Abstract
Stereotactic body radiotherapy (SBRT) is an important option for prostate cancer treatment. It involves the delivery of a high dose of radiation to the malignant tissue without affecting the neighboring healthy tissue to achieve a high therapeutic index. As the cells of prostate cancer are radiobiologically similar to a late-responding tissue with a low 𝛼/𝛽 ratio, the dose escalation approach has a better disease control probability than conventional dose fractionation. Therefore, considering the availability of intensity-modulated RT and possibility of dose escalation (>70 Gy), ultrahypofractionated SBRT is now being recommended in select patients with prostate cancers of low and intermediate risk. Although adverse events following SBRT are a concern, acute toxicity can be limited to a certain extent by adjusting the overall length of treatment time by spacing out RT over multiple fractions. Modern radiation planning and delivery methods used in conjunction with cutting-edge imaging techniques have led to the increased adoption of SBRT in prostate cancer. Thus, from a practical standpoint, it is noninvasive and effective for treating prostate cancer, leading to increased access and higher patient satisfaction.
Keywords
Introduction
Prostate cancer, the risk of which increases with increasing age, is common in men worldwide. 1 As per Globocan (2018), its incidence and mortality in India were 4.4% and 2.9%, respectively 1 ; the incidence rose to 5.5% while mortality reduced to 2.7% in 2020. 2 The cancer stage and aggressiveness as well as patient preference and their life expectancy decide the treatment choice. In prostate cancers of low and favorable intermediate risk, external beam radiotherapy (EBRT) is an integral therapeutic option either alone or in combination with other therapies. 3 Research has revealed that the overall survival in prostate cancer can be enhanced by combining RT with androgen-deprivation therapy (ADT). 4 In contrast to conventional RT, it has been shown that biochemical control after EBRT can be enhanced with dose escalation via stereotactic body radiotherapy (SBRT). 5 SBRT refers to a stereotactic RT procedure for treating tumors with ultrahigh doses in a single fraction in a total of 5 or fewer hypofractionated fractions. 6 It has been shown that SBRT can achieve dosimetric parameters similar to high-dose-rate brachytherapy. Hence, it can deliver high radiation doses with a sharp dose falloff and has been increasingly used in prostate cancer. 7 Indeed, the outcome of SBRT in prostate cancer of low and intermediate risk is well established.8–10 The present review summarizes the therapeutic role of SBRT in prostate cancer.
Radiobiology of the Prostate Tissue
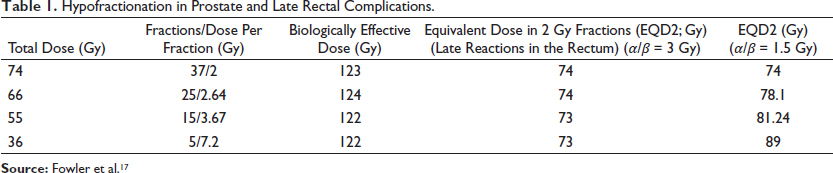
Compared to other tumors that proliferate faster, the intrinsic radiosensitivity to repair capacity ratio (𝛼/𝛽 ratio) in prostate cancer is lower. Thus, prostate cancer appears radiobiologically similar to healthy tissue. The 𝛼/𝛽 ratio for tumors and early responding tissues (skin and mucosa) is estimated to be approximately 8 to 10 Gy, whereas that for late responding tissues (bladder or rectal mucosa, muscle, and connective tissue) is approximately 3 to 5 Gy. 11 The considerably low prostate cancer 𝛼/𝛽 ratio of 1.5 Gy (95% confidence interval [CI], 0.8-2.2 Gy) was derived by Brenner and Hall 12 . Fowler et al. 13 obtained a ratio of 1.5 Gy (1.25-1.75 Gy), which was confirmed by Miralbell et al. 14 This phenomenon of prostate cancer behaving similar to healthy tissue has been studied using DNA labeling, which revealed hypoxic cells. The hypoxia was attributed to poor tumoral blood supply, thereby slowing the proliferation and reducing the 𝛼/𝛽 ratio of the tumor. 15 The low 𝛼/𝛽 ratio of prostate cancer suggests that hypofractionation is a better therapeutic option, but late rectal complications need to be accounted for implementing hypofractionation. Of note, the 𝛼/𝛽 ratio of late rectal complications has been found to be approximately 3 Gy. 16 Table 1 presents the late complications in the rectum after RT and the required doses for prostate cancer treatment. The table shows that with a decrease in the total RT dose with increasingly large but few fractions, the effect of radiation in the tumor increases considerably with a similar late complication rate in the rectum, suggesting that hypofractionation is beneficial in prostate cancer treatment.
