Abstract
Objective:
Sexual dysfunction amongst persons with opioid use disorder (OUD) is quite prevalent. Research about the effects of abstinence from opioids on sexual functioning is scarce. The aim of this study is to assess the sexual functioning among men with OUD and examine the effects of abstinence from opioids on sexual functioning.
Methods:
An observational study was conducted at a tertiary-level multi-specialty public-funded hospital in northern India. A total of 50 men with OUD underwent a self-report–based assessment of sexual functioning using the International Index of Erectile Function (IIEF-15). Five sub-domains of IIEF-15 (i.e., erectile function, orgasmic function, sexual desire, intercourse satisfaction and overall satisfaction) were assessed at baseline, two weeks and eight weeks of abstinence from opioids.
Results:
A total of 44 participants out of 50 reported significant dysfunction. Most of them reported dysfunction in the domains of overall satisfaction (88%,
Conclusion:
Abstinence from opioids leads to improvement in sexual functioning over a period of eight weeks of abstinence in men with OUD.
Introduction
Sexual dysfunction amongst persons with opioid use disorder (OUD) is quite prevalent. Most studies assessing the epidemiology of sexual dysfunction have reported the prevalence rates for sexual dysfunction in patients with OUD to range between 48% and 92%.1,1 Research available from Western countries is predominantly focused on the sexual side effects of opioid substitution therapy.1,1 A cross-sectional study from northern India exploring sexual dysfunction in men seeking treatment for opioid dependence reported the prevalence of significant dysfunction in various domains of sexual functioning, such as sexual desire (41%–86%), arousal (29%–78%), erectile dysfunction (36%–45%), orgasm (21%–76%), overall satisfaction (74%) and sexual pleasure (90%). 1 But still, the assessment and management of sexual dysfunction during treatment of OUDs in patients are often overlooked or neglected. As per the available research, sexual dysfunction was reported in about 34%–85% of patients with OUD who were actively using opioids, about 14%–81% of abstinent patients on methadone maintenance treatment, about 36%–83% of abstinent patients on buprenorphine maintenance treatment and about 90% of abstinent patients on naltrexone maintenance treatment. 5 Persons with OUD have a 2 to 3.4 times higher risk of decline in sexual functioning than the general population. 6 Though there is sufficient literature reporting a significant association between opioid use in a dependent manner and sexual dysfunction in them, there is a lack of research examining the effects of varying periods of abstinence from opioids on sexual functioning in this population. To the best of our knowledge, there has been no longitudinal study conducted till now which has evaluated the effect of abstinence from opioids on sexual functioning among men with OUD. The index study was carried out with the aim of assessing sexual functioning among men with OUD and examining the effects of abstinence from opioids on sexual functioning.
Materials and Methods
This was an observational study conducted in the outpatient, community clinics and emergency departments of psychiatry at a tertiary-level multi-specialty public-funded hospital located in northern India. Participants were recruited through convenience sampling. Ethical approval was obtained from the institutional ethics committee prior to the beginning of recruitment for the study and registered with the Clinical Trial Registry of India (CTRI/2021/05/033633, approved on 17 May 2021). The study sample consisted of 50 male participants in the age range of 18–55 years fulfilling the diagnostic criteria for OUD as per the
Statistical Analysis
Data was analysed using the Statistical Package for the Sciences Software (SPSS, version 22.0).
9
Continuous variables were analysed using the mean and standard deviation (SD). Categorical variables were analysed using frequency tabulation. A repeated measures analysis of variance (ANOVA) test was applied to examine the change in IIEF-15 scores (i.e., at baseline, two weeks and eight weeks) over the course of the study. All the test results were considered statistically significant at a
Results
Out of the 60 participants screened for this study, 50 completed the baseline and follow-up assessments and were included in the analysis. A total of nine patients were excluded; seven patients dropped out due to their inability to maintain abstinence during the first two weeks itself and two patients reported their inability to follow up for assessments during the intake interview. The socio-demographic and clinical profiles of the study population have been described in Table 1. The mean age of the participants was 27.44 (standard deviation: 6.43) years. About four-fifths of the study population (
Socio-demographic and Clinical Characteristics of the Study Population (

aMean ± standard deviation (in years); bFrequency (percentage); cSeverity of opioid dependence questionnaire.
