Abstract

Introduction
Vision realised
In 2000, a pivotal short paper in Nature 1 described the successful use of a wireless capsule to image the gastrointestinal (GI) tract, marking the birth of a new diagnostic era. The idea of swallowing a miniature camera capable of traversing the length of the small bowel (SB) was revolutionary at that time. Professor Paul Swain, a central figure in this development, had long envisioned overcoming the ‘black box’ of the GI tract. His vision and clinical knowledge, combined with the engineering ingenuity of Gavriel Iddan et al., led to the creation of the first capsule endoscope (CE). 2 Before this innovation, examining the SB was technically challenging, and panendoscopy of the small intestine was rarely possible. Clinicians were limited to radiologic studies with low yield or invasive enteroscopy, with associated morbidity. CE emerged as a technical innovation and a paradigm shift, allowing direct, high-resolution and non-invasive visualisation of the whole length of the small intestine for the first time.
From concept to clinical tool – Clinical impact and standardisation
Initial trials of CE were conducted with caution, prioritising proof-of-concept and safety. This prudent approach was exemplified by Given Imaging Ltd (Yokneam Illit, Israel), which strategically provided system access and capsules to key opinion leaders in gastroenterology worldwide, enabling them to conduct independent studies and gain first-hand experience with the technology and procedure. However, by 2001, with the presentation of the first study demonstrating the capsule’s meaningful contribution in the evaluation of suspected SB bleeding, the U.S. Food and Drug Administration (FDA) approved the first CE system for clinical use. 2 Despite initial scepticism from some within the gastroenterology community, the clinical impact of CE soon became undeniable, particularly in the evaluation of suspected SB bleeding, iron deficiency anaemia (IDA) and suspected Crohn’s disease (CD). Moreover, it is now being used in newly diagnosed CD patients to assess the true extent of the disease, and in some countries, reimbursed for use after biologic treatment, to assess mucosal healing.
The use of CE quickly expanded.3,4 Three versions of the original SB capsule have been developed with a longer battery life, wider angle of view and adaptive frame rate, making it perform better. Competitive SB capsules were approved as well. The capsule’s use quickly expanded beyond the small intestine. Within a decade, two subtypes of an oesophageal and colonic capsule platforms were developed. The oesophageal capsule was proven to be useful in diagnosing Barret’s oesophagus and oesophageal varices5–7 in a relatively short, simple and patient-friendly procedure, but due to economic reasons, it has disappeared from the market. The colonic variant, though technically more challenging due to the need for longer battery life, rapid transit times in some colonic segments, and bowel preparation requirements, extended capsule technology’s role in colorectal cancer (CRC).8–10 The second-generation colon capsule endoscope (CCE) with its dual cameras, adaptive frame rate up to 35 frames/s, and 172° angle of view, performed significantly better than the first-generation capsule 11 and as good if not better than virtual computed tomography (CT). 12 Over the last few years, magnetically guided capsules have been developed for use in the upper GI tract – mainly oesophagus and stomach – and a panenteric capsule was released for use, at this stage aimed at CD/inflammatory bowel disease (IBD). 13 (Figure 1).
