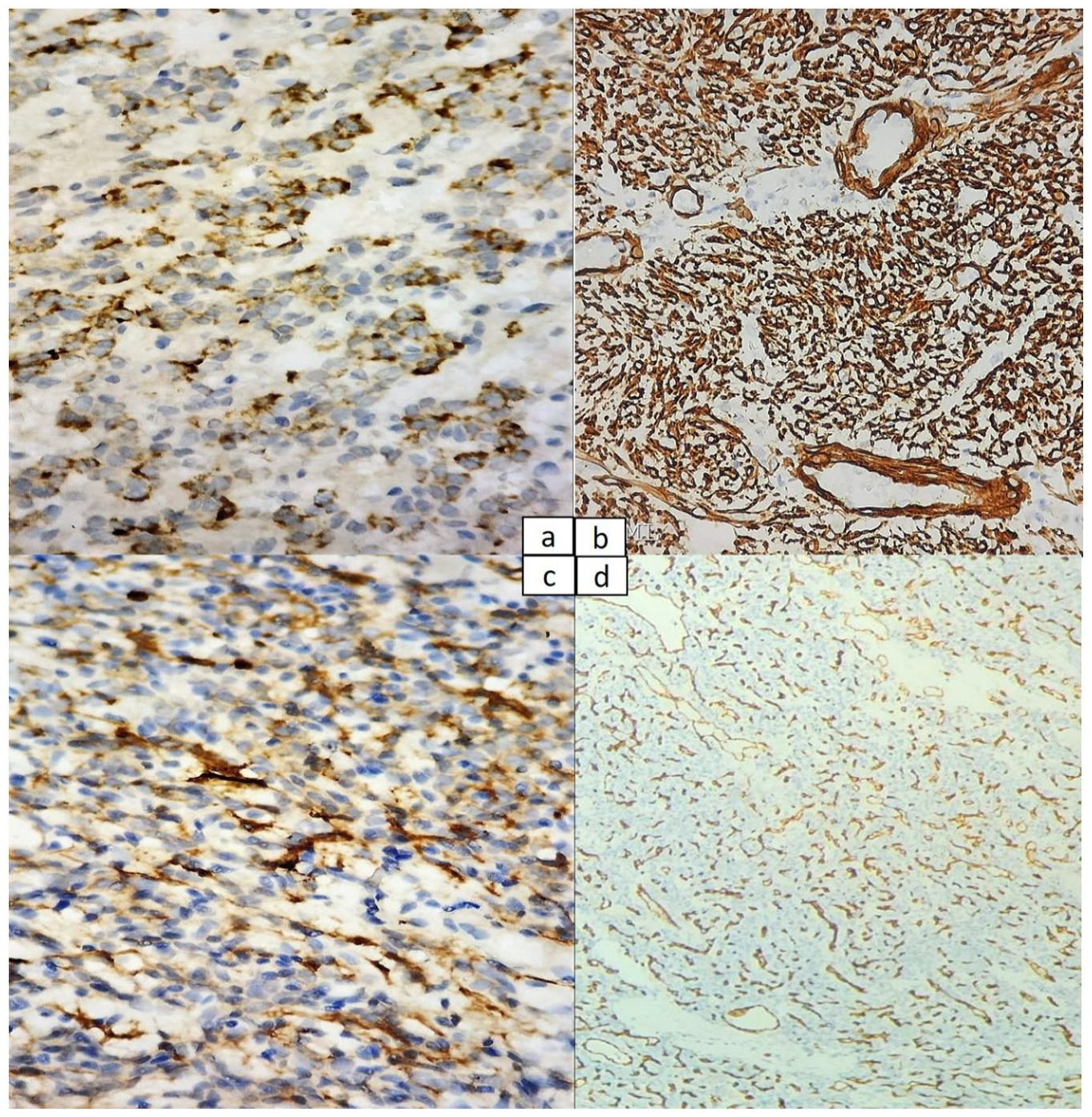
Immunohistochemical staining (a) cytoplasmic positive staining of Melan A (×400) (b) membranous and cytoplasmic positive staining of smooth muscle actin (SMA) (×400), (c) membranous positive staining of CD 10 (×400). (d) Positive staining of CD34 highlighting numerous thin-walled capillaries between tumor cells (×100).
Based on histological and immunohistochemical features, the diagnosis of primary cutaneous malignant perivascular epithelioïd cell tumor (PEComa) was made. The large size (7 cm), the count of mitoses (6 mitotic figures/10 HPF), and the nuclear pleomorphism argued for malignancy. The absence of soft tissue or visceral localization argued for the cutaneous primitive origin.