Hypofractionation in Prostate and Late Rectal Complications.
Fractionation in Prostate Cancer RT
According to the American Society for Radiation Oncology (ASTRO), the fraction size is 1.8 to 2 Gy for conventional fractionation, 2.4 to 3.4 Gy for moderate hypofractionation, and ≥5 Gy for ultrahypofractionation. 3 Since the 1980s, RT delivery has evolved, with treatment regimens moving from standard or conventional fractionation to ultrahypofractionation. During the 1980s, 3-dimensional conformal radiotherapy (3DCRT) or conformal treatment techniques became available. Conventional fractionation was used in these techniques, and the prescription doses ranged from 60 to 78 Gy.
The most common method in the early- and mid-1990s for delivering radiation was a 4-field 3DCRT technique that produced 4 beams in the right and left lateral directions as well as the anteroposterior direction. However, >78-Gy doses and hypofractionated regimens require a higher dose conformity than that possible with 3DCRT. 18 Intensity-modulated radiotherapy (IMRT), a form of EBRT introduced in the early 1990s, uses a multileaf collimator to improve highly conformal radiation delivery. Owing to the lack of randomized controlled trials, studies conducted retrospectively compared the outcomes and toxicities between IMRT and 3DCRT and reported similar outcomes with a superior toxicity profile in the case of IMRT.19, 20 At present, IMRT is recommended for dose escalation (>70 Gy). 21
Many studies have evaluated dose escalation using EBRT. Kuban et al. 16 studied patients with prostate cancer who were treated with EBRT between 1986 and 1995. The results showed that in patients with the pretreatment prostate-specific antigen (PSA) values of 0 to 4, 4 to 9.9, 10 to 20, and >20 to 30 ng/mL, the 8-year PSA control rates were 80%, 60%, 46%, and 34%, respectively. A Phase III trial 22 from MD Anderson Hospital included patients with T1-3 prostate cancer (n = 301) who were classified into 2 arms: 1 arm received 70 Gy (conventional EBRT) and the other arm received 78 Gy (3D boost after conventional EBRT). The results revealed 70% and 64% PSA relapse-free survival (RFS) rates, respectively, for the 78 and 70 Gy arms. A subsequent update to the study revealed an improvement in the 8-year biochemical control at 88% over 63%. Zelefsky et al. 23 studied 2,551 prostate cancer (T1-3) cases who received radiation doses increased systematically via 5.4-Gy increments from 64.8 to 86.4 Gy in consecutive patient groups. The results showed that the 8-year PSA RFS rates were 85% for patients with favorable risk, 76% for those with intermediate risk, and 72% for those with unfavorable risk. Similar results were obtained in the Radiation Therapy Oncology Group (RTOG) 94-06 trial. 24 In this study, patients were treated with 5 sequential doses of 68.4, 73.8, 79.2, 74, and 78 Gy. The study results suggested an advantage of dose escalation in patients with prostate cancer of low risk.
The CHHiP trial 25 classified 3,216 cases of mostly intermediate risk into 3 arms. The first arm was administered 7,400 cGy in the form of 200 cGy in 37 fractions across 7.4 weeks. The second arm was administered 6,000 cGy in the form of 300 cGy in 20 fractions across 4 weeks. The third arm was administered 5,700 cGy in the form of 300 cGy in 19 fractions across 3.8 weeks. The patients were followed-up for a median period of 5.2 years. The results revealed noninferior biochemical failure for the 6,000 cGy arm compared with the 7,400 cGy arm. Of note, the 5,700 cGy arm was not noninferior to the 7,400 cGy arm. The PROFIT 26 trial included 1,206 men with intermediate-risk prostate cancer who were randomized to 2 treatment arms: 6,000 cGy in the form of 300 cGy in 20 fractions over 4 weeks or 7,800 cGy in the form of 200 cGy in 39 fractions over 7.8 weeks. The participants were followed-up for a median period of 6 years. The results revealed similar biochemical–clinical failure rate for both arms. The RTOG 0415 8 study allocated 1,115 cases with prostate cancer of low risk into 2 arms: 7,000 cGy in the form of 250 cGy in 28 fractions across 5.6 weeks or 7,380 cGy in the form of 180 cGy in 41 fractions across 8.2 weeks. These patients were followed-up for a median period of 5.8 years. Similar to the PROFIT trial, this study also showed similar disease-free survival (DFS) in both the arms. The Dutch HYPRO study 27 assigned 820 men with prostate cancer, with a majority being high risk, to 2 arms: 6,460 cGy in the form of 340 cGy in 19 fractions for 3 days/week across 6.3 weeks or 7,800 cGy in the form of 200 cGy in 39 fractions for 5 days/week across 7.8 weeks. No significant difference was observed in the RFS rates between the 2 treatment arms over a follow-up period of 5 years. Taken together, these 4 large prospective randomized clinical trials (RCTs) demonstrated similar rates of early prostate cancer control using hypofractionated and conventional EBRT.
