Abstract
Sexual function/dysfunction are crucial factors that determine the quality of life of an individual. In order to avoid hypoactive sexual desire and lack of sexual satisfaction, many people consume aphrodisiac agents which enhance libido and sexual desires. Psychoactive plants are claimed to have aphrodisiac properties. In folklore, a number of natural products derived from these plants, including crude extracts and aphrodisiac concoctions, are consumed to mediate sexual motivation, increased desire, prolonged duration, and alertness. Psychoactive plants possess chemical compounds that most likely evolved as allelochemicals but, when taken by humans, target specific neural receptors, affecting emotion, perception, and cognition. Due to the diverse psychotropic effects of these plants, they have been utilized as medicines and in religious rites from ancient times. The historic use, toxicological profile, and effects of psychoactive plants on sexual function were briefly examined in this article.
Introduction
Penile erection is achieved by the integration or modulation of psychological factors, neurological, as well as vascular processes which initiate a cascade of reactions within the penile smooth muscles (Figure 1). 1 Psychological and hormonal factors affect penile erection. Nerve impulses release neurotransmitters from cavernous nerve terminals and relax factors from endothelial cells in the penis, relaxing smooth muscle in arteries and arterioles, supplying erectile tissue and increasing penile blood flow. 1 Relaxing the trabecular smooth muscle increases sinusoidal compliance, allowing for rapid filling and expansion. Subtunical venular plexuses are thus compressed between the trabeculae and tunica albuginea, occluding venous outflow. This traps blood in the corpora cavernosa and erects the penis. 1
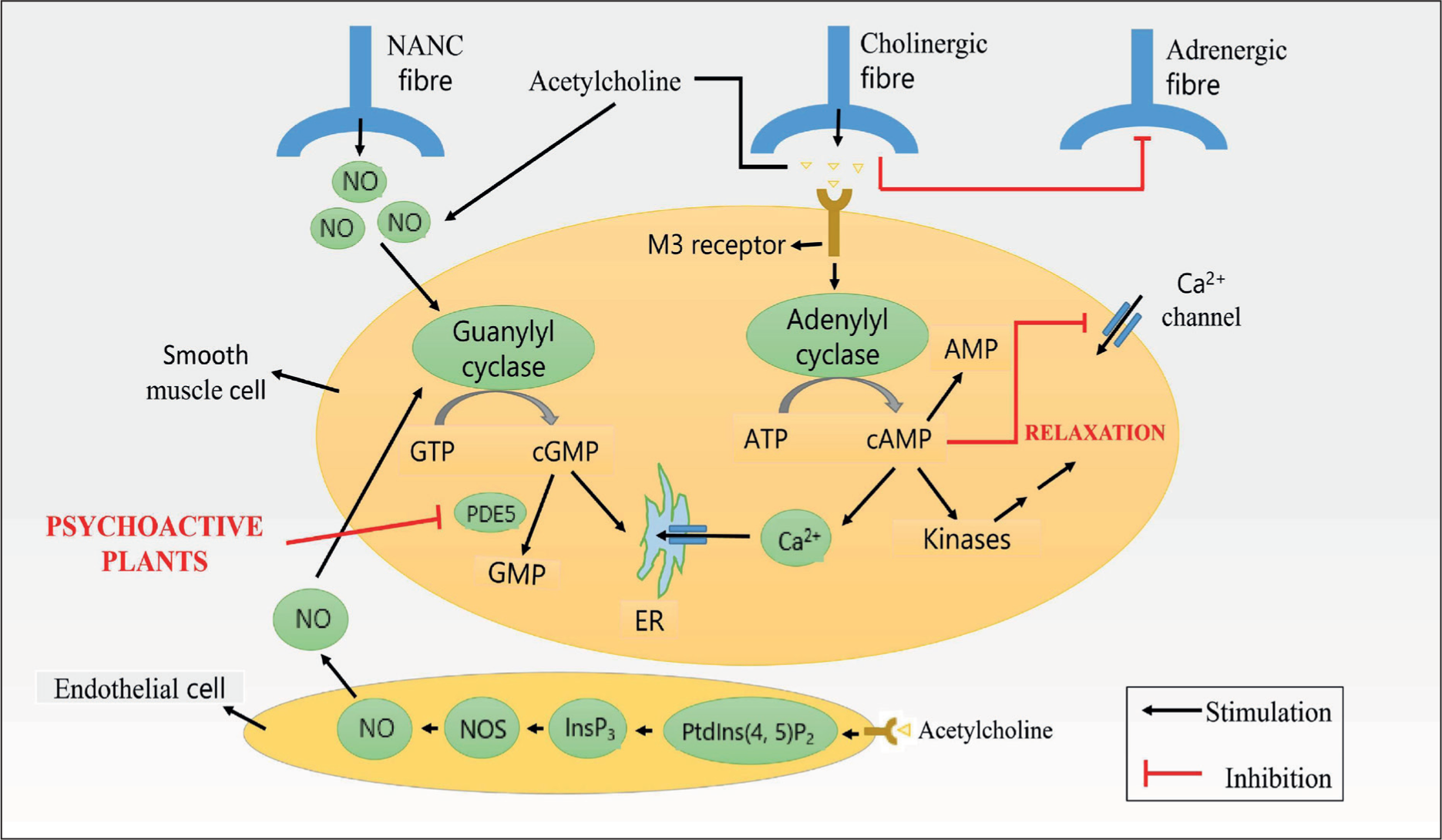
Male sexual dysfunction is an abnormality or absence of sexual psychology and physiology. It includes erectile dysfunction (ED) (inability to attain and maintain an erection for sexual satisfaction), premature ejaculation, sexual failure, and loss of libido/desire. Male sexual dysfunction is not a single disease. It includes male sexual arousal, penis erection, penis insertion into the vagina, ejaculation, and any obstacle to achieving any of the above-mentioned can result in male sexual dysfunction. Male sexual dysfunction affects a man’s quality of life, including loss of self-esteem, avoidance of intimacy, anxiety, and depression. It also affects sexual partners’ desire and satisfaction.2,3 There are several causes of male sexual dysfunction, including physical and psychological factors. Physical causes can include conditions such as diabetes, heart disease, high blood pressure, hormonal imbalances, and neurological disorders. Psychological factors can include anxiety, depression, stress, and relationship issues. 4 Treatment options for male sexual dysfunction can depend on the underlying cause of the condition. For example, treating an underlying medical condition or addressing psychological issues through therapy may improve sexual functioning. In some cases, medications such as phosphodiesterase-5 (PDE-5) inhibitors (e.g., sildenafil, tadalafil) or other therapies (e.g., vacuum devices, penile injections, surgery) may be recommended. 5
Traditional medicines derived from plants have been used in the treatment and management of male sexual dysfunction especially in developing countries with varying degrees of success due to their natural phyto-contents. Plants that have the ability to alter an individual’s consciousness, mood or mental processes and are related to dose tolerance and dependence are called psychoactive plants. Since ancient, these plants have been employed for their varied psychotropic properties in religious rites and as medicines. 6 While there is a well-established mythology relating to the connection between consuming some of these plants and sexual function, psychoactive plants are thought to be aphrodisiacs. The aim of the article is to review and summarize the scientific literature on the influence of psychoactive plants on male sexual function.