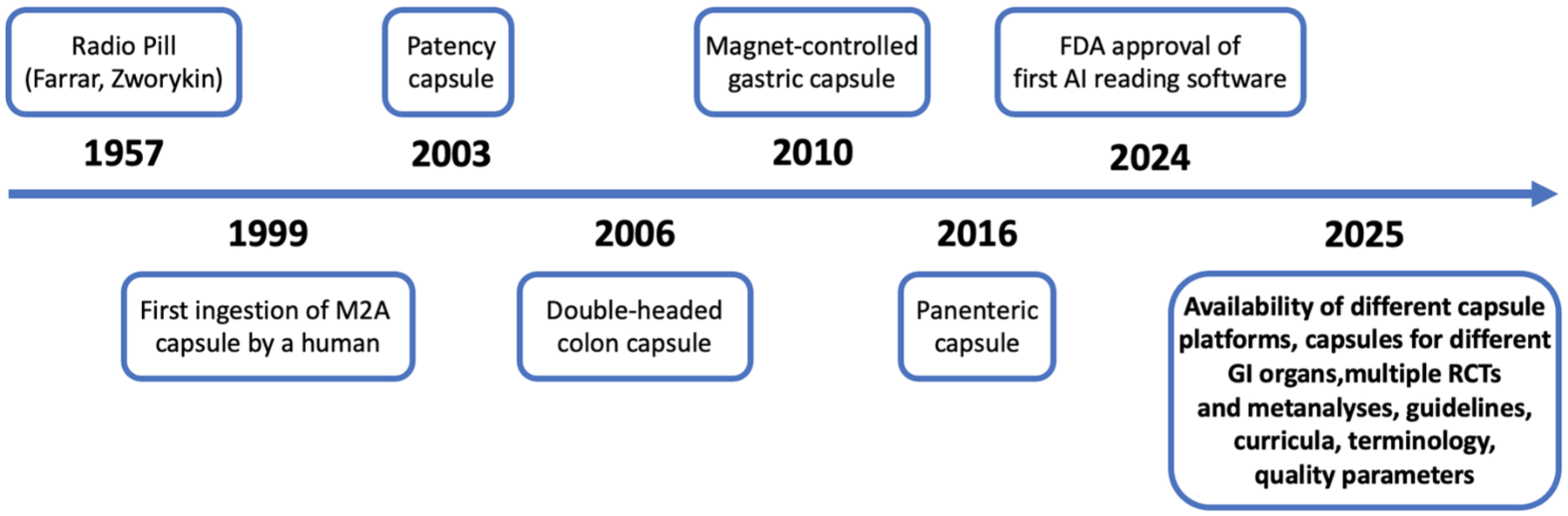
History timeline of the development of capsule endoscopy.
CE’s high diagnostic yield in these conditions has led to its integration into national and international guidelines. The European Society of GI Endoscopy (ESGE) and the American Society for Gastrointestinal Endoscopy (ASGE) have endorsed CE as a first-line investigation for SB evaluation in selected clinical scenarios.3,4 Direct access to the entire length of the small intestine has made it possible, as never before, to approach initially ill-defined anomalies. These intestinal drones have made almost commonplace lesions that until a few years ago were reserved for rare, uncertain, invasive diagnoses. In the early 2000s, a consortium of leading experts therefore came together to develop a common terminology and language under the name of CEST (Capsule Endoscopy Structured Terminology). 14 This approach has continued to be refined, culminating in the 2020s in semantic descriptions and atlases of vascular, 15 inflammatory 16 and atrophic 17 lesions via Delphi processes. This step came along with the establishment of a widely used score of pertinency (known as Saurin’s classification) and its tuning.18,19
With increased usage of CE, different approaches to improve and standardise training in the evaluation of CE studies 20 have been developed. Face-to-face courses, online tools and courses have been established, and their benefit validated. SB Curricula (e.g. of the ESGE and ASGE) outline the content of training courses and suggestions for assessing competency.4,21 Standards have also been published 22 and updated 23 based on consensus on expected quality, together with key performance parameters. These include, for instance, adherence to accepted indications and technical recommendations on the conduct of a CE procedure, 24 image quality, reading speed, retention rates of capsules and usage of a Patency capsule in patients with high risk for retention.
