Abstract
Scientists identified SARS-CoV-2 in December 2019 in Wuhan city of China. Soon after its identification, Covid-19 spreads almost everywhere. The World Health Organization (WHO) declared the Covid-19 outbreak as a pandemic on March 11, 2020. Countries are facing multiple waves due to the different variants of the coronavirus. Personal preventive measures, vaccines, and antiviral drugs are the approaches to control Covid-19. However, these approaches are being implemented in different countries at different levels because of the availability of personal protective measures and antiviral agents. The objective of this study was to evaluate the effectiveness of practicing measures to fight the Covid-19 pandemic. Here we searched relevant literature from PubMed and Scopus using the keywords such as personal protective measures, antiviral agents, and vaccine effectiveness. According to the present findings, protective measures were found comparatively less effective. Nevertheless, these measures can be used to limit the spreading of Covid-19. Antiviral agents can reduce the hospitalization rate and are more effective than personal protective measures. The most effective strategy against Covid-19 is early vaccination or multiple vaccination dose. The respective authorities should ensure equal distribution of vaccines, free availability of antiviral drugs, and personal protective measure in poor and developing countries. We recommend more studies to describe the effectiveness of practicing preventive measures and antiviral agents against recent variants of the coronavirus.
Keywords
Introduction
The world has faced a public health threat with the rise of the coronavirus pandemic. 1 So far, the world recorded more than 649 million confirmed cases with more than 6.6 million deaths. 2 Signs and symptoms are fever, taste or smell loss, cough, headache, fatigue, difficulty in breathing, and muscle aches are the most prevalent in the case of Covid-19. A coronavirus is an encapsulated virus with a positive RNA genome. 3 We see coronavirus is continuously mutating to produce its variants. The first variant of coronavirus was Alpha found in the UK in September 2020. The Beta variant was first to come into view in May 2020 in South Africa. B.1.351 (20H/501Y.V2) was renamed on December 18, 2020, as a Beta variant by the World Health Organization (WHO) and registered as a variant of concern (VOC). The Gamma variant was detected in November 2020 in Brazil. The WHO entitled the Gama variant on January 11, 2021. It is also recognized as lineage P.1. 4 The Delta variant was first found in India in October 2020. It was considered a VOC (a variant of concern) on May 11, 2011, and was also specified as a variant of interest (VOI) on April 4, 2021, by WHO. It is also known as lineage B.1.617.2. The Lambda variant was first recognized in December 2020 in Peru. The WHO designated the Lambda variant on June 14, 2021. It is also noted as lineage C.37. Mu variant was first found in January 2021 in Colombia. It was entitled as a VOI by the WHO on August 30, 2021. It is also identified as lineage B.1.621. 5 The Omicron strain was first detected in South Africa in November 2021. It was recognized as a VOC on November 26, 2021. It is often referred to as lineage B.1.1.529. 6 A wide range of strategies, including vaccines, anti-viral agents, and health safety measures (quarantine, proper masking, proper coughing, hand sanitizing, hand washing, social distancing, etc.) are being followed by the healthcare authorities to fight the Covid-19 outbreak. Vaccines are now available to come in different forms, for example, AstraZeneca- AZD1222 (Viral vector based), Moderna- mRNA-1273 (RNA based), Pfizer- BNT16b2 (RNA based), Johnson & Johnson- Ad26.COV2.S (Viral vector-based), Sinovac- CoronaVac (Inactivated virus-based), Novav-ax- NVX-CoV2373 (Protein based), Sinopharm- BBIBP-COrV (Inactivated virus-based), and Bharat Biotech-BBVV152 (Inactivated virus-based) to prevent Covid-19.1. 7 As SARS-CoV-2 changes over time, it generates several strains that the WHO has categorized as Alpha, Beta, Gamma, Delta, Mu, and the most recent Omicron variant. 8 The vaccines showed satisfactory efficacy against earlier strains, but the ability has declined against the newer variants like Omicron. 9
Antiviral drugs nirmatrevir-ritonavir,molnupiravir, and remdesivir have obtained FDA approval to treat SARS-CoV-2 infection. Remdesivir is employed to suppress the viral RNA-dependent RNA polymerase (RdRp). 10 Both the RdRp and the primary protease can mutate SARS-CoV-2, making them antiviral drug targets. A protease inhibitor called nirmatrevir-ritonavir can prevent the virus from replicating by blocking the major protease (3CL protease) activity. 11 The main protease of SARS-CoV-2 is considered a drug target because it is different from human proteases. 12 Non-pharmacological interventions (NPIs) can lower the spreading rate of coronavirus. They are quality mask use, physical distancing, teleworking, proper coughing, handwashing, and isolation. 7 Depending on how well the mask fits, procedural and surgical mask-wearing can decrease the risk of SARS-CoV-2 infection. 13
Achieving zero Covid (elimination technique) is not the ultimate goal for living a normal life with Covid-19. It needs to accept that novel variants may cause new waves of the ongoing pandemic. Thus, it is required to modify vaccines/medications to fight against the disease. There is no evidence that the vaccine will provide lifetime protection. 14 It would be ideal if be alert and take precautions to defend against coronavirus strains. We searched related articles in Scopus and PubMed. We read relevant articles and extracted the required information from them. This study aims to evaluate the effectiveness of personal protective measures and antiviral agents against the Covid-19 pandemic.
