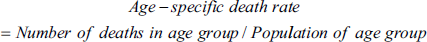Abstract
Importance
Existing literature provides only sparse data concerning US death trends for skin melanoma.
Objective
To evaluate the trends in skin melanoma-related mortality in the United States.
Design, Setting, and Participants
A retrospective cohort study was conducted using mortality statistics spanning from January 1999 to December 2020 from the CDC Wide-Ranging Online Data for Epidemiologic Research (WONDER) database. The data included all US age cohorts where cutaneous melanoma was recorded as an underlying or contributing cause of death. Statistical review was performed in March 2024.
Exposure
All age groups with skin melanoma recorded as a contributing and underlying cause of mortality.
Main Outcomes and Measures
Primary outcomes were the Age-Adjusted Mortality Rates (AAMR) for cutaneous melanoma per 100,000 US citizens, differentiated by sex, race/ethnicity, and geographical region
Results
Between 1999 and 2020, a total of 184,416 melanoma-related deaths occurred in all age groups. Of these, 119,557 (64.8%) were men and 64,859 (35.16%) were women, and 2,756 (1.5%) were Black, 18,262 (97.7%) were White, 352 (0.19%) were Alaskan Indian or American Native, and 1,046 (0.56%) were Asian or Pacific Islanders. The overall AAMR during the study duration was 2.52; AAMR was higher for men compared with women (2.52 vs. 1.6) and higher for white adults compared with non-Hispanic and Black adults (2.89 vs. 2.69 vs. 0.38, respectively). Across most demographic and regional subgroups, AAMR increased in non-metropolitan areas from 2.86 in 1999 to 3.04 in 2007 and then decreased from 2008 to 2020. There were significant regional differences in the burden of melanoma-related mortality, with states in the upper 90th percentile (such as Utah, Idaho, Wyoming, Oklahoma, and West Virginia) having a higher burden than those in the lower 90th percentile.
Conclusion and Relevance
The skin melanoma-related mortality rate decreased among all age groups between 1999 and 2020. White adults had a 0.2- and 2.51-fold increase in AAMR from 1999 to 2009 compared with non-Hispanic and Black adults, respectively, and then it decreased for all races from 2009 onwards. Specific health policy interventions are necessary to assess the growing death rate from cutaneous melanoma across all age ranges. Public health approaches, driven by data, must prioritize prevention, early detection, effective treatment, and equitable reduction of disparities.
Introduction
Melanoma, a type of skin cancer stemming from uncontrolled growth of melanin-producing cells in the dermis, ranks as the third most prevalent skin cancer and the primary contributor to skin cancer-related deaths. Within the demographic most susceptible to melanoma—older fair-skinned males—the mortality rate is thought to have risen over time. 1 It originates from melanocytes, specialized cells that safeguard the skin from UV radiation by producing pigment. When mutations occur in the DNA of melanocytes, it can cause a loss of regulation in genes controlling growth, the release of self-stimulating growth factors, and a decrease in adhesive molecules. These disruptions disturb the normal balance of cells, leading to unchecked melanocyte growth and potentially the development of a mole. 2
A phenotypic risk factor for melanoma is skin that is prone to sunburn, while the most significant genetic factors studied in recent years revolve around variations in the melanocortin-1 receptor (MC1R) gene. A major external factor contributing to melanoma is exposure to UV radiation, particularly intermittent exposure to sunlight.3, 4 Melanoma rates are on the rise among white populations, particularly in regions where fair-skinned individuals receive significant sun exposure, as evidenced by skin cancer data from Australia. In Europe, the incidence rate is approximately <25 new cases per 100,000 people, while in the United States, it is around 30 per 100,000. Recent trends show a notable increase in melanoma cases among individuals aged over 60 and across all age groups. Projections based on incidence curves indicate that this trend is likely to persist in the years ahead. 4
During the 1990s and 2000s, significant progress was made in the field of melanoma treatment with the introduction and acceptance of sentinel lymph node biopsy and the conduct of pivotal clinical trials such as the Multicenter Selective Lymphadenectomy Trials (MSLT-1 and MSLT-2) and the Dermatologic Cooperative Oncology Group trial (DeCOG). More recently, there have been notable advancements in utilizing immunotherapies to treat Stage 3 and 4 melanomas. Analyzing data trends related to disease burden will offer valuable insights into the global impact of these interventions. Additionally, evaluating recent data will establish a foundation for assessing the effectiveness of emerging therapies as they become more widely accessible. 5 In the context of increased resources and advancements in healthcare, melanoma incidence rates have shown a decrease in the United States. However, despite these improvements, disparities persist in screening and treatment access. This descriptive study aimed to evaluate malignant melanoma mortality rates in the United States spanning from 1999 to 2020. It primarily focused on investigating trends across various demographic factors such as sex, race/ethnicity, region, gender, and location of death. The objective was to analyze variations in melanoma mortality trends among different population groups, offering insights into the occurrence of melanoma-related deaths.