Most of these studies evaluated the biochemical measures of cancer control; however, these measures cannot be considered as standard for disease-specific survival and overall survival, which are long-term outcomes. Thus, it is important to conduct extended follow-up studies to evaluate the impact of hypofractionation regimens on long-term cancer outcome. 6
Research has shown that increasing the dose per fraction significantly improves prostate cancer outcomes, including local and distant failure as well as freedom from biochemical failure. 5 Moreover, conventional fractionated EBRT for 8 weeks could be logistically challenging for patients and hospitals. From a practical standpoint, SBRT is noninvasive, quicker, and cost effective compared with conventional fractionated RT, brachytherapy, and surgery. 28 Departmental efficiency has been reported to increase because of the increased cost effectiveness. 29 All these factors lead to increased access and higher patient satisfaction. Thus, it may be reasonable to assume that ultrahypofractionation would be beneficial, considering the myriad reasons of improved logistics, reduced cost, and increased treatment speed.
Current Evidence for SBRT in Prostate Cancer
Observational Studies
Loblaw et al. 30 administered stereotactic ablative RT (35 Gy/5 fractions/once weekly) in 84 patients with prostate cancer of low risk. In the 55-month median follow-up, 96% patients were biopsy negative after treatment and 98% patients achieved 5-year biochemical control. Bolzicco et al. 31 evaluated SBRT (35 Gy/5 fractions) in 100 patients with localized prostate cancer and noted the 3-year biochemical DFS as 94%. Bernetich et al. 32 studied 142 patients with localized prostate cancer who were treated with various dose levels ranging from 35 to 37.5 Gy over 5 fractions. The 5-year actuarial freedom from biochemical failure was 100% for very low-, 91.7% or low-, 95.2% for favorable intermediate-, 90% for unfavorable intermediate-, and 86.7% for high-risk disease. These studies indicate that hypofractionation is effective in prostate cancer.
RCTs
Several RCTs have validated the efficacy of hypofractionation in prostate cancer RT. These have been summarized in Table 2. Hypofractionated SBRT for localized prostate cancer was first evaluated by Madsen et al. in their stereotactic hypofractionated accurate RT for the prostate cancer (SHARP) study. 33 They delivered SBRT (33.5 Gy/5 fractions) using noncoplanar conformal fields in addition to the daily stereotactic localization of implanted fiducials in 40 patients. At the end of 48-month follow-up, they reported 90% freedom from biochemical failure. A multicentric Phase 1 feasibility study in prostate cancer of low risk was conducted by McBride et al. 34 who studied 2 different dose schedules of 7.5 Gy/5 fractions and 7.25 Gy/9 fractions and assessed biochemical progression-free survival (PFS) along with toxicities. Over a 45-month follow-up period, the median percentage PSA declines at 12 months and biochemical PFS were 80% and 97.7%, respectively. The study concluded that in the selected population, prostate cancer of low risk can be treated safely and effectively with ultrahypofractionated RT.
Randomized Clinical Trials on Ultrahypofractionated Stereotactic Body Radiotherapy for Prostate Cancer.
