Abstract
Introduction
Renal cell carcinoma (RCC) accounts for 2% of all cancers globally. Majority of RCC are sporadic, and only 2%-5% are associated with inherited tumor syndromes. The latest World Health Organization (WHO) classification of renal cell tumors (5th edition) has introduced a new category of molecularly defined renal tumors, which include fumarate hydratase (FH)-deficient RCC and succinate dehydrogenase (SDH)-B-deficient RCC among others. These entities can be screened in tumor tissue using immunohistochemistry (IHC). Ours is the first study from India documenting screening of renal neoplasms for FH and SDH deficiency, using IHC.
Materials and Methods
In our study, we screened 297 cases of renal cell neoplasms using IHC “SDH-B” for SDH-B deficiency, and “FH” & 2-succino-cysteine (“2-SC”) for FH-deficient RCC.
Results
We found 0.7% (2/297 cases) of FH-deficient RCC. Both these patients were of young age (20 and 37 years), with stage III disease showing early metastasis. The common feature of vesicular nuclei with conspicuous eosinophilic nucleoli was seen in both cases. We could not perform the mutational analysis in these cases. We did not find any case with SDH-B deficiency.
Conclusion
One should keep a low threshold in diagnosing the aggressive FH-deficient RCC, and use IHC panel for the same, especially in cases with papillary architecture and grade 3 nuclei. These patients also require germline testing and surveillance of other family members.
Introduction
Majority of renal cell carcinomas (RCC) are sporadic, and 2%-5% are associated with inherited tumor syndromes. The inherited tumors develop at an earlier age which can be multifocal and bilateral, with multiorgan involvement. 1 The latest World Health Organization (WHO) classification of renal cell tumors (5th edition, 2022) 2 has introduced a new category of molecularly defined renal tumors, which include fumarate hydratase (FH)-deficient RCC and succinate dehydrogenase (SDH-B)-deficient RCC.
FH-deficient RCC arises in approximately 15%-30% of patients with hereditary leiomyomatosis and renal cell carcinoma syndrome (HLRCC), which is associated with early age of onset and aggressive disease. 3 Autosomal dominant germline mutation in any of the SDH subunit predisposes to “SDH-deficient” tumors, which include RCC, pheochromocytoma, paraganglioma, gastrointestinal stromal tumor (GIST), and pituitary adenoma. Counseling and germline testing of family members in such cases should be advised.4, 5
Ours is the first study from India documenting screening of renal neoplasms for FH and SDH deficiency, using immunohistochemistry (IHC).
Materials and Methods
Study Design
An observational study was undertaken at a premier oncology tertiary care and research center, after due approval from the Institutional Review Board (IRB) {Res/SCM/43/2020/123}. Tumor tissue blocks of cases diagnosed as renal cell neoplasms over a period of three years (2017 to 2019) were retrieved, and IHC for SDH-B, FH, and 2-succino-cysteine (2-SC) performed. A total of 297 cases were analyzed, which included cases diagnosed as clear cell RCC, papillary RCC (types 1 and 2), chromophobe RCC, unclassified RCC, and oncocytoma.
Methodology
Archival tumor tissue paraffin blocks of these 297 cases were retrieved, and tissue microarrays (TMA) of 3 mm diameter were prepared, using Qick-Ray Manual Tissue Microarrayer (IHC World, Life Sciences Products & Services). IHC for FH (Monoclonal, Abbkine, 1:300), 2-succino-cysteine (2-SC, Polyclonal, Discovery antibodies, 1:2000), and succinate dehydrogenase (SDH-B, EP288, Master Diagnostics, RTU) was performed.
Immunohistochemical procedure using mouse monoclonal antibodies was performed on Ventana Benchmark XT automated immunostainer (Ventana Medical Systems, Inc. [F.Hoffmann-La Roche Ltd.]). The chromogenic signal was generated using the Ventana UltraView universal 3,3’-diaminobenzidine (DAB) detection kit (Ventana Medical Systems, Inc. [F.Hoffmann-La Roche Ltd.]). All IHC stains were applied with appropriate positive controls.
Interpretation
Interpretation of IHC was done on glass slides by light microscopy and on whole slide imaging (WSI) scanned by digital slide scanner (Hamamatsu S210), independently by two pathologists—GG and MK.
