Abstract
Endoscopic eradication therapy for Barrett’s esophagus has been established as an effective management strategy for patients with Barrett’s esophagus with dysplasia and early esophageal cancer. Among the endoscopic therapies, ablation techniques such as radiofrequency ablation and cryoablation are effective primary treatment interventions with acceptable low complication rates forming the spectrum of a multimodal approach. Appropriate selection of patients, high-definition endoscopic evaluation, and dedicated histological assessment are important cornerstones to help navigate to the best effective treatment method. Carefully structured surveillance programs and preventive measures will be needed to provide long-term durability for maintaining complete remission.
Introduction
Barrett’s esophagus (BE) is a commonly addressed condition in gastroenterology given the association with gastroesophageal reflux disease (GERD). Prevalence of BE ranges widely from 0.4% to more than 20% depending on the population studied and the diagnostic criteria used in the study.1–4 The goal of this article is to provide an overview of BE and discuss the available ablative treatment options.
According to current guidelines, BE should be diagnosed when salmon colored mucosa extends ⩾1 cm proximal to the gastroesophageal junction with histology proven columnar-lined intestinal metaplasia. If BE is suspected during esophagogastroduodenoscopy (EGD), obtaining 4-quadrant random biopsies, every 1 to 2 cm of the segment is required throughout the columnar-lined esophagus per Seattle protocol (Figure 1(a) and (b)). 5 Risk factors associated with BE include long-standing GERD, male gender, Caucasian race, central obesity, current or previous history of smoking, age over 50 years, and confirmed first degree family history of BE or esophageal adenocarcinoma (EAC). Screening and surveillance for BE should be considered in men with greater than 5-year history and/or weekly symptoms of GERD with two more risk factors as previously stated to prevent progression to EAC. 6 Screening in females is not generally recommended but should be considered in individual cases based on the presence of multiple risk factors. 6 In patients with risk factors, EGD should be performed, and surveillance is dependent on the histological findings. If Los Angeles classification B, C, or D esophagitis is found, a repeat EGD is recommended after 8–12 weeks of proton pump inhibitor (PPI) therapy to ensure healing and exclude underlying BE. 6 A repeat EGD in 3–5 years is recommended surveillance for BE without dysplasia, and with indefinite for dysplasia is recommended to treat with PPI for 3–6 months then repeat EGD. When pathology reveals low-grade dysplasia (LGD), it is recommended to perform endoscopic therapy or surveillance EGD every 12 months, but if high-grade dysplasia (HGD) is identified then endoscopic therapy is recommended. 6
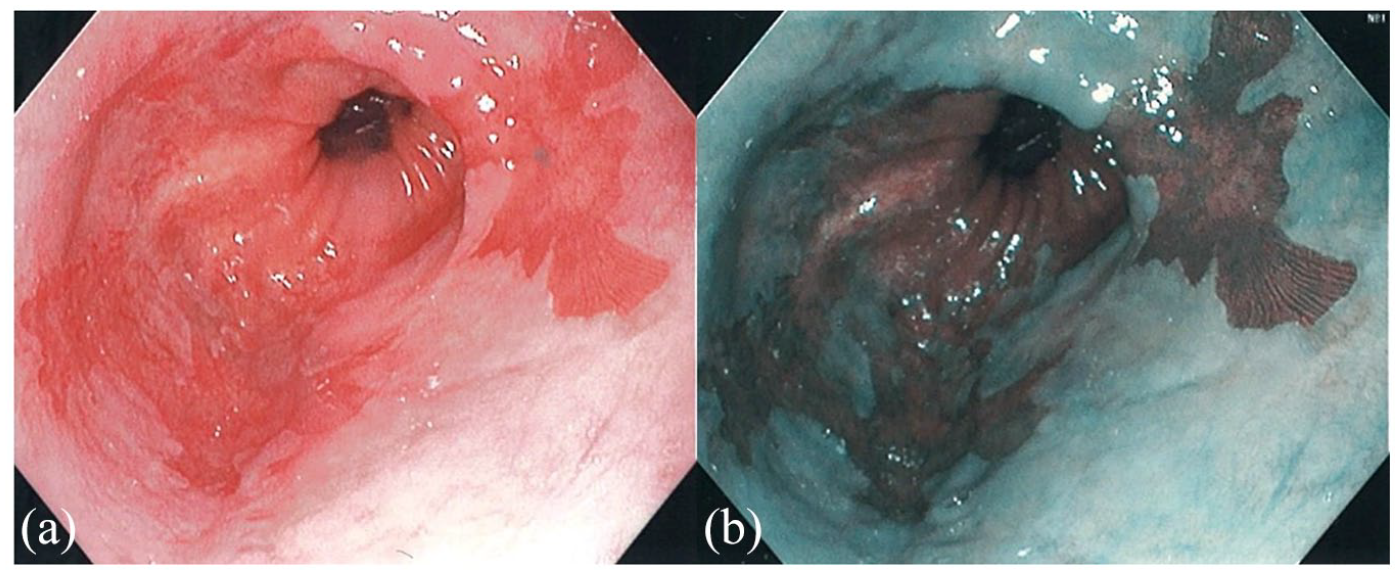
(a) Endoscopic view of salmon colored mucosa extending from the top of the gastric folds to the proximal esophagus and (b) Endoscopic view under narrow band imaging detailing the salmon colored mucosa.
