Abstract
After failed biliary cannulation via standard endoscopic retrograde cholangiography approach, endoscopic-ultrasound-based rendezvous-endoscopic retrograde cholangiography (EUS-RV-ERC) is a valid alternative. One of the challenging factors in this setting is the management of the guidewire. Here, we propose a method, where a slim endoscope is used to stabilize the guidewire and optimize wire manipulation in a patient who underwent EUS-RV-ERC via a transgastric approach. This was executed in a patient suffering from severe alcoholic pancreatitis presented with a severely narrowed duodenum due to extrinsic compression and inflammation in the setting of cholangitis Tokyo Grade III.
Introduction
Rendezvous-endoscopic retrograde cholangiography (RV-ERC) is known for a long time as a valid alternative in patients where the standard retrograde ERC approach is not feasible. In fact, it has been reported to be safe and superior to precut sphincterotomy for single-session biliary access. 1 One of the possible challenging factors in performing endoscopic ultrasound (EUS)-based RV-ERC (EUS-RV-ERC) is the manipulation of the guidewire before it can be securely retrieved after transpapillary advancement particularly in conditions of minimal endoscopic maneuverability in a compressed duodenum. Here, we report a novel technique in order to avoid guidewire dislodgement by using a slim endoscope to stabilize the guiding wire in an RV-ERC, where we had to opt for EUS-transgastric access to the biliary system. The reporting of this study conforms to the Consensus-based Clinical Case Reporting guidelines. 2
Patient information/clinical findings/diagnostic assessment
A patient in his 50s with a severe alcohol-induced acute pancreatitis was admitted to us from the intensive care unit (ICU) of a regional hospital for emergency endoscopic retrograde cholangiopancreatography (ERCP) due to mechanical obstruction of the bile duct and associated cholangitis Tokyo Grade III. He was consuming 80–100 g of alcohol per day for approximately 15 years and during the last 22 years only occasionally. The patient was admitted to the regional hospital because of acute pancreatitis with an initial recovery after a conservative treatment with fluid resuscitation, analgetics, and antibiotics. The first laboratory investigations after the admission showed an elevation of the inflammatory parameters and lipase, whereas the liver function tests were completely normal [white blood cells (WBC) 23.13 G/L (3.50–10.0), lipase 12,189 U/L (13–60) C-reactive protein (CRP), aspartate aminotransferase (AST), alanine aminotransferase (ALT), gamma glutamyl transferase (g-GT), alkaline phosphatase, and bilirubin were normal]. The primary imaging with a CT scan as well as a magnetic resonance cholangiopancreatography (MRCP) showed an edematous interstitial pancreatitis without signs of a chronic pancreatitis (which could not be ruled out), biliary obstruction, or a cholecystolithiasis.
One week later, the clinical state of the patient was worsening with severe abdominal pain, tachycardia, and confusion. He was transferred to the ICU. A blood analysis showed an elevation of WBC as well as of the cholestatic enzymes [WBC 36.58 G/L, AST 602 U/L, ALT 403 U/L, alkaline phosphatase 1057 U/L, g-GT 2167 U/L, and bilirubin 168 μmol/L (<21)]. A CT scan was repeated, and this time revealed dilation of the intra- and extrahepatic bile ducts due to a compression of the common bile duct (CBD) from the head of the pancreas, intramural air in the gastroduodenal wall, and ascites (Image 1). Cholangitis was most likely present, 3 and hence, biliary drainage was indicated.4,5 The patient was referred to our unit, and an ERC was attempted. However, severe duodenal swelling and external compression of the duodenal lumen resulted in failure to identify the papillary orifice while severely limiting the scope maneuverability. Nevertheless, the scope could still be passed through parts II and III of the duodenum.

CT before the ERCP showing swelling of the head of the pancreas and dilated intrahepatic ducts.