Complete loss of nuclear expression of FH is considered as deficiency of FH enzyme. Positive internal control in nuclei of adjoining endothelial cells, inflammatory cells, stromal cells, and adjacent nontumoral renal parenchyma was ensured for accurate expression. If any case showed negative internal control, the IHC procedure was repeated.
Strong and diffuse nuclear and cytoplasmic expression of 2-SC in the tumor cells is also a feature of FH enzyme deficiency. 2-SC-negative staining in the adjacent non-neoplastic cells (internal negative control) was ensured. Cases showing diffuse strong 2-SC expression with or without loss of FH were diagnosed as FH-deficient RCC. Presence of only nuclear expression or only cytoplasmic expression and/or cytoplasmic granularity was considered negative.
Complete loss of SDH-B expression in cytoplasm is considered as SDH-B enzyme deficiency. Positive internal control in cytoplasm of adjoining endothelial cells and adjacent unremarkable renal parenchyma is essential for accurate expression. Many cases show scant weak granular expression, which is considered positive. Hence, careful examination of area under higher magnification is always recommended. If any case showed negative internal control, then IHC procedure is repeated.
Results
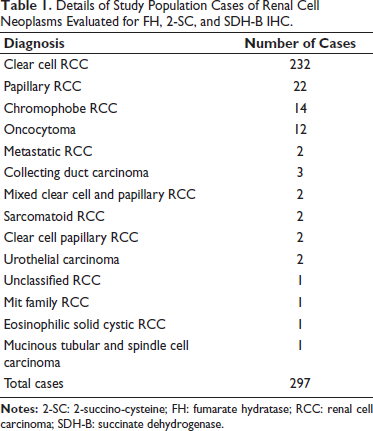
A total of 297 cases of renal cell tumors were analyzed for IHC expression of FH, 2-SC, and SDH-B. A detail of histopathologic diagnosis of all cases has been tabulated in Table 1.
Details of Study Population Cases of Renal Cell Neoplasms Evaluated for FH, 2-SC, and SDH-B IHC.
Two cases (0.7%) of FH-deficient RCC were identified, of which one case showed loss of FH with diffuse strong nuclear-cytoplasmic 2-SC expression, and one case showed positivity for both FH and 2-SC (Figure 1). Our results confirm that 2-SC is a more sensitive marker to diagnose this entity. Demographic details of patients finally diagnosed with FH-deficient RCC have been enumerated in Table 2.
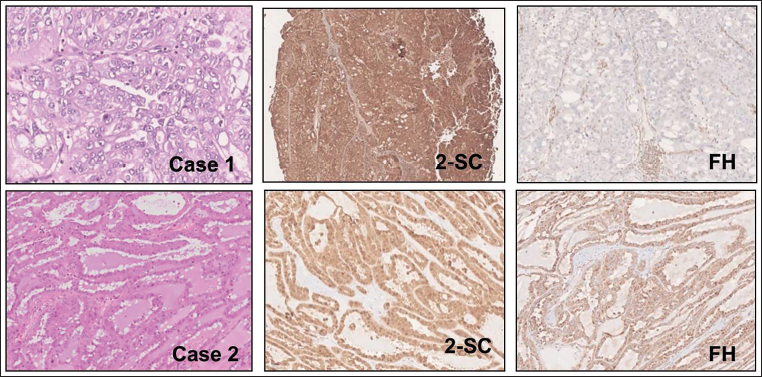
° Case 1: Section Showing Sheets of Large Polygonal Cells, with Diffuse Strong Nucleocytoplasmic Expression of 2-SC and Negative for FH (Internal Positive Control Seen in Blood Vessels).
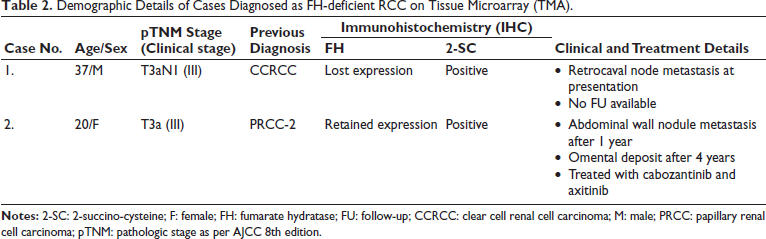
Demographic Details of Cases Diagnosed as FH-deficient RCC on Tissue Microarray (TMA).