Performance of Personal Protective Measures Against Coronavirus Spread
The WHO urged people to take precautions (such as keeping physical distance, washing hands, using well-fitting masks, leaving crowded areas, getting immunized, etc.) to lower their chance of getting Covid-19 due to different variants. Implementation of the Covid-19 preventive and control recommendations is necessary to combat SARS-CoV-2. Delay in treating the Omicron infection might create waves of different strains, hopefully, with reduced severity but the risk of new mutations.15,16 Based on the quality and fitting of masks, procedural and surgical masks limit virus transmission by 40.0% to 60.0%. However, facemasks use can reduce the viral transmission risk but not provide complete protection against Covid-19.13,17,18 A recent investigation reported that wearing masks has been shown to stop the transmission of Covid-19 and surgical masks are much more effective than fabric ones. The study demonstrates that the preventive effects of facemasks may vary based on the density of the population and the quality of personal protective equipment. 19 A randomized study reported that the prevalence ratio (PR) for Covid-19 decreased by 11.0% with the appropriate use of surgical masks. However, the study observed the highest reduction (35.0%) of PR for Covid-19 for those above 60 years. 20 Researchers recommend a regular change of facemasks for improved effectiveness. 21 A study found that wearing a surgical mask can decrease the probability of Covid-19 disease by 56%. 22 A meta-analysis reported that N95 or similar masks have an effectiveness of up to 70.0% because of the risk of SARS-CoV-2 disease in comparison to approximately 30.0% for surgical or medical masks. 23 N95 breathing masks reduce exposures to aerosol or similar particles by 98.5%, providing protection that is significantly superior to surgical masks. 18 An advantage of N95 breathing masks is to protect people from the nosocomial transmission. 17 A recent study demonstrated that individuals with the Omicron variants have 4 times higher viral load (nasopharyngeal) than the other variants. 24 A study has been conducted on SARS-CoV-2 patients and reported that disinfectant reduced the incidence of Covid-19 by about 77.0%. 25 A bivariate analysis found that the risk ratio of 3.53, 2.44, and 2.63 for acquiring Covid for not washing hands, proper coughing practice, and maintaining social distancing, respectively. 26 It is reported that physical distancing and staying at home can decrease the risk rate of Covid-19 by 12.0% and 51.0%, respectively.27,28 Personal protective measures help to limit viral infections in our bodies.