Male Sexual Dysfunction and Depression
Multiple developmental, cognitive, affective, and interpersonal characteristics that predispose men to sexual dysfunction have been outlined in historical ideas explaining the psychological components of ED. At the moment, it is believed that a set of risk, precipitating, and maintenance factors are principally responsible for psychogenic ED.7,8 Psychological factors such as lack of sexual arousability, performance anxiety, stress, strained relationship, schizophrenia, and depression with or without hormonal, vascular, and neurological pathologies have been the leading cause of male sexual dysfunction 9 inhibiting the process of erection.
Physical alterations linked to depression cascades include altered eating habits, low libido or energy levels, disruptions in daily and ultradian rhythms, body temperature, or numerous endocrine functions, as well as hypersomnia or insomnia. 10 Male sexual dysfunction and depression are closely related. Major depression is primarily related to decreased sexual desire, fear of failure or performance anxiety. 25% of male depressive patients also report ED. 11 There is a bidirectional link between depression and ED: sad mood can reduce sexual desire and result in ED, while decreased sexual activity and dissatisfaction with one’s sexual life can promote depressive symptoms. 12 The well-known sexual adverse effects of antidepressants further exacerbate this association. 11 Even in the absence of syndromal depression, ED is frequently associated with depressive symptoms. Depression has also been linked to premature ejaculation 13 and might be a reflection of the lower self-esteem brought on by this disorder.
Particularly, anxiety and depression are thought to have a significant impact on the emergence and maintenance of problems with sexual functioning. Several research have established a link between anxiety and ED,14-16 as well as a link between anxiety and premature ejaculation.16,17 However, other investigations have discovered that anxiety may actually promote sexual desire. 18 Depression and ED have a high correlation, and the intensity of the correlation rises with the severity of the depression 19 premature ejaculation has also been associated with depression, and more recent research has indicated that this association is stronger the longer the premature ejaculation lasts. 13 Recent research has shown that feelings of anxiety and sadness may influence ED and premature ejaculation differently. For example, performance and free-floating anxiety appear to play a major impact in the development and sustenance of ED, while genetic rather than psychological factors are increasingly acknowledged as important contributors to the development of premature ejaculation.16,20
There is a significant association between male and female sexual problems. Men with ED have been proven to significantly affect the sexual health of their female partners, and the opposite is also true. Women are more likely than men to have melancholy, anxiety, and dysfunctional sexual behavior. 21 Depression may harm sexual health by decreasing the desire for or satisfaction from partaking in enjoyable activities, interfering with intimate relationships, or raising the risk of smoking or substance abuse, particularly with widely available psychoactive plants. Female sexual dysfunction may be further exacerbated by other illness processes, such as metabolic syndrome, which depression and its accompanying behavioral patterns may also contribute.22,23
Psychoactive Plants
Some plants are classified as “psychoactive” because when consumed by humans, they can modulate noradrenergic, dopaminergic, Ƴ-butyric, serotonergic, cholinergic, and purinergic pathways, as well as neuronal receptors, affecting emotion, state of consciousness, and perception. 6 The present review therefore defined a psychoactive plant as “any plant, capable of modulating or affecting the mind or the mental processes.”