Indications for ablative therapy
Based on current guidelines, indications for endoscopic ablative therapy include nonvisible lesions with dysplastic BE, confirmed LGD, and HGD.6–8 In patients with visible T1a lesions, endoscopic mucosal resection (EMR) is the preferred modality followed by ablation therapy. In patients with T1b EAC (submucosal), a multidisciplinary team should discuss if endoscopic therapy is an alternative therapy to esophagectomy in patients with superficial disease, well-differentiated neoplasm without lymphovascular invasion, or poor surgical candidates. The T staging is usually accomplished using EMR techniques. 6
Principles of ablative therapy
The goal of ablative therapy in BE is to allow for complete eradication of intestinal metaplasia (CEIM) resulting in squamous cell re-epithelialization of the esophagus. This is achieved by inducing superficial tissue necrosis through photochemical, thermal, or freezing injury. When the patient is acid suppressed, as when taking a PPI, the damaged tissue is then replaced by normal squamous mucosa. While the exact mechanism is unknown, it is thought that neighboring squamous cells and progenitor cells result in the squamous regeneration.9,10 The current ablative endoscopic treatment options for BE include thermal: radiofrequency ablation (RFA), argon plasma coagulation (APC), and less commonly used laser and multipolar electrocoagulation, and nonthermal: cryotherapy, photodynamic therapy (PDT). Nonablative endoscopic treatment includes EMR, and a hybrid or two-step therapy involving EMR followed by RFA. This review article will focus on RFA, cryotherapy, and PDT (Archived).
Techniques, outcomes, and complications
Preparation needed prior to ablative therapies include standard EGD pre-procedure considerations such as sedation and procedural risks. The choice of sedation is mostly driven by institutional preferences; however, it must be noted that the demographics of patients with BE are typically those with comorbid conditions such as obesity and smoking, among others which may place them at slightly higher sedation risk compared with the general population. Anesthesia provider-based sedation has been used increasingly for endoscopic procedures for BE. Overall, ablative therapies are considered low risk for bleeding; therefore, the decision on withholding antiplatelet or anticoagulants should be weighed against the risk of exacerbating the underlying indication for their use. 11
Prior to ablation therapy, it is essential that accurate endoscopic pretreatment staging is obtained using the Prague C & M classification which incorporates the length of the circumferential (C) segment and the maximal (M) extent of the BE segment. 12 Once BE with dysplasia is found on EGD, it is recommended to be reviewed by two pathologists, at least one that specializes in gastrointestinal (GI) pathology. The esophagus should be carefully inspected for the presence of a hiatal hernia, strictures, ulceration, previous scaring from EMR, and importantly visible lesions or nodularity which would preclude ablation given the risk of occurrence of buried BE post ablation. If strictures are present, dilation should be performed ideally 2–3 weeks prior to ablation. 13
Radiofrequency ablation
RFA is a frequently used and an effective treatment for BE. The treatment uses a bipolar electrode array which delivers a definite amount of thermal radiofrequency energy to uniformly disrupt tissue at a depth of 0.5 mm. Within RFA, there are unique techniques using devices with electrode dimensions such as the circumferential (c-RFA): Barrx™ 360 Express RFA Balloon Catheter (4 cm) or focal (f-RFA): Barrx™ 90 RFA Focal Catheter (20 mm length × 13 mm width), Barrx™ 60 RFA Focal Catheter (15 mm length × 10 mm width), Barrx™ Ultra Long RFA Focal Catheter (40 mm length × 13 mm width), and through the channel ablation device, Barrx™ Channel RFA Catheter (7.5 mm × 15.7 mm).