Therapeutic Effectiveness of Antiviral Drugs Against Covid-19
Remdesivir
Remdesivir is a monophosphoramidate prodrug of the nucleoside GS-441524. It was first approved on October 22, 2020, by Food and Drug Administration (FDA). 29 Remdesivir is available as a single dose of 100 mg vial (5 mg/mL after reconstitution). Usually, the course of treatment takes 3 days. A single 200 mg loading dose for the first day and 100 mg were given once daily for the second and third days. 30 It is used to prevent the RNA-dependent RNA polymerase of SARS-CoV-2. It was the first authorized antiviral for emergency use to treat Covid-19 in various countries. 10 Several variants of SARS-CoV-2 (Alpha, Beta, Gamma, Delta, and Omicron) are susceptible to remdesivir. 31 Both adult and pediatric mild to moderately ill Covid-19 patients can be treated with remdesivir to decrease the risk of getting severe infection and hospitalization or even death. 32
PINETREE clinical trial found that remdesivir reduces the hospitalization or mortality rate by 87% associated with Covid-19. 33 The findings of the WHO-sponsored solidarity studies played a vital role in the conditional recommendation for using remdesivir in severe Covid-19. It has shown a significant reduction (17.0%) in relative risk of dying or progressing to require ventilation in individuals using supplementary oxygen at baseline, in comparison to the standard for care (relative risk (RR): 0.83; 95%). Additionally, according to solidarity, remdesivir therapy had a significant reduction rate (13.0%) of mortality risk for patients admitted to the hospital on supplemental oxygen and without mechanical ventilation than standard care (RR: 0.87; 95%). According to the same study, Covid-19 patients with ventilation support do not get enough benefits from remdesivir therapy. Furthermore, in a post hoc subgroup analysis, remdesivir-treated patients who received lower flow oxygen at baseline had a decreased mortality compared to placebo (HR: 0.30; 95%). 34
Nirmatrelvir and ritonavir combination
It is effective to combine nirmatrelvir and ritonavir in treating infections due to the Omicron variant. 35 Emergency Use Authorization (EUA) of PAXLOVID is issued by the FDA on December 22, 2021, for the treatment of Covid-19. 36 This combination is available in the market as a trading name of Paxlovid. Nirmatrelvir is a primary protease inhibitor of SARS-CoV-2. Ritonavir is a protease inhibitor of HIV-1 and an inhibitor of CYP3A. Ritonavir has not demonstrated any efficacy against SARS-CoV-2. However, it can stop nirmatrelvir from being metabolized by CYP3A, increasing its plasma concentrations to prevent SARS-CoV-2 replication. 37 The medicines are combined and given twice daily for 5 days. Ritonavir 100 mg one tablet and nirmatrelvir 150 mg 2 tablets are administered together. 38 Clinical data support that nirmatrivir-ritonavir decreases Covid-19 related hospitalization or mortality by 89.0% relative to placebo in individuals who have risk factors for disease development and are unvaccinated. 39 According to a study, a 5-day therapy of nirmatrivir-ritonavir is effective and reduces hospitalization and mortality among pediatric Covid-19 patients. 40 Geriatric Covid-19 patients are at higher risk of developing severe respiratory symptoms. Moreover, a 5-day course of Paxlovid helps to reduce the disease severity and hospitalization among the older population in China. 41 However, some side effects can be happened after taking Paxlovid including, allergic reactions, liver problems, resistance to HIV medicines, high blood pressure, etc. 38 There is a chance of reducing nirmatrelvir or ritonavir plasma concentration if Paxlovid co-administer with potent CYP3A inducers. 7
Molnupiravir
