Abstract
The assessment of opioid withdrawal symptoms is common in both clinical and research settings. The Clinical Opiate Withdrawal Scale (COWS) is among the most frequently used instruments for the assessment of signs and symptoms associated with opioid withdrawal. The COWS is a validated, clinician-administered instrument initially developed and validated for English-speaking populations. To date, however, the COWS has yet to be linguistically and culturally adapted for French-Canadian populations.
Objective
The main objective of the present study was to develop a French-Canadian translation and adaptation of the COWS (i.e., the COWS-FC) for the assessment of opioid withdrawal symptoms in clinical and research settings.
Methods
The French-Canadian translation and cultural adaptation of the COWS was performed following guidelines for the translation and cross-cultural adaptation of self-report measures. The steps consisted of (1) initial translation from English to French, (2) synthesis of the translation, (3) back-translation from French to English, (4) expert committee meeting, (5) test of the prefinal version among healthcare professionals and (6) review of final version by the expert committee. The expert committee considered four major areas where the French-Canadian version should achieve equivalence with the original English-version of the COWS. These areas were (1) semantic equivalence; (2) idiomatic equivalence; (3) experiential equivalence and (4) conceptual equivalence.
Results
Rigorous steps based on the guidelines for the translation and cultural adaptation of assessment tools were followed, which led to a semantically equivalent version of the COWS. After a pretest among healthcare professionals, members from the expert committee agreed upon slight modifications to the French-Canadian version of the COWS to yield a final COWS-FC version.
Conclusions
A French-Canadian translation and adaptation of the COWS (i.e., the COWS-FC) was developed. The COWS-FC could be used for the assessment of opioid withdrawal symptoms in clinical and research settings.
Introduction
Opioids refer to a class of natural, synthetic or semi-synthetic drugs that bind to opioid receptors. Opioids are commonly used for medical reasons (e.g., pain relief), but they are also used illicitly for recreational reasons. Whether used for medical or recreational reasons, it is well recognized that the prolonged use of opioids may lead to physical dependence due to opioid-induced neurobiological adaptations in the central nervous system.1–5 Signs of physical dependence on opioids are typically manifested by opioid withdrawal symptoms following cessation of opioids, decreases in opioid blood levels or administration of an opioid antagonist.2,3,6–8 Opioid withdrawal may be experienced with varying intensity and may include signs and symptoms such as tremors, sweating, yawning, piloerection, lacrimation, rhinorrhea, nausea, achiness, restlessness and dysphoria.2,3,6–8 Opioid withdrawal symptoms are mainly observed among illicit opioid users presenting with opioid use disorder (OUD),7–11 but transient opioid withdrawal symptoms may also be experienced towards the end of dosing intervals among patients prescribed daily opioid therapy,2,12–18 or when patients undergo opioid dose reduction.19–23
The assessment of opioid withdrawal symptoms is common in both clinical and research settings. In the clinical settings, the assessment of opioid withdrawal symptoms is routinely performed for diagnostic or treatment planning purposes3,7,24,25 as well as in the context of buprenorphine–naloxone induction.26–30 It is also important to assess opioid withdrawal symptoms because they lead to psychological distress and contribute to decreased quality of life.18,31–34 In research settings, opioid withdrawal symptoms are commonly assessed in studies testing the effectiveness of pharmacological26,35,36 and non-pharmacological37–39 interventions for individuals with OUD.
A number of instruments have been developed and validated for the assessment of opioid withdrawal symptoms, including the Opiate Withdrawal Scale (OWS 40 ), the Objective Opiate Withdrawal Scale 41 , the Subjective Opiate Withdrawal Scale 42 , the Short Opiate Withdrawal Scale (SOWS-G 43 ), the Subjective Opiate Withdrawal Questionnaire (SOWQ 44 ), the Adjective Rating Scale for Withdrawal (ARSW 45 ) and the Clinical Opiate Withdrawal Scale (COWS 46 ). To date, however, none of these instruments have been linguistically or culturally adapted for French-Canadian populations. Among these instruments, the COWS is one of the most commonly used. The COWS is a validated, clinician-administered instrument that can be used to quantify the frequency and severity of eleven common signs and symptoms associated with opioid withdrawal. It was developed for American English-speaking populations in the early 2000s by Wesson and Ling 46 and later validated by Tompkins and colleagues. 9 Because of its good psychometric properties, clinical utility and ease of application, the COWS has become widely used for the assessment of opioid withdrawal symptoms, both in clinical and research settings.47–52
The main objective of the present study was to develop a French-Canadian translation and adaptation of the COWS (i.e., the COWS-FC) that can be used for the assessment of opioid withdrawal symptoms in clinical and research settings. As recommended, 53 linguistic translations of instruments also need to be culturally adapted to the target population. Given that up to seven million of Canadians report French as their first official spoken language, 54 there is a need to develop a culturally adapted version of the COWS that could be used by French Canadians in the province of Quebec as well as in other Canadian provinces and territories.
