Abstract
Objective:
Vaccination is one of the effective ways to fight against COVID-19 disease. Various vaccines have been designed during the coronavirus pandemic. Each of the used vaccines has beneficial effects as well as side effects. Healthcare workers were among the first vaccinated persons against COVID-19 in different countries. The current study aims to compare the side effects of AstraZeneca, Sinopharm, Bharat, and Sputnik V on healthcare workers in Iran.
Methods:
This descriptive study was conducted from July 2021 to January 2022 on 1639 healthcare workers who received the COVID-19 vaccines. Data were collected using a checklist that contained questions related to systemic, local, and severe side effects of the vaccine. The collected data were analyzed using the Kruskal-Wallis, Chi-square, and trend chi-square. P < .05 was regarded as a significant statistical difference.
Results:
The most commonly injected vaccines were Sinopharm (41.80%), Sputnik V (36.65%), AstraZeneca (17.75%), and Bharat (3.80%), respectively. At least 37.5% of participants reported one complication. The most common side effects after 72 hours of the first and second doses were as follows: injection site pain, fatigue, fever, myalgia, headache, and chill. Overall complication rates were reported as follows: AstraZeneca (91.4%), Sputnik V (65.9%), Sinopharm (56.8%), and Bharat (98.4%). Bharat showed the highest overall side effects, while Sinopharm had the lowest overall side effects. Also, our results indicated that individuals with a previous history of positive COVID-19 infection had a higher rate of overall complications.
Conclusions:
The majority of participants did not show life-threatening side effects after the injection of 1 of the 4 studied vaccines. Since it was well accepted and tolerable by the participants, it can be used widely and safely against SARS-CoV-2.
Introduction
The coronavirus (COVID-19) is a deadly disease that has plagued the world as a global pandemic. 1 The disease spread has been more prevalent in Europe than in Africa, West Pacific, and Southeast Asia. 2 In Iran, the spread of COVID-19 was high, so after 15 days, all the provinces were involved. 3 Different studies on this infectious disease have shown that coronavirus can damage multiple organs of individuals. 4 Due to the intracellular state of viruses and their dependence on eukaryotic cells, many drugs failed to be effective enough to confront or control viruses. As a result, the design of effective vaccines is one strategy for controlling viral diseases. 5 The most launched drugs proved unstable, inducing side effects and demanding the development of a vaccine crystallized in a short time. 6 World Health Organization (WHO) has recommended public vaccination against the SARS-CoV-2 coronavirus to ensure immunization against the COVID-19 pandemic mainly because SARS-CoV-2 is highly contagious and infects people. 7 Among the most popular candidate vaccines that prevent COVID-19 spread are SARS-CoV2 component antigens, genetic materials, or inactivated SARS-CoV-2 virus. Efforts are still being made to develop effective vaccines against SARS-CoV-2, including almost 200 vaccines in various stages of development and 30 in clinical trials. 1 AstraZeneca, Sinopharm, Bharat, and Sputnik are the most common vaccines used for immunization against COVID-19 in Iran.
Sputnik V is a heterologous COVID-19 vaccine injected in 2 doses separated by at least 21 days. This vaccine consists of 2 immunogenic components including the vectors of recombinant adenovirus serotype 26 (rAd26) and adenovirus type 5 (rAd5). 8 Researchers have concluded that this vaccine can be a safe and efficacious solution to the prevention of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). The AstraZeneca vaccine was made available to the public after receiving official approval from WHO. Characterized by an acceptable safety profile, ChAdOx1 nCoV-19 can stimulate the immune system by making the body produce its protection (antibodies) against the virus. CoronaVac (Sinovac Life Sciences, Beijing, China) is an inactivated vaccine candidate for preventing COVID-19. According to the implementation results, it can provide partial or complete protection against severe interstitial pneumonia after the SARS-CoV-2 challenge without observable antibody-dependent infection enhancement that prevents its progression in human beings to clinical trials. In this regard, intramuscular injection of the Sinovac-CoronaVac vaccine in 2 doses (0.5 ml) is highly recommended. 9 Covaxin known as BBV152 is another COVID-19 vaccine that was developed by Bharat Biotech, an Indian biotechnology company and Indian Council of Medical Research. It is a type of whole-virus vaccine called an inactivated vaccine with 78% efficacy, containing a modified or dead version of the SARS-CoV-2 virus. 10 The possible side effects after vaccination are common. Indeed, the side effects indicate that the vaccine teaches the body’s immune system against a foreign antigen.