Discussion
To date and to the best of our knowledge, this is only the eighth case of a primary cutaneous malignant perivascular epithelioïd cell tumor (PEComa) reported in the literature. We have realized a literature review using PubMed/Medline, Scopus, and Web of Science databases, with the search terms “PEComa, malignant, immunohistochemistry, perivascular epithelioïd cell, primary.” On literature review, we found 68 cases of cutaneous PEComa including 59 benign cases, 7 malignant cases, and 2 cases of skin metastasis of primary uterine and adrenal gland PEComas. The first case was published by Calder in 2007, the second one was published in 2013 by Greveling as an abstract, then a third case was reported in 2017 by Haiges, a fourth case reported in 2021 by Cole and finally, 3 cases were published in 2022 separately by Cohen, Cornell, and Neumann. We summarized all clinical, histological, and immunohistochemical features of these cases including our current case in Tables 1 to 3.
Clinical features of reported primary cutaneous malignant PEComa cases.

Abbreviation: NA, not available; NED, no evidence of disease; SCC, squamous cell carcinoma.
Histopathological features of reported primary cutaneous malignant PEComa cases.
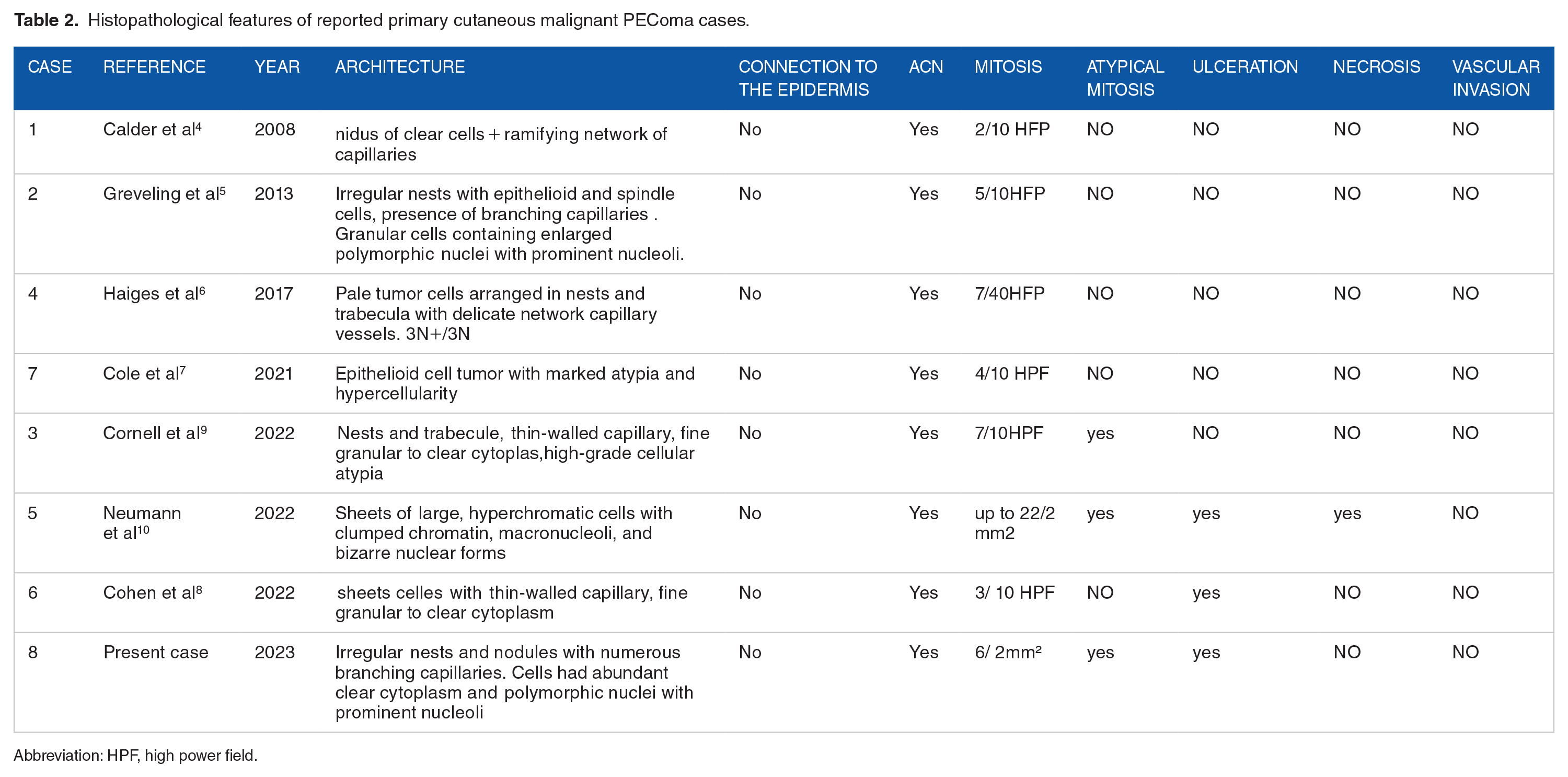
Abbreviation: HPF, high power field.