Abuse of psychoactive substances is an emerging issue for public health and socioeconomic burden on a global scale. Inadequate information on the pattern of psychoactive substance usage in underdeveloped and emerging worlds and the high incidence of drug-related health issues are two factors that highlight this worldwide problem. 24
The use of these plants is categorized as licit and illicit use. Nicotiana tabacum, Datura stramonium, and Carica papaya are examples of licit ones. Because the illicit varieties, such Cannabis sativa and Catha edulis, are so addictive, most countries have agreed to criminalize their sale and non-medical use. Despite being illegal, this herb is nonetheless widely used illicitly in many civilizations.24,25
Psychoactive plants contain several phyto-content especially alkaloids, which have been observed to act as antidepressants, sedatives, memory enhancers, psychedelic agents, soporific, euphoriant, and stimulatory agents thereby modulating neurotransmitter functioning systems resulting in psychoactivity. 26 Psychoactive plants have been reported to be involved in traditional festivals in many cultures across the world, they are used in recreational activities, some are potent aphrodisiacs, they are also use as an antidepressant agent and they are actively involved in spiritual rites in many cultures.6,24,25,27
Effect of Psychoactive Plants on Male Sexual Function
Marijuana
C. sativa of the family of Cannabaceae whose local names are wi-wi (Hausa), Igbó (Yoruba), Nwonkaka (Ibo), and generally called marijuana or Indian hemp in the English language is a native plant of Nigeria, and nearly everywhere. It is usually administered through inhalation, smoking, chewing, dipping, and drinking in folklore. The plant is said to have stimulation of appetite, aphrodisiac, sedation, and hallucination effects. 24 Several indigenous tribes treat numerous ailments with C. sativa derivatives. C. sativa leaves are used for wound healing in the Miandam region of Swat and North Pakistan. Powdered leaves are used as an anodyne, sedative, tonic, and narcotic. 24 C. sativa is also used by small ethnic groups in rural regions in Nepal, Arunachal Pradesh, and Haryana to treat pain, nausea, and snake bites in humans and animals. 28 Furthermore, C. sativa is used as an ethnoveterinary medicine by some livestock producers in Kenya and Uganda. 28
According to studies,29,30 nearly all cultures have some form of drug culture that relies on psychoactive substances for recreational, ritual, and/or therapeutic purposes. From Asia to Africa, many shamanistic religions hold the view that psychotropic herbs have the divine capacity to heal. One of the most common hallucinogenic plants employed by Old World shamans is C. sativa. 6 C. sativa was used as a painkiller during surgeries and for a variety of illnesses in ancient China, including malaria and constipation. The herb was revered in India and used to produce pleasurable feelings in the user.6,31
C. sativa have more than 750 identified chemical compounds. 24 C. sativa’s psychoactive ingredient, ∆-9-tetrahydrocannabinol (∆-9-THC), acts by binding to cannabinoid receptors and mediating pleasure-inducing sensations. THC’s absorption into the body increases dopamine release, which has sedative effects and euphoric effects. 32
As observed in folklore medicine, these psychoactive plants’ crude extracts and aphrodisiac mixtures can be used to treat and manage sexual dysfunction. 24 To mediate sexual enthusiasm, prolonged duration, better desire, and alertness, Nigerians use a variety of natural remedies composed of crude extracts and aphrodisiac concoctions from plants. The majority of these items are derived from psychoactive plants including C. sativa. 28 In a study, 5, 50, and 500 mg/kg alkaloid extracts from C. sativa were administered to Wistar albino rats in order to ascertain its aphrodisiac effect. The result of the study revealed that the Nitric oxide (NO) level was significantly elevated compared to the control group; the mounting and intromission latencies decreased at lower doses indicating the potential of C. sativa; the alkaloid extracts from C. sativa were also observed to inhibit PDE-5 activities thereby reducing the hydrolysis of cGMP and enhancing prolonged erection. 24 THC reduces psychological inhibitions, and some people may feel that this increases desire. It is possible to perceive that the sexual stimulants that cause sexual arousal are heightened since the perception of senses (smell, touch, taste, hearing, etc.) is sharpened. The aphrodisiac-like properties of cannabis are also exploited. These effects have led to its long-standing use in shamanic and pagan societies as well as in tribal sex rituals. 33
Exposure to C. sativa at higher doses or by heavy users has been observed to affect the normal functioning of the brain; dopamine production is increased and acetylcholine secretion is inhibited in the process resulting in to decrease in glutamatergic synaptic transmission, affecting the brain function. 34 Working memory, intelligence quotient, cognitive performance and task execution are affected by chronic exposure to the plant. 35 Pregnant women who are exposed to C. sativa suffer from impaired dexterity and vision, increasing the probability that their unborn children will exhibit aberrant behaviors. 36
Yohimbe
Pausinystalia yohimbe of the family Rubiaceae is a native plant of Cameroon, Gabon, Congo, and Nigeria among several African countries. Traditionally, the plant is utilized as a stimulant to prevent sleep and as an aphrodisiac. 37 Yohimbine, a tryptamine alkaloid extracted from the stem back of P. yohimbe is responsible for the aphrodisiac effects of the plant, increasing libido and ejaculation. 38 Several studies have reported that extracts from the plant increase the mounting frequency, intromission frequency, ejaculatory frequency, ejaculatory latency, testosterone level and completely relax the corporal cavernosa smooth muscle. Interestingly, delayed ejaculation and other orgasmic problems have been successfully treated with yohimbine.39,40 Yohimbine has a strong affinity for alpha-2 adrenoceptors in humans, a moderate affinity for alpha-1 adrenoceptors, and a weak affinity for certain serotonin and dopamine receptors in the central and peripheral nervous systems. 41 Alpha-2 adrenoceptors control erection by preventing central nervous system (CNS) impulses. The alpha-2 adrenoceptors in the brain’s locus coeruleus are thought to be blocked by yohimbine, which is typically thought to increase central sexual urge. 42 Yohimbine may block alpha-1 and alpha-2 adrenoceptors in the periphery while also promoting cavernosal endothelial cells’ production of NO.43,44
The toxicological profile of yohimbine, as reported by ref.38,45, includes increased heart rate, insomnia, shivering, nausea, sweating, high blood pressure, and sleeplessness which has been attributed to its central adrenergic activity.
