Abstract
Introduction:
Cutaneous metastases in patients with incurable cancer can severely impact quality of life, highlighting the need for minimally invasive treatment options. Electrochemotherapy (ECT) effectively treats tumors across various cancer types by combining a standard chemotherapy dose (15