Therapeutic intervention
After failure to cannulate the CBD via standard ERC (Figure 1), an EUS-guided biliary access was favored. This was not possible via the transduodenal approach due to an unstable position of the scope. A left-sided dilated intrahepatic biliary duct in liver segment II was transgastrically punctured under EUS-guidance (EG 3870UTK Linear Array Ultrasound Gastroscope, Pentax) using a 19-gauge needle (Firma Boston 19 Ga Expect™ flexible needle) in order to access the biliary system (Figures 2 and 3). A 0.025-inch guidewire (Visiglide, Olympus) was advanced through the needle into the CBD and subsequently into the duodenum (Figure 4). The absence of biliary concrements enabled the advancement of the wire easily into the duodenum; otherwise, a biliary catherization with an ERCP cannula would have been necessary in order to advance the wire passing biliary concrements in the duodenum. The needle and the echoendoscope were, in turn, removed while maintaining the wire position. In order to stabilize the wire, a slim endoscope with a diameter of 5.4 mm (GIF-XP-180N, Olympus) was back-loaded onto the guidewire using a sphincterotome (TRUEtome 44, Boston Scientific) as a bridge between the distal tip of the scope and the biopsy channel. The exchange was performed as follows: the sphincterotome was passed through the biopsy channel of the slim endoscope and then the edge of the wire, which was already outside of the patient, was passed retrogradely through the sphincterotome. The slim endoscope (being sprayed with silicone, dimethyl polysiloxane) was then advanced into the stomach along the esophagus. The risk of dislodgment of the guidewire is very minimal during this procedure due to the tube-shaped anatomy, limited length of guiding the slim endoscope along the puncture site, and the fact that the sphincterotome inside the working channel did also stabilize the guidewire avoiding banding and buckling. The slim endoscope with the tip of the sphincterotome just at the tip of the endoscope was brought in close contact to the cardiac puncture site in order to stabilize the guidewire at its best (Figure 4).

Edematous papilla.

EUS-guided transgastric puncture for accessing biliary tree.
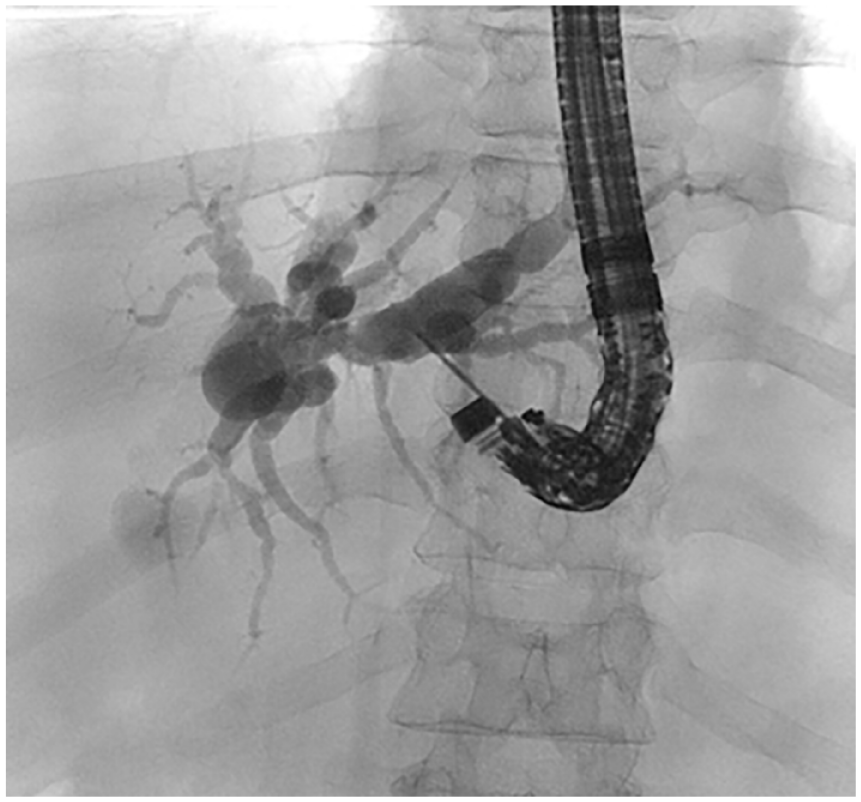
Cholangiogram via transgastric EUS access.