There is increasing data on the use of CE not only for the diagnosis but also monitoring of small-bowel CD post-therapy. Recent studies in IBD and focusing on ‘treat to target’ have demonstrated the key role of CE in this strategy to reduce the impact of disease burden on patient populations. 25 In the setting of overt GI bleeding, CE offers a rapid diagnosis, often with a significantly higher yield than radiology/triple-phase CT. 26 The panenteric CE, which allows visualisation of both the small and large bowel in one sitting, has shown promise in this setting too.27,28 Patient preference has also influenced its adoption: CE is generally well tolerated, minimally invasive and does not require sedation or hospital admission. This ease of use makes it particularly appealing to elderly and frail populations, even in the out-of-clinic context, with adequate completion rates and patients’ satisfaction. 29 Application of double-headed capsules, together with extended bowel cleansing and ingestion of boosters for propulsion, has made CCE feasible.30,31 National pilot programs have been set up to reduce the workload for traditional colonoscopy by CCE, triggered by the pandemic.32,33 However, trials in screening the population or in FIT-positive patients point out the need for still further improvement.9,34 External magnetic steering of capsule movement finally enables inspection of the stomach, for example, in screening programs.12,35 More recently, a cloud-based program has allowed CE to be administered in the community and remotely read and reported by clinicians. 36
Technological evolution
Over the past 25 years, CE has undergone a profound technological evolution, with several companies now competing in this field. These early CE systems were restricted by low-resolution imaging, a limited field of view and minimal battery life. 37 Constraints significantly impacted both diagnostic yield and clinical utility. Today, state-of-the-art capsule devices incorporate high-resolution imaging sensors with a wider field of view up to 175° and refined optical systems. 37
CE devices now employ advanced imaging sensors, with pixel resolutions vastly improved over earlier models. These upgrades enable detailed visualisation of mucosal structures, allowing for the detection of subtle findings. Enhanced LED-based illumination, combined with more sophisticated image processing algorithms, further improves clarity, contrast and visibility. Recent developments in deep learning have also introduced super-resolution techniques, such as the EndoL2H framework, which potentially enhances image quality by up to 12 times, facilitating better diagnostic accuracy. 37 Battery performance has also improved significantly. While earlier capsules offered only 6–8 h of operational time, newer systems have an extended battery time of 10–14 h, ensuring complete SB coverage in most patients and enabling full GI tract visualisation in selected cases. This extension of battery life has been made possible through energy-efficient chipsets, smarter data transmission protocols and lower-power consumption imaging components. Longer operational durations increase diagnostic completeness and reduce the need for repeated procedures.
Another major development has been the implementation of adaptive frame rate technology. Traditional CE systems employed a fixed frame rate (e.g. 2–3 frames per second), which could either miss important findings during rapid transit or lead to redundant image capture during slower movement. Newer devices have a bi-directional communication between the capsule and the recorder. This allows an adaptive increase of frame rate up to 10 fps in SB CE and to 35 fps in colon capsules when real-time recorder analysis shows a rapid change of images during fast capsule motion. The angle of view has also expanded considerably. While early-generation capsules had a field of view of approximately 140°, modern designs offer viewing angles ranging from 156° to nearly 180°, providing near-complete circumferential visualisation of the intestinal lumen. This broader perspective minimises blind spots and enhances the detection of lesions located on mucosal folds or lateral walls.
Incorporation of panoramic lateral-view optics and application of dual-headed capsule also for the SB are further attempts to increase mucosal coverage and improve lesion detection. Real-time viewing capabilities have become another important feature of contemporary CE systems. These technologies allow for on-the-fly visualisation of capsule progression, particularly useful in delayed gastric transit, where clinicians can administer prokinetics as required. 24 Another significant development is the external control of the CE in the upper GI tract. With the use of an external magnet, clinicians can guide the capsule’s position and orientation to obtain targeted views of specific regions of the stomach, mimicking some functions of traditional gastroscopy without the need for sedation or intubation. Recent studies have demonstrated the feasibility of autonomous magnetic systems navigating a capsule within the stomach controlled by AI based on the real-time analysis of obtained images. 35
Standard transmission of image data is by radio frequency, while some platforms use electric current or onboard data storage. Recent prototypes using a bi-directional Bluetooth connection with a smartphone could avoid the need for additional dedicated equipment. 30 Despite these advancements, several limitations persist. Capsule retention, while less frequent due to improved pre-procedure screening, the use of patency capsules, more precise patient selection criteria and new testing protocols, remains a potential complication, especially in patients with known or suspected strictures.38,39 Moreover, the interpretation of CE recordings continues to be a time-intensive and operator-dependent task. AI tools are emerging as promising aids to reduce reading time, as shown by recent meta-analyses.40,41 However, their future role needs to be clarified further based on pre-defined technical and ethical requirements. 33 Although CE offers unparalleled, non-invasive visualisation of the GI tract mucosa, it has inherent limitations. It is impossible to navigate and manoeuver the CE within the SB in cases where greater scrutiny is required on pathology with uncertain significance. It also still cannot perform biopsies or deliver therapeutic interventions, such as to bleeding lesions. Whilst there have been many trial iterations of such a CE, none have reached the mass market for routine use. As such, CE remains primarily a diagnostic tool and may need to be complemented by traditional endoscopic procedures in cases requiring histological analysis or therapeutic management.