Molnupiravir is another oral antiviral drug for treating Covid-19. 42 It helps to prevent the SARS-CoV-2 replication and gives activity against different variants (Alpha, Beta, Gamma, Delta, and Omicron) of SARS-COV-2.43 On November 4, 2021, molnupiravir received its initial approval for treating mild to moderate Covid-19 patients. 42 Usually, 800 mg is taken every 12 hours for 5 days. 43 A study found that molnupiravir reduces the hospitalization or mortality rate by 50.0% associated with Covid-19. 44 Patients from Shenzhen’s Third People’s Hospital participated in an experiment. This study shows that molnupiravir increases viral RNA clearance in Covid-19 patients due to the Omicron variant (Molnupiravir group: 76.3% against control group: 51.6%). 45
Monoclonal antibody
Monoclonal antibodies have been considered for the treatment since the beginning phases of Covid-19. There are 4 types of monoclonal antibodies- bamlanivimab (DB15718) plus etesevimab (DB15897), casirivimab (DB15941) plus imdevimab (DB15940), sotrovimab (DB16355) and tixagevimab (DB16394) plus cilgavimab (DB16393). 46 It is usually used to treat non-hospitalized individuals who are at high hospitalization risk. Casirivimab plus imdevimab or bamlanivimab plus etesevimab are recommended for non-Omicron VOCs. A longer duration of action is achieved by sotrovimab. It also has therapeutic activity against a wide range of VOCs. Because of the decreased susceptibility to omicron, the FDA postponed the use of bamlanivimab plus etesevimab and casirivimab plus imdevimab. 10
Casirivimab plus imdevimab
Imdevimab and casirivimab are non-competitive monoclonal antibodies. It can interact with the receptor binding domain (RBD) of the coronavirus spike-glycoprotein and prevent viral entrance into host cells. A combination of casirivimab and imdevimab is used in Covid-19 hospitalized patients. 47 Patients with mild to severe Covid-19 who are at high hospitalization risk, a single intravenous dose (600 mg of casirivimab plus 600 mg of imdevimab) was introduced. If IV infusion is not possible, the USFDA has approved casirivimab plus imdevimab in the amount of 2.5 mL for each injection subcutaneously. 10
Bamlanivimab plus etesevimab
Bamlanivimab and etesevimab bind to the distinct area but overlap in the RBD of spike-protein (SARS-CoV-2). It works by blocking its binding affinity to the human Angiotensin-converting-enzyme 2 receptor. In clinical trials, 700 mg of bamlanivimab with 1400 mg of etesevimab and 2800 mg of bamlanivimab plus 2800 mg of etesevimab were given to non-hospitalized individuals with mild to moderate Covid-19. 48
Tixagevimab plus cilgavimab
Tixagevimab and cilgavimab bind to the distinct area in nonoverlapping epitopes of the spike protein RBD of SARS-CoV-2. The initial dosage in adults and pediatric patients (⩾12 years weighing at least 40 kg) is tixagevimab 300 mg and cilgavimab 300 mg provided as 2 distinct repeated injections (IM). 49
Sotrovimab (DB16355)
Sotrovimab binds to the RBD epitope of the SARS-CoV-2 spike protein, suppressing viremia and triggering clearance of infected cells. Sotrovimab is recommended to be delivered as an intravenous (IV) infusion at a dose of 500 mg; the solution must be adjusted with 0.9% 5% dextrose or sodium chloride injection before use. 10
Preventive Performance of Covid-19 Vaccines Against SARS-CoV-2 Variants
Concern over the efficacy of the vaccine against the variants has increased due to the global spreading of Covid-19 mutations. The WHO has approved the following vaccines: AstraZeneca-Vaxzevria, Pfizer-BioNTech-Comirnaty, Moderna-mRNA-1273, and Janssen Ad26. COV2. S, Sinovac-CoronaVac, Sinopharm BBIBP-CorV, Novavax NVXCoV2373.1 AstraZeneca-Vaxzevria, Janssen Ad26. COV2. S, Sinovac-CoronaVac, and Sinopharm BBIBP-CorV are effective and safe for all people aged ⩾18 years. Novavax NVX-CoV2373 is effective and safe for all people aged ⩾12 years. Pfizer-BioNTech-Comirnaty and Moderna-mRNA-1273 are effective and safe for all people aged ⩾6 months. 49