Objectives
Considering the lack of accurate information on the side effects of COVID-19 vaccines and highlighting the necessity of immunization coverage, the current study puts its main focus on the side effects of 4 vaccine candidates, namely Sputnik V, Sinovac, AstraZeneca, and Covaxin, which are the most common vaccines used against COVID-19 in Iran.
Methods
Study setting
The present research is a descriptive-analytic study conducted from July 2021 to January 2022 on 1639 participants with the objective of comparing the side effects of the most commonly used COVID-19 vaccines against SARS-CoV2 in the workers of hospitals and healthcare centers in Shoushtar, a city located in the southwest of Iran. Followed by clarifying the objectives of the study to the eligible cases, the written informed consent forms were gathered from the participants who volunteered for this study. Data collection was done based on a checklist of vaccine side effects such as local reactions, systemic reactions, and serious side effects in the first and second doses. The content validity checklist was approved by 10 faculty members in Public health. This checklist was finalized after going through 2 stages by applying experts’ opinions. The checklist was pilot tested for 10 participants to assess the reliability of the checklist items by internal consistency (Alpha Cronbach = .822). The whole process of the present study was carried out under the requirements of the Helsinki Declaration. Institutional review board approval was received from the Shoushtar Faculty of Medical Sciences (Ref. No.: IR.SHOUSHTAR.REC.1400.003).
Statistical analysis
Descriptive statistics of both qualitative and quantitative variables were shown as the frequency (percentage) and mean ± Standard Deviation (SD), respectively. The Kruskal-Wallis test was also carried out to assess the association between the quantitative characteristics of subjects and vaccine types. Moreover, Chi-square and trend chi-square tests were done to evaluate the association between the qualitative variables and vaccine types. The normality of data was evaluated using the Shapiro-Wilk test. In this study, P < .05 was regarded as a significant statistical difference. Further, data analysis was done using the SPSS software.
Results
A total of 1639 participants over 19 to 69 years old with a mean age of 34.69 ± 9.73 cooperated in this study. Among them, 1025 (62.53%) and 614 (37.47%) were female and male, respectively. The demographic and some information related to vaccination are illustrated in Table 1.
Some demographic and information related to vaccination from the participants.

The most commonly injected vaccines were Sinopharm (41.80%), Sputnik V (36.65%), AstraZeneca (17.75%), and Bharat (3.80%), respectively. Overall, 37.5% of the participants reported at least one complication, with an average onset of complications of less than 5 hours and persistence of fewer than 24 hours. The most common side effects after 72 hours of the first and second doses were as follows (Table 2): injection site pain, fatigue, fever, myalgia, headache, and chill.
Prevalence of side effects according to the type of vaccine.