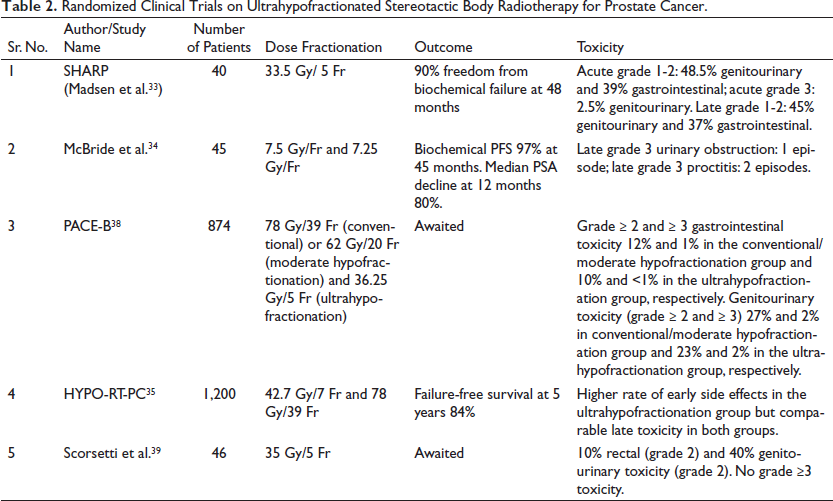
A randomized Phase III noninferiority trial 35 that included 12 centers from Sweden and Denmark (HYPO-RT-PC) compared ultrahypofractionated and conventionally fractionated RT in 1,200 patients. The patients were randomly assigned into 2 arms: the first arm was administered conventional RT (78 Gy/39 fractions, 5 days/week across 8 weeks) and the second arm was administered 42.7 Gy/7 fractions, 3 days/week across 2.5 weeks. Ultrahypofractionation arm had higher early side effects than the conventional fractionation arm, and both groups exhibited similar late toxicity. The PACE-C trial 36 is an ongoing trial that has planned the randomization of 1,182 patients who were either declined surgery or were not candidates for the same into 2 groups: conventional radiotherapy and SBRT (i.e., 60 Gy/20 fractions and 36.25 Gy/5 fractions, respectively). NRG-GU 005 37 is another ongoing clinical trial that will compare 2 doses (36.25 Gy/5 fractions and 70 Gy/28 fractions) in prostate cancer of low risk. The endpoints of this study are DFS and 2-year bladder/rectal toxicity.
Meta-Analyses
A meta-analysis including >6,000 SBRT-treated cases of localized prostate cancer included data from 41 clinical trials and revealed that SBRT was associated with significant biochemical control and low side effects. 40 The study reported 5‑ and 7‑year bRFS rates and toxicities, with a median follow-up of 39 months. The results showed that the combined acute ≥G3 toxicity was <1%. Further, 2.0% and 1.1% late ≥grade 3 toxicities, respectively, were observed in the genitourinary and gastrointestinal systems. On including studies with a median follow-up period of ≥5 years, the toxicity rates remained unchanged. Of note, late genitourinary (≥grade 3) toxicity, but not gastrointestinal (≥grade 3) toxicity, was associated with the dose. Thus, ultrahypofractionation was recommended in localized prostate cancer. However, this conclusion might be premature as high-risk patients were underrepresented in the study.
Kishan et al. 41 assessed 12 Phase II trials containing the data of 2,142 cases with prostate cancer of low and intermediate risk treated with either conventional linear accelerator (5/12 studies) or CyberKnife (7/12 studies). Of the 2,142, 55.3%, 32.3%, and 12.4% had low-risk, favorable intermediate-risk, and unfavorable intermediate-risk disease, respectively; the patients were followed-up for a median of 6.9 years. The study results revealed that the 7-year cumulative rates of biochemical recurrence were 4.5% (95% CI, 3.2%-5.8%), 8.6% (95% CI, 6.2%-11.0%), 14.9% (95% CI, 9.5%-20.2%), and 10.2% (95% CI, 8.0%-12.5%) for low-risk, favorable intermediate-risk, unfavorable intermediate-risk disease, and all intermediate-risk disease, respectively. Acute grade ≥3 genitourinary and gastrointestinal toxic event rate was 0.6% and 0.09%, respectively, and their 7-year cumulative incidence was 2.4% and 0.4%, respectively. King et al. 42 analyzed pooled Phase III data from 8 centers including 1,100 patients with localized prostate cancer who were treated with CyberKnife (36.25 Gy in 4-5 fractions). Of the 1,100 patients, 58% and 30% had low- and intermediate-risk cancer, respectively, and the patients were followed up for a median of 36 months. The results showed that 4.55% had PSA failure, with 5-year biochemical RFS rates of 93%, 95%, 84%, and 81% biochemical RFS rates for all, low-risk, intermediate-risk, and high-risk patients, respectively.