One case (case 1) showed sheets of large polygonal cells, with vacuolated eosinophilic cytoplasm, and occasional papillary tufts, initially reported as CCRCC. The other case (case 2) showed tubulo-cystic pattern with large cuboidal cells having eosinophilic cytoplasm, exhibiting oncocytic appearance, initially reported as PRCC type 2. It also showed intraluminal eosinophilic and pale blue secretions, with scalloping towards lumen, similar to that seen in colloid filled thyroid follicles. The common characteristic seen in both cases was vesicular nuclei, prominent eosinophilic nucleoli and perinucleolar halo, with grade 3 nuclei as per World Health Organization/International Society of Urological Pathology (WHO/ISUP) grading system.
The second case received cabozantinib systemic therapy and later axitinib. This patient had recurrence as abdominal wall nodule and omental deposit after one year and four years of surgery, respectively. No follow-up details could be obtained for the first case.
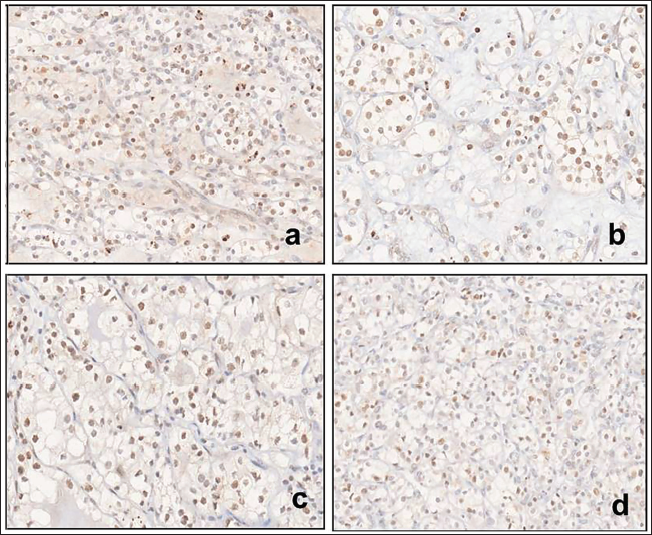
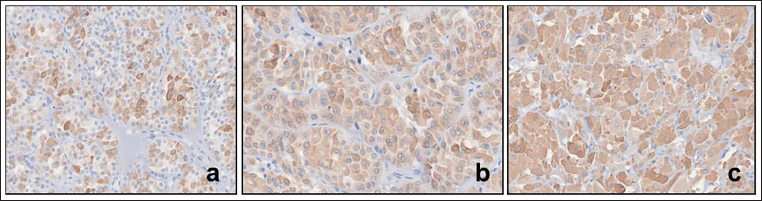
Other patterns of 2-SC expression (cytoplasmic only or nuclear only) were also observed, which were considered negative (Table 3). Diffuse cytoplasmic, only granular cytoplasmic stain (moderate to strong) and dot like expression was seen in 20.8% of CCRCC (48/231), and 41.7% of oncocytomas (5/12), (Figures 2 and 3). Nuclear expression was seen in 6% of CCRCC (14/231) (Figure 4). We did not find any case with SDH-B deficiency.
Negative Expression of 2-SC as Cytoplasmic, Granular, and Nuclear Expression.

Negative Expression of 2-SC in CCRCC Seen as Cytoplasmic Expression (a-c) and Cytoplasmic Granular Expression (d-f).
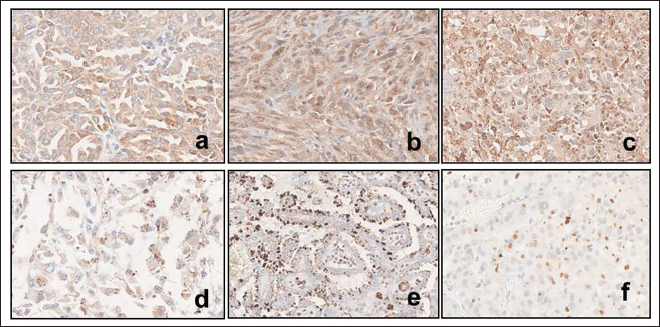
Negative Expression of 2-SC in Clear Cell Renal Cell Carcinoma (CCRCC) Seen as Nuclear Expression (a,b,c,d) in Different Cases.