Frequency of Sexual Dysfunction on the IIEFa-15 Scale in Three Assessments (
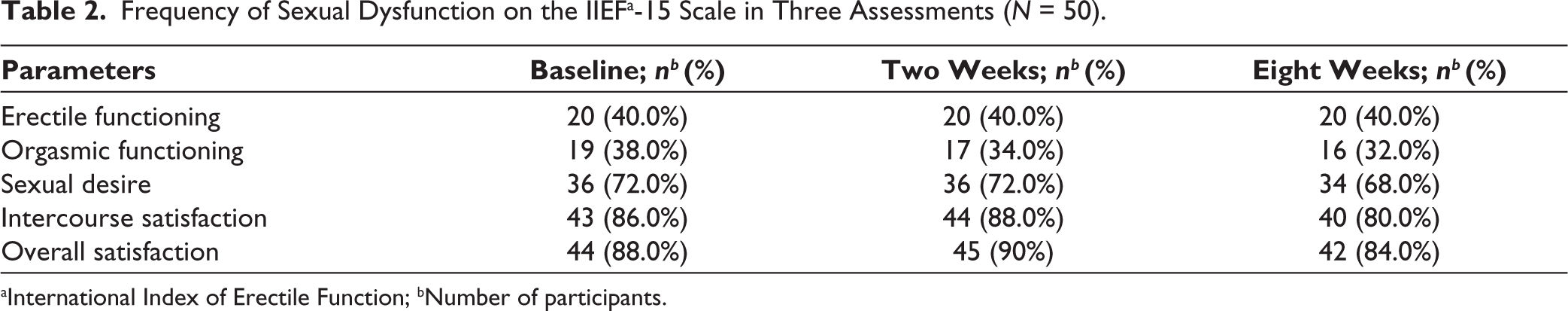
aInternational Index of Erectile Function; bNumber of participants.
Comparison of Scores on the IIEFa-15 Scale at Three Assessments (
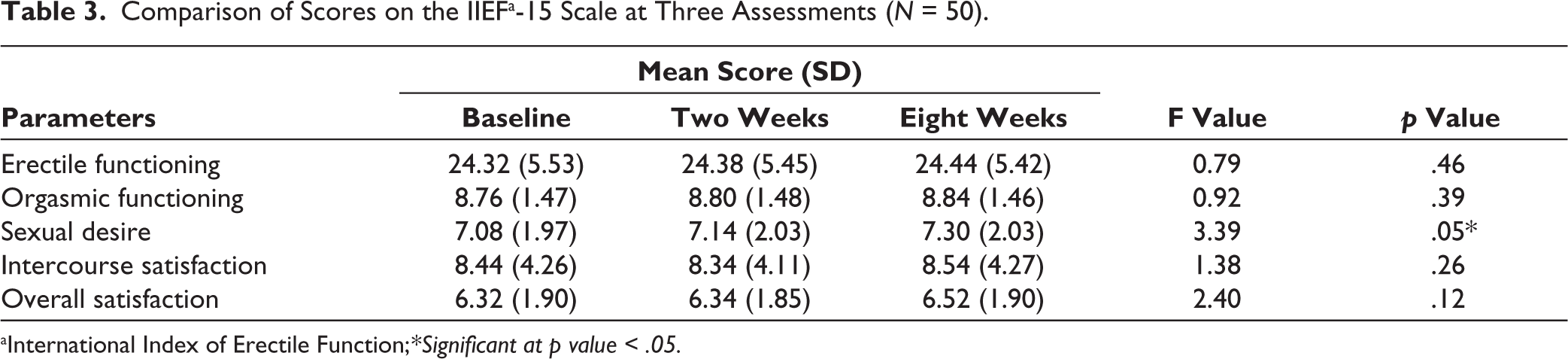
aInternational Index of Erectile Function;
Over the period of eight weeks, it was noted that a small number of participants had a decline in scores of erectile function (8%), orgasmic function (2%), sexual desire (8%), intercourse satisfaction (8%) and overall satisfaction (16%) as compared to baseline. A significant negative correlation was found between age (in years) and scores for erectile function, orgasmic function, sexual desire and overall satisfaction at baseline (all
Discussion
The prevalence of sexual dysfunction reported by men with OUD in our study was between 38% (for orgasmic functioning) and 88% (for overall satisfaction with sexual activity), depending upon the area of sexual functioning under assessment. The widespread reporting of dysfunction in the overall satisfaction (88%) and intercourse satisfaction (86%) domains of sexual functioning by about four-fifth of the study population in this study was similar to that reported in a few previous studies conducted among patients with OUD from India.1,1,1 A study by Venkatesh and colleagues reported significant dysfunction in intercourse satisfaction and overall satisfaction (74% each) domains on the IIEF scale, which was higher than the prevalence reported in the other three domains on the IIEF scale. 1 During the initial two weeks of abstinence, there was no improvement, but rather some worsening reported by a few study participants. An increased proportion of participants reported significant dysfunction at two weeks of abstinence in the overall satisfaction (90% vs. 88%) and intercourse satisfaction (88% vs. 86%) domains of sexual functioning when compared to the baseline. This could be attributed to the effects of the acute withdrawal phase on sexual functioning. However, after eight weeks of abstinence, either a reduction or no change was reported in the proportion of study participants who reported significant dysfunction across all five domains of sexual functioning assessed using the IIEF-15 scale in this study. There was significant improvement in the IIEF-15 sub-domain score representing sexual desire over eight weeks of abstinence. These findings describing the effect of the duration of abstinence from opioids on sexual functioning in men with OUDs provide an important contribution to our understanding of this important area. To the best of our knowledge, no prior study from India had reported the effect of abstinence from opioids on sexual functioning in persons with OUD. A study conducted in China reported a similar significant improvement in all domains of IIEF among patients with opioid dependence who were maintaining abstinence from illicit opioids while being on methadone maintenance therapy for at least one month duration. 