Areca catechu
Areca catechu of the family Arecaceae is a native plant of Sri Lanka, Malaysia, New Guinea, Indonesia, and Thailand. The plant is also widely distributed in Africa, America, and Europe. A. catechu has long been used to eliminate parasites and improve digestion. The areca nut was listed in Mingyi Bielu as an herbal remedy with potent diuretic, digestion-improving, and anti-parasitic qualities. The areca nut is a well-known emmenagogue, astringent, and digestive aid in Indian traditional medicine. The areca nut is reportedly used as a traditional medicine in Cambodia to treat liver diseases, diarrhea, and dysentery. In Malaya, the areca nut is also employed as a folk remedy for the management of lumbago and abdominal distension.46,47 The fruit of this plant, known as an areca nut, has long been used in ceremonies and as a folk medicine. 48 This fruit is often chewed and can be taken as a “betel quid” with other ingredients. Areca nut is the fourth most popular narcotic in the world, maybe because of its energizing, calming, or aphrodisiac properties. 48
From A. catechu, numerous chemical substances have been identified. Condensed tannins and pyridine-type alkaloids have been identified as the distinctive components of the more than 59 chemicals that have been isolated and characterized from this plant. 48 The primary psychoactive substance found in A. catechu fruit is arecoline. This substance is an alkaloid that functions as a non-selective nicotinic and muscarinic agonist as well as a competitive inhibitor of gamma-aminobutyric acid (GABA). 49 Arecoline readily crosses the blood-brain barrier once within the body, having an impact on the parasympathetic nervous system. 49
In a study, 50 and 100 mg/kg body weight ethanolic extract of Areca fruit was administered to Male Rattus, Wistar strain. The result of the result revealed that the extract increases the levels of follicle-stimulating hormone (FSH), luteinizing hormone (LH), libido, and sexual activities in the experimental rats. 50 Interestingly, the result from this study was supported by Anthikat’s 51 research. The aphrodisiac effects of areca nut ethanol extracts were assessed using an animal model of mating behaviors, including mounting latency and frequency, intromission latency, and frequency. The findings demonstrated that mounting frequency, intromission latency, and frequency were all considerably increased by the extract (150 mg/kg, for 28 days). The testis, cauda epididymis, ventral prostate, and seminal vesicle weights in the rats were all significantly higher. Additionally, there is an increase in the quantity of sperm cells. 51
The A. catechu fruit is addictive and has a number of negative effects, including abstinence syndrome (insomnia, mood swings, irritability, and anxiety) and effects on the digestive system.48,49 Aside from these consequences, eating this fruit has also been linked to myocardial infarction, severe extrapyramidal syndrome, and asthma.52,53 However, Hindu and Buddhist populations have documented the usage of this fruit for therapeutic purposes since antiquity. Areca nut eating has been linked to a variety of general benefits, including contentment, well-being, psychostimulating effects, stress relief, gum strength, and breath sweetening. 54
Tobacco
N. tabacum of Solanaceae family, commonly called Taba (Yoruba), Utaba (Igbó), Taba (Hausa), and tobacco in the English language is a native plant of Nigeria and other African countries as well as European, and Asian countries. It is normally smoked, chewed, inhaled, drunk, and snuffed in folklore. The plant is commonly grown commercially in southern Africa and diviners in these regions use N. tabacum as a snuff to begin their divination and as a traditional offering to the ancestors. 55 Among the Matsigenka Indians of Amazonian Peru, tobacco smoke is closely linked to the concept of the shaman’s magical breath, which is breathed on patients to heal them or at adversaries to kill them. 56
The pharmacologically active component of N. tabacum is nicotine, a tropane alkaloid that interacts with a particular subset of acetylcholine-mediated sites called nicotinic receptors. Nicotine affects the brain in a variety of ways, including stimulation, improved memory and awareness, reduced anxiety, and pleasure. 56
Smoking has a negative effect on sexual function, which is understandable given that nicotine is a strong vasoconstrictor. Nicotine decreases the effectiveness of blood circulation to the genital area, which is necessary for the physical aspects of erection. Smoking affects the vascular endothelium and peripheral nerves, which results in vasculogenic ED in men. 57 Furthermore, smoking causes an increase in blood nicotine levels, which may enhance the sympathetic tone in the penis by interfering with the relaxation of the smooth muscles in the body necessary to achieve and sustain an erection. 58 In their analysis of the causes of nicotine-related sexual dysfunction, Wolf and Shulman 59 speculate that alterations in endothelium-derived relaxing factor, NO, prostaglandins, prostacyclins, and thromboxanes may be involved in smoking-related impotence.