Immunohistochemical features of reported primary cutaneous malignant PEComa cases.
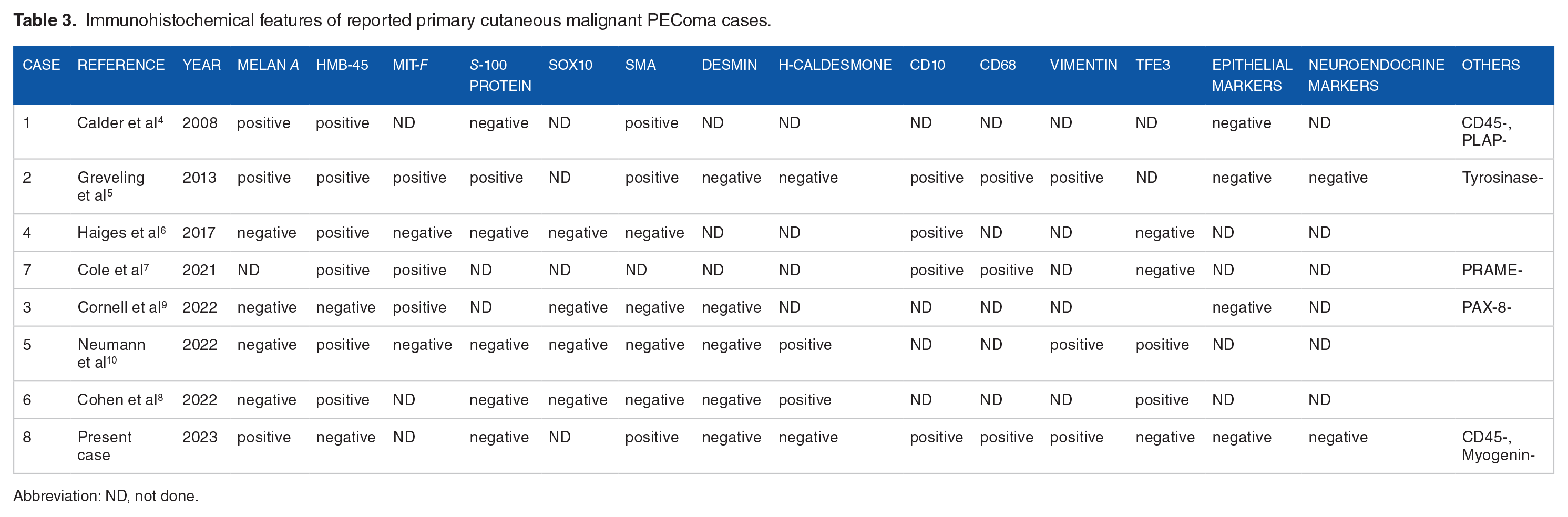
Abbreviation: ND, not done.
Through the reported cases of primary cutaneous malignant PEComas, we had not noted a predilection of sex with a sex ratio of 1 (4 women/4 men). The mean age of patients was 56 years. Clinical presentation was predominated by a growing, exophytic, and nodular tumor. Extremities were the site the most found on this review. The mean size was 40 mm (10-120 mm).
On histologic examination, all these reported tumors involved the dermis at least in part without connection to the epidermis, had infiltrating growth pattern and were characterizing by nodules, fascicles, and nests of epithelioïd or spindled cells. The cells had a clear or granular, eosinophilic cytoplasm. The nuclei were round or oval vesicular with a nucleolus. A ramifying network of capillaries between cells was noted. Mitotic count was ranging from 2 to up to 22 per 10 HPF with atypical mitoses in 3/8 cases. Necrosis was found in 1/8 case, ulceration found in 3/8 case, and no vascular invasion was noted.
Histologically, PEComa should be differentiated from any skin tumor with a clear cell change. This includes metastatic clear cell renal carcinoma, sebaceous carcinoma, clear-cell sarcoma, balloon-cell melanoma, balloon-cell nevi, clear cell hidradenoma, clear cell xanthoma, and the list is long. 11 For a precise diagnosis, a wide immunohistochemical analysis with a panel of multiple antibodies is required.