Selection Criteria for SBRT
As per ASTRO guidelines, 3 ultrahypofractionation (SBRT) is recommended in intermediate-risk and low-risk prostate cancer not under active surveillance. However, ultrahypofractionation has been suggested to avoid in cancer of high risk unless within a clinical trial and that it should be offered only in cancer of low and intermediate risk with <100 mL prostates. Owing to the risk of late toxicity, doses >3,625 cGy in 5 fractions to the planning target volume (PTV) are not recommended. In addition, owing to the increased risk of late urinary and rectal toxicity, the administration of the same fractionation doses daily should not be considered. Finally, patients with large prostates, those who have undergone transurethral resection of the prostate, and those with major urinary symptoms at presentation may be poor candidates for SBRT in view of limited data. 3
Contouring
For prostate SBRT, target and organs at risk (OAR) volume segmentation is critical with regard to the dose and OAR proximity. The rectum and bladder being distensible organs, simulation should be representative of the daily setup in terms of consistent bladder filling. In addition, delineation should consider contouring uncertainty, variable filling, and organ motion. Magnetic resonance imaging (MRI)-based RT planning for prostate cancer is becoming increasingly common in clinical practice 43 . Prostate delineation in computed tomography (CT) is difficult and may require co-registration with MRI to better delineate the adjacent soft tissues along with the prostate gland. The development of MR simulators may enable a better delineation of the prostate without the need for CT. 44 The clinical target volume (CTV) incorporates the extracapsular extension areas and the prostate, including or excluding the proximal seminal vesicles. To incorporate PTV, CTV is expanded by 3 to 5 mm. Tighter constraints may be required for sparing the rectum posteriorly. At least 95% PTV is covered when the dose is 90% of the isodose. 29 OAR that need delineation include the bladder neck, neurovascular bundle, penile bulb, rectum, and femoral heads. It is important to carefully consider the interfraction deformation/motion and intrafraction motion of the prostate. Multiple methods are available to position and track the prostate motion; these methods must be finalized before RT simulation. 45 The most commonly used method is to implant gold fiducials evenly throughout the prostate.46, 47 In addition, when technically feasible, an injectable rectal hydrogel spacer, functioning as an anatomic modulator, may be used to help reduce gastrointestinal toxicity.48, 49
Before contouring, it is necessary to prepare the bowel and bladder before simulation, and patients may be administered enema/stool softeners before simulation to keep the rectum as empty as possible. 50 The bladder is moderately filled by asking the patient to first empty their bladder and then consume water (500 mL), and the CT/MR images are acquired 45 min after; patients are advised to follow the same protocol during RT days. Foley’s catheter may be used to visualize and delineate the urethra. Patients, covered with thermoplastic mold extending from the mid-abdomen to mid-thigh, are usually simulated in a headfirst supine position using 1-mm-slice thickness CT/MR images from L1 to mid-thigh.
Studies conducted on ultrahypofractionation vary significantly with respect to the target volumes employed. Of note, for CTV, nearly all prostate cancer RT studies considered the prostate alone in low risk and prostate along with varying seminal vesicle portions in intermediate risk. Finally, compared with PTV in moderate hypofractionation, narrower PTV margins (isotropic 5 mm) have been employed in ultrahypofractionation.
Among all available techniques, volume-modulated arc therapy is a widely adopted and preferred method for delivering SBRT. At the minimum, the following two dose–volume constraint points should be used for moderately or ultrahypofractionated EBRT for the rectum and bladder: the mid-dose range (middle of the total dose) and the high-dose end (near the total dose). Considering toxicity risks, it is not recommended to use normal tissue constraints applied in moderate or ultrahypofractionated EBRT that differ from those used previously. 3 As few randomized controlled trials have compared the parameters of treatment toxicity, toxicity reported by physicians, and quality of life outcome reported by patients, standard normal tissue constraints have not been devised. 3
Dose Constraints
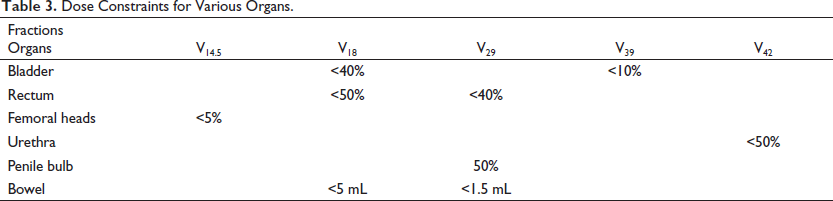