Numerous studies have examined the effects of tobacco use on sexual behavior, notably in male sexuality.60,61 Compared to nonsmokers, smokers have a 1.5 times higher risk of having ED. Men with ED who smoke regularly have ultrastructural damage found in their body tissue, and 86% of smokers have abnormal penile vascular assessments. When compared to nonsmokers, smokers’ sperm density is found to be lower.57,58
In addition to being obviously associated with a higher risk of ED, smoking has an immediate detrimental effect on a man’s capacity to achieve and sustain a powerful erection. 62 According to a 1992 study by Hirshkowitz et al. 63 on 314 smokers with ED, smoking several cigarettes per day was linked to lower levels of a number of erectile function indicators, such as reduced penile stiffness and impaired penile blood pressure measurements. 63
In another study, Fasakin et al. revealed that the alkaloid extracts of N. tabacum significantly elevate the NO level and other sexual parameters at lower dosages (5 and 50 mg/kg bw) when compared with the control group. The extracts were also observed to inhibit DE-5 activities thereby reducing the hydrolysis of cGMP and enhancing prolonged erection. 24
In an in vitro study by Slotkin et al. 64 it was discovered that N. tabacum smoke extracts had about ten times the potency of nicotine in upsetting neurotoxicity, indicating that other N. tabacum constituents such as anabasine and polycyclic aromatic compounds, may also be involved in the neurotoxicity. Hence exposure to N. tabacum at higher doses or by heavy users (smokers) may affect the normal functioning of the brain and impair sexual functions.
Jimson Weed
D. stramonium belongs to the Solanaceae family. The plant originates from tropical America and is nearly everywhere throughout the world. In Nigeria, the plant is called Gegemu (Yoruba) or Zakami (Hausa) and is commonly called jimson weed, devil trumpet, angel trumpet or tear, thorn apple, or green dragon. 24 Typically, this plant is taken by eating the seeds, blossoms, or infusions made from the leaves or crushed seeds. Smoked forms of dried leaves, flowers, and seeds are also consumed.
Due to its effectiveness as a painkiller and sleep aid, D. stramonium has been used as a medicine since ancient times. The god Shiva’s dependence on D. stramonium and C. sativa smoking has also been reported.65,66 Jimson weed is used locally to treat a variety of illnesses and is used as a psychedelic to produce powerful visions. In the past, it was used to store components for love potions and witches’ brews. 67 Armed robbers have historically used D. stramonium combined with N. tabacum to sedate and induce anterograde amnesia in their robbery victims. 68
Due to the presence of the alkaloids scopolamine and atropine dispersed throughout the plant, D. stramonium also has hallucinatory effects when consumed. These substances pass the blood-brain barrier quickly because they are tertiary amines. While atropine has been seen to activate the CNS, scopolamine operates at the level of the CNS and has antimuscarinic effects (Table 1). 69 Psychomotor agitation, Tachypnea, pupillary dilatation, delirium, impaired vision, or photophobia are side effects of Jimson weed use. 70
Psychoactive Plants and their Effects on Male Sexual Function.
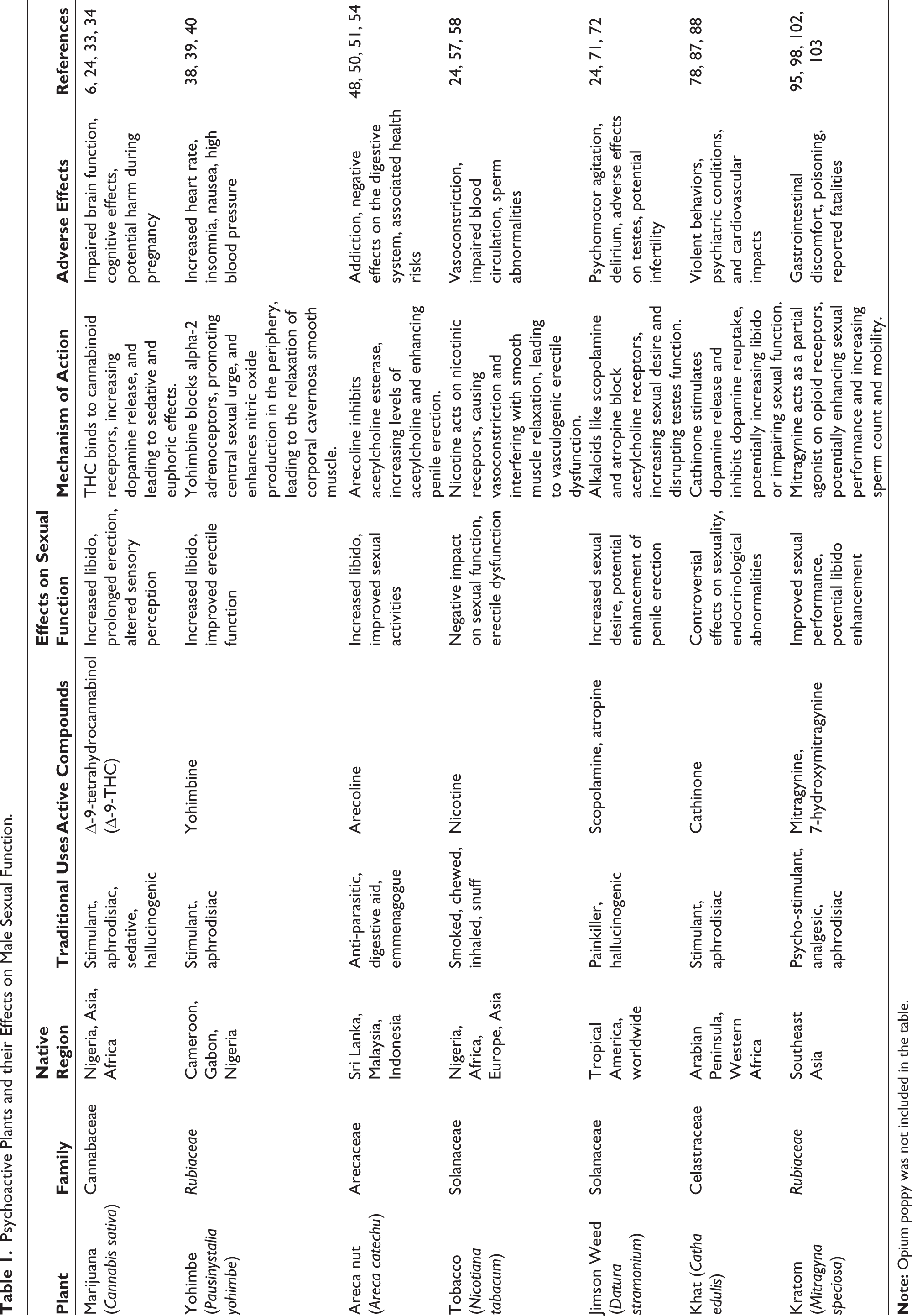
According to Enema et al. 71 D. stramonium arouses sexual desire. This may be due to the pharmacological activities of alkaloids present in the plant. These alkaloid extracts of D. stramonium significantly inhibit the activities of Acetylcholine esterase, the enzyme that hydrolyzes acetylcholine to choline and acetate, thereby increasing the levels of Acetylcholine and enhancing penile erection. In the same study, the dose-dependent extracts increase the activity of PDE-5, the enzyme that causes the rapid hydrolysis of cGMP to GMP thereby reducing penile erection and lowering sexual functions. 24
