Abstract
Introduction
A 24-year-old male undergoing treatment for carcinoma at the base of the tongue developed invasive mucormycosis (IM), a rare and severe fungal infection. While common in haematological malignancies, this condition is rarely reported in solid cancers.
Primary Diagnoses, Interventions and Outcomes
Diagnosis was established through radiological investigations, potassium hydroxide staining and histopathological examination of the infected tissue. He was treated with liposomal amphotericin B, surgical debridement and supportive care measures.
Conclusion
This case highlights the critical need for early recognition and prompt management of IM in patients with solid malignancies undergoing intensive cancer therapy, where the index of suspicion is low. Enhanced clinical vigilance and a multidisciplinary approach are essential for improving outcomes.
Introduction
Invasive mucormycosis (IM) represents an aggressive, rapidly progressing and potentially lethal opportunistic fungal infection caused by saprophytic fungi belonging to the order Mucorales. This disease was documented by Fürbinger in Germany, in a cancer patient in the year 1876. 1 Characterised by angioinvasion, IM induces tissue infarction and necrosis due to the infiltration of hyphae into vasculature. 2 Although commonly encountered in immunocompromised individuals with uncontrolled diabetes mellitus, prolonged neutropenia, extended use of steroids, chemotherapy, haematological malignancies and organ or stem cell transplants, the association of IM with solid organ malignancies remains an underexplored aspect. In the realm of haematological malignancies, invasive fungal infections like zygomycosis and mucormycosis pose significant threats, given the presence of multiple high-risk features. The immunocompromised state in malignancies, induced by factors such as altered immune systems, cytotoxic drugs, broad-spectrum antibiotics and other immunomodulatory therapies, heightens susceptibility to these infections. 3 Patients with haematological malignancies are particularly prone to granulocytopenia and febrile neutropenia, increasing the likelihood of fungal infections, as neutrophils play a pivotal role in defending against fungal elements.4, 5 Despite the well-established association between solid organ malignancies and fungal infections, there is a dearth of data illustrating a clear incidence of mucormycosis in these patients. Consequently, we present what appears to be the inaugural case of IM in a patient with a solid malignancy affecting the head and neck. This case emphasises the necessity of further exploration into the occurrence and clinical implications of mucormycosis in individuals with solid organ malignancies, shedding light on a potential area of concern in this patient population.
Case Report
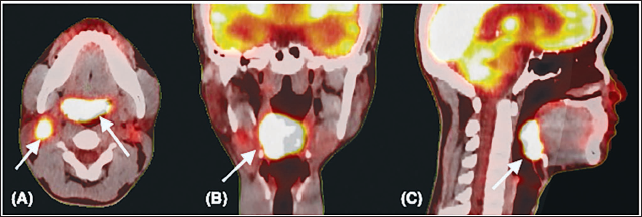
A 24-year-old medical student of North-East Indian ethnicity, with no known addictions, presented to the head and neck cancer (HNC) department with complaints of painful swelling on the left side of the neck and difficulty in swallowing for the last two months. On examination, an exophytic growth was identified on the right base of the tongue along with a fixed, tender right level II node. Fine-needle aspiration cytology (FNAC) and cell block from node indicated p16 negative moderately differentiated squamous cell carcinoma (SCC). F-18 fluorodeoxyglucose positron emission tomography-computed tomography (PET-CT) scan was done for further investigation (Figure 1). The disease was staged as cT3N2cM0 following the American Joint Committee on Cancer (AJCC) 8th edition criteria. The patient was scheduled for definitive concurrent chemoradiation to the head and neck, employing image-guided radiation therapy (IGRT) with a simultaneous integrated boost (SIB). The planned dose gradient was 70 Gray (Gy), 63 Gy and 56 Gy to high, intermediate and low-risk primary target volumes, respectively. Concurrently, intravenous three-weekly cisplatin (100 mg/m 2 ) was administered.
(A-C) 8 Fluorodeoxyglucose Positron Emission Tomography Computed Tomography (PET-CT) Images in Axial, Coronal and Sagittal Sections, Respectively. The Baseline Malignant Metabolically Active Soft Tissue Lesion in Base of Tongue, Right Tonsillar Fossa, Glossotonsillar Sulcus, Bilateral Vallecula, Median Glossoepiglottic Fold and Lingual Surface of Epiglottis* Measuring 4.2 × 2.0 cm, Maximum Standardised Uptake Value (SUVmax) 17.7. Axial Section (A) Shows Metabolically Active Right Level II Lymph Node Measuring 2.1 × 1.6 cm, SUVmax 9.7 and a Smaller Left Lymph Node Measuring 1.2 × 1.0 cm.
After 11 fractions of radiation and one cycle of concurrent cisplatin, the patient presented to the emergency room with severe, persistent throat pain, dysphagia, poor oral intake and low-grade fever. Evaluation revealed febrile neutropenia, leading to the deferral of chemoradiation. He was managed conservatively with intravenous fluids, antacids, broad-spectrum antibiotics, analgesics, antipyretics, Ryle’s tube insertion and strict aseptic precautions. Additionally, he experienced heightened chemoradiation-related toxicities, including Common Terminology Criteria of Adverse Events (CTCAE) v4.1 grade 2 oral mucositis, grade 3 oral pain, grade 3 dysphagia and grade 1 dermatitis. A multidisciplinary team, including a haematoncologist, infectious disease specialist, general physician and radiation and medical oncologists, managed the patient.