Oxford-AstraZeneca vaccine
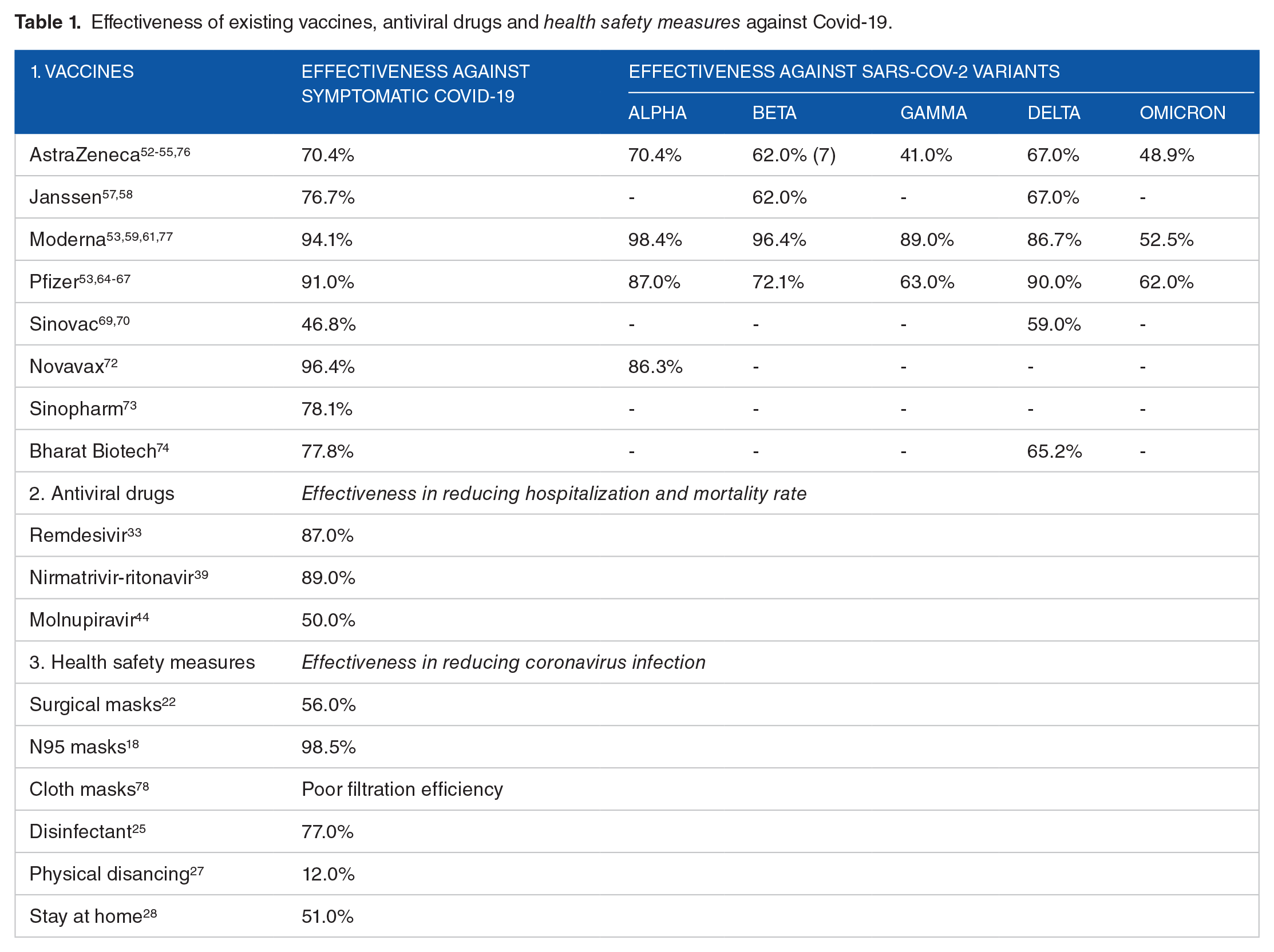
Oxford University and AstraZeneca jointly developed the recombinant Covid-19 vaccine named ChAdOx1 nCoV-19 (AZD1222). AstraZeneca has granted a license to the Serum Institute of India (SII) to produce the SII-Covishield vaccine. Oxford-AstraZeneca used non-replicated Chimpanzee adenoviruses to develop their Covid-19 vaccine. 50 To trigger an immune response, a protein is produced by the DNA vector that resembles the viral s-peptide. Following the introduction of the DNA into the cytoplasm, it enters the cell nucleus. It is turned into mRNA by the host enzymes rather than being incorporated into the cellular DNA and then tends to way back into the cytoplasm where it interacts with the ribosomes of the host cell to yield translated proteins. The stimulation of antibodies, B-cell, T-cells, and plasma cells are occurred at this phase due to similarities in the mechanisms of DNA and RNA vaccines. 51 To determine the vaccine effectiveness, a randomized controlled study was conducted that showed the Ox-ford-AstraZeneca Covid-19 vaccine efficacy was 70.4% against symptomatic Covid-19. 52 According to the clinical study results in Ontario, the effectiveness was 62.0% against the Beta strain (B.1.351) of AZD1222 vaccine ⩾14 days after administration of the first dose Same report showed that one dose of ChAdOx1 prevented 41% of symptomatic infections by Gamma variant, ⩾14 days after administration of the first dose. 53 A test-negative case-control study was conducted in England that showed vaccine effectiveness was approximately 67% with 2 doses of the vaccine against the Delta variant. 54 Another case-control study was conducted in England to assess the efficacy of the vaccine against symptomatic disease caused by the omicron variant. The effectiveness was 48.9% after the administration of the second dose of the vaccine. 55
Janssen Ad26.COV2.S vaccine
The Janssen Ad.26.COV2.S is a recombinant adenoviral vector vaccine designed by Johnson & Johnson. After exposure to SARS-CoV-2, it generated potent responses of neutralizing antibodies and offered nearly complete protection. 56 To trigger an immune response, a protein is produced by the DNA vector that resembles the viral S-peptide. Following the introduction of the DNA into the cytoplasm, it enters the cell nucleus. It is turned into mRNA by the host enzymes rather than being incorporated into the cellular DNA and then tends to move back into the cytoplasm where it interacts with the host cell ribosomes to yield translated proteins. The stimulation of antibodies, T-cells, B-cell, and plasma cells are occurred at this phase due to similarities in the mechanisms of DNA and RNA vaccines. 51 To estimate the vaccine effectiveness, a phase III clinical study was done to obtain the effectiveness among the people whose SARS-CoV-2 testing was negative. The vaccine efficacy of a single dose was 76.7% against severe critical Covid-19 at 14 days after administration. 57 Another study showed the vaccine effectiveness related to hospital admission conducted in South Africa. The Effectiveness of that study was 62.0% and 67.0% against the Beta and Delta variants, respectively. 58
Moderna-mRNA-1273 vaccine