Methods
Study Setting and Population
This descriptive study retrieved death certificate data from the CDC WONDER (Centers for Disease Control and Prevention Wide-Ranging Online Data for Epidemiologic Research) database, analyzing the period 1999–2020 for skin melanoma mortality. To identify relevant deaths, we used the International Statistical Classification of Diseases, 10th Revision (ICD-10) code C43 (Malignant Melanoma of the Skin). 6 The multiple cause-of-death public use records were screened to select deaths where melanoma was noted as either an underlying or contributing cause. The study included all ages, defined as descendants ranging from <1 to >85 years. As this research used a de-identified, government-issued public dataset, it was exempt from local institutional review board (IRB) approval and adhered to the STROBE guidelines for reporting.
The age-specific mortality rate for each age group was multiplied by the equivalent weight from the given standard population to determine the age-adjusted rate. This result was then added up for all age groups and multiplied by 100,000. Here is the exact formula that determines the age-adjusted rate: The sum of (Age-specific death rate × Standard population weight) × 100,000 is the age-adjusted mortality rate. The number of deaths for a particular age group divided by the population of that age group is the age-specific death rate.
The “standard population weight” for an age group is calculated by (Standard population weight = Population for age group/Sum of age group populations) for all age groups in the query.
Moreover, the research employed the Joinpoint Regression Program (version 4.9.0.0, National Cancer Institute) to examine the annual national patterns in skin melanoma mortality. Utilizing log-linear regression models that considered temporal variations, the Joinpoint Regression Program facilitated the computation of the annual percent change (APC) in Age-Adjusted Mortality Rate (AAMR), accompanied by 95% confidence intervals (CIs). This methodology allowed for the detection of noteworthy fluctuations in AAMR across different periods.
Data Abstraction
We collected data encompassing population size, year, death locale, demographic factors, urban-rural status, region, and state. Demographics included gender, age, and race/ethnicity. Death locations were categorized as medical facilities (outpatient, emergency room, inpatient, death on arrival, or unknown status), home, hospice, and long-term care facilities. The analyzed race/ethnicity groups were African American, American Indian/Alaskan Native, Asian/Pacific Islander, Hispanic/Latino, non-Hispanic, and White individuals. This death certificate-based data has been routinely used in WONDER database research. Urbanicity was defined using the 2013 US census criteria from the NCHS Urban-Rural Classification Scheme: urban (large metropolitan [≥1 million], medium/small metropolitan [50,000-999,999]) or rural (<50,000). Regions adhered to the U.S. Census Bureau’s definitions: Northeast, Midwest, South, and West. 7