In another in vivo study, the smoke extracts of D. stramonium were observed to have adverse and severe effects on the testes of male Sprague Dawley rats. In the same study, the germ cell lining and the seminiferous tubules were disrupted and the degeneration of the Leydig cells was observed. The author concluded that the adverse effect observed resulted in a reduction of viable sperm cells and may lead to infertility. 72
High dosages of the plant have been known to cause human insanity and impair sexual function.24,73 Thus, the World Health Organization (WHO) identified D. stramonium as a significant contributor to mental illnesses and psychiatric illness. Even popular poisons for suicide and murder are made from it. 74 Scopolamine, hyoscyamine, and atropine, three tropane alkaloids that block the neurovegetative cholinergic system by acting competitively antagonistically on both central and peripheral muscarinic cholinergic receptors, have been found to be present in lethal concentrations in all plant parts. 75
Khat
C. edulis of the family Celastraceae is a native plant of the Arabian Peninsula and Western Africa, including Ethiopia. The plant is commonly called Khat, cat, qat among other names. The plant is usually administered or consumed by chewing the fresh leaves or smoking the dried plant materials. 49 Infusions of the leaves are used to cure asthma, coughs, and other respiratory conditions in southern Africa. 45 In South Africa, the Xhosa chew the leaves and twigs or prepare them as an infusion for its stimulating effects on the CNS. 76 Young leaves are chewed in East Africa and Arabia to reduce feelings of hunger and exhaustion and to benefit from their stimulating effects on the CNS. The phenethylamines in the fresh leaves, especially cathinone, which has amphetamine-like features, are what give them their stimulating effects. 77
Upon ingestion, Cathinone affects neurotransmission by causing presynaptic dopamine release and inhibiting dopamine reuptake in order to produce its euphoric effects. 78 Although cathinone has the highest affinity for norepinephrine receptors, it can also bind to dopamine and 5-HT receptor sites. Cathinone has also been demonstrated to stimulate serotonin release and prevent its reuptake.79,80
Male sexual potency has been linked to khat and its alkaloid cathinone.81,82 Reports on the relationship between khat use and sexuality are controversial. Khat, however, has reportedly also been used as a remedy for early ejaculation and as an aphrodisiac.83,84 On the other side, it has been noted that sexual function is impaired, erections cannot be maintained, libido is lost, and semen production is diminished. Khat users have been found to experience endocrinological abnormalities, including changes in their sex hormone levels.85,86
In an in vivo study by Abou-Elhamd et al. 87 100, 200, and 300 kg/bw ethanol extracts of Khat were administered to a group of male albino rats. The result of the study revealed that the sexual parameters are severely impaired especially at higher dosage when compared with the control group. In contrast, 100 mg/kg bw extracts of Khat were found to improve male sexual behavior while 200 and 400 mg/kg bw impaired sexual behavior in male rats in the same study. 88 Interestingly, these findings correlate with the findings of Nyongesa, 89 as well as Mohammed and Engidawork. 90 Ultimately, Khat can be said to have positive effects on sexual function at low dosage.