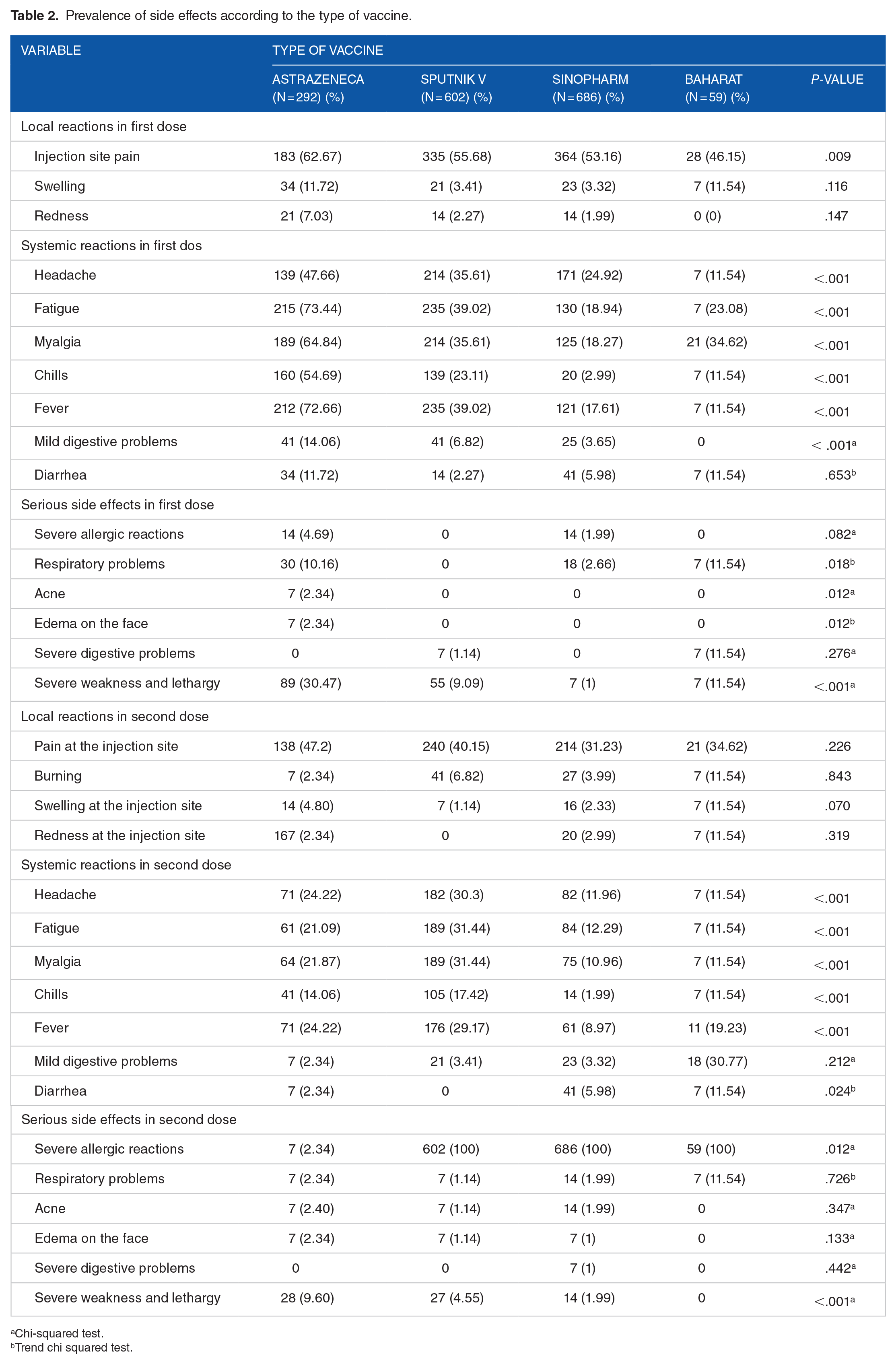
Chi-squared test.
Trend chi squared test.
The most complications in the first dose of vaccine injection were related to Bharat (95%), AstraZeneca (92.7%), Sputnik (68.5%), and Sinopharm (58.9%) (Table 3). Moreover, the most side effects in the second dose of vaccine injection were related to Bharat (97.4%), AstraZeneca (86.2%), Sputnik (72.5%), and Sinopharm (54.2%) (Table 3).
The prevalence of side effects of each vaccine in the first, second, and both doses.

Overall complication rates were reported as follows: AstraZeneca (91.4%), Sputnik (65.9%), Sinopharm (56.8%), and Bharat (98.4%). Bharat showed the highest side effects, and Sinopharm illustrated the lowest side effects (Figure 1).

Frequency of complications of vaccine injections, grouped by type of vaccine.