It is an RNA-based vaccine designed by Moderna. A lipid nanoparticle is encapsulated to produce the mRNA-1273 vaccine. The mRNA-1273 Vaccine contains the full-length spike protein of the SARS-CoV-2 virus, which is prefusion stabilized and induces Covid-19. 59 The SARS-CoV-2 spike glycoprotein is encoded by a nucleoside-modified mRNA in the vaccine, which is enclosed in lipid nanoparticles for more effective distribution inside the host cells. The vaccine aims to stimulate both T-cell and B-cell defenses against the spike protein. The vaccine uses a potent lipid nanoparticle delivery mechanism to prevent initial stimulation of interferon-associated genes are significant characteristics that boost its effectiveness. 60 To assess the efficacy of the vaccine, a phase III clinical trial among the participants who were 18 years of age or older showed that the effectiveness against the Covid-19 disease was 94.1% after a double dose. 59 A case-control study performed among adult participants of Kaiser Permanente Southern California (KPSC) that showed the efficacy of the vaccine was 98.4% against the Alpha variant within 2 weeks or more after the second dose. 61 An investigation from the Qatar population particularly working-age adults revealed that the vaccine offered 96.4% effectiveness against disease for the Beta strain. 62 The study demonstrated that the efficacy of the vaccine was 89.0% against the Gamma strain after administration of the first dose. 53 The case-control study on adult KPSC participants reported that the vaccine efficacy was 86.7% against the Delta strain after the second dose. 61 In Canada, a retrospective cohort analysis showed that the Moderna vaccine was able to reduce infection and hospitalization rates by 92.0% and 97.0%, respectively. 63 A report from Spain demonstrated that booster doses of Moderna vaccines were given among the participants aged ⩾40 years, and showed 52.5% effectiveness against the Omicron strain
Pfizer-BioNTech-Comirnaty vaccine
Pfizer-BioNTech has designed an RNA-based Covid-19 vaccine named Comirnaty. The Pfizer vaccine has similar mechanisms of action as Moderna. 60 In Israel, a third dose vaccine of Pfizer-BioNTech was pushed into their citizens (age: 37-52 years) to evaluate the effectiveness. Both male and female participants were taken to conduct the study. The third dose was administered to the participants who had taken their second dose at least 5 months ago. It showed 91% effectiveness against symptomatic diseases of Covid-19. 64 In Qatar, a retrospective cohort study conducted on their inhabitants reported that the vaccine effectiveness was 87.0% against the Alpha strain and 72.1% against the Beta strain. 65 An observational analysis was conducted on symptomatic community-dwelling individuals in Canada which reported that this vaccine has 63.0% efficacy against Gamma strains. 53 In the United Kingdom, an investigation was done in their populations (60 years of age or older) and demonstrated that the vaccine effectiveness was 90.0% against the Delta variant within 2 weeks or more after the second dose. 66 Another investigation was performed in Canada in individuals (aged: 12-17 years) who reported that the vaccine effectiveness of the third dose was 62.0% against the symptomatic disease of the Omicron variant. 67
Sinovac-CoronaVac vaccine
This Covid-19 vaccine is designed by Sinovac R&D Co., Ltd. Typically, this kind of vaccination is intramuscularly given and needs an adjuvant to stimulate an immune response that is targeted against various viral proteins. 1 The SARS-CoV-2 virus is introduced into the African kidney cells (Vero-Cell) of the green monkey. It is mixed with aluminum hydroxide (adjunct) and inactivated using β-propiolactone. Antigen-presenting cells which are introduced into the body engulf inactive viruses. It incorporates antigenic markers that the immune system recognizes and responds to by producing antibodies. 68 To estimate the vaccine effectiveness, a case-control investigation was done in adults aged ⩾70 years in Brazil that revealed the effectiveness of the 2 doses was 46.8% and 55.5% against symptomatic disease and hospitalization, respectively, because the Gamma variant within 2 weeks or more after the second dose. 69 A retrospective cohort analysis from Colombia reported that this vaccine can reduce hospitalization and death rates by 67.2% and 77.1%, respectively, due to the Mu variant of coronavirus. 1 During the outbreak of the Delta variant in May 2021 in China, an investigation performed among the participants aged 18 to 59 years showed 59.0% effectiveness for 2-dose vaccination against the Delta variant. 70