Statistical Analysis
We calculated crude and age-adjusted mortality rates (AAMRs) per 100,000 residents from 1999 to 2020 to assess national trends in skin melanoma deaths. The analysis generated 95% confidence intervals (CIs) and was segmented by year, sex, race/ethnicity, state, and urban-rural classification. Crude mortality rates were ascertained by dividing the total deaths attributed to melanoma by the corresponding annual US population. AAMRs were determined by standardizing the melanoma-specific deaths to the relevant US population for that year. 8
For a comprehensive understanding of national yearly trends in Skin melanoma cancer-related mortality, we employed the Joinpoint Regression Program (Joinpoint V 5.0.2, National Cancer Institute). This program determined the APC in AAMR along with a 95% CI. The method involves fitting log-linear regression models to identify significant changes in AAMR over time where temporal variation is observed. The APCs were categorized as increasing or decreasing based on whether the slope depicting the change in mortality significantly differed from zero using 2-tailed t-testing. A significance level of P < .05 was considered statistically meaningful. 9
Results
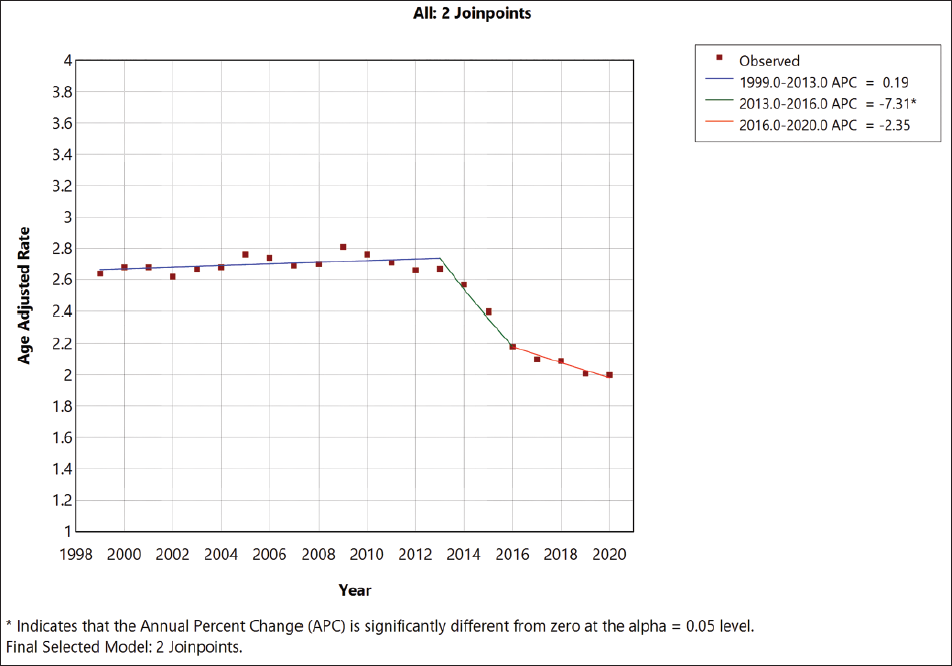
Between 1999 and 2020, a total of 184,416 melanoma-related deaths occurred in all age groups (Table S1 in the supplemental material). Of these, 119,557 (64.8%) were men and 64,859 (35.16%) were women, and 2,756 (1.5%) were Black, 180,262 (97.7%) were White, 352 (0.19%) were Alaskan Indian or American Native, and 1,046 (0.56%) were Asian or Pacific Islanders. Out of 184,416 non-Hispanic individuals were 179,598 (97.38%), Hispanics were 4,529 (2.4%) and not stated were 289 (0.15%) (Table S4 in the supplemental material). Of 184,416 deaths that had information available on the location of death, 45,692 (24.77%) occurred within medical facilities, 25,392 (13.7%) occurred in nursing homes/long-term care facilities, 16,257 (8.81%) occurred in hospice, 85,624 (46.42%) occurred at home and other locations were 10,962 (5.94) (Table S6 in the supplemental material). The overall AAMR during the study duration was 2.52; AAMR was higher for men compared with women (2.52 vs. 1.6) and higher for white adults compared with non-Hispanic and Black adults (2.89 vs. 2.69 vs. 0.38, respectively). Overall, the AAMR for skin melanoma mortality in the population decreased from 2.64 in 1999 to 2.0 in 2020 (Figure 1; Table S1). AAMR increased from 1999 to 2013 (APC, +0.19% [95% CI, −0.02 to 0.464]) and there was a sharp decline from 2014 to 2016 (APC, −7.30% [95% CI, −8.30 to −5.8 ]) and from 2016 to 2020 it remained steady (APC, −2.35% [95% CI, −3.5to 0.48]).
