Abstract
Leukemoid reaction (LR), which is defined as leukocytosis with a white blood cell (WBC) count above 50 000/µL, can be caused by various conditions, while paraneoplastic leukemoid reaction (PLR), a rare type of paraneoplastic syndrome, occurs in cases of solid tumors. Here we report 2 cases of high-grade urothelial carcinoma (HGUC) with PLR accompanied by rapid tumor progression after complete resection of the primary tumor. We reviewed the patient’s clinical history, histopathology, and the results of laboratory tests to rule out LR induced by non-tumor causes. In both cases, PLR appeared after primary tumor resection, and the patients died of disease at the peak of PLR at 6 and 8 weeks. Immunohistochemistry for granulocyte colony-stimulating factor and its receptor was performed on tumor tissues. Patients with HGUC and PLR are rare and have an extremely poor prognosis. The mechanism by which solid tumors are associated with PLR and rapid tumor progression after surgical resection of the primary tumor is incompletely understood and will be discussed here.
Keywords
Introduction
Leukemoid reaction (LR) is defined as leukocytosis with a WBC count above 50 000/µL, mainly comprising mature neutrophilia or neutrophil precursors. This phenomenon may occur under various conditions including infections, therapeutic drug use, intoxication, hemolysis, or malignant tumors.1-3 Paraneoplastic syndrome occurs in patients with malignancy and is not related to tumor invasion or metastasis but by reactions to hormones or cytokines secreted by tumor cells. Paraneoplastic leukemoid reaction (PLR) is diagnosed when patients present with paraneoplastic syndrome with LR. PLR is rare and occurs in a small subset of patients with solid tumors, including lung cancers, gliomas, bladder cancers, colorectal cancers, melanomas, and skin carcinomas, among others.4-15 The frequency of PLR in different solid tumors has not been well studied. Granger et al reported an incidence rate of 10% in a series of 758 patients with nonhematologic solid tumors. Most of their cases with LR were not tumor-related. 4
PLR in patients with solid tumors always predicts a poor outcome, and most patients die within 6 months. In most cases, increased white cells are mature neutrophils, and granulocyte colony-stimulating factor (G-CSF) secreted by these tumors is responsible for the generation of PLR and the tumor progression cascade.5,7,8,10,15 Recently, a new understanding of tumor-associated neutrophils (also designated myeloid-derived suppressor cells (MDSCs)) has revealed the role of neutrophils (PMN-MDSCs) in the mechanism of tumor progression. Some evidence indicates that a subset of pro-tumor neutrophils could be PMN-MDSCs and are likely key players in PLR and tumor progression.16-18
Here, we present 2 additional cases of high-grade UC (HGUC) with PLR. In the first case, the tumor was located at the left renal pelvis, while in the second case, the tumor was in the urinary bladder. Interestingly, both patients had initial mild leukocytosis before primary tumor resection. However, after resection of the primary tumor, their leukocytosis progressed rapidly to PLR in parallel with tumor metastases. Case 1 and case 2 died of their disease at the peak of leukocytosis at 6 and 8 weeks, respectively, after primary tumor resection. Our cases demonstrated a markedly positive correlation between PLR and tumor progression. Furthermore, resection of the primary tumor seemed to trigger rapid tumor progression through a mechanism that is not fully clear.
Case Reports
Case 1
A 54-year-old woman present with hematuria for more than 3 years with complaint of left side flank pain in June, 2017. Physical examination showed tenderness over the left flank region. Laboratory data consisting of routine blood cell count revealed slightly elevated WBCs at 14.8 × 103/µL (normal range 4-10 × 103/µL) with 82.7% segmented cells (normal range 40%-60%), anemia with hemoglobin 11.2 g/dL (normal range 12-16 g/dL), and a slightly elevated eosinophil count at 5.7% (normal range 1%-5%). Peripheral blood smears revealed predominantly mature neutrophils with frequent cytotoxic granulations, occasional Dӧhle bodies, and cytoplasmic vacuoles but no immature cells. Infection-related leukocytosis was excluded by clinical symptoms and negative blood culture. Sonography and computed tomography (CT) scans showed a 5-cm left upper renal mass (Figure 1A). Atypical cells were found in urine cytology before operation. Ureteroscopic biopsy was initially performed but failed to obtain a diagnostic tissue. CT-guided biopsy and subsequent surgery consisting of left nephroureterectomy and hilar lymph node excision were performed on August 4, 2017 and led to a diagnosis of HGUC without hilar lymph node involvement (stage III, pT3N0M0). The immediate operation after the diagnosis of malignancy might minimize the possible risk of seeding related to the CT-guided biopsy. The postoperative course was uneventful. Unfortunately, the 5-week follow-up after surgery revealed a leukemoid reaction (LR) with a markedly elevated WBC count at 91.7 × 103/µL with 93.2% segmented cells. Further CT scans showed widely disseminated tumors in the abdominal cavity, pelvis, para-aortic lymph nodes, and lungs (Figure 1B). Her WBC count increased to a peak of 191.8 × 103/µL within 10 days before the date of death (Figure 2). She did not receive chemotherapy due to rapid course of the disease. Bone marrow biopsy 5 days before her death revealed hypercellularity with increased matured neutrophils without left shift. Cytogenetic analysis for the BCR-ABL1 translocation was negative. Flow cytometry on bone marrow aspirate excluded the possibility of leukemia. The features were compatible with PLR. Immunohistochemistry (IHC) showed that granulocyte colony-stimulating factor (G-CSF) was weakly expressed in the primary tumor and that G-CSF receptor (G-CSFR) expression was demonstrated in the metastatic tumor (Figure 3).