11 Though our study participants were abstinent from opioids and did not receive any substitution or maintenance treatment with methadone or buprenorphine, unlike that reported in the previous study, this is important since the available literature on the effects of maintenance treatment with methadone or buprenorphine on sexual functioning in patients with OUD has been mixed, with both positive and negative effects reported.1,1 Further, the majority of the participants reported either improvement or no change following abstinence from opioids in sexual functioning assessed in the present study. The negative correlation between age and sexual functioning of participants was in line with a previous meta-analysis published in 2014, which identified eight risk factors for sexual dysfunction, namely age, medical and psychiatric co-morbidities, hormone assays, duration of treatment, methadone dose, other current substance use and family status. 14 The index study has the strengths of using the self-administered scale for sexual functioning on three different point periods, negative urine sampling to ensure abstinence and excluding patients with other substance use disorders and major mental illnesses. The index study also had certain limitations in the form of using purposive sample of treatment-seeking population comprising exclusively of male gender and a short duration of follow-up, hence caution should be exercised while generalising it to the wider population.
Conclusion
Abstinence from opioids leads to improvement in all five domains of sexual functioning on the IIEF-15 scale, with statistically significant improvement in the sexual desire domain over a period of eight weeks. Future research needs to be done in a more representative community sample comprising participants from both genders with different treatment modalities, that is, opioid substitution therapy versus naltrexone, over a longer follow-up period.
Supplemental Material
Supplemental material for this article is available online.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
The ethics approval was duly obtained from institutional ethics committee of Government Medical College and Hospital, Chandigarh . Approval ID: GMCH/IEC/2020/458/38 and trial was registered with the Clinical Trial Registry of India (CTRI/2021/05/033633, approved on 17 May 2021).
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed Consent
Informed consent was obtained from study participants.