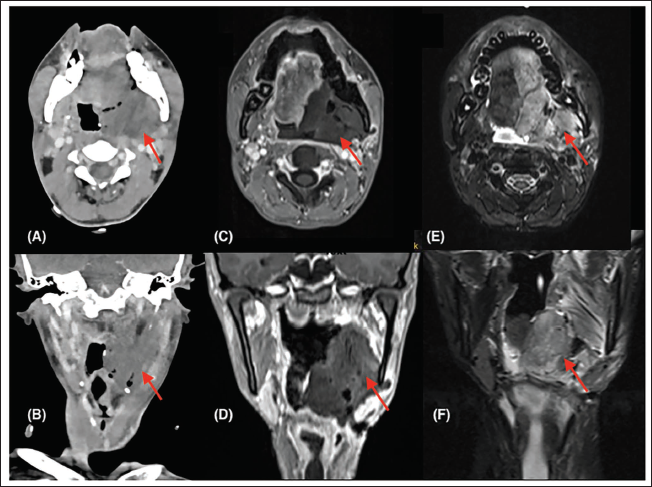
On the ninth day of admission, the patient developed minor oral bleeding followed by mild nasal bleeding. Examination of the oral cavity revealed clots with black necrotic tissue, which upon potassium hydroxide (KOH) staining, exhibited scanty broad, aseptate, right-angled branching filamentous fungi resembling Mucor. Isolation on Sabouraud Dextrose Agar (SDA) resulted in rapidly growing white-raised cottony filaments (Figure 2A). Contrast-enhanced CT of the head and neck revealed a large hypodense collection extending from the left infratemporal fossa to the floor of the mouth, involving the left parapharyngeal space (Figure 3). Biopsy from the invasive front of the lesion confirmed IM.
(A) and (B) Contrast-enhanced CT Images; (C) and (D) MRI T1 Contrast Sequences; and (E) and (F) Shows MRI T2 Fat-suppressed Images in Axial and Coronal Sections, Respectively. A T1 Isointense/T2 Hyperintense Nonenhancing Lesion Measuring 6.9 × 4.9 × 6.8 cm Along Left Base of Tongue, Oral Tongue, Oro-nasopharynx Extending Till Ipsilateral Carotid Space in the Posterior Triangle, Infratemporal Fossa to Floor of Mouth, Parapharyngeal Space, Pterygoids and Soft Palate.
(A) White Cottony Colonies of Mucorales After KOH Staining of Oral Swab Specimen. (B) Per-operative Surgical Field Highlighting the Necrotic Slough in the Floor of Mouth Involving Parapharyngeal Space. (C) Histomorphology Depicting Infiltration by Multiple Broad Aseptate Fungal Hyphae with Right Angle Branching Consistent with Mucor Species (haematoxylin and eosin (H&E); 200×). (D) Microscopic Section with Vascular Invasion Within Lumen Filled with Fungal Hyphae (H&E; 400×).
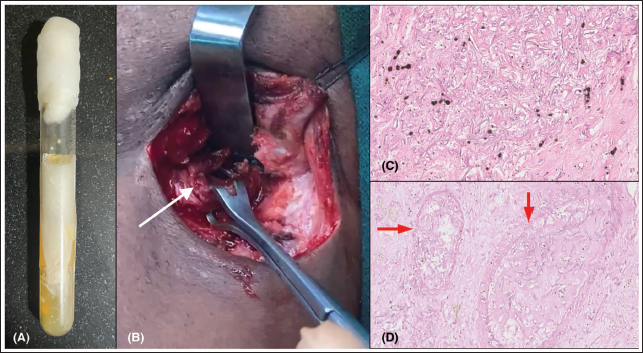
Subsequently, the patient underwent extensive debridement along with a temporary tracheostomy, revealing blackish discoloration in the region of the premaxilla, hard palate and soft palate, with extension along the glossotonsillar sulcus to the left base of the tongue, left buccal mucosa and retromolar trigone. Notably, per-operative examination revealed no exophytic growth or ulceration along the base of the tongue (Figure 2B). Histopathological examination confirmed the diagnosis of IM (Figure 2C). The patient was initiated on intravenous liposomal amphotericin B (LAmB) to a dose of 5 mg/kg per day and antibiotics. Due to continued bone marrow suppression and thrombocytopenia, the patient was deemed unfit for further radical excision. Conservative medical management for mucormycosis was continued until further optimisation for radical surgery. After two weeks of medical management and stabilisation, contrast-enhanced magnetic resonance imaging revealed the left base of tongue lesion, primarily suggestive of mucormycosis, with no radiological evidence of malignancy (Figure 2). Patient had another episode of oronasal bleed during hospitalisation. He underwent CT angiography which showed a leak in the left external carotid artery. He was advised angioembolisation, but the family did not consent for the same. Despite being advised to undergo radical surgery, the patient chose not to provide consent and instead opted for exclusive medical management. Expressing a preference for home-based best supportive care, the case was then transferred to his hometown to honour the wishes of the patient and his family. Three months later, it was discovered that the patient had succumbed to his prolonged illness, specifically due to hepatorenal failure.