Novavax NVX-CoV2373 vaccine
The Collaboration for the foundation of Epidemic Preparedness Innovations and American biotechnology Novavax combinedly designed NVX-CoV2373. The vaccine incorporates spike proteins into a nanoparticle (knuckle-shaped) which is injectable in combination with the patented Matrix-M adjuvant. The adjuvant is shown to have a well-tolerated and potent impact by promoting the passage of antigen-presenting cells through the site of injection and boosting immune response. 71 Following a post hoc study, 29 participants had the symptomatic disease (non-B.1.1.7 variant), while 66 participants had the Alpha variant. This investigational report of Phase III clinical trials reported that the vaccine gave 96.4% efficacy against symptomatic disease and 86.3% against the Alpha strain. In late 2020, investigations from South Africa indicated the appearance of B.1.351 (or beta variants), which had been linked to higher transmission, more severe infection, and varying degrees of immune resistance to Covid-19 vaccinations. This survey was conducted among the adult population and the effectiveness of the vaccine was found 51.0% against the Beta variant. 72
Sinopharm BBIBP-CorV vaccine
Beijing Institute of Biological Products has designed Sinopharm, an inactivated BBIBP-CorV vaccine for treatment against Covid-19. 7 Once inactivated viruses get into the body, the immune system of the body stimulates the production of antibodies and make the body ready to respond against SARS-CoV-2. The SARS-CoV-2 dead copy in the form of a vaccine is administered intravenously. The immune system is then boosted using the dead antigens from the virus against further viral attacks. 71 There are many studies conducted to know the efficacy of this vaccine. An investigational phase III study was performed in UAE and Bahrain among adults (18 years). One of the 2 inactivated vaccines made from the SARS-CoV-2 HB02 and WIV04 strains, or aluminum hydroxide (alum) was given to each participant. Vaccine efficacy was found 78.1% for the HB02 group and 72.8% for the WIV04 group, compared with the alum group. 73
Bharat Biotech BBV152/Covaxin vaccine
It is a Vero cell-based inactivated SARS-CoV-2 vaccine designed by Bharat Biotech. To enhance immune response and promote more durable immunity, the vaccine is used in conjunction with immune stimulants which are referred to as vaccine adjuvants (Alhy-droxiquim-II). The inactivated virus is combined with the Alhydroxiquim-II adjuvant from Kansas-based ViroVax to produce the vaccine candidate. 71 A study was performed in 25 Indian hospitals on randomized people who were aged ⩾18 years. This investigational Phase III study reported that the vaccine efficacy was 77.8% against symptomatic disease and 65.2% against the Delta strain. 74 An investigational report demonstrated that the efficacy of the vaccine showed a 26-fold reduction against the Omicron variant, 6 months following the second dose. Contrary to Covishield vaccinated patients, those who received the BBV152 vaccine maintained anti-nucleocapsid antibodies for a long duration. 9 Among these vaccines, AstraZeneca, Janssen, Moderna, and Pfizer have shown better efficacy than others against Delta, Mu, and Omicron variants to prevent the pandemic situation. Vaccination should be widely promoted in several poor and developing nations to achieve the preventative goal.