Negative Expression of 2-SC in Oncocytomas (a,b) and Chromophobe RCC (c) Seen as Diffuse Cytoplasmic Expression.
Discussion
Majority of RCCs are sporadic, and 2%-5% are associated with inherited tumor syndromes. Patients with inherited syndromes develop tumor at an earlier age which can be multifocal, bilateral, and heterogeneous. 1 Determination of any genetic tumor syndrome requires clinicopathological and molecular analysis. Recently few immunohistochemical markers have been recognized to diagnose syndromic disease. 2
HLRCC is an autosomal dominant cancer susceptibility syndrome characterized by the development of multiple cutaneous and uterine leiomyomas, renal cancer, and, rarely, pheochromocytoma, due to inactivating pathogenic germline mutations in FH gene, located at 1q42.3-q43.4, 6, 7 Renal carcinomas associated with HLRCC syndrome are now classified as a distinct entity termed FH-deficient RCC. 2
FH-deficient RCC arises in approximately 15%-35% of patients with HLRCC (germline FH mutation), with mean age of 40 years, but metastatic renal cancer can present in teenage.4, 7 However, they can also arise sporadically secondary to loss of function in both FH alleles. 5 The Cancer Genome Atlas Research (TCGA) in study of papillary RCC identified FH-deficient cases clustered with the CpG island methylator phenotype subgroup of tumors, characterized by multiple metabolic alterations and an aggressive clinical course.3, 8 Five of these cases were found to harbor germline or somatic FH mutation. 9
The FH gene codes for enzyme FH (that converts fumarate to L-maleate) in Krebs cycle; however, when mutated with biallelic inactivation in HLRCC, it triggers oncometabolic fumarate accumulation, favoring aerobic glycolysis, decreasing oxidative phosphorylation (Warburg effect), and aberrant succination with accumulation of 2-SC. This upregulates downstream hypoxia inducible factor genes leading to cell proliferation and tumorigenesis. 4
The prevalence of FH-deficient RCC is unknown. It is more common in males (M:F ratio: 1.9:1) with median age of 44 years. A study reclassified 12% of unclassified RCCs in children and young adults as FH-deficient RCCs, with median age of 18 years. 2
FH-deficient RCC are classically characterized as type 2 papillary RCC-like architecture with prominent eosinophilic inclusion-like nucleoli; however, now other morphologies including solid, cribriform/sieve-like, tubular, cystic, low-grade oncocytic, and sarcomatoid are also being recognized. 6 A series of four cases with low-grade oncocytic morphology, resembling SDH-B-deficient RCC, were found to have FH loss with strong diffuse nucleocytoplasmic 2-SC IHC. Of these, two cases were multifocal. Two cases also had high-grade FH-deficient RCC (one synchronous, and one after 4 years), and both showed tumor progression.2, 10
The morphological clues may be subtle or absent in many cases, thus, a low threshold for IHC screening using FH and/or 2-SC should be done, especially in cases with difficult classification. 2 It is now known that papillary RCC of unspecified type, collecting duct carcinoma, tubulocystic carcinoma, and unclassified RCC can also show evidence of FH deficiency. 11
From an IHC perspective, as a consequence of FH gene mutation and loss of function, staining for FH protein expression is often lost (FH negative), and succination with 2-SC accumulation seen as S-(2-succino)-cysteine (2-SC) positive. FH can be retained in a subset of cases in which a nonfunctional protein is expressed, and has been associated with missense FH mutation.11, 12
IHC for FH has low sensitivity of ~80%-90%, and positive diffuse nuclear and cytoplasmic staining for 2-SC is a highly sensitive marker of FH deficiency.2, 5, 7 Observation of germline FH mutation remains true gold standard for HLRCC diagnosis.
In our study, we observed cases with cytoplasmic granular expression in 20.8% (48/231) CCRCC and 41.7% (5/12) oncocytomas. These should not be mistaken for positive expression.