The equipment needed for c-RFA includes an endoscope plus equipment, Barrx™ Flex RFA Energy Generator console, HALO360 express balloon ablation catheter (new generation circumferential device incorporating the esophageal sizing and ablation into one catheter), HALO cap, savary spring-tipped guidewire of 0.025 or 0.035 in diameter, at least 260 cm long or Nitinol-based guidewire. 14 The equipment needed for f-RFA includes an endoscope plus equipment RFA energy generator console and the focal ablation device of choice.
Contraindications for RFA include prior radiation to the esophagus which increases the risk of stricture formation and poor wound healing, esophageal varices, and in nodular or ulcerated BE as these patients would benefit from EMR for diagnosis, staging, and treatment. 15
Techniques
The choice of device used depends on the distribution of the BE segments. For patients with circumferential BE longer than 2 cm, the c-RFA would be the ideal device of choice. For shorter segments and islands, the f-RFA devices and catheters would be preferred.
Circumferential radiofrequency ablation techniques
The classic protocol uses a guidewire and sizing balloon, the sizing balloon has largely been replaced by the HALO360 express balloon. After the landmarks are recorded, a guidewire is passed to the gastric antrum and the endoscope is exchanged. The HALO360 catheter is than advanced over the wire into the esophagus and guided toward the Barrett’s segment (Figure 2(a)–(c)). The endoscope is intubated alongside it to allow for visualization of the proximal balloon. By using the foot pedals attached to the control unit, the balloon is inflated and the energy is delivered. The energy delivered uniformly has a density of 12 J/cm2, a power of 40 W/cm2, ablating a depth of 700–1000 µm over 3 cm area. 13 A second ablation is applied, and the balloon subsequently deflates, and the catheter is repositioned to a different segment carefully avoiding >1 cm of overlap. The catheter and the endoscope are exchanged over the guidewire and the catheter is cleaned. A second ablation pass is performed as described above. A second pass may be circumvented given the effectiveness of a single pass with a considerably shorter procedure time. 16

(a) The HALO360 balloon catheter passed over the guidewire under endoscopic guidance toward the circumferential Barrett’s segment. (b) The HALO360 balloon catheter is inflated and RFA is being delivered under endoscopic guidance. (c) The balloon catheter is deflated, and the mucosa is inspected.
Focal radiofrequency ablation techniques
The f-RFA catheters including those that are through the scope (Halo TTS) are utilized following initial c-RFA to tackle shorter segments and smaller islands (Figure 3(a) and (b)). Other f-RFA catheters are attached to the tip of the endoscope at the 12-o’clock position (Figure 4). Lubricant jelly should be avoided to allow contact of the catheter with the mucosa with all RFA therapies. Once the esophagus is intubated, the targeted area of BE esophagus is oriented toward the 12-o’clock position and the mucosa is opposed (Figure 5). Using the foot pedal, energy is delivery; this is repeated twice. Once the area of BE is ablated, gentle scraping is performed, and the area is irrigated, suctioned, the endoscope is then withdrawn. The catheter is cleaned using dampened gauze, and the esophagus is re-intubated, and the prior steps are repeated. The cleaning stage can also be circumvented by using three applications without cleaning with noninferior results compared with the standard regimen. 17

(a) Narrow band image view of the TTS RFA catheter which is passed through the channel of a standard endoscope and rotated toward the target Barrett’s segment. (b) Narrow band image view of the Barrett’s island using the TTS RFA catheter.

(a) The f-RFA catheter (HALO 90), attached to the tip of the endoscope at the 12-o’clock orientation. (b) f-RFA catheter (HALO 90) at the 12-o’clock position opposing the Barrett’s segment.

(a) The CryoBalloon system consisting of a handheld trigger, the nitrogen oxide canister, a foot pedal, and different types of balloon catheters (focal for esophageal and pear shaped for gastroesophageal junction (GEJ). (b) System being activated and targeting a Barrett’s segment.