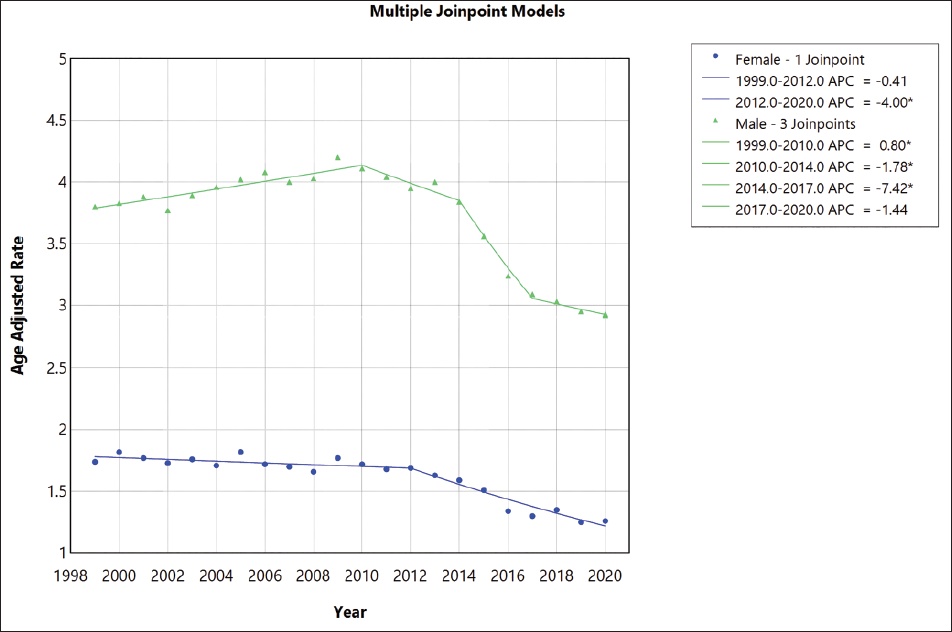
Skin melanoma mortality decreased in men and women. Specifically, the AAMR in men decreased from 3.8 in 1999 to 2.93 in 2020. {APC (1999-2010),0.79% [95% CI, 0.57 to 1.14]}, {APC (2010-2014), −1.77% [95% CI, −2.81 to −0.59]}, {APC (2014-2017),-7.41% [95% CI, −8.331 to −5.96]}, {APC (2017-2020), −1.43% [95% CI, −2.61 to 0.97]}, while the AAMR in women decreased from 1.74 in 1999 to 1.25 in 2020 {APC(1999-2012), −0.40% [95% CI, −0.82 to 0.14]} and {APC (2012-2020), −3.99% [ 95% CI, −5.11 to −3.15]}. White adults had the highest AAMR which increased from 2.97 in 1999 to 3.16 in 2009 (APC, 0.74% [95% CL 0.46, to 1.52]) then it decreases from 2010 to 2014 (APC, −0.93% [95% CL −1.82, to −0.005]) and there is a rapid decline from 2014 to 2017 (APC, −7.14% [95% CL −8.39, to −5.32]) and it remained steady from 2017 till 2020 (APC, −0.97% [95% CL −2.84, to 2.57]).
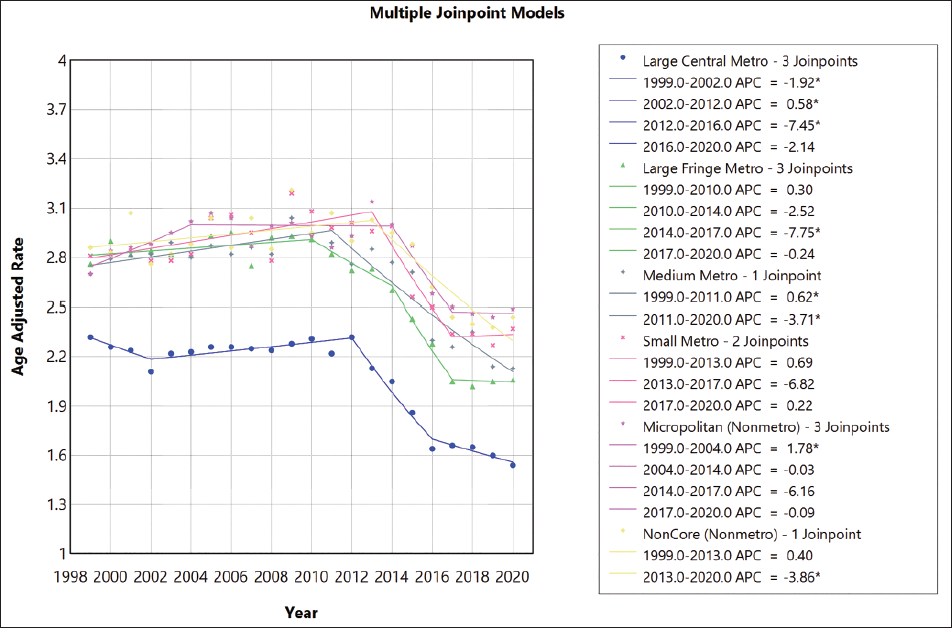
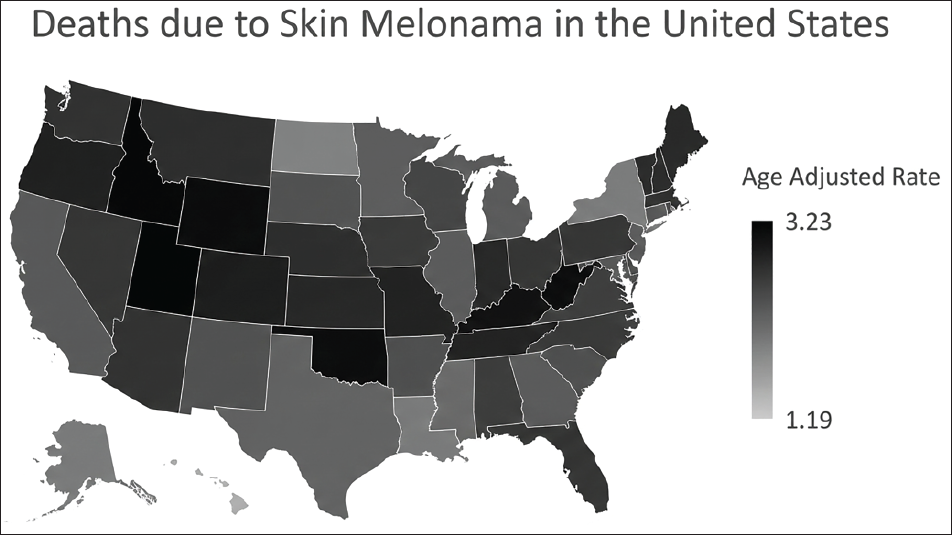
For non-Hispanic individuals, AAMR increased from 1999 to 2013 (APC, 0.39% [95% CL 0.16 to 0.66]), and then there is a rapid decline from 2013 to 2017 (APC −6.51% [95% CL −8.44 to −4.66]), and a steady decline is seen from 2017 to 2020 (Figure 2). For White adults, AAMR increased from 1999 to 2009 (APC 7.5% [95% CL 0.46 to 1.52]) and slightly decreased from 2009 to 2014 (APC 0.93 [95% CL −1.82 to −0.005]). There is a rapid