A pertinent question has been – is the utilisation of CE green? The direct CO2 emissions have been compared across different CE systems, and patient travel contributes the most to emissions, accounting for more than 95% of the total emissions. 42 A CE service predominantly based on the patient’s home and a cloud-based download and reporting of CE would help make the process and procedure more sustainable. In tandem, it is the responsibility of CE manufacturers to prioritise the recycling of CE components.
The Horizon: AI, therapeutics and autonomy
The integration of artificial intelligence (AI) promises to address many of CE’s current limitations. Deep learning algorithms 40 can now identify common lesions, such as angioectasias, ulcers and polyps, with high sensitivity and specificity, reducing the burden on clinicians and the potential of standardising interpretation in the future.43,44 Some systems have demonstrated the ability to triage normal studies automatically, cutting reading time up to 12-fold. 41 Beyond diagnostics, CE technology is exploring therapeutic frontiers. Engineers are developing capsules capable of targeted drug delivery and biopsy acquisition, further enhancing the capabilities of this innovative tool. 45 Locomotion systems, whether based on magnetic fields or micro-robotic appendages, suggest a future where capsules can be steered, paused or reoriented in vivo. 45 These advancements are no longer theoretical; studies have demonstrated their feasibility in animal models and early human trials. The convergence of gastroenterology, robotics and computer science has the potential to create a fully autonomous, multifunctional capsule platform within the next decade. 46
Reflections from a pioneer
It is impossible to discuss CE without recognising the pivotal role of Paul Swain. His belief in the feasibility and clinical utility of a swallowable camera led to one of the most significant innovations in GI medicine in the last 50 years. He not only advocated for this technology but also participated in its early trials, promoted its acceptance and mentored a generation of researchers in CE. Reflecting on the journey, Professor Swain has often emphasised the interdisciplinary nature of this success, which is a combination of clinical insight, engineering expertise and entrepreneurial courage. ‘The real triumph of capsule endoscopy’, he once remarked, ‘is not just that it sees what we couldn’t see – it changed how we think about accessing the body’.
Looking ahead: Tradition meets transformation
As we look toward the silver anniversary, CE stands at the intersection of tradition and transformation. It has evolved into a standard tool in many gastroenterology units, integrated into workflows and relied upon for complex diagnostic cases (Table 1). However, its foundational simplicity – a swallowed camera – belies the extraordinary potential that still lies ahead. Challenges persist. Equitable access to CE remains limited in many low-resource settings. Training for CE interpretation is inconsistent, and regulatory frameworks for AI-assisted diagnostics are still evolving. Nevertheless, the trajectory is clear: CE is not an endpoint, but a foundation, a platform of sorts. As we teach our trainees to interpret CE images, we also encourage them to envision better tools. While we analyse AI-assisted studies, we remind ourselves of the human intuition that remains essential to our profession. As we honour the past, we commit to the same dedication, humility and collaboration that sparked this technology.
Key landmark studies in capsule endoscopy.