Introduction and stabilization of the guidewire by the slim endoscope.
After disconnecting the slim endoscope from the image processor while maintaining its position also with the help of the fluoroscopy, we switched to a duodenoscope, which was advanced besides the slim endoscope to the fourth part of the duodenum. Also, the duodenoscope was sprayed with silicone (dimethyl polysiloxane) before insertion in order to avoid an interference between the two endoscopes being in place at the same time. Next, the transpapillary end of the guidewire was grasped with a snare and pulled out through the working channel enabling full RV. The slim endoscope had to be removed in order to free enough wire length, so that a standard sphincterotome could be introduced over the wire and advanced to the tip of the duodenoscope. Subsequently, biliary cannulation and sphincterotomy could be performed over the wire. Due to the severity of cholestasis and grade of stenosis at the distal part of the CBD and in anticipation of possible late complications as a result of the pancreatitis, a fully covered metallstent (Wall Flex biliary RX fully covered 40 mm length, 10 mm diameter, Boston Scientific) was inserted with excellent biliary decompression (Figure 5), also confirmed by the laboratory investigations on the day after the intervention (bilirubin 85 μmol/L, AST 56, ALT 201, and g-GT 1013 U/L). Although sufficient biliary drainage is most of the times achieved after placement of a single plastic stent, our own experience in previously treated cases made us decide to use a self expandable metallic stent (SEMS). In fact, in selected cases, we faced the need for sequential re-ERCs applying either multiple plastic stents or fully covered metal stent because the drainage from a single plastic stent was not adequate. Indeed, the treatment with a plastic stent is also associated with a higher risk of occlusion or migration as compared to SEMS. 6 Given the complexity of the case and the fact that we could not place more than one plastic stents due to limited visibility and required rendezvous approach, we opted for a SEMS despite its higher cost in order to ensure the best possible biliary decompression.

Rendezvous ERC and transpapillary stenting.
Follow-up and outcomes
After 6 months, we performed an ERC with the extraction of the metal stent and small bile duct stones. Two months later, the patient was evaluated in our outpatient clinic. He was completely asymptomatic with unremarkable liver funktion test (LFT).
Discussion
ERCP was initially performed in 1968 by Dr. William S. McCune, an obstetrician, using a fiber duodenoscope with an external accessory channel. Initially, it was developed as a diagnostic tool. In 1973, the first biliary sphincterotomies were reported in Japan and in Germany. 7 Nowadays, ERCP is an established method for the treatment of diseases of the biliary and pancreatic duct (Table 1), which can, however, lead to serious complications (Table 2).


Even in standard situations (Figure 6), ERCP faces several challenges associating with a failure rate ranging between 3% and 10% of cases, even when experienced endoscopists use advanced techniques such as precutting or the double-wire technique. However, in cases of ampullary hindrance caused by tumor, diverticula, or even severe inflammatory duodenal stenosis, ERCP is even more challenging (Figure 7), and an alternative access site is often required.

Challenges in ERCP in standard situation.