decline from 2014 to 2017 (APC −7.14% [95% CL −8.39 to −5.32]), and it remained steady from 2017 to 2020 (APC −0.97% [95% CL −2.84 to 2.57]) (Figure 3; Tables S4, S5 in the Supplemental). Non-metropolitan areas had the highest AAMR, which increased from 2.86 in 1999 to 3.04 in 2007 (APC 1.76% [95% Cl 0.66 to 5.56]), and there was a decline in mortality rate from 2008 to 2020. Mortality rates similarly decreased in large and medium/small metropolitan areas during the study period (Figure 4; Table S8 in the supplementary material). There were significant regional differences in the burden of melanoma-related mortality, with states in the upper 90th percentile (such as Utah, Idaho, Wyoming, Oklahoma and West Viginia) having a significantly higher mortality burden compared with those in the bottom 10th percentile (Louisiana, New York, North Dakota, Hawaii, District of Columbia) (Table S7 in the supplementary material).
Discussion
Analysis of the two-decade mortality data for cutaneous melanoma from the CDC WONDER database yielded key observations. The mortality rate showed a steady rise from 1999 to 2013 (APC: 0.19%). This was succeeded by a rapid decrease from 2013 to 2016 (APC: −7.3%), before a further modest decline occurred from 2016 to 2020 (APC: −2.35%). Furthermore, throughout the two-decade span, it was consistently observed that men had a higher annual mortality rate (AMR) in comparison to women.