Loss of FH is associated with an epithelial-to-mesenchymal transition, a phenotypic switch associated with cancer initiation which may explain why HLRCC-associated RCC are high-grade tumors, and why they often and rapidly metastasize. 11
FH-deficient RCC is a high-grade, aggressive carcinoma arising in HLRCC syndrome, which occurs more frequently at a younger age, with an advanced stage at presentation, and a dismal prognosis. 4 High-grade FH-deficient RCC has been associated with progression and/or death from disease in 30%-80% cases, 10 whereas tumors with low-grade oncocytic type have much better prognosis with no recurrence or metastasis.
The recognition of FH-deficient RCCs is important, not only because of their aggressive behavior but also because of the hereditary association, which should prompt genetic counseling and FH mutational analysis in these patients and their families.4, 11 Cases found to have FH mutation should undergo annual abdominal magnetic resonance imaging (MRI) to detect renal tumor, and if found should undergo prompt surgical excision with wide margins and retroperitoneal lymph-node dissection (RPLND).2, 11 Our study was limited by lack of available germline testing.
Trpkov et al. 6 identified 19% FH-deficient RCC of total 124 tumors, in cases previously diagnosed as “unclassified RCC with papillary pattern,” and 2 (0.5%) tumors as “papillary RCC type 2.” FH mutations were found in 19/21 of these FH-deficient tumors (with confirmed germline mutations in all 9 tumors assessed) and in 1/26 FH-indeterminate tumors identified by IHC.
In a recent screening study by Gupta et al. 13 in consecutively diagnosed 1,009 renal cell neoplasms on TMAs, FH-deficient RCCs were identified in the papillary (0.5%) and unclassified RCC cohorts (4.35%).
SDH-deficient RCCs occur in the setting of germline mutations of SDH-B. The SDH complex, comprising SDH-A, SDH-B, SDH-C, and SDH-D subunits, is located in inner mitochondrial membrane, and the enzyme SDH participates in Krebs cycle and transport chain. Inactivation of this enzyme leads to aerobic glycolysis (Warburg effect) which has carcinogenic effect, with predisposition to “SDH-deficient” tumors, which include RCC, pheochromocytoma, paraganglioma, GIST, and pituitary adenoma.4, 5 Thus, counseling of family members for germline testing for SDH subunit mutation should be advised in such patients.1, 4, 5
SDH-deficient RCC forms 0.05%-0.2% of all RCCs, with characteristic granular eosinophilic cytoplasmic flocculations, and low nuclear grade (WHO/ISUP grade 2). Differentials include oncocytoma, chromophobe RCC and CCRCC, which can be resolved on IHC.4, 5, 14 They carry a favorable prognosis, and can be diagnosed consistently by SDH-B protein loss on IHC.1, 4, 5, 10 Thus, IHC can be used as a screening tool for this hereditary disease (true positive IHC shows strong cytoplasmic granular positivity, and positive internal control must be ensured).4, 5 However, we did not find any case. In a screening study conducted to evaluate incidence of SDH deficient RCC using IHC, 1.1% of originally diagnosed oncocytomas showed loss of SDH-B. 13 The confirmatory gold standard molecular test for germline SDH mutation can be done. 1
Conclusion
As most of syndromic and familial tumors show varying and overlapping histology, the pathologist needs to keep a low threshold and work up all cases adequately with immunohistochemical or molecular work up, and appropriate genetic counseling.
Footnotes
Authors’ Contributions
Meenakshi Kamboj, Gurudutt Gupta, Amitabh Singh, Sunil Pasricha, and Garima Durga contributed to concept and design of study or acquisition of data or analysis and interpretation of data.
Meenakshi Kamboj, Gurudutt Gupta, Anurag Mehta, Sudhir Rawal, Amitabh Singh, and Anila Sharma helped in drafting of the article or revising it critically for important intellectual content.
Meenakshi Kamboj, Gurudutt Gupta, Anurag Mehta, Sudhir Rawal, and Amitabh Singh provided final approval of the version to be published.
Meenakshi Kamboj and Gurudutt Gupta were in agreement to be accountable for all aspects of the work are appropriately investigated and resolved.
The manuscript has been read and approved by all the authors, and the requirements for authorship have been met. Each author believes that the manuscript represents honest work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the Institutional Review Board (IRB), Res/SCM/43/2020/123.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