Computed tomography scan of case 1 before surgery showed a left upper renal tumor (A) and gross picture of the renal tumor (B), tumor metastasis in the pelvis after primary tumor resection (C) and the computed tomography scan image of the bladder tumor in case 2 (D) were also shown.

In case 1, the patient underwent major surgery on August 4 with PLR found 5 weeks later; peak white cell count was 191.8 × 103/µL. This patient died due to rapid tumor progression.
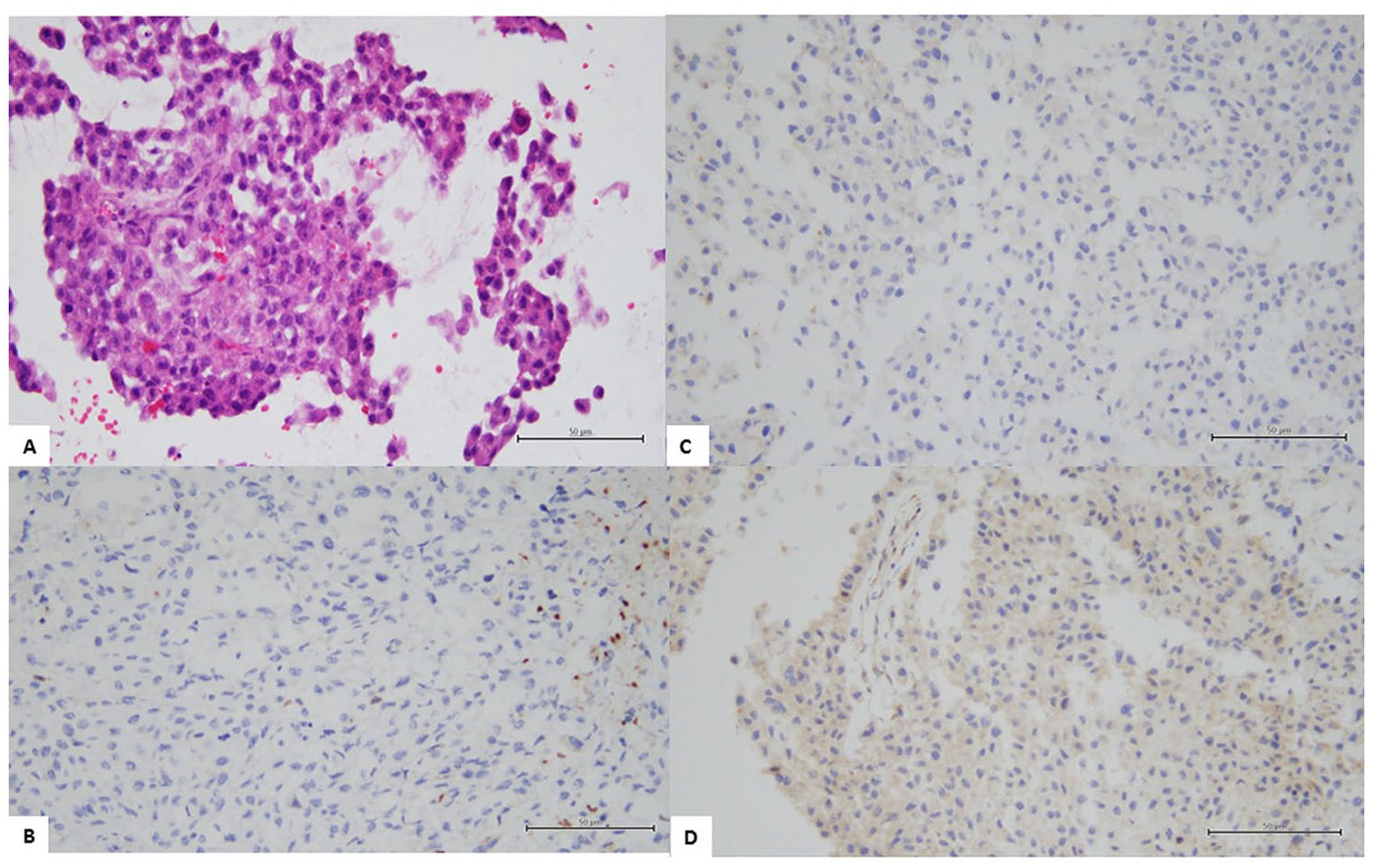
In case 1 (A), the primary tumor was diagnosed as HGUC (Hematoxylin and Eosin stain 200×), (B) negative G-CSFR expression in the primary tumor (IHC 200×), (C and D) expression of G-CSF and G-CSFR in the metastatic tumor (IHC 200×).