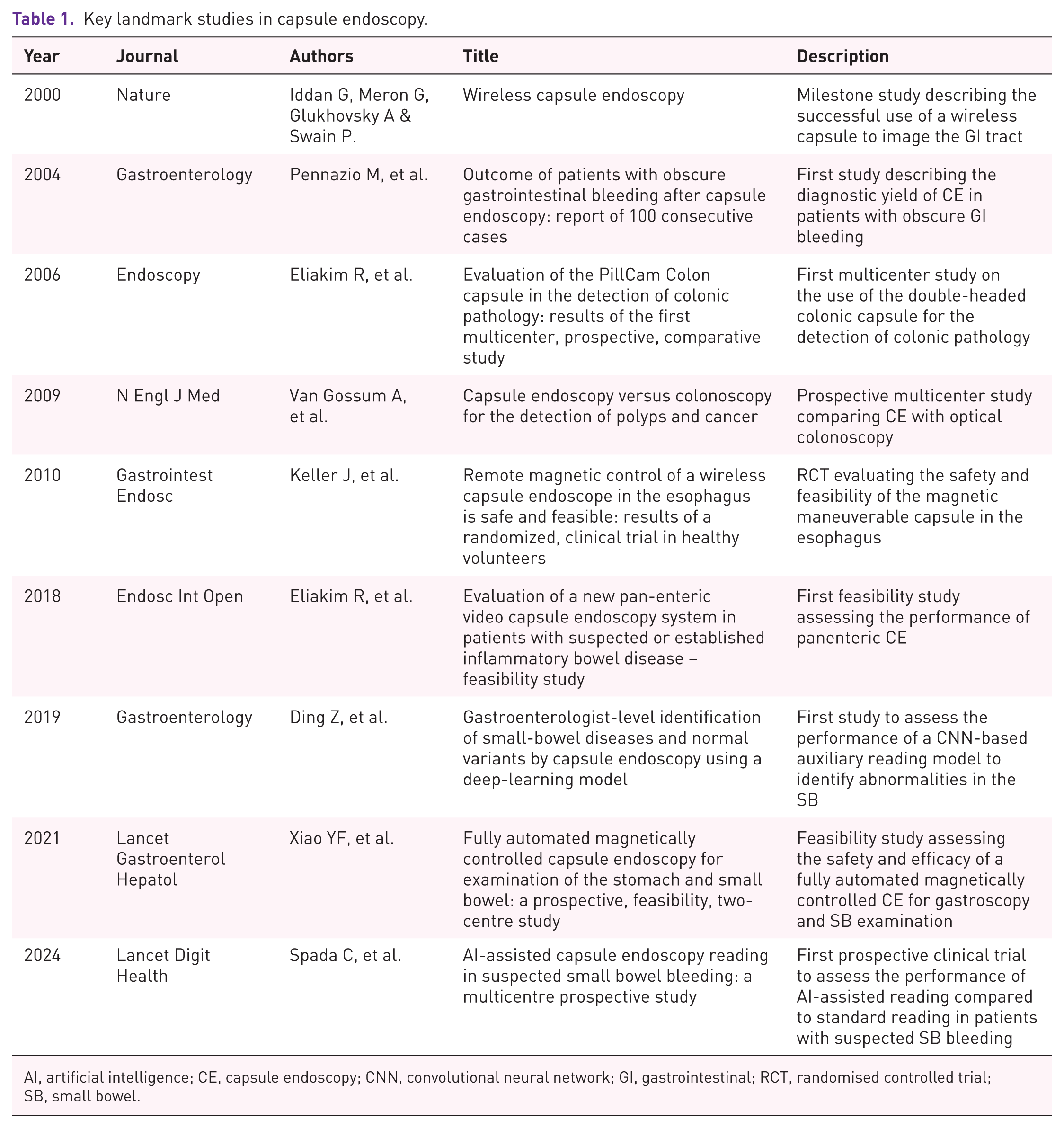
AI, artificial intelligence; CE, capsule endoscopy; CNN, convolutional neural network; GI, gastrointestinal; RCT, randomised controlled trial; SB, small bowel.
Conclusion
Since its inception at the dawn of the new millennium, CE has transformed from a visionary concept into a cornerstone of GI diagnostics. What began as a novel solution to overcome the ‘black box’ of the small intestine has evolved into a versatile, non-invasive tool, reshaping clinical practice with unparalleled impact. Technological advancements throughout the years – such as higher-resolution processors, adaptive frame rates, AI-assisted analysis – have dramatically improved diagnostic accuracy and efficiency, while newer innovations (e.g. magnetically controlled capsules, therapeutic capsules, non-imaging sensor-based devices) and expanded clinical scenarios (e.g. first-line capsule pan endoscopy in acute bleeding patients) continue to define new horizons of application.
Yet, the journey is far from complete. Challenges remain, including equitable access, global standardisation of training and the need for cost-effective solutions. The future holds even greater promise, from autonomous robotic capsules to AI-driven real-time diagnostics and precision-targeted therapies. As we celebrate this milestone in its silver anniversary, we honour the pioneers whose collaboration made CE possible and look ahead with anticipation to the next era of innovation. CE is not just a diagnostic tool: it is a testament to how far we have come and a beacon for where we are headed, ensuring its enduring place at the forefront of GI diagnostics.