Discussion
Comparative effectiveness of existing vaccines, antiviral drugs, and health safety measures against Covid-19 have been presented in Table 1. In this study effectiveness means, protection from infection in case of health safety measurements, less hospitalization, and reduced mortality rate in the matter of antiviral agents. This result does not indicate that mask use is useless in fighting Omicron waves. Therefore, mask use remains efficient in reducing the spreading of coronavirus risk as this approach is non-specific to variants. It appears sensible to indicate that more effective respirators for example N95 or equivalent would be preferred to provide significant defense against the Omicron variant (Table 1). Even after vaccination, people should wear masks or keep doing so, particularly in crowded areas. As it is an airborne disease, people should follow social distancing, handwashing, and proper coughing guidelines. We observed that the antiviral drugs are effective in decreasing Covid-19 related hospitalization and mortality rates (Table 1). 75 Also, these oral medications did not increase the incidence of adverse effects. However, oral antiviral agents for Covid-19 are still under investigation. Covid-19 vaccines showed better efficacy against Alpha, Beta, Gamma, and Delta Variants (Table 1). 75 The effectiveness of vaccines has reduced significantly against Omicron variants (Table 1). Astra-Zeneca and Janssen’s vaccines have shown minimum effectiveness against Beta, Gamma, Delta, and Omicron variants. Moreover, all the existing vaccines showed significantly reduced efficacy against the Omicron variant (Table 1). The Omicron variant was not sufficiently prevented by 2 doses of vaccination. Therefore, we require an additional third or booster dose for better performance against the Omicron strain. Also, we noticed inadequate or limited data about the effectiveness of other vaccines against the latest SARS-CoV-2 variants (Table 1). Among these vaccines, AstraZeneca, Janssen, Moderna, and Pfizer have shown better efficacy than others against different variants to prevent the pandemic situation. Vaccination should be widely promoted in several poor and developing nations to achieve the preventative goal. The WHO has repeatedly advised nations to start mass vaccination programs.
Effectiveness of existing vaccines, antiviral drugs and
People should take some precautions like social distancing, using the mask, routinely washing hands, ensuring rooms are well-ventilated, avoiding close contact and crowds, and coughing into an elbow (bent) or tissue. The healthcare authorities should provide Covid-19 vaccination to every eligible individual. Governmental and non-governmental organizations should create more awareness about protective measures, spreading information, health messaging, getting funds, and emergency relief for vulnerable communities. Political leaders should aid in the uniform distribution of vaccines across the country. Policymakers should reform, reorganize and strengthen health system studies and clinical services to manage Covid-19. WHO should keep updating the current incidents and country-specific data. Researchers and scientists should emphasize research and innovation to minimize adverse reactions and improve the efficacy of higher levels of existing vaccines against recent variants. Additionally, they should assess the effects of control and mitigation strategies.
As of December 21, 2022, 72.83% of individuals (3 in 4) have been vaccinated with at least one dose in high-income countries. However, only 29.37% of people in low-income countries have received at least one dose of the Covid-19 vaccination. 79 Vaccination should be widely promoted in several poor and developing nations to achieve the preventative goal.80 -86 Countries across the world need to accelerate their vaccination campaign at least 20 times to reach 70% global coverage.87-90
Conclusion
Antiviral drugs and other non-pharmaceutical interventions (NPIs) are still efficient preventive measures that are affordable and accessible regarding the ongoing Covid-19 outbreak. In case of non-pharmaceutical interventions, masks are more effective to prevent the spreading rate of COVID-19. Medical grade masks are more efficacious than cloth face masks. The researchers across the world are working to develop anti-COVID-19 therapeutics that have shown valuable outcomes. Among the antivirals Remdesivir, Nirmatrivir-ritonavir, and Molnupiravir might be effective against coronavirus. It is crucial to vaccinate a significant population in poor and developing nations and underprivileged areas. The outcomes from clinical trials of existing vaccines might trigger the need for developing customized COVID-19 vaccines against new variants. The combined effects of preventive and therapeutic approaches can be effective against the Covid-19 pandemic. Also, enhanced testing, vaccinations, and safety measures can suppress the infection rate. We recommend clinical trials of existing vaccines against recently identified corona-virus variants.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MAH, MT and BA, and conceived the study and wrote the first draft. MJH, AR, MS, and MAB revised and gave intellectual inputs in the manuscript. MRI conceived and supervised the work. All the authors approved the final version for submission.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Ethics Statement
It was an analysis of online available aggregate data. No Ethical approval was needed.