Efficiency of the different techniques
The standard protocol for focal RFA uses a cleaning step in between two ablations (EURO II). To lessen the time, a simplified protocol was developed to skip the cleaning step. A multicenter trial from Pouw et al. assessed if the cleaning step during a focal ablation could be omitted by randomly assigning patients to the standard group (2 × 15 J/cm2 with cleaning) to the simplified group (3 × 12 J/cm2 without cleaning). The study showed that the simplified protocol was noninferior to the standard protocol, without an increased risk of strictures and a reduction of 7 min of average procedure time. 18 Furthermore, van Vilsteren et al. 16 studied a similar concept for circumferential ablation with 12 J/cm2, which also showed the simplified protocol had similar safety and efficacy as compared with the standard protocol.
However, with the current use of new-generation circumferential ablation device, HALO express, which utilizes esophageal sizing and ablation in one catheter, this concept does not seem to apply. According to Belghazi et al., 19 using 10 J/cm as a standard setting (1 × 10 J/cm2– clean - 1 × 10 J/cm2) in the new generation circumferential ablation device allows energy delivery to be more efficient as the device has better apposition to esophageal mucosa allowing for more scarring in 23% of patients after one session. In addition, a simplified protocol (without cleaning) had an increased risk of stenosis and inferior efficacy when using the new generation device. 20 Therefore, it is currently recommend to use the simplified protocol when performing focal ablation, and the standard protocol for the new generation circumferential ablation devices.
Complications
RFA is generally a well-tolerated procedure with low complication rates. Common complications on post-procedure day 3–4 include chest pain and dysphagia, which typically return to baseline without intervention.21,22 According to a meta-analysis by Qumseya et al., 23 the rate of stricture development was 5.6% of patients. However, there is a higher risk of development of RFA preceded by EMR. If strictures do develop, they are managed with endoscopic dilation. Less than 1% of patients develop bleeding from RFA, and the risk is high in patients on antiplatelet or anticoagulation therapy. According to current literature review, there have been no reported cases of esophageal perforations or death associated with RFA.
Outcomes and recurrence after radiofrequency ablation
RFA is an effective treatment for BE in patients with HGD and LGD. According to the landmark randomized controlled trial by Shaheen et al., complete eradication of HGD was achieved in 81.0% of patients in the RFA group compared with 19.0% of patients in the control group. After 12 months, complete eradication of LGD was achieved in 90.5% of patients in the RFA group compared with 22.7% in the control group. 24 It has been shown that 3 years following RFA procedure, 98% and 91% of patients have continued eradication of dysplasia and metaplasia, respectively. 25 With optimal endoscopic treatment of BE, RFA has shown to have long-lasting durability, or maintaining neosquamous epithelium after reaching CEIM, as seen in the recent meta-analyses discussed below.
Recurrence is defined as the development of BE after achieving CEIM. Current literature shows no statistically significant difference when defining CEIM after one versus two negative biopsy sessions.26,27 Endoscopic surveillance is recommended for patients after receiving endoscopic therapy for BE, due to the risk of recurrence. A meta-analysis performed by Krishnamoorthi et al. looked at incidence rates of IM after achieving CEIM using RFA. The secondary outcomes measured incidence rates after use of all endoscopic modalities, and the incidence rates of recurrent dysplastic BE, HGD/EAC. The study included 41 studies which reported a total of 795 cases of IM recurrence after CEIM in 4443 patients over 10,427 patient years of follow-up, the average follow-up after CEIM was 2.5 years with individual studies ranging from 1 to 10.5 years. The study included 21 RFA studies with the pooled incidence of IM recurrence was 9.5% per patient year, with individual studies rates ranging from 0.9% to 28.8%. The pooled incidence rate was 2.0% and 1.2% per patient year for dysplastic BE and HGD/EAC, respectively. Of the 41 studies reviewing all endoscopic modalities, the pooled incidence of IM recurrence was 7.1% per patient year with individual studies rates ranging from 0.7% to 28.8%. The pooled incidence rate for dysplastic BE was 1.3% per patient year and 0.8% per patient year for HGD/EAC. Interestingly, the recurrence rate was numerically lower with hybrid therapy (9.2%) compared with RFA alone (14.3%), but this finding was not statistically significant. In addition, the recurrence rate was higher in RFA studies (9.5%) compared with APC studies (2.9%), but no randomized control trials directly compared these therapies. 28