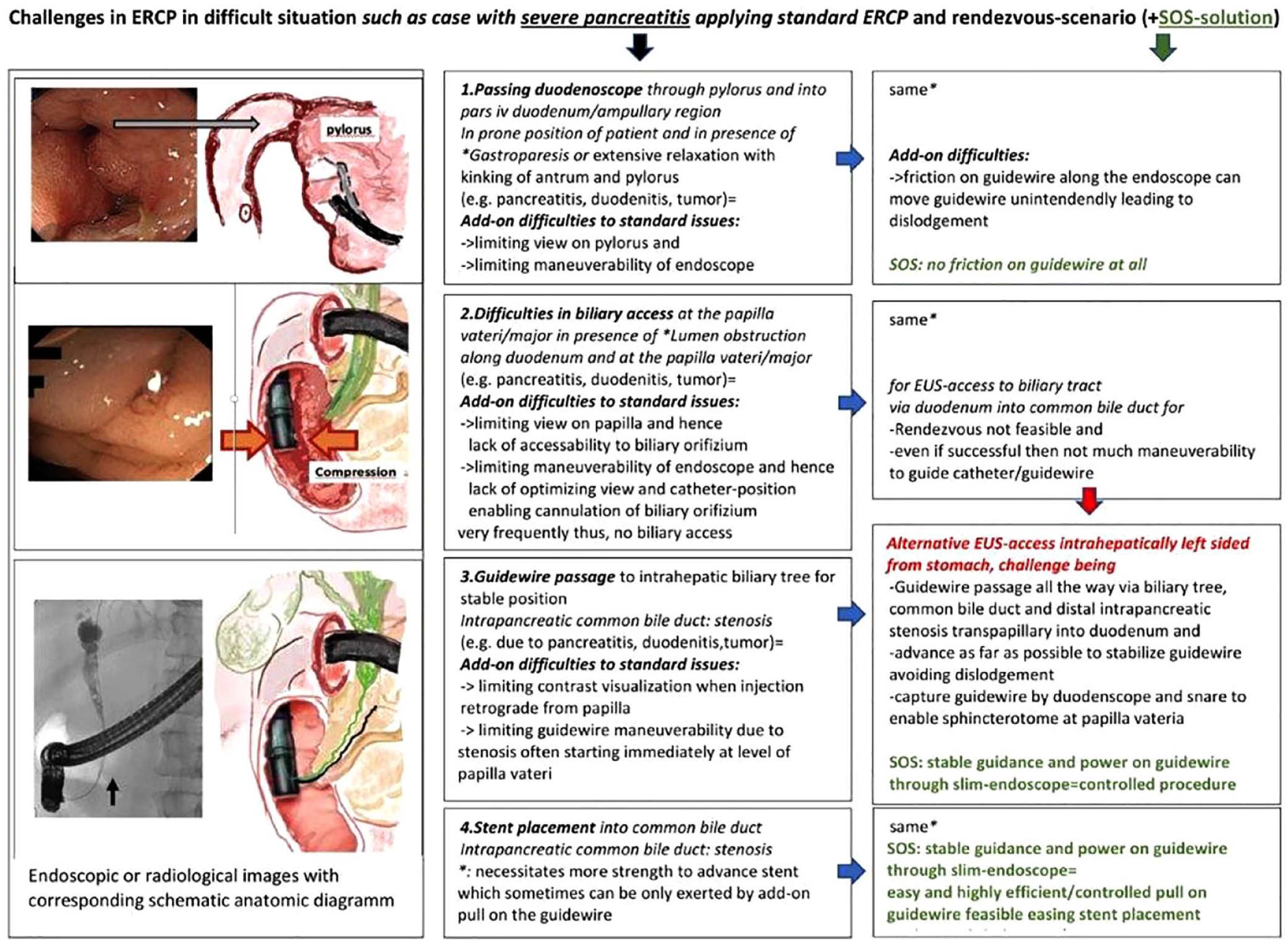
Challenges in ERCP in difficult cases.
For many years, percutaneous transhepatic cholangiography and drainage (PTCD) was the most established alternative; however, EUS-based approaches have gained much attraction recently. 8 These include EUS hepaticogastrostomy, EUS choledochoduodenostomy, EUS-antegrade stenting, or EUS-RV-ERC.