As previously reported, PEComas have characterized by co-expression of at least 1 melanocytic marker (HMB-45, Melan A, MiTF, tyrosinase) and 1 smooth muscle marker (SMA, Desmin, Hcaldesmone), while epithelial markers, neuroendocrine markers, SOX10 were never expressed. HMB-45 has seemed the most sensitive, it was positive in 6/8 cases (Table 3). On the other hand, it has noted that the smooth muscle markers had a lower rate of positivity in the primary cutaneous PEComas. Others non-specific markers which found positive in primary cutaneous PEComas were CD10 in 100% (4/4) tested cases), CD68 in 100% (3/3) tested cases) (Table 3). Usually, immunohistochemical CD10 positivity has been often allied to cutaneous metastases of clear cell renal carcinoma, however, 2 authors12,13 had reported 3 cases and 5 cases of CD10-positive cutaneous PEComa, respectively, as well as our case was also CD10 positive.
In a recent publication, 14 the authors had tested the usefulness of the anti-PRAME antibody in the diagnosis of tumors with melanocytic differentiation. They had found a positivity of PRAME in a single case of PEComa among the 9 cases tested, then they concluded in this work that immunostaining for PRAME may be useful to support diagnosis of melanoma in the setting of difficult dermal melanocytic neoplasms and other epithelioïd neoplasms with melanocytic differentiation as PEComa. 14
In contrast to systemic PEComa, TFE3 positivity had not observed in the 17 primary skin PEComas, reported by Llamas-Velasco et al., 15 and FISH assay for TFE3 rearrangement yielded negative results in this same study. However, 2 primary cutaneous malignant PEComas have previously reported, they showed TFE3 positivity (2/6) (Table 3).
Genetically, primary cutaneous malignant PEComas have a different molecular signature than their systemic counterparts. Loss of function in tuberous sclerosis complex 1 (TSC1) or 2 (TSC2) in systemic PEComas cause an activation of the mTOR pathway; however, primary cutaneous malignant PEComas are unrelated to tuberous sclerosis complex and show overactivation of the mTOR pathway by a different mechanism. So overexpression of 4EBP1, which is an effector protein in the mTOR pathway, supports mTOR overactivation independent of TSC1 or TSC2 mutations in primary cutaneous malignant PEComas. 16
Given the rarity of primary cutaneous PEComas, treatment options and prognostic factors are not well established. In all published cases of primary cutaneous PEComas, radical surgery with free margins was the gold standard treatment followed by radiotherapy in 2 patients.7,8 Some authors report that treatment with mTOR inhibitor (nab-sirolimus) seems to be useful in the treatment of cases with mTOR overactivation.
Follow-up data were available for 7 of 8 patients and ranged from 6 to 24 months with a mean of 10 months. All of these reported primary cutaneous malignant PEComas showed no evidence of metastatic disease and none of the patients presented recurrence after therapy (Table 1). Our patient has followed in oncology after the surgical excision and adjuvant thetapy, and no evidence of recurrence or metastasis have been revealed to date.
Conclusions
This case highlights an additional case of a primary cutaneous malignant PEComa, which clinically seemed an aggressive skin malignancy. On histologic analysis, we found a dermal clear cell epithelioïd neoplasm with marked atypia, hypercellularity, and high mitotic activity. On immunohistochemistry, tumor cells co-expressed muscle and melanocytic markers, they expressed also CD10. Therefore, cutaneous tumors that express CD10 should not only raise the possibility of metastasis from renal cell carcinoma but for a precise diagnosis, a wide immunohistochemical analysis with a panel of multiple antibodies is required. Diagnosing primary cutaneous PEComas is challenging given their rarity, on the 1 hand, pathologists have to bring to mind this diagnosis facing a dermal clear-cell tumor, and on the other hand, they should argue for malignancy based on the features described above.
Footnotes
Acknowledgements
The authors would like to thank all the technicians in our department for their enthusiasm in contributing to the management of the specimens.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Layla Tahiri Elousrouti and Laila Chbani did the histological and imunohistological diagnosis, conceived the study, participated in its design and coordination, and drafted the manuscript. N Hammas, A Mouadden, and I Fadlalalh participated in pathological process. S elhitmi, S Eloudi and FZ Mernissi did clinical examination and biopsy specimen. M Elidrissi resected chirugically the tumor. S Arifi and T Bouhafa indicated the systemic treatment and radiation.
Consent
Written informed consent has been obtained from the patient to publish this paper.