Another meta-analysis performed by Fujii-Lau et al. included 39 published studies. The primary outcome was the pooled incidence of IM and early neoplasia (dysplasia and EAC) after patients achieved CEIM after endoscopic therapy (RFA and stepwise complete EMR). The average follow-up was at least 1 year after the first endoscopy confirming complete eradication via pathologic abscess of IM and/or dysplasia. The meta-analysis found the pooled incidence of recurrence was 7.5% per 100 patient years with recurrence of IM was 4.8% per 100 patient years and dysplasia was 2.0% per 100 patient years. In patients who received RFA, the incidence of recurrence in total, IM, and dysplasia per 100 patient years was 8.6%, 23%, and 25%, respectively. While in the stepwise complete EMR group, the incidence of recurrence in total, IM, and dysplasia was 13%, 12%, and 13%, respectively. The overall rate of recurrence and IM recurrence was significantly higher in the RFA group compared with step wise complete EMR, 8.6 versus 4.9% per 100 patient years (p = 0.007). However, the majority of recurrences were without dysplasia and were amenable to repeat endoscopic therapy. 29
The importance of adherence to acid control with high-dose PPI is relevant for prevention of recurrence and effective treatment following RFA. Other factors for ineffective eradication therapy can be related to presence of a hiatal hernia, length of BE segment >5 cm, and other relative factors such as stricture of the BE segment before RFA, squamous regeneration within EMR scar, longer duration of dysplasia or neoplasia before therapy, non-Caucasian race, smoking history, and obesity. Buried BE glands rate after RFA is rare (<1%) but is noteworthy to be aware especially during surveillance for Barrett’s following RFA. 30
Pouw et al. observed the long-term outcomes from the Surveillance versus Radiofrequency Ablation study, or SURF study. The original SURF study was a randomized controlled study of 136 patients – 68 RFA and 68 surveillance, which demonstrated that in patients with LGD, RFA significantly reduced the rate of progress to HGD/EAC when compared with the surveillance group. 31 However, after the complete of the SURF study, 15 patients originally in the surveillance group underwent RFA based on patient preferences and study outcomes. They observed an additional 40 months of follow-up for a total median timeframe of 73 months. During this time, HGD/EAC was diagnosed in 1 patient in the RFA group (1.5%) and 23 patients in the surveillance group (33.8%) (p = 0.000). The absolute risk reduction was 32.4% with a number needed to treat of 3.1. Of the 83 patients treated with RFA, 75 patients (90%) achieved complete remission, 7 patients (9%) developed BE recurrence, and 3 patients (4%) developed LGD. This study illustrates a significantly reduced risk of progression to EAC with sustained clearance in patients who receive RFA for BE with confirmed LGD. 32
Cryoablation
While RFA uses thermal energy, cryoablation applies freezing temperatures to induce tissue injury. Rapidly alternating freezing and thawing results in denaturation of proteins, fracturing cell membranes, and tissue destruction. 33 Initially, there are minimal changes to the tissue endoscopically, and after cryoablation the tissue will develop a cherry red appearance with minimal oozing blood. However, the tissue will slough off with time and ideally heal with neosquamous epithelium. Cryoablation can be performed using cryospray with liquid nitrogen or carbon dioxide as cryogenic fluid, CryoBalloon focal ablation, or circumferential techniques. The equipment needed for cryospray include the console unit, two-foot pedals, spray catheter, and decompression tube. Depending on the type of cryogenic fluid used, liquid nitrogen (truFreezeTM) or carbon dioxide (Polar Wand) will determine the type of console unit as it houses the fluid. truFreezeTM has the ability to freeze the mucosal tissue to −196°C and the Polar Wand can freeze to −78°C. The cryoballoon focal ablation requires a portable commercially available system consisting of a handheld controller which houses liquid nitrogen. 34
Current relative contraindications include ulcerative, eosinophilic esophagitis, and mucosal breaks. In patients with surgically altered anatomy, there is an increased risk of perforation, and cryotherapy is therefore not recommended.35,36
Cryospray techniques
After esophageal mucosa is inspected and the landmarks are measured and photographed, the truFreezeTM 213-cm-long catheter is inserted through the working channel of the endoscope. In addition to the catheter, a 20 F dual-channel decompression tube is placed beside the endoscope and placed in the stomach, over a guidewire to allow for decompression when the flow of liquid nitrogen is initiated, preventing luminal perforation. The catheter is placed a few millimeters away from the Barrett’s segment, and the foot pedal is initiated resulting in flash freezing. Two to three cycles may be applied between each thaw.13,34