Rectal dose constraints from multiple trials have suggested that rectal volume should be <50% for a 20-Gy dose and should be as minimum as possible for >30 to 35 Gy. Bladder dose constraints followed in the randomized controlled trials suggest keeping volume receiving 20 Gy <40% to 50% and volume receiving >35 Gy to <5 to 10 mL. A meta-analysis by Wang et al. 51 showed that the following dose constraints should be included: bladder V(Rx dose) Gy <5 to 10 mL, rectum Dmax <35 to 38 Gy, and urethra Dmax <38 to 42 Gy. To calculate the dosimetric predictors of late urinary flare specific to SBRT, Kole et al. 52 performed probability modeling. An RT of 35 to 36.25 Gy/5 fractions was administered to 216 patients. The 2-year urinary flare-free survival was 95.7% in patients older than 65 years whose bladder D12.7% was ≤33.5 Gy compared with 74.5% who did not meet these criteria. Seymour et al. 53 evaluated 56 patients who received 38 Gy/4 fractions and assessed the relationship of genitourinary toxicity with dose and volume. The following were associated with any grade 2+ genitourinary toxicity: 44-Gy dose to the urethra, 19-Gy dose to the bladder, ≥50 mL prostate volume, and >7 baseline International Prostate Symptom Score. Using a dose of 36.25 Gy in 5 fractions on 67 patients with prostate cancer, King et al. 54 suggested rectal dose constraints of V50% < 50%, V80% < 20%, V90% < 10%, and V100% < 5%. Dose constraints were V50% <40% and V100% <10% for the bladder and V40% <5% for the femoral head dose–volume histogram. It has been suggested that nonmodulated 3DCRT should be avoided and IMRT, if available, should be selected for the delivery of SBRT. The safe and effective SBRT delivery requires image-guided RT. As per UK consensus guidelines, 55 normal tissue dose constraints are V18.1 Gy <50% and V29 Gy <20% in the rectum and V18.1 Gy <40% and V37 Gy <10% in the bladder. Moreover, the optimal dose constraints for prostatic urethra and bowel are V42Gy <50% and V18.1 Gy <5 mL or V30Gy <1 mL, respectively. Finally, the consensus guidelines suggest V14.5 Gy <5% for femoral heads and V29.5Gy <50% for penile bulb. These have been summarized in Table 3.
Dose Constraints for Various Organs.
Toxicity and Concerns
In prostate cancer treatment planning, one of the most significant issues is quality of life. One of the drawbacks of delivering high doses to achieve tumor control is the limitation posed by the adjacent OAR. In addition, prostate movement, either interfractional or intrafractional, may occur during RT. Research has shown that side effects are related to prescription doses. 51
Genitourinary and Gastrointestinal Toxicity
The genitourinary system is the most affected organ system after prostate RT. The common urinary side effects include dysuria, weak stream, and urinary frequency in addition to incontinence and hematuria, which are rare. 56 The etiology of these toxic effects may be related to the dose to the urethra and bladder as well as the prostate size. These events that resolve over time without any medical intervention are usually self-limiting. Most symptoms can be managed with alpha-adrenergic antagonists or nonsteroidal anti-inflammatory drugs; in unresponsive patients, a short course of steroids may be prescribed. Acute toxicity can be limited to a certain extent by adjusting the overall length of treatment time by spacing out treatment over multiple weeks. In the majority of patients, once the acute urinary symptoms resolve, return to baseline or improved urinary function can be expected. Approximately 10% to 20% patients may experience late urinary flare syndrome, which is usually noted around a year after the completion of SBRT. This syndrome is usually self-limiting and managed medically. High doses to small bladder volumes (e.g., hot spots) may predict late urinary flare syndrome. 57 Bladder neck dosimetry and inhomogeneous plans are also considered important in the development of late flare.
Acute gastrointestinal symptoms appear a week following treatment, persist for a month, and subside at 3 months. The urgency and frequency of defecation are the most common acute effects. The most common toxicities include hematochezia in addition to rectal ulcers or rectourethral fistula. These symptoms are usually rare with 35 to 40-Gy SBRT doses and more common in 50-Gy SBRT doses. 51 Late gastrointestinal toxicity can be reduced by tight treatment margins and image-guided therapy. Rectal toxicity can be minimized using enema prior to CT simulation and by reducing stool and gas production with the help of a low-residue diet. Moreover, a bioabsorbable hydrogel spacer between the prostate and rectum can help reduce toxicity.