Also, we evaluated the relationship between the incidence of side effects caused by vaccination and previous infection with COVID-19. As shown in Table 4, our results indicated that individuals with a previous history of positive COVID-19 infection had a higher rate of overall complications than participants with a previous history of negative COVID-19 infection.
Prevalence of total side effects based on previous history of positive and negative infection with COVID-19.

Chi-squared test.
Discussion
Different vaccines have been introduced to the world to take control over the COVID-19 disease pandemic. Among the most common vaccines used against COVID-19 in Iran are AstraZeneca, Sinopharm, Bharat, and Sputnik. The present research aimed to assess the side effects of these vaccines in 1639 healthcare workers who received both doses.
The AstraZeneca vaccine was developed by an Indian Serum Institute and was made available to the public after receiving official approval from WHO. The most common side effects caused by this vaccine were injection site pain, Chills/ fever, fatigue, headache, and even digestive and allergic side effects with limited prevalence. In line with our study, an observational study in the United Kingdom reported local reactions, headaches, and fatigue. 11 In our study, 62% of individuals who had received their first dose reported pain in the injection site, 73% fever, 74% fatigue, as the most common complications, 65% myalgia, and 48% headache. Of note, the obtained results were in line with other studies.12 -14 In different studies, similar complications have been reported for the AstraZeneca vaccine; however, the frequency of these side effects was considerably different. To justify this difference, the author would like to refer to the differences in the types of study design, evaluation method, and target population. Some studies expressed concerns over the occurrence of some unpleasant side effects such as thrombosis after the injection of this vaccine. 15 Considering the mentioned concern, the current research reported no cases with such complications. This finding is in agreement with those reported by Babaee et al. 16 However, Houshmand et al reported cases with mild thrombosis and one with moderate thrombus after receiving AstraZeneca vaccine. 17 This difference can be caused by the difference in the sample size (292 vs 578) and target population (health workers vs dental workers and students).
Sinopharm, the most commonly used vaccine in Iran, showed that it produces effective antibodies against coronavirus with limited side effects in the host after a clinical trial study. The most important self-limiting side effects were fever and mild pain at the injection site. 18 In our study, the most commonly observed side effects of the mentioned vaccine were injection site pain, headache, fatigue, myalgias, and fever. These findings are consistent with those from several studies conducted in some other countries such as Iraq, Jordan, Turkey, and the United Arab Emirates.19 -21 Among the vaccines evaluated in this study, Sinopharm exhibited the least side effects among other vaccines with 56% incidence of the overall complications. As evident from the results obtained from most studies as well as clinical trial phases, Sinopharm, among the currently available vaccines, has the least side effects.20,22 In addition, the findings, which were in agreement with the reports of the Centers for Disease Control and Prevention )CDC( and other studies, indicated that the incidence of general complications (59% vs 54%, respectively) at the first dose was lower than those at the second dose.16,23 In line with a study conducted in the UAE, some rare side effects caused by Sinopharm vaccine such as allergic reactions were observed in 1.9% of the recipients (14 cases) of the first dose; however, such complications caused by the second dose injection were not reported. 24
Sputnik V, as the first registered vaccine in the world against COVID-19, was reported to be 91.6% effective. 25 Irrespective of the vaccine type, occurrence of the side effects caused by the host body reactions, can be expected, and a majority of them do not seem to be unpleasant and frightening. 26 Based on the CDC reports that were consistent with our findings, the most common complications of Sputnik V were fatigue, headache, and injection-site pain. 25 Studies done by Pagotto et al in Argentina and Zare and Babaee et al in Iran evaluated the side effects of the mentioned vaccine.16,27,28 Our study proved that Sputnik V with 65.9% of all complications after Sinopharm represented the second vaccine that enjoyed the least side effects. Our result is in close agreement with the findings of the phase-III trial for 64.7% of general complications. Moreover, these findings are consistent with those presented by Pagotto. However, studies of Babaee and Zare reported the rates of 82.7% and 81.9%, respectively, being higher than the rates found in the current study. The difference between the obtained rates results from various factors including study design, age of participants, pain threshold based on genetics or race, physical and psychological conditions of the person when reporting the results, and sample size. 29 For example, most of the participants were of Bakhtiari ethnicity, followed by Shushtri in our study.