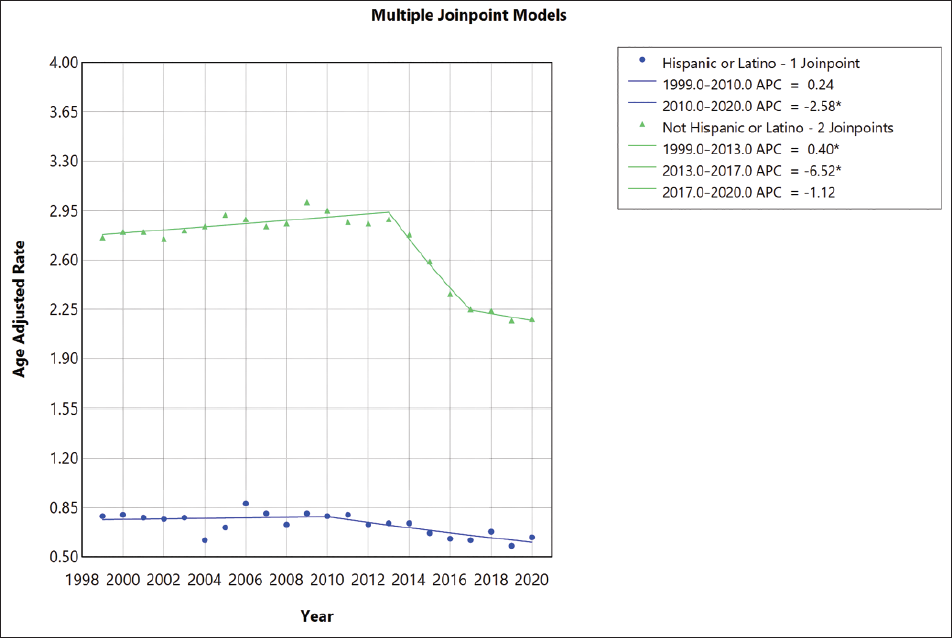
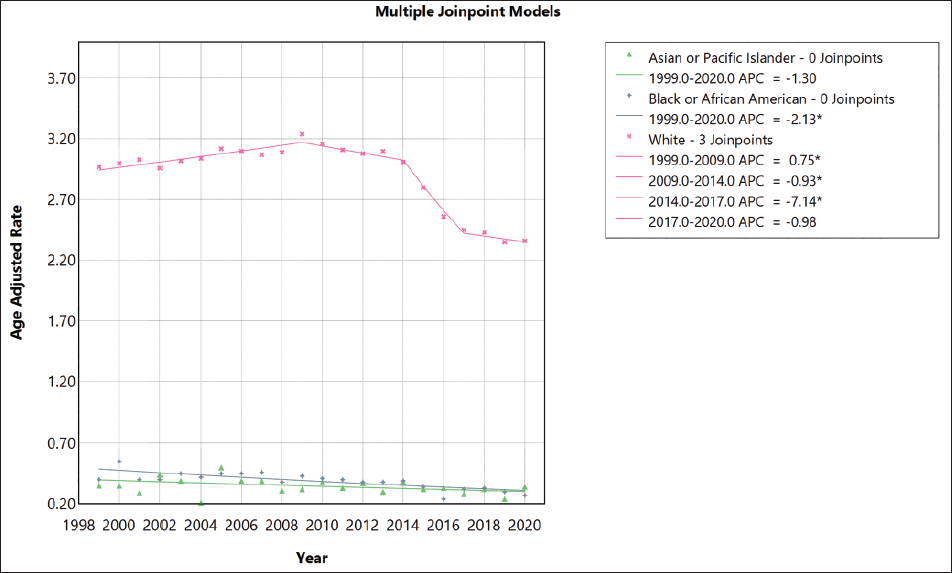
Different racial and ethnic populations showed diverse mortality results across the two decades. White patients consistently registered the highest AAMR. Following them were non-Hispanic individuals, then Black or African American and Asian or Pacific Islander individuals, and finally, American Indian or Alaska Native populations. 10 The higher melanoma mortality incidence among White men compared to other ethnic groups likely results from a complex interplay of genetic, lifestyle, socioeconomic, and health disparity factors. Current data suggests a historical gap between skin melanoma screening rates among Black individuals and White individuals, 11 although recent trends indicate a gradual narrowing of this disparity. The persistent higher incidence of melanoma among White individuals is associated with factors like blistering sunburns after acute sun exposure, ever-sunburn, a family history of melanoma, and a personal history of non-melanoma skin cancer. 12 Additionally, White individuals are more likely to be diagnosed with melanoma before age 50, underscoring the limitations of previous screening guidelines.