CryoBalloon techniques
The CryoBalloon Focal Ablation System is composed of a 175-cm balloon-tipped catheter that is attached to a handheld trigger. The balloon is compatible with endoscopes that have ⩾3.7 mm working channel. Esophagus is intubated, the catheter is then passed through the working channel, the trigger is used to inflate the 3-cm balloon, and continued activation of the trigger will result in cryogen delivery through a 1 mm side opening in the catheter within the balloon. The inflated balloon is cooled by spraying nitrous oxide via the diffuser within the inflated balloon, which subsequently freezes the targeted mucosa to −80°C. The site of targeted delivery for the cryotherapy can be controlled by rotating the catheter in addition to advancing and withdrawing it within the balloon using the foot pedals using the Next-Generation C2 CryoBalloon™ Ablation System. 34
Complications
Cryoablation therapies are generally well tolerated. Most often reported adverse events include self-limited chest pain, esophagitis, esophageal ulcers, and dysphagia.37–39 Stricture development has been reported in 3–13% of patients which were amenable to endoscopic dilation.33,40 However, more significant complications have been reported. According to Verbeek et al., 41 in a prospective single center case series using carbon dioxide cryospray, one patient developed a gastric perforation believed to be attributed to learning curve and catheter positioning. In another study using liquid nitrogen cryospray, one patient with Marfan syndrome developed a gastric perforation. 42 No mortality was reported in the use of endoscopic cryoablation. While there is a risk of perforation, appropriate supervision and development of skills should minimize poor outcomes.
Outcomes and recurrence after cryoablation
While RFA is a widely used ablative therapy with studies supporting its efficacy and safety, cryotherapy is frequently used as salvage therapy. According to a meta-analysis by Visrodia et al., 43 cryoablation can be used as a second line option for patients who failed RFA as 45.9% of patients achieve CEIM and 76% achieve complete eradication of dysplasia after failing RFA. Previous studies did not evaluate the use of cryoablation as a primary treatment. However, a meta-analysis by Hamade et al. evaluated the use of cryoablation as primary treatment of BE showing 69.35% of patients achieved CEIM, 97.9% complete eradication of neoplasia, 7.3% had persistent dysplasia, and 4% progressing to cancer. The recurrence rate of IM in patients who underwent primary cryoablation therapy was 19.1 per 100 patient years, 10.4 per 100 patient years recurrence rate of neoplasia, and 5 per 100 patient years for recurrence of dysplasia. 44
Recent prospective clinical trial involving 11 academic and community hospitals studied the use of multifocal cryoablation for BE eradication. The inclusion criteria included “treatment naïve” BE of 1–6 cm length with pathology confirmed LGD, HGD, or intramucosal adenocarcinoma. If visible lesions were seen, patients underwent EMR prior to enrollment. A nitrous oxide cryoballoon focal ablation system was used for all visible columnar mucosa up to five sessions. The primary outcome was CEIM at 1-year follow-up. Of the 120 patients enrolled, 20% had intramucosal adenocarcinoma, 56% HGD, and 23% LGD. In the intention to treat analysis, CEIM was achieved in 72%, while the per-protocol analysis found that 91% of patients achieved CEIM. 45 Further comparative trials, comparing RFA and cryoablation, are needed to assess the effectiveness of cryoablation as primary treatment for BE.
Photodynamic ablation
Historically, PDT used systemically administered chemical agents, or photosensitizers, approximately 2 days prior to the endoscopic procedure to treat BE. The photosensitizers are retained at higher concentrations in neoplastic tissue which are then activated by endoscopically delivered red light of specific power and wavelength resulting in cytotoxicity due of free radical formation inducing cell damage and apoptosis allowing for significant depth of tissue penetration and wide application. The results of PDT have been variable with decreased rates of eradication as compared with RFA including cost, and therefore have fallen out of favor and not widely used.46,47
Complications
While 4-aminolevulinic acid has less side effects compared with porphimer sodium due to the shorter half-life and less mucosal penetration, the rate of stricture formation has caused this therapy to largely drop out of favor. The rate of stricture formation has been shown to be approximately 36%. 48 In addition, 60% of patients who received porphimer sodium as the photosensitizers develop photosensitivity. 49 This procedure has currently been archived for the abovementioned reasons.