Methods
Original Instrument: The COWS
The COWS was first published as a training manual for OUD treatment with buprenorphine. 46 It was developed by Wesson and Ling in response to increased rates of opioid prescription among pain patients, as well as a gain in popularity of sublingual buprenorphine in the United States. It was designed as a clinician-administered instrument that rates 11 common opioid withdrawal signs and symptoms 46 and took into consideration that each sign and symptom may occur along a spectrum, as reflected by the use of graded responses. The COWS consists of 11 items, including 1 subjective symptom item, 6 objective sign items and 4 items that included subjective and objective components. The final score is a sum of all items, with the following cut-off points: 5–12 = mild; 13–24 = moderate; 25–36 = moderately severe; more than 36 = severe withdrawal. The scale was validated in a sample of 46 out-of-treatment opioid-dependent volunteers. 9 The initial validation revealed a good internal consistency, with a Cronbach α of 0.78. 9
The COWS was specifically selected by our expert committee among the various scales for linguistic and cultural adaptation given its psychometric properties, its previous validation as an instrument for the assessment of withdrawal symptoms among various populations of opioid users,55–62 as well as for its ease of administration by clinicians.
Procedures
The translation and cultural adaptation of the COWS was performed following guidelines for the translation and cross-cultural adaptation of self-report measures published by Beaton and colleagues. 53 All the translation and cultural adaptation procedures are described below.
Step 1: Initial (French) translation
The first step of the translation process was the forward translation from the original language (i.e., English) to the target language (i.e., French-Canadian) by two bilingual individuals from the province of Quebec, speaking French-Canadian as their native language. These two translators had the ability to speak both languages with the facility of a native speaker. The first translator was an individual working in the field of pain and opioids (T1), and the second translator was a certified French linguist with no biomedical background and without any a priori knowledge of the concepts being measured (T2). Each individual involved in the translation process produced a Canadian French version of the COWS and a detailed report summarizing the rationale for their choices of words, as well as comments regarding potentially challenging phrases and concepts.
Step 2: Synthesis of the translations
A synthesis of the two translated versions (from T1 and T2) was made following the independent translations of the COWS. The two independent translators and the research coordinators met through a video conference platform to examine the results of the first step and to reconcile any discrepancies. At the end of the meeting, the team agreed upon a common French-Canadian version and provided a unified preliminary version of the COWS.
Step 3: Back translation
The third step consisted of a back-translation of the questionnaire (i.e., from French to English) by bilingual translators from Canada, speaking English as their native language. One of the back-translators (BT1) was an individual working in the field of pain and opioids, and the other back-translator (BT2) was a certified English linguist with no biomedical background and/or knowledge of the field. Both individuals were blinded to the original version of the COWS. As in the first step, each individual involved in the back-translation process generated an individual back-translated version of the COWS accompanied by a detailed report of their rationale for the choice of words and comments regarding potentially challenging phrases or concepts.
Following the back translation, a synthesis meeting was held between the two individuals involved in the back translation and the research coordinator to compare the two back-translated versions and to reconcile any discrepancies. At the end of the synthesis meeting, a global report was created, which included the original English version of the COWS, the two French-Canadian versions, and the reports of the forward translators, the unified T-12 version, the two English back translations, and the reports from the back translators.
Step 4: Expert committee
The fifth stage involved an expert committee composed of healthcare professionals (i.e., physicians, nurses and clinical psychologists), researchers working in the field of opioids and research coordinators. The global report of the preceding step (i.e., Step 3) was sent to all committee members before the meeting. A teleconference (using a web-based screen-sharing system) was then organized with the objective of generating a prefinal French-Canadian version of the COWS. As recommended by Beaton et al., 53 the experts considered four major areas where the French-Canadian version should achieve equivalence with the original English version of the COWS. The areas are (1) semantic equivalence (i.e., the meaning of words); (2) idiomatic equivalence (i.e., the meaning of expressions or idioms); (3) experiential equivalence (i.e., the adaptation or replacement of items that are specific to the target culture); and (4) conceptual equivalence (i.e., the definition of concepts that are common to both cultures).