Various additional elements contribute to the risk of skin melanoma among White individuals, such as genetic susceptibility and variations in how the disease manifests. The increased susceptibility to melanoma observed in non-Hispanic, White, and multiracial populations is correlated with the presence of phenotypes in White individuals that are more prone to sunburn. Studies suggest that despite similar levels of DNA damage from ultraviolet radiation (UVR) between darker-skinned individuals and White individuals, the incidence of melanoma varies and is more prevalent in White individuals. 13 This could be attributed to the potential for more efficient repair of UVR-related DNA damage in non-White individuals and/or its confinement to the superficial layers of the skin. 14
A 2009 review by the U.S. Preventive Services Task Force (USPSTF) on skin cancer screening concluded that insufficient evidence existed to confirm that full-body skin checks performed by primary care providers were effective for the early discovery of cutaneous melanoma. 15 Due to the significance of melanoma, the USPSTF revisited this topic in a 2015 Draft Recommendation Statement. Once more, they concluded that the existing evidence did not sufficiently support the practice of skin cancer screening, mainly because there were no randomized controlled trials assessing the efficacy of whole-body skin examination. 16 However, adults with a family history, genetic predisposition, pertinent past medical history, or history of sun exposure and fair skin are recommended to be regularly screened. Collaborative efforts and advancements in treatment modalities have significantly improved AAMR, particularly in White patients, within the last two decades. Advancements in screening techniques, regular skin self-examinations, physical check-ups, using dermoscopy, and being vigilant with early intervention are the key reasons for declining AAMR.
The lower AAMR consistently observed in women throughout the study period compared to men may be attributed to multifaceted factors (Figure 5). The influence of sex steroid hormones on melanoma remains uncertain. 17 A higher expression of estrogen receptor beta (ER-ß) is linked to lower invasiveness and thickness of melanoma tumors. 18 Adding estrogen externally has been found to impede tumor growth both in laboratory settings and in metastatic tumors. 19
Variations in autosomal genetic traits play a crucial role in elucidating the reasons behind the gender gap observed in melanoma. Earlier research has highlighted notable differences in genetic effects for melanoma between males and females. One non-synonymous single nucleotide polymorphism (SNP), identified as rs16891982 in the SLC45A2 gene, shows a markedly elevated association with melanoma risk in men versus women. The calculated odds ratios underscore this disparity, being 5.5 for males and 2.37 for females. 20 Variations in specific genes (TYR, GPR143, and F2RL1) were observed to heighten the susceptibility to melanoma more in males compared to females. Moreover, these variations were linked to darker pigmentation and increased sun tolerance specifically in females, with no such associations found in males. 21 Disparities in outcomes among cutaneous melanoma patients based on gender are linked to inherited mutations within genes involved in the apoptosis pathway, namely TP53, MDM2, and BCL2. Polymorphisms in the MC1R gene, associated with red hair phenotypes, were observed to have a differing impact on melanoma susceptibility when comparing male and female patients. Melanoma has a high rate of missense mutations. 22
Certain health factors which contribute to less AAMR in females like skin awareness, self-examination, and professional examination are all linked to detecting thinner lesions in females. 23 Brady et al. demonstrated that females exhibit a higher propensity for self-detecting melanoma lesions compared to men. Additionally, other studies have indicated that females demonstrate greater skin awareness and are more inclined to use sunscreen. 24
This study shows that the AAMR in the population has increased with the passing years till 2013. Between 2013 and 2020, the rate of mortality decreased, indicating a positive outcome possibly linked to the introduction of effective treatments during this period. This trend implies that the adoption of new pharmaceutical interventions correlates with a reduction in mortality rates across the United States. These findings provide strong encouragement for ongoing research and development of similar therapies. However, it is crucial to also consider the accessibility of these treatments and their associated healthcare expenses. After newer therapeutic options were introduced near 2011, with peak adoption occurring post-2013, the United States witnessed a substantial decline in melanoma death rates between 2013 and 2020. This reduction strongly suggests the beneficial effect of more efficacious, recently available treatments. The fact that even older individuals, aged over 75 years, experienced extended survival with melanoma suggests that these therapies are not only effective but also well tolerated in this demographic. 25
There is a growing necessity to enhance health guidelines to address the rising risk factors associated with skin melanoma by implementing measures that can be modified and limited to reduce the occurrences of this disease. Additionally, screening should commence for individuals under the age of 50 especially males who exhibit risk factors for developing skin melanoma. This information will raise awareness among the public and help allocate resources more strategically to achieve improved outcomes.