Discussion
IM, commonly known as zygomycosis or ‘black fungus’, represents a life-threatening, ubiquitous opportunistic infection caused by fungi of the order Mucorales. Clinical significance is primarily observed in immunosuppressed individuals, given its extrinsic nature to humans. Uncontrolled diabetes mellitus stands out as the most common cause of IM in developing countries, accounting for approximately 50% of cases. 6 In contrast, in the developed world, haematological malignancies and solid organ transplants take precedence as the leading causes, ranging from 38% to 62%.7, 8 Acute leukaemias and lymphomas are the most frequently associated malignancies, and other well-known risk factors include haematopoietic stem cell transplants, prolonged neutropenia, corticosteroid use, voriconazole prophylaxis, burns, neonatal prematurity, malnutrition, iron overload, illicit intravenous drug usage, natural disasters and recent coronavirus disease 2019 (COVID-19) infection.4, 5, 9
Mucormycosis manifests in various forms based on anatomic distribution and clinical presentation, including pulmonary, rhino-orbital-cerebral mucormycosis (ROCM), gastrointestinal, cutaneous, central nervous system or disseminated. 5 Pulmonary mucormycosis is predominant in patients with haematological malignancies and stem cell transplants, while ROCM is more common in individuals with diabetes mellitus, followed by haematological malignancies. 4 The inhalation of sporangiospores leads to infection, with subsequent hyphal development causing angioinvasion and thrombus formation, resulting in tissue necrosis. Common symptoms include fever, headache, facial pain, nasal discharge, epistaxis and nasal crusting. Necrosis progresses, leading to the formation of a hallmark black eschar visible on the palate, nasal mucosa or orbital skin. 10
Despite the well-established association of haematological malignancies and solid organ transplants with IM, literature regarding its link with solid organ malignancies is limited, constituting approximately only 0.01% of IM cases. 6 The compromised immune system in solid malignancies, especially squamous cell cancer of the head and neck, contributes to carcinogenesis. Cancer treatment modalities, particularly cytotoxic chemotherapy and new immunomodulating agents, heighten susceptibility to opportunistic infections due to neutropenia and cell kill.1, 11 Neutropenia, an independent risk factor, exposes patients to polymicrobial bacterial and fungal infections, impacting various bodily systems. 4 Prolonged use of antibiotics and antifungals in treating these infections further compromises the immune response.
Early diagnosis of ROCM is crucial for optimal outcomes, translating to less extensive treatment, reduced physical disfigurement and improved survival. 12 However, diagnosing ROCM poses challenges, necessitating standard mycological testing. The condition can be suspected in high-risk individuals exhibiting sinusitis and infarcted tissue in the nose or palate. Immunohistopathological evaluation, demonstrating typical fungal hyphae amid necrotic debris, with culture confirmation, provides a definitive diagnosis. 4 Molecular biology tools, including polymerase chain reaction (PCR), enable noninvasive diagnosis by confirming the presence of fungal elements in histologic specimens or blood samples. Serological testing using mucor immunoglobulin E (IgE) antibodies is employed in rare cases.
Gold-standard treatment for ROCM involves aggressive surgical debridement initiated promptly after diagnosis. Concurrent systemic antifungal therapy and addressing predisposing factors are integral to effective management. Early commencement of antifungals, particularly amphotericin B, enhances outcomes. 7 LAmB and cochleate-containing amphotericin B (CAmB) are preferred formulations. LAmb is usually started at a dose of 5 mg/kg daily and is administered for three to six weeks. Posaconazole and isavuconazole serve as newer adjuncts or alternatives, with intravenous administration considered for patients experiencing toxicity or inadequate response to amphotericin. Oral forms are primarily reserved for step-down approaches in responsive patients.
Conclusion
ROCM presents a substantial overall mortality rate ranging from 25%-62%, a figure that escalates significantly due to delayed diagnosis, delayed medical intervention and the inherent challenges associated with managing advanced cases. The existing body of evidence indicating the occurrence of mucormycosis post-chemotherapy predominantly stems from observations in haematological malignancies. Regrettably, the incidence of mucormycosis in the context of solid malignancies, such as HNCs, lacks clear evidence. 8
The imperative for further investigation and research into the pathophysiology and preventive strategies surrounding this fatal condition within the realm of solid malignancies, particularly SCC, is evident. Such endeavours are crucial for developing effective management strategies tailored to the unique challenges presented by patients with solid malignancies. As we advance our understanding of mucormycosis in this specific patient population, we can enhance diagnostic capabilities, expedite therapeutic interventions and ultimately improve patient outcomes. The knowledge gained from focused research in this area will contribute to a more comprehensive approach to the prevention, early detection and treatment of mucormycosis, offering a valuable resource for clinicians and researchers alike.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article.