Case 2
A 61-year-old man with a history of UC was diagnosed in March 2017 and received 4 courses of neoadjuvant chemotherapy with MEC (Mitoxantrone, Etoposide, and Cytarabine) at another hospital. He presented to our hospital with hematuria in July 2017. Physical examination on admission showed no fever or other symptoms of infection. Laboratory data consisting of routine blood cell count were initially normal, but an elevated WBC count of 24.6 × 103/µL (normal range 4-10 × 103/µL) with 76% segmented cells (normal range 40%-60%) was noted after the first surgery, which was transurethral resection of bladder tumor (TURBT) that was performed at our hospital. He also had anemia with a hemoglobin level of 11.6 g/dL (normal range 12-16 g/dL) and slightly elevated eosinophil count at 5.9% (normal range 1%-5%), which increased to 15.9% after cystoprostatectomy was performed on August 10, 2017, but gradually returned to normal along with an increased WBC count. Peripheral blood smears showed predominantly mature neutrophils without blasts. An infection-related LR was excluded by a low WBC count and the presence of segmented WBCs before surgery. At his second admission on September 4, 2017, the patient presented with fever. Blood culture showed no bacterial growth, but 1 urine culture showed Pseudomonas aeruginosa and Klebsiella pneumoniae growth, which was controlled with antibiotics treatment. However, the WBC count continued to increase to a peak of 90.0 × 103/µL with 97.1% segmented cells until the end of his hospitalization on October 10, 2017.
Grossly, the urinary bladder during cystoprostatectomy contained a 7.5 × 4.7 cm tumor invading the prostate, and a pathologic diagnosis of HGUC with focal squamous differentiation was made (Figure 4A). Additional IHC for G-CSF and G-CSFR was positive (Figure 4B-D). The tumor stage was stage IV (pT4bN1Mx). CT performed 1 month after surgery revealed extensive tumor disseminations in the pelvis, abdominal cavity, mediastinal lymph nodes, vertebral bones, and the lungs. Bone marrow biopsy revealed hypercellularity with an increased number of mature neutrophils. Cytogenetic analysis for the BCR-ABL1 translocation was negative. Flow cytometry on bone marrow aspirate and the bone marrow biopsy excluded myeloproliferative neoplasms. The patient was discharged with terminal disease, extensive tumor dissemination, and multiple organ failure and died 8 weeks after major surgery.

In case 2 (A), the urinary bladder tumor was diagnosed as HGUC with squamous metaplasia (Hematoxylin and Eosin stain 200×), (B) tumor seen in the first surgery exhibited G-CSFR expression (IHC 200×), (C and D) tumor removed by cystoprostatectomy showed G-CSF and G-CSFR expression (IHC 200×).