We also report significant racial and ethnic disparities as AAMR among White individuals increases almost 0.2% and 2.51% times higher than non-Hispanic and Black adults respectively, and the AAMR is higher among white adults and non-Hispanic adults, from 1999 to 2009 for the White population and from 1999 to 2013 for the non-Hispanic population. Disparities in socioeconomic status and health insurance coverage have been identified as underlying causes of healthcare discrepancies between non-Hispanic people and the White population. 26
Between 2011 and 2015, the cancer incidence rate among non-Hispanic Black (NHB) men was 9% higher compared to non-Hispanic White (NHW) men. However, by 2016, the Black-White disparity in overall cancer mortality had decreased to 14% when considering both men and women combined (183.6 vs. 160.7 per 100,000 population). This trend toward a narrower racial gap in cancer mortality has been a positive development over the recent decade. Nonetheless, there is a concerning widening of socioeconomic disparities, particularly evident in regions with higher low income and lack of insurance coverage. This exacerbates the burden of preventable cancers, including melanoma, on individuals residing in such areas. 26
Substantial regional differences were observed in the mortality rates associated with skin melanoma, with states ranking in the upper 90th percentile for skin melanoma mortality (such as Utah, Idaho, Wyoming, Oklahoma, West Viginia) bearing a significantly heavier mortality burden compared to those in the bottom 10th percentile (including Louisiana, New York, North Dakota, Hawaii, District of Columbia). This wide geographical divergence could be attributed to factors such as access to outpatient melanoma screening facilities, the influence of state legislation on the expansion of Medicare and Medicaid, and the presence of comorbid conditions that greatly affect disease outcomes (Figure 6).
Effective health policy is essential for improving public health outcomes related to skin melanoma mortality. Screening, diagnosis, and treatment initiatives are vital in reducing the burden of this disease. Screening efforts, when implemented effectively, have the potential to detect melanoma at earlier stages, leading to better prognosis and reduced mortality rates. 27
Despite efforts to improve access to screening and treatment, disparities in melanoma outcomes persist, particularly among underserved populations. Health policies need to address these disparities by ensuring equitable access to screening, regular self-skin examination, physical examination by dermoscopy, and treatment services for all individuals, regardless of socioeconomic status or geographical location. Furthermore, public health campaigns and educational programs can play a crucial role in raising awareness about the importance of sun safety practices, early detection, and seeking medical attention for suspicious skin lesions. 28 Organizations like the CDC and the IARC should champion effective strategies to mitigate the negative effects of UVR exposure. These measures encompass avoiding direct sunlight, especially during midday peaks; using a wide-brimmed hat to protect the head, face, ears, and neck; wearing clothes to cover exposed skin; using UV-blocking sunglasses; and applying SPF 15+ sunscreen. In conclusion, the current era holds great promise for treating and preventing melanoma. The necessity is plain: prompt intervention is crucial to lessen the disease burden across the United States. 29
Limitations
The use of ICD codes and death certificates to determine the cause of death will likely result in a very incomplete representation of melanoma mortality. Additionally, the data set is highly dependent on information provided by healthcare providers, which may introduce potential errors or inaccuracies, particularly concerning essential demographic variables such as race and ethnicity. Moreover, greater digital health record diagnosis of skin melanoma mortality may prompt higher reporting on death certificates. This increased reporting causes a rise in death certificate trends for melanoma mortality. Such an increase may not reflect the factual trends of actual melanoma mortality within the population, distorting the true statistics or associated comorbidities. Furthermore, the lack of detailed data about individual risk factors or potential confounders influencing the development or progression of skin melanoma will limit a comprehensive analysis of complex relationships among variables. Significantly, our data shows a distinct increase in AAMR among White individuals. Our findings displayed a notable upswing and were included in the manuscript, even if they did not fully meet the criteria. We recognize the challenges associated with grouping diverse populations, such as categorizing Pacific Islanders and Asians and non-Hispanics under the same umbrella, as distinct subgroups within this classification may exhibit differing mortality rates. However, we must recognize that this study might not completely reveal all these differences because of the specific grouping used.
Footnotes
Data Availability Statement
The CDC WONDER database, created by the CDC in the United States, is an openly available online system. It supplies a very extensive range of public health data that allows users to monitor disease outbreaks, identify patterns, and assess the effectiveness of public health measures. The database is free, and individuals can readily access this information through the CDC WONDER website.
Declaration of Conflict of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
NA.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Patient Consent
NA.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.