Since the first description of EUS-RV in 2004, 9 several techniques have been proposed, but none has been considered as a gold standard. The most common technique is accessing the extrahepatic bile duct (EHBD) under EUS guidance via transduodenal puncture (short and long scope positions) or the intrahepatic bile ducts via transgastric puncture (short scope position) with a 19-gauge fine needle aspiration (FNA) needle. After injection of contrast to visualize the biliary system, a guidewire is advanced in the duodenum transpapillary. Next, the FNA needle and the echoendoscope are removed while keeping the guidewire in place, and a duodenoscope is advanced parallel to the wire in the duodenum (Figure 8). Finally, the wire is grasped with a snare and pulled out through the accessory channel of the duodenoscope.
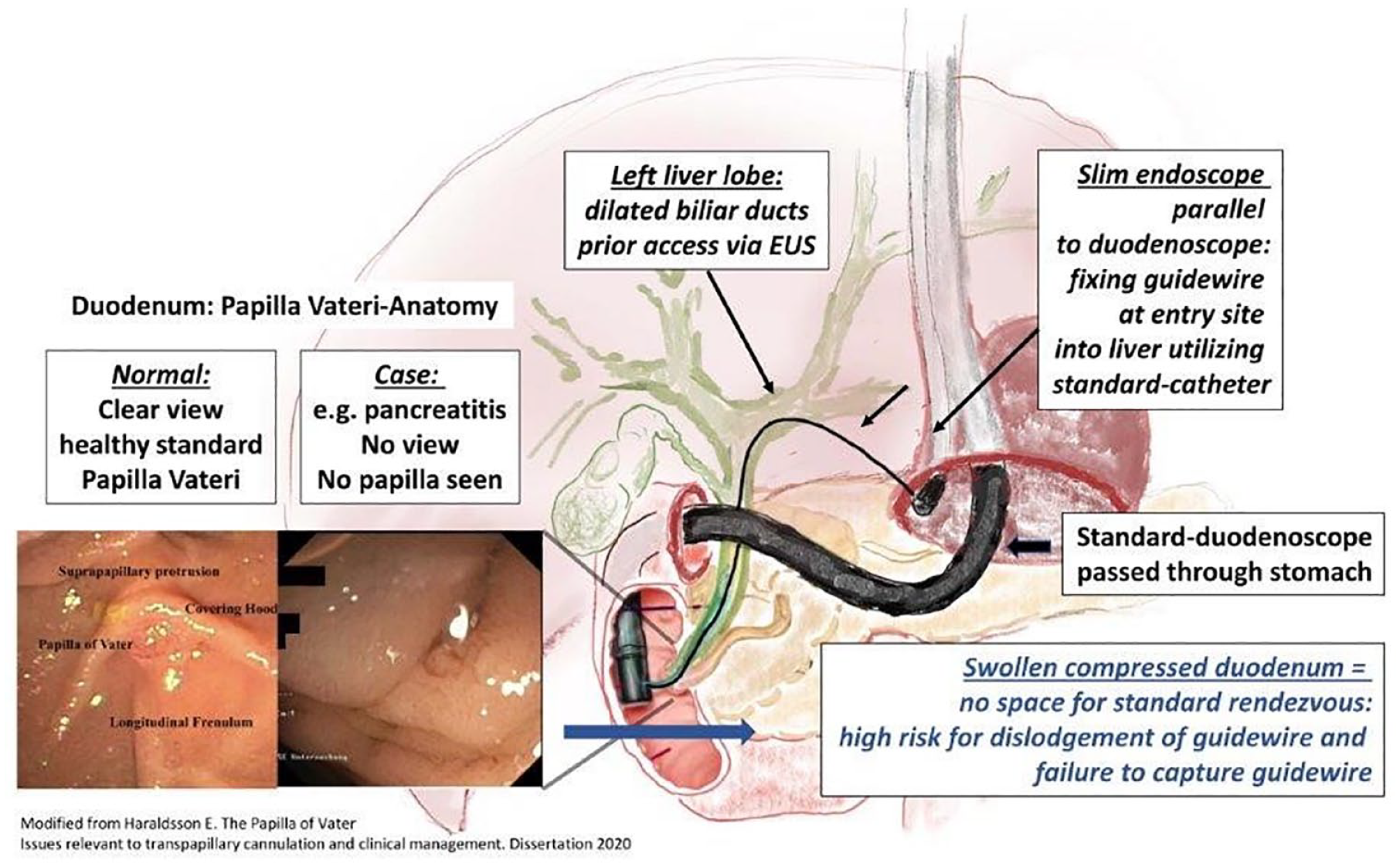
Graphical abstract.