Argon plasma coagulation
APC delivers an electrical energy through ionized plasma of argon gas through a contact-free probe to the targeted tissue. Typically, APC is applied with energy settings ranging from 30 to 90 W at a rate of 1–2 liters/min. Wronska et al. performed a randomized clinical trial to study the impact of PPI dose (40 mg or 120 mg of omeprazole) and energy settings (60 W or 90 W) on the rate of complete ablation of LGD BE at 6 weeks. The primary outcome was complete endoscopic and histologic ablation of BE. A total of 71 patients were randomized into 90 W/120 mg, 90 W/40 mg, and 60 W/120 mg groups with complete ablation rates at 6 weeks of 78%, 60%, and 74%, respectively. At 2 years post treatment, the complete ablation rates were 70% for 90 W/120 mg, 52% for 90 W/40 mg, and 65% for 60 W/120 mg. However, the differences were not significant, illustrating the APC power setting and PPI dose did not impact efficacy of BE ablation. 50
Complications
While standard APC was one of the first techniques for nondysplastic BE, significant rates of complications are observed including bleeding, pneumomediastinum, perforation, and buried BE glands.50,51 In the study mentioned, they found chest pain and discomfort was a more frequent adverse event in the 90 W group compared with the 60 W group (p < 0.001). Of the 71 patients who were randomized, 1 patient (1.4%) developed an esophageal perforation and 2 patients (2.8%) developed esophageal stenosis. 50
Outcomes and recurrence after argon plasma coagulation
New modifications such as hybrid-APC are becoming a widely used modality as supplemental treatment for residual small BE islands after initial RFA and have reported less complications of bleeding and perforation. Hybrid-APC uses the standard APC application followed by high-pressure needleless submucosal injection of saline via built-in water jet within the APC channel. A randomized controlled trial from Manner et al. studied the efficacy and safety of Hybrid-APC for BE. Patients were selected if they had residual BE segment of at least 1 cm after EMR. A total of 50 patients were included for Hybrid-APC ablation, and 48 patients (96%) achieved macroscopically complete remission after a median of 3.5 APC sessions. In addition, 39 patients (78%) achieved histopathological remission of BE. Complication rates included 1 patient with esophageal stricture (2%) and 11 patients (22%) observed minor adverse events including retrosternal pressure/pain, heartburn, and/or odynophagia. 52 While Hybrid-APC is effective in treating BE, ongoing comparative studies are needed to compare RFA with Hybrid-APC.
Immediate post-procedure care
After endoscopic therapy, patients may experience dysphagia or chest discomfort which is treated with viscous lidocaine or liquid acetaminophen. Patients should be advised to adhere to a liquid diet for approximately 24 h post procedure, then advance their diet as tolerated. Typical post-procedure maintenance therapy includes a PPI and sucralfate. Often, patients are able to discharge home after the procedure, and rarely require inpatient admission for pain control and observation. 13
Surveillance following endoscopic therapy
According to a meta-analysis performed by Krishnamoorthi et al., 28 the pooled incidence of intestinal metaplasia recurrence was 7.1% per patient year rate when reviewing all endoscopic treatment modalities for BE. Therefore, the current recommendations for endoscopic surveillance in patients with HGD or intramucosal carcinoma following CEIM is recommended every 3 months for 1 year, every 6 months during the second year, and yearly afterwards. In patients with LGD prior to therapy and CEIM is achieved, endoscopic surveillance is recommended every 6 months for 1 year and yearly thereafter.
Learning curve of advanced ablation therapies
American Society for Gastrointestinal Endoscopy (ASGE) core curriculum guidelines for endoscopic ablation therapy training recommends proficiency in upper endoscopy, colonoscopy, and hemostasis prior to beginning training in mucosal ablation techniques as well as knowledge of management for potential complications. In order to have the capability of performing advanced procedures, institutions should have high-definition endoscopy, narrow band imaging, blue light imaging, and chromoendoscopy available to evaluate for mucosal enhancement. 53 Current studies reviewed outcomes of patients who were treated at high-volume centers (>100 enrolled patients), medium-volume centers (51–100), and low-volume (<50) centers. There was no correlation with the center volume and CEIM or complete eradication of dysplasia. However, there is a statistically significant difference in high-volume centers resulting in lower recurrence rates compared with low-volume centers. According to Lipman et al., 54 endoscopists need 18 supervised cases of endoscopic ablation to achieve competency in endoscopic treatment of Barrett’s dysplasia. Another study found that the number of treatment sessions to achieve CEIM decreases as the center’s number and experience of the endoscopist increases suggesting a learning curve effect, which was shown to occur at approximately 30 patients. 55 When reviewing a patient for endoscopic therapy, we recommend a multidisciplinary approach including an advanced endoscopist, thoracic surgeon, oncologist, and GI pathologist, as well as reviewing the endoscopist experience and volume of the ablation program to provide the most appropriate therapy for the patient. We have suggested an approach to the management of BE with neoplasia in Algorithm 1.