The SBRT toxicity profile has been evaluated extensively. Rucinska et al. 58 studied SBRT toxicity and quality of life after treatment in 68 localized prostate cancer cases who received the SHARP regimen. Grade III acute bladder toxicity was observed in 1 patient (1.5%) and Grade II toxicity in 24 patients (35.3%). Grade II acute rectal toxicity was observed in 7 patients (10.3%), whereas none developed Grade III toxicity. There were no Grade III or IV late gastrointestinal or genitourinary toxicities. Eight (11.8%) and three (4.4%) patients, respectively, developed Grade II late urinary symptoms and gastrointestinal symptoms. The quality of life/global health status was improved during the observational period. Hence, hypofractionated stereotactic RT in prostate cancer of low and intermediate risk was considered safe and convenient treatment with regard to its duration, toxicity, PSA response, and short-term quality of life. Quon et al. 59 evaluated the effect of overall RT time on the quality of life in cases with cancer of low and intermediate risk. The patients (n = 152) received 40 Gy/5 fractions either every other day or once per week. The acute bowel quality of life was better, with fewer moderate–severe problems with bowel quality of life and better acute urinary quality of life, in the once per week treatment group than in the every other day treatment group. However, no significant differences were detected in the late urinary or bowel quality of life at the 2-year follow-up stage. Van Dams et al. 60 prospectively evaluated the SHARP regimen on 344 patients with localized prostate cancer of high risk who were followed-up for a median of 49.5 months. The results showed 81.7% 4-year biochemical recurrence-free survival and 89.1% distant metastasis-free survival. Furthermore, late grade ≥3 genitourinary and gastrointestinal toxicity were found in 2.3% and 0.9% patients, respectively. Lucchini et al. 61 assessed dose-intensified linac-based SBRT using novel real-time organ motion tracking in unfavorable prostate cancer (n = 13). The patients were delivered SBRT in 4 or 5 fractions, and preliminary findings revealed that reliable real-time prostate monitoring ensures no increased toxicity on dose intensification. These data suggest that SBRT for prostate cancer of high risk has a favorable toxicity and efficacy profile. However, the optimal dose and target volume of SBRT for prostate cancer of high risk need to be validated via additional studies.
The PACE-B trial, 38 a randomized noninferiority Phase III trial with 2-year toxicity data, evaluated acute toxicity with fractionated IMRT and SBRT in 874 men. They were randomly assigned to 2 arms: 62 Gy in 20 fractions across 4 weeks (moderate hypofractionation) or 78 Gy in 39 fractions over 7.8 weeks (conventional fractionation) and 36.25 Gy in 5 fractions over 1 to 2 weeks (SBRT). RTOG grade ≥2 gastrointestinal toxicty occurred in 53 (12%) and 43 (10%) patients, respectively, in the first arm and SBRT arm. RTOG grade ≥2 genitourinary toxicity was observed in 27% and 23% patients, whereas RTOG grade ≥3 gastrointestinal toxicity occurred in 1% and <1% in the 2 groups, respectively. RTOG grade ≥3 genitourinary toxicity was observed in 2% and 2% patients in the 2 groups, respectively, with no treatment-related deaths. Thus, the 2-year toxicity rates were similar for both groups, suggesting that SBRT to the prostate is safe and associated with few side effects. The biochemical outcomes of the study are yet to be published.
Some studies have recommended vigilance owing to significant toxicity observed after RT for prostate cancer. Bauman et al. 62 evaluated brief androgen suppression and SBRT in 15 high-risk patients who were administered androgen suppression for 12 months and RT of 25 and 40 Gy/fraction/week across 5 weeks, respectively, to the pelvic nodes and prostate gland. At 6 weeks, 4/15 patients developed grade ≥2 toxicity. At 6 months, however, 9/15 (60%), 4/15 (26%), and 5/15 (30%), respectively, developed grade ≥2 gastrointestinal/genitourinary toxicity, ≥3 gastrointestinal/genitourinary toxicity, and grade ≥2 gastrointestinal and genitourinary toxicity, suggesting late toxicities that were significantly higher than hypothesized. The high toxicities prompted study termination before Phase 2 patient recruitment. Scorsetti et al. 39 assessed the early side effects of SBRT in a prospective Phase I and II study. The study included 26 and 14 patients with prostate cancer of low and intermediate risk, respectively, who were treated with 35 Gy in 5 fractions every 2 days. The results revealed 10% Grade 2 rectal and 40% Grade 2 genitourinary toxicity; however, no grade ≥3 toxicity was detected. Koh et al. 63 evaluated rectal toxicity in 91 patients who received 45, 47.5, and 50 Gy in 5 fractions. High-grade rectal toxicity was observed in 6.6% patients; 5 of these patients required colostomy. When the volume of rectal wall receiving 50 Gy was >3 mL and circumference of rectal wall receiving 39 Gy was >35%, grade ≥ 3 delayed rectal toxicity was observed. Moreover, when the circumference of rectal wall receiving 24 Gy was >50%, grade ≥2 acute rectal toxicity was significantly observed. These results suggest that caution is necessary when considering high-dose SBRT and certain threshold dose constraints should be followed.