Step 5: Test of the prefinal version
For the fifth step, the prefinal French-Canadian version of the COWS was pretested in a sample of 28 healthcare professionals working in francophone university-affiliated hospitals in the province of Quebec (see Table 1). The healthcare professionals that were recruited for the pretest were clinician working in the area of chronic pain or substance use (anesthesiologists, nurses, psychiatrists, pharmacists, family physicians). The healthcare professionals also had to be aged 18 years old or above and speak French as their native language. All procedures related to the pretest were approved by the Ethics Review Board of the Centre hospitalier de l’Université de Montréal.
Characteristics of the Health Care Professionals Participating in the Study (N = 28).
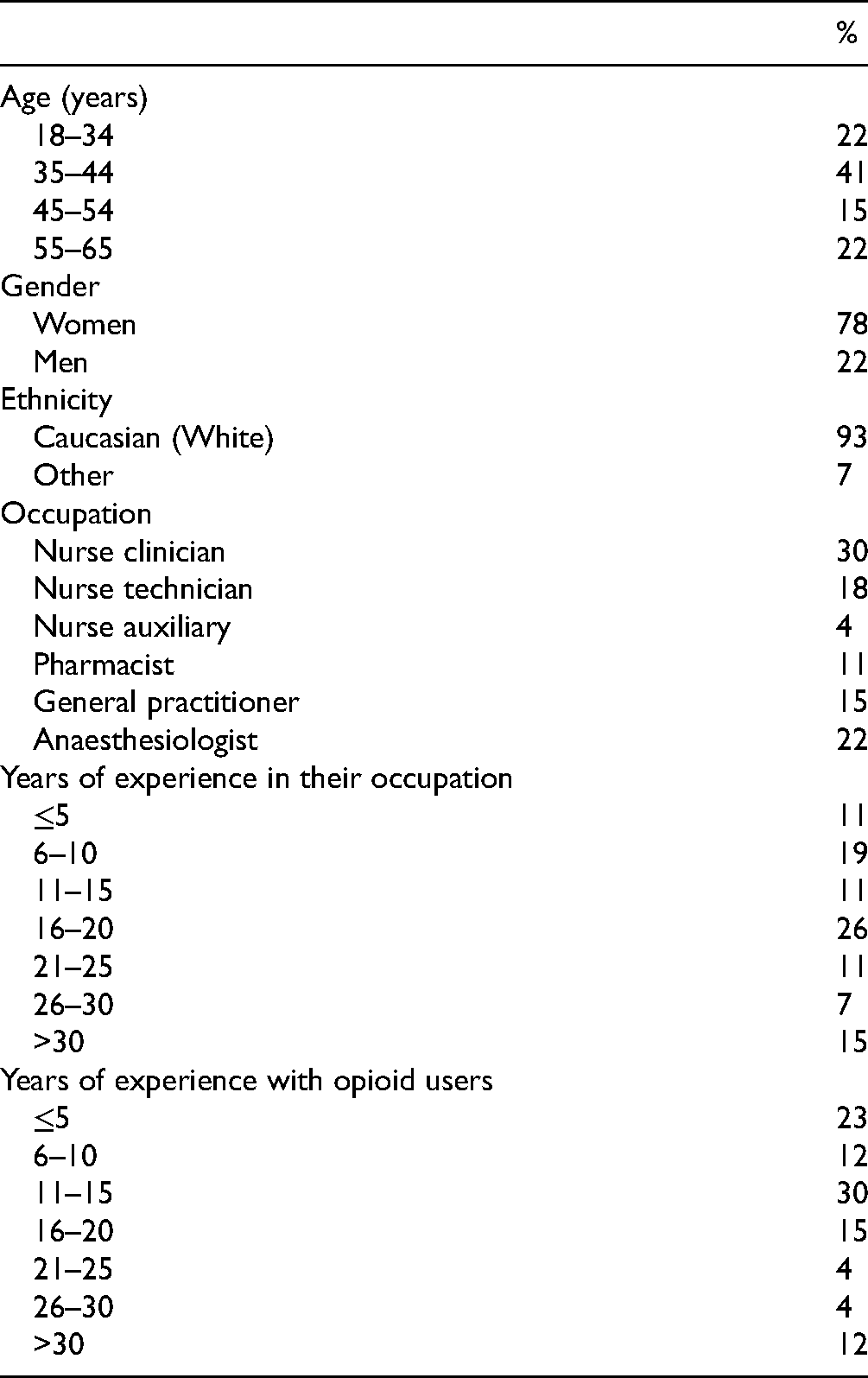
Between October 2019 and December 2019, the convenience sample of healthcare professionals was contacted through institutional emails and invited to participate in the pretesting step, which took place through a web-based questionnaire. After providing consent, healthcare professionals were asked to read all instructions and items from the prefinal French-Canadian version of the COWS. They were then asked to provide open-ended comments regarding the clarity of each item. After providing comments on the prefinal French-Canadian version of the COWS, participants (i.e., healthcare professionals) were asked to provide sociodemographic information (i.e., age, gender, ethnicity) as well as information about their occupation and years of experience working with opioid users. The pretest took approximately 15 minutes in total, and no financial compensation was offered.