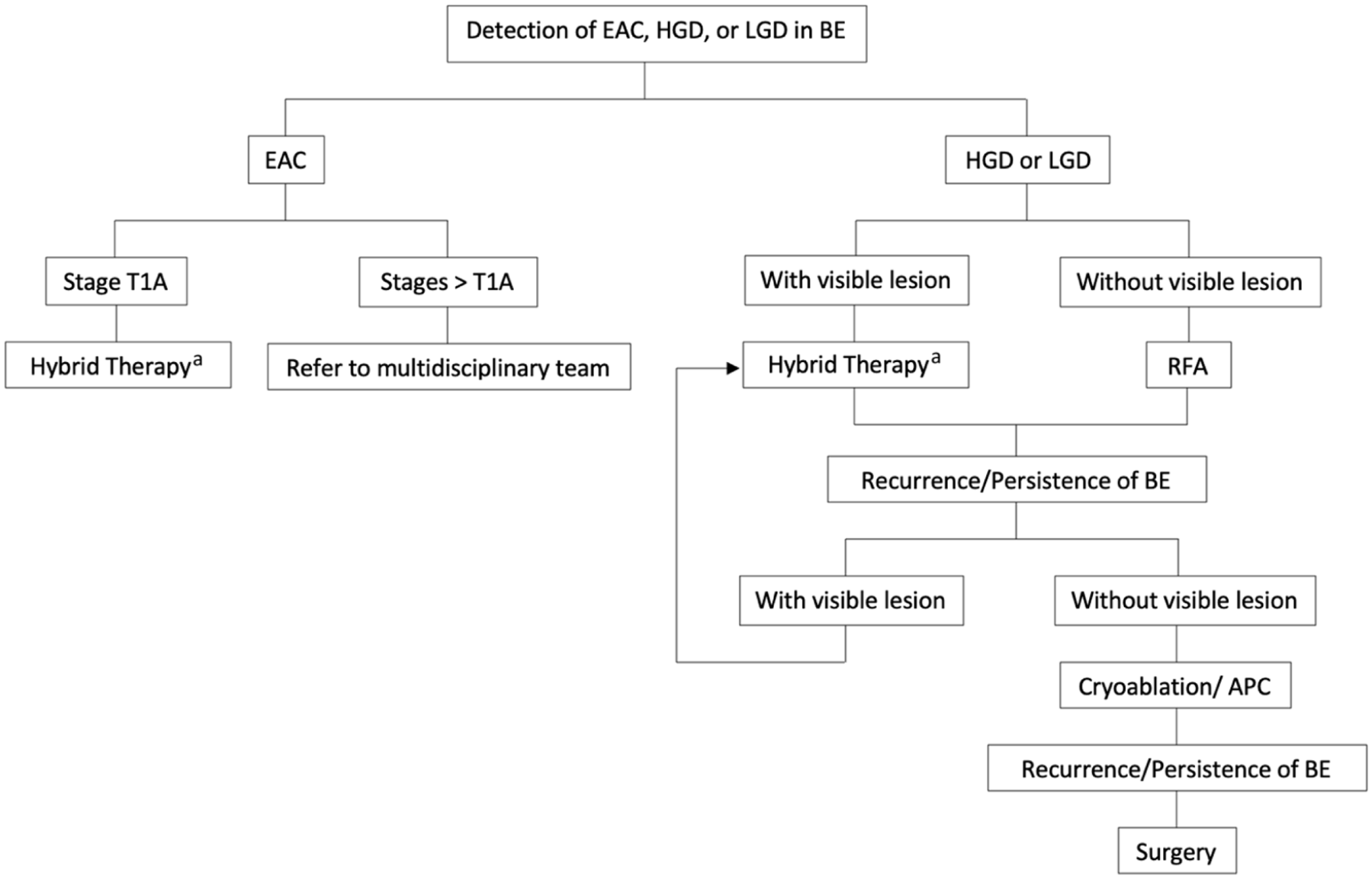
Suggested approach in the management of confirmed BE with dysplasia.
Footnotes
Author contributions
RS & RM complied the data, drafted and edited the mansucript. SP edited the manuscript and critically reviewed it for intellectual content.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