Heavy khat users, in particular, are more likely to experience violent behaviors as well as serious psychiatric conditions including paranoid psychosis and schizophrenia. Khat also negatively impacts the cardiovascular, endocrine, gastrointestinal tract, respiratory, and genito-urinary systems, causing sleeplessness, constipation, tachycardia, overall malaise, migraines irritability, and increased impotency in men.25,78,91
Kratom
Mitragyna speciosa Korth belongs to the Rubiaceae family and it is a native plant of Southeast Asia, especially in Malaysia and Thailand. In Malaysia, the plant is popularly called Kas Biak-Biak or Ketum and Kakuam, Kraton, Kratom, Thom or Ithang in Thailand. 25 With the vein removed, kratom leaves can be chewed fresh, dried, and taken as a powder with tea or coffee, or steeped and ingested as an herbal decoction. Its stimulating qualities have historically been sought after to improve difficult work under challenging seasonal conditions and to lessen exhaustion, pain, cough, fever, and diarrhea. It has also been used as a mild psycho-stimulant.92,93 When people cannot drink alcohol because of their religious views, they sometimes utilize kratom in cultural performances, tea shops, or as a substitute beverage. In addition to being utilized to enhance sexual performance, kratom is also used socially and recreationally.94,95
Indole alkaloids, particularly mitragynine and 7-hydroxymitragynine, which act as partial agonists on opioid receptors implicated in analgesia, are the primary active components of kratom. 96 The CNS inhibition of serotonergic 5-HT2A receptors and postsynaptic activation of α2 adrenergic receptors (α2R) may be the cause of mitragynine’s potential stimulant and libido-enhancing actions. 97 In a study, LaBryer et al. proposed that kratom may have improved the levels of prolactin, FSH, LH, and testosterone in a patient with hypogonadotropic hypogonadism. 98
Southeast Asia uses kratom leaves as an aphrodisiac. Male Kratom addicts in Thailand chew fresh leaves to prolong sex. 99 According to research by Ahmad and Aziz 95 as well as Vicknasingam, 100 consuming kratom tea or juice can improve sexual performance. An in vivo study reported that the administration of 100 and 200 mg/kg bw extracts of M. speciosa increases sperm mobility and sperm count in the treated mice. These effects could result from increased spermatogenesis, which is triggered by the hypothalamus-pituitary-gonad axis. 101 Interestingly, a clinical investigation study among 19 regular kratom users in Malaysia revealed that long-term consumption of kratom juice does not impair the male sexual hormone. 102 According to Singh et al. 103 72 participants out of a total of 92 kratom users, in a field survey in Malaysia reported that the consumption of kratom juice not only enhances sexual performance but also delays ejaculation, gives energy during sexual intercourse, Stimulate sexual desire, and help to maintain an erection during intercourse.
The use of kratom has been linked to a number of negative health outcomes, such as intrahepatic cholestasis, gastrointestinal discomfort, and poisoning or fatalities. 103 Heavy users have also mentioned experiencing side effects such as nausea, weight loss, exhaustion, constipation, sleeplessness, dry mouth, frequent urination, and cheek hyperpigmentation.104,105
Opium poppy
The opium poppy (Papaver somniferum), belonging to the Papaveraceae family, has garnered significant attention due to its pharmaceutical, ornamental, and nutritional applications. Over 2,500 distinct bioactive compounds have been identified in this plant, spanning various biochemical categories, many of which are utilized in pharmaceutical industries. Among these, opioids represent a major class of medicinally important compounds derived from the opium poppy, valued for their analgesic properties. Opioids act by binding to specific opioid receptors, including μ (mu), κ (kappa), and δ (delta) receptors, located predominantly in the central nervous system (CNS) and gastrointestinal tract, thereby modulating nociceptive transmission and producing pain relief. 106 Although some opioids can be synthesized chemically, several alkaloids, such as phenanthrenes (e.g., morphine, codeine, thebaine), benzylisoquinolines (e.g., papaverine), and phthalideisoquinolines (e.g., narcotine), are exclusively obtained from opium. These compounds place the opium poppy among the most significant medicinal plants globally. 107
The opium poppy is native to temperate and subtropical regions and is cultivated under strict legal control in countries such as India, China, and several European and South American nations.108-109 Licit cultivation is regulated globally by the International Narcotics Control Board, with oversight centralized in Vienna, Austria.