Sexual Dysfunction and Quality of Life
Sexual dysfunction is another area of concern in prostate RT. However, with regard to male sexual health and well-being, SBRT has an excellent toxicity profile. 64 Acute toxicity is rare, although sperm count may be affected. During and in the months following prostate SBRT, the risks of conceiving should be explained. ADT is more associated with sexual symptoms than RT alone. The long-term toxicity of SBRT include the inability to achieve erection. The cause of erectile dysfunction after SBRT is unclear. Erectile dysfunction has been attributed to damage to the posterior structures of penile bulb and neurovascular bundles. Elias et al. 65 evaluated SBRT using 35 Gy in 5 fractions on 84 patients and observed poor sexual quality of life associated with penile bulb V20 Gy >40% and V35 Gy >4%. Boyer et al. 66 evaluated the toxicity and quality of life in 60 patients with prostate cancer of low and intermediate risk who received 37 Gy/5 fractions. The patients were followed-up for a median period of 27.6 months, during which there was no genitourinary toxicity of Grade 3 or higher. Four and five patients, respectively, developed late Grade 2 toxicities in the genitourinary and gastrointestinal systems. Besides, only one patient experienced late Grade 3 gastrointestinal toxicity. The median score of the American Urological Association symptom increased to 11 while on treatment (p < .01) from 4.5 prior to treatment, but 36 months posttreatment, the score was 5 (p = .65). Moreover, the median score of The International Index of Erectile Function declined to 17 from 19 during the follow-up period (p < .01). The expanded prostate cancer index composite short form score of the sexual domain was reduced at 36 months following intervention (67.9 vs. 45.2). Katz and Kang 67 conducted a 7-year CyberKnife SBRT study on genitourinary and rectal toxicity as well as sexual quality of life in 515 patients with prostate cancer of localized, low, intermediate, and high risk who were followed-up for a median period of 72 months. Acute Grade 3 or 4 complications were not observed, whereas acute Grade 2 urinary or rectal toxicity was observed in <5% patients. Grade 2 rectal and urinary toxicity was observed in 4% and 9.1% patients, respectively, but Grade 3 urinary toxicity was detected in only 1.7% patients, indicating low late toxicity. The mean of the expanded prostate cancer index composite that included the quality of life of the urinary system and bowel reduced a month after RT. The index composite was at baseline at the end of 2 years and stabilized thereafter. With respect to the sexual quality of life, the expanded prostate cancer index composite reduced by 23% at 6 to 12 months and was steady subsequently. During the last follow-up, 67% of all patients potent at baseline remained potent. 67
Future Directions
Numerous randomized controlled trials, including 2 PACE trials (PACE-A and PACE-C), are currently ongoing to assess SBRT toxicity and efficacy. PACE-A will compare SBRT with radical prostatectomy and analyze bowel and bladder toxicities, DFS, biochemical PFS, and biochemical DFS parameters. Multiple trials are also analyzing the utility of emerging advanced imaging techniques (such as MRI) for dose escalation. 68 MRI facilitates safe dose escalation to MR-identifiable lesions and allows accounting for changes in rectal and/or prostate anatomy for accurate dose delivery. Moreover, the combination of SBRT and ADT is being studied in prostate cancer.69, 70 Finally, SBRT in patients with prostate cancer of high risk is being evaluated. 71 It is unclear whether pelvic lymph nodes should be targeted with radiation. In this context, RTOG 09-24 study results may help in the counseling of patients with regard to the increased toxicities and potential benefits of targeting the lymph nodes.
Conclusion
Prostate cancer is increasingly being treated with SBRT. While similar to conventional fractionation with regard to outcome, it reduces the overall treatment time. Indeed, most studies have reported high biochemical recurrence-free rates (90%-100% for low risk and 84%-100% for intermediate risk). With regard to the toxicity profile, most studies have reported no significant late toxicities (1%-3% Grade 3 gastrointestinal or genitourinary toxicity), with quality of life being similar to conventional fractionation. MR simulation and MR-guided RT systems may help in better conformity with dose delivery to the target.
Footnotes
Author Contributions
KG, NB, and AM performed literature review and drafted and revised the manuscript. KT and HT supervised the literature review and revised the manuscript. All authors agree to the final version of the manuscript.
Ethical Approval
This is a review article, and no patient data have been used in this article.
Informed Consent
No patient data have been used in this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