Step 6: Final version
During the final step, members from the expert committee agreed upon slight modifications to the French-Canadian version of the COWS after reviewing the results obtained in the pretest (see Table 2). The final version of the COWS-FC is presented in Online Appendix 1.
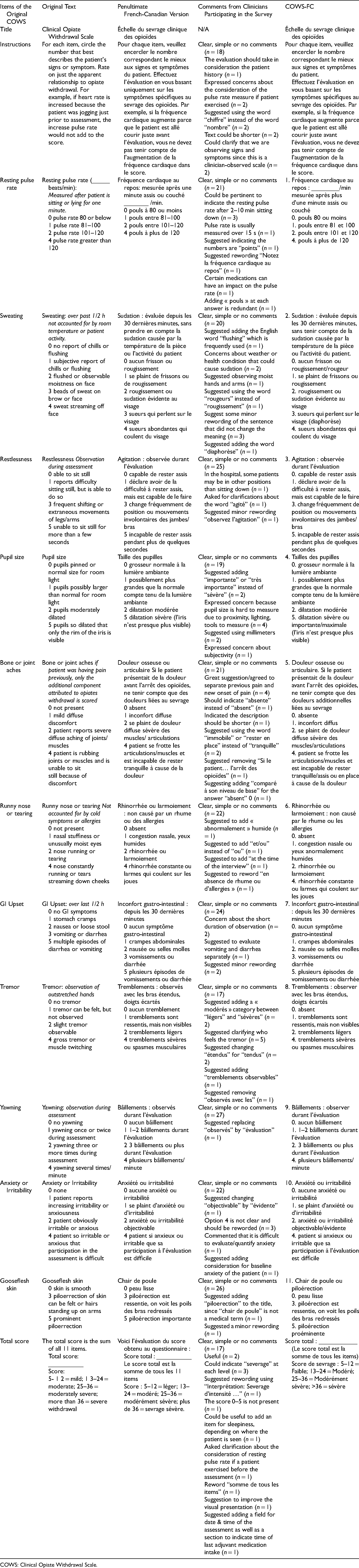
Overview of Results from the Pretest and Subsequent Changes Made to Yield the COWS-FC.
COWS: Clinical Opiate Withdrawal Scale.
Results
All the steps (i.e., Steps 1–6) involved in the French-Canadian translation and cultural adaptation of the COWS took place between June and November 2019. Descriptive statistics for the sample of healthcare professionals who participated in the pretest of the COWS-FC are presented in Table 1. The majority of participants were women (78%), aged between 35 and 44 years (41%), and Caucasians (93%). Among these participants, nursing (52%) and medicine (37%) were the most frequent healthcare disciplines, and close to 90% of participants reported having worked with opioid users for more than 5 years.
Table 2 presents an overview of the results from the pretest (i.e., Step 5) conducted among healthcare professionals. The minor adjustments that were made to yield the final French-Canadian version of the COWS (i.e., COWS-FC) are also described in Table 2. Results from the pretest indicated that the vast majority of items from the prefinal version of the COWS-FC were clear and well understood by participants (i.e., healthcare professionals). However, based on participants’ suggestions, minor adjustments to certain words or statements were made to enhance clarity. For instance, this occurred for item # 4 (adding the words “rougeurs” and “diaphorèse”), item # 10 (adding the words “assis en place”) and item # 22 (adding the word “piloérection”). The wording of item # 12 in the final version was also slightly modified to more accurately represent one of the response choices (i.e., by adding the word “anormalement”). For the “total score” section, three participants suggested adding the word “sevrage” to the scoring legend, which was done in the final version to enhance clarity.
Some participants (n = 2) suggested adding one response choice (i.e., “modérés”) to item # 16, but this suggestion was not retained because it would have altered the scoring system implemented in the original version of the COWS. Similarly, suggestions were made to add additional items (e.g., an item assessing sleep disturbance and an item assessing the date/time of last adjuvant medication intake), but these suggestions were not retained because these items were not included in the original version of the COWS and this would have altered the original scoring system.