COVAXIN® (BBV152) is another inactivated vaccine developed by Bharat Biotech at the National Institute of Virology in India. Few studies have evaluated the complications caused by this vaccine and consequently, few findings are available. In our study, the overall percentage of complications caused by this vaccine was 98.4%, which is in close agreement with the findings of another study conducted by Houshmand et al. 17 Similar to the results of the phase-II clinical trial, the most common side effects of pain at the injection site included headache, fatigue, and fever, which were reported in our findings. In addition, such complications as myalgia (35%), skin redness (31%), and digestive problems (30%) were evident in our study. Despite the high incidence rate of general side effects caused by BBV152, fortunately, serious and life-threatening side effects were not evident as reported in other studies.30,31
In general, our findings indicate that the side effects of evaluated vaccines are moderate to mild and transient. Fortunately, no unpleasant and life-threatening side effects were observed. Therefore, with the arrival of a new wave of corona disease in many different countries, it is recommended that new and more efficient vaccines be produced and injected as one of the ways to fight against COVID-19.
One of the strengths of this study is that the diagnosed and evaluated people were healthcare workers and they had enough knowledge or expertise to report any complications. Moreover, our study covered the simultaneous evaluation of the side effects of 4 vaccines. This study is subject to a number of limitations. For instance, throughout the several waves of COVID-19 pandemic in Iran, the participants in our study received a third dose of vaccines and we could not evaluate the corresponding side effects in the case of these people. In addition, we failed to take into account the long-term complications resulting from the vaccines received in this study. Finally, despite the simultaneous assessment of the 4 vaccines, the sample size in our study was relatively small.
Conclusions
This comprehensive study aimed to evaluate the side effects of the coronavirus vaccines in the case of workers in hospitals and health centers. From the obtained results, it can be concluded that regardless of the age and gender of the participants, most of them did not exhibit significant side effects after the injection of 1 of the 4 vaccines, that is, Sputnik V, Sinovac, AstraZeneca, and Covaxin. Besides, the effects of complications did not last more than 24 hours, on average. Since the vaccines were well accepted and tolerable by the participants, they can be used widely and safely against SARS-CoV-2, although their long-term side effects should be investigated in future studies.
Supplemental Material
sj-docx-1-pat-10.1177_2632010X231166798 – Supplemental material for Side Effects Comparison of Coronavirus Vaccines Among Healthcare Workers in Shoushtar, Iran
Supplemental material, sj-docx-1-pat-10.1177_2632010X231166798 for Side Effects Comparison of Coronavirus Vaccines Among Healthcare Workers in Shoushtar, Iran by Edris Nabizadeh, Fatemeh Honarmandpour, Rezvan Mashhadizade and Azam Honarmandpour in Clinical Pathology
Footnotes
Acknowledgements
We thank all the participants who volunteered for this study; this study is derived from research project Shoushtar Faculty of Medical Sciences in the southwest of Iran (Shoushtar). All expenses of this study were provided by Shoushtar Faculty of Medical Sciences.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported financially by Shoushtar Faculty of Medical Sciences, Shoushtar, Iran.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ Contribution
A.H and E.N, Conceptualization; Data curation; Formal analysis; A.H, Funding acquisition; Supervision Investigation; E.N, Methodology; Project administration; Resources; Supervision; Writing—original draft; Writing—review & editing. A.H, E.N, F.H, and R.M, Formal analysis;
Availability of Data and Materials
All data included in this study are available upon request by contact with the corresponding author.
Consent for Publication
Not applicable.
Ethics Approval and Consent to Participate
The whole process of the present study was carried out under the requirements of the Helsinki Declaration. Institutional review board approval was received from the Shoushtar Faculty of Medical Sciences (Ref. No.: IR.SHOUSHTAR.REC.1400.003). Followed by clarifying the objectives of the study to the eligible cases, the written informed consent forms were gathered from the participants who volunteered for this study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