Opium and its derivatives, including heroin and methadone, exhibit profound effects on sexual function, mediated by their interaction with the hypothalamic-pituitary-gonadal (HPG) axis. Acute opioid use often induces a transient euphoria that may enhance sexual arousal and delay ejaculation in men; however, chronic use is strongly associated with sexual dysfunction, including reduced libido, erectile dysfunction, and anorgasmia. 110 These effects stem from the suppression of gonadotropin-releasing hormone (GnRH) secretion at the hypothalamus, leading to downstream reductions in luteinizing hormone (LH), follicle-stimulating hormone (FSH), and testosterone levels. 111 Chronic heroin use, for instance, significantly reduces free testosterone levels, impairing spermatogenesis and sexual performance, though normalization of hormonal profiles is observed following discontinuation. 111
Methadone and buprenorphine, often used in opioid substitution therapy, demonstrate differential impacts on sexual function. Methadone is associated with a higher prevalence of sexual dysfunction compared to buprenorphine, likely due to its greater suppression of HPG axis activity. 112 Ajo et al. 113 found that men receiving higher morphine-equivalent doses reported a greater prevalence of sexual dysfunction (33%) compared to women (25%), highlighting a dose-dependent relationship between opioid use and sexual health.
Tramadol, a synthetic opioid with mixed μ-opioid agonist and serotonin-norepinephrine reuptake inhibition properties, has been proposed as a treatment for premature ejaculation due to its ability to delay ejaculation latency. 114 However, its impact on broader sexual health remains controversial. Chronic tramadol use has been associated with reduced libido, hypogonadism, and anorgasmia.115 Furthermore, tramadol dependency carries risks of adverse effects, including tonic-clonic seizures, particularly when co-administered with alcohol.116
Psychoactive Plants and Sexual Functions
Male sexual function includes controlling penile hemodynamics, which is regulated by neurotransmitters, vasoactive substances, and endocrine components under the control of signal input from the central and peripheral neural systems. 106 The two synergistic neurophysiologic pathways that mediate reflexogenic erections and psychologically induced sexual stimulation, respectively, in the thoracolumbar region and sacral region, respectively, are in charge of controlling penile tumescence. Erections are caused by the corpora cavernosa’s smooth muscles relaxing, which increases the flow of blood to the penile region. 107 The release of a relaxing component originating from the endothelium, which has been identified as NO, is also assumed to govern the relaxation of the cavernosal smooth muscles. 108
Chemicals that affect the nervous system, sometimes known as psychoactive or psychotropic substances, have an impact on thought and behavior. They include substances that alter reality, such as hallucinogens, as well as sedatives and opioids, tranquilizers, anxiolytics, antidepressants, and stimulants that stimulate the mind.6,30 To produce different psychoactive effects, psychoactive substances interact specifically with neuronal receptors in the nervous system where they have distinct molecular targets. 109
In general, situations that lower brain serotonin or raise brain dopamine, or the opposite, encourage sexual behavior. The limbic system is the region of the brain that is most closely linked to sexual behavior.110,111 For instance, THC’s absorption into the body increases dopamine release, which has sedative effects and euphoric effects, 32 Cathinone affects neurotransmission by causing presynaptic dopamine release and inhibiting dopamine reuptake in order to produce its euphoric effects, arousal and attention-related cholinergic regions of the brain are stimulated by stimulants like nicotine, an alkaloid that mimics the endogenous neurotransmitter acetylcholine.78,109
The Mechanisms of Sexual Response in Males
Psychogenic cues, such as those that are auditory, visual, olfactory, gustatory, tactile, or creative, can cause erection. 112 These could activate sexual brain regions, most likely in the limbic system, sending signals to the sacral and thoracolumbar erection areas via the spinal cord. The reflexogenic stimulus, which results from tactile stimulation of the genital area or from impulses from the bladder or rectum, is the other erection-inducing factor. These produce impulses that mostly flow to the sacral erection center along the pudendal nerve. The autonomic nervous system balances the sympathetic (adrenergic) and parasympathetic (cholinergic) systems to regulate the body’s automatic operations.110,113 The two, corpora cavernosa and corpus spongiosum, vascular structures that are typically deflated, are filled with blood during an erection by the right and left internal pudendal arteries. When the penis is flaccid, blood is forced out of the corpora cavemosa and into the veins. 114
The autonomic nervous system has control over the shunting procedure and this happens when impulses from the thoracolumbar and sacral erection centers are received, increasing blood flow to the vascular spaces of the erectile tissues and resulting in an erection. 112 The erectile tissue veins contract, reducing venous outflow and increasing turgescence and might be managed by the sympathetic nervous system. It is conceivable that sympathetic nerve impulses cause erection by narrowing the penile arterioles that do not supply the erectile tissues with blood. 110
Conclusion
A psychoactive plant is a plant capable of modulating or affecting the mind or the mental processes. When administered at a certain dose, these plants elicit their aphrodisiac effect by improving the sex hormone, sexual enthusiasm, prolonged duration, better desire, and alertness. The indole alkaloids in these plants often triggered a cascade of reactions that resulted in neurotransmitter release mediating an erection. The thoracolumbar region and sacral region of the brain are the two synergistic neurophysiologic pathways that mediate reflexogenic erections and control penile tumescence. Due to the detrimental effects of psychoactive plants on human health when consumed, they should be discouraged as an aphrodisiac agent as they can result in mental illness and organ damage.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Ethical permission was not applicable to this article, as this is a review article drafted from various research articles and not from patients directly.
Funding
The authors received no financial support for the research, authorship and/or publication of this article
Informed Consent
All the authors contributed substantially to the work, partici-pated in the writing, and have seen and approved the submitted version.