Discussion
The main objective of the present study was to develop a French-Canadian translation and adaptation of the COWS (i.e., the COWS-FC) for the assessment of opioid withdrawal symptoms in clinical and research settings. A number of instruments have previously been developed and validated for the assessment of opioid withdrawal symptoms, but none have been culturally adapted for French-Canadian populations. Opioid withdrawal symptoms are frequently assessed both in clinical and research settings, and there was a need to develop a culturally adapted version of the COWS that could be used by French Canadians.
Because of its psychometric properties, ease of application and clinical utility, the COWS has become widely used for the assessment of opioid withdrawal symptoms among various populations of opioid users.47–52 Results from the pretest conducted among healthcare professionals with experience working with opioid users showed that the French-Canadian version of the COWS was generally well understood by participants, both in terms of general instructions and item wording. Given the very few adjustments that needed to be made following the pretest, it is unlikely that the clarity and understandability of items from the COWS-FC meaningfully varied as a function of healthcare professionals’ demographic or occupational characteristics. Finally, the pretest did not reveal any specific challenge that would need to be addressed. Overall, the pretest led only to minor modifications to the wording of certain items to enhance clarity, without any alterations to the meaning of items included in the original (i.e., English) version of the COWS. The comparability and absence of any meaningful differences between the original English and French-Canadian versions of the COWS is expected to be useful not only for clinical purposes but also to facilitate comparisons across studies involving the assessment of opioid withdrawal symptoms using the COWS.
Strengths and Limitations
Rigorous steps based on the guidelines for the translation and cultural adaptation of assessment tools were followed, which led to a semantically equivalent version of the COWS. However, future studies that will use the COWS-FC should report its psychometric properties and compare them with those from the original version. Second, data from the pretest were collected among healthcare professionals from various disciplines having worked with opioid users for several years, but the distinction between healthcare professionals’ experience working with medical or nonmedical users was not made. It is unlikely that this distinction would have impacted healthcare professionals’ understandability of the COWS-FC, but this needs to be considered when interpreting results from the pretest. Finally, there are limitations associated with the initial version of the COWS that need to be considered. For instance, although the COWS has been used in clinical and research settings among patients prescribed opioids,47,61,63–65 it was primarily developed and validated for illicit opioid users. Further studies are needed to test the psychometric properties of the COWS among patients using opioids for medical reasons.
Despite these limitations, this study led to a linguistically and culturally adapted version of the COWS that is expected to be useful for the assessment of opioid withdrawal symptoms by French-Canadian populations. The linguistic and cultural adaptation conducted in the present study has broad applicability given that the COWS-FC is frequently used in both clinical and research settings. Clinicians and researchers working in francophone settings across Canada should now consider using the COWS-FC for the assessment of opioid withdrawal symptoms.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437221087066 - Supplemental material for French-Canadian Translation and Cultural Adaptation of the Clinical Opiate Withdrawal Scale: The COWS-FC
Supplemental material, sj-docx-1-cpa-10.1177_07067437221087066 for French-Canadian Translation and Cultural Adaptation of the Clinical Opiate Withdrawal Scale: The COWS-FC by Alice Bruneau, Clarice Poirier, Mélanie Bérubé, Aline Boulanger, Céline Gélinas, Line Guénette, Anaïs Lacasse, David Lussier, Yannick Tousignant-Laflamme, M. Gabrielle Pagé, Marc O. Martel and in The Canadian Journal of Psychiatry
Footnotes
Authors’ Note
Members of the Quebec Pain Research Network Strategic Initiative on Opioids are (in alphabetical order): Archambault Léonie, Beaudet Nicolas, Beaudry Hélène, Bérubé Mélanie, Boulanger Aline, Bruneau Julie, Choinière Manon, Gauthier Lynn, Gélinas Céline, Gendron Louis, Guénette Line, Lacasse Anaïs, Lavigne Gilles, Lussier David, Martel Marc O., Pagé M. Gabrielle, Perreault Michel, Pinard Anne Marie, and Tousignant-Laflamme Yannick.
Acknowledgements
The authors would like to thank members from the Addictions Unit of the Centre hospitalier de l’Université de Montréal (CHUM), who provided input on our work and facilitated the translation and cultural adaptation process of the COWS. The authors also thank healthcare professionals who took the time to participate in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Quebec Pain Research Network (Opioid Strategic Initiative), a network funded by the Fonds de recherche du Québec-Santé (FRQ-S).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