Discussion
PLR in urothelial carcinoma is extremely rare, as only approximately 30 cases have been reported up to 2020 in the English-language literature. 5 Interestingly, Izard et al reported a 0.6% incidence rate of paraneoplastic leukocytosis in patients with UC based on a WBC count >20.0 × 103/µL on more than 2 occasions 30 days apart. 6 In practice, PLR is a diagnosis of exclusion. The use of recombinant G-CSF or high-dose corticosteroids, or leukemia may cause extreme leukocytosis and must be excluded before a diagnosis of PLR is made. In our cases, we excluded the above causes by medical history and clinical records. Chronic myeloid leukemia and other myeloproliferative neoplasms were excluded by negative BCR-ABL1 translocation, absent clones in flow cytometry analysis of bone marrow aspirate, and histological evaluation of a bone marrow biopsy specimen. Infections may cause leukocytosis but rarely to the degree of LR and can be excluded by clinical symptoms and laboratory data. A peripheral blood smear, bone marrow biopsy, and a clinical history of underlying high-grade malignancy are essential for the diagnosis of PLR. 3
In terms of prognosis, many reports demonstrated that patients with solid tumors and associated PLR present very poor outcomes and usually die of their disease within 6 months of diagnosis. In our 2 patients, PLR appeared after surgical resection of the primary tumor, and the WBC count persistently increased in parallel with wide tumor dissemination. The high stage in case 2 indeed indicates a poor prognosis of the disease. However, such an early dissemination with tumor metastases soon after the operation was thought unusual, especially when it was accompanied by leukocytosis/ leukemoid reaction that was suggested to be a poorly prognostic factor. Furthermore, it seems that rapid tumor progression is related to resection of the primary tumor. The clinical presentations in our cases are consistent with previous reports, which found that a subset of HGUC patients with PLR exhibited rapid tumor progression.5,8,10,15 The pathogenesis of tumor spread after resection of the primary tumor is not well defined. Perez et al proposed that undetected micrometastases before surgery could be responsible for tumor spread. Interestingly, this phenomenon was observed in animal models. 7 Dormancy of disseminated tumor cells and their reactivation could result in metastases after surgical removal of the primary tumor through mechanisms that are not yet clear. 19
An autocrine loop or paracrine stimulation by G-CSF and G-CSFR secreted by the tumor cells has been proposed to induce rapid tumor growth. 8 Some cytokines, such as interleukins and TNFs, might contribute to tumor growth in patients with PLR. 9 Mizutani et al 10 reported that 9.2% (13/141) of patients with UC tumors had secreted G-CSF in their serum. Furthermore, the serum G-CSF level was correlated with higher tumor grade and stage, and consequently, shorter survival time. Tachibana et al. proposed the presence of high affinity G-CSF binding receptor on cultured cancer cells. 11 In their series, 6 of 26 (23.1%) patients with primary bladder cancer presented with positive G-CSFR mRNA expression. 12 Their reports indicated a significant subgroup of patients with HGUC that secreted G-CSF and expressed G-CSFR, which led to PLR and rapid tumor progression.8,13,14
Using IHC in a case of urothelial carcinoma of the renal pelvis with PLR, Lin et al demonstrated the expression of G-CSF, vascular endothelial growth factor (VEGF), and hypoxia-inducible factor 1 (HIF-1) in tumor cells. 15 Their results indicated that these factors might be associated with leukemoid reaction in G-CSF-producing tumors. VEGF and HIF-1 expression may exert a proangiogenic effect to promote invasion and metastasis in HGUC. IHC of the surgical specimens from our 2 cases demonstrated G-CSF and/or G-CSFR expression in the primary and metastatic tumors.
Recently, accumulating evidence has revealed a significant role of tumor-associated neutrophils in PLR and tumor progression. A subset of pro-tumor neutrophils, that is, granulocyte-myeloid-derived suppressor cells (G-MDSCs or PMN-MDSCs) was suggested to suppress anti-tumor immune reactions and promote tumor proliferation, invasion, and metastasis through the G-CSF-G-MDSC-tumor axis.16-18 The interactions among tumor cells, the tumor microenvironment, MDSCs, and tumor-suppressing immune cells may explain the mechanisms of PLR and tumor progression.
For therapeutic purposes, understanding the effects of G-CSF and G-CSFR in patients with solid tumors is important. In the report by Granger et al, solid tumors and extreme leukocytosis in up to 69% of patients were caused by the use of exogenous hematopoietic growth factors (ie, recombinant G-CSF) for chemotherapy-related leukopenia. 4 The risk associated with recombinant G-CSF is still controversial.
In conclusion, we present 2 patients with HGUC who experienced PLR and rapid tumor progression after surgical resection of the primary tumor. G-CSF and G-CSFR expression in the primary and metastatic tumors in our cases may be significant in the etiology of tumor progression. We noted that interactions among G-CSF- secreting tumor cells, G-MDSCs, and suppressive anti-tumor immune cells might trigger the process. More studies are required for a better understanding of this rare and intriguing phenomenon.
Footnotes
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Contributions
S.-T.P. initiated the study. S.-P.T. and C.-H.W. did the experiments and analyzed the data. S.-T.P reviewed and investigated the study cases. S.-T.P. wrote the manuscript. S.-T.P. and Y.-T.C. revised and edited the manuscript. All authors approved the manuscript.