Iwashita et al. 10 showed in a multicenter prospective study evaluating the safety and efficacy of EUS-RV, which included 20 patients, that puncturing from the second part of the duodenum close to the papilla (possible in 10/20 patients) seems to yield a higher success rate of 100% in these patients (10/10) compared to that from the stomach 75% (3/4) or 60% (3/5) from the bulb (short scope position). However, accessing the distal EHBD from the duodenum is not always feasible because of the unstable scope position or in case of a duodenal stenosis. Should the transgastric puncture be the only viable approach, as in our case, a common problem that can occur is the difficulty in advancing the wire through the papilla because of the long distance from the entry point in the intrahepatic bile duct.
Our method offers a more elegant way for stabilizing the guidewire in cases where increased manipulation capabilities are required to overcome obstacles hindering transpapillary wire advancement such as stenosis of the distal CBD, since the wire is stabilized at the puncture site in the stomach wall. Another risk factor when using the transgastric approach is that of the dislodgement of the guidewire during the duodenoscope insertion parallel to the guidewire. Indeed, occasionally, only a few centimeters of the wire can be advanced distal to the papilla, and even a small displacement of the wire into the CBD during duodenoscope insertion would require the whole procedure to be repeated from the beginning. In the proposed slim-endoscope-stabilized (SES) approach, the guidewire is protected with the tip of the sphincterotome and the slim endoscope itself. This minimizes the risk of accidental guidewire dislocation while advancing the duodenoscope.
Due to this advantage, it is tempting to speculate that this SES approach may increase the success rate of the EUS-RV-ERC. This may help to establish EUS-RV-ERC in difficult cases particularly considering the known lower rates of complication risks and morbidity as compared to EUS-based transmural stenting procedures such as hepaticogastrostomy or choledochoduodenostomy. 11 Finally, compared to PTCD-based RV-ERCP, which requires two sessions, only one session is needed, which results in shortened postprocedural hospital stay while maximizing patient comfort and quality of life since no external drainage is needed. 1
The potential disadvantages of the presented ‘SES’-method include (i) insertion of two endoscopes at the same time, (ii) necessity of highly experienced personal, and (iii) multiple operators in place to execute all steps appropriately. As for the insertion of the duodenoscope in parallel to a slim endoscope already in place, this sums up to about 18 mm in total diameter. This is 3 mm more than, for instance, large-diameter endoscopes such as some older version linear EUS or enteroscopes. In our hands, in the presence of normal and compliant esophageal anatomy and structure with optimal extension of the neck, this is feasible without any need for prior dilation. In terms of team effort, the procedure does necessitate a highly experienced endoscopist as well as at least two highly efficient assistants for the guidewire manipulation and fixation of the slim endoscope at the same time. Possible complications include perforation, peritonitis, and bleeding.
Conclusion
Utilizing a slim endoscope to stabilize the guidewire (SES) in EUS-RV-ERC via transgastric access may help increase the success rate of the intervention by offering better long guidewire manipulation while minimizing the risk of guidewire dislocation. Compared to percutaneous biliary drainage rendez vous ERCP (PBD-RV ERCP), only one session is needed. EUS-RV-ERC is a technically challenging procedure that requires a highly experienced endoscopist ideally in a tertiary center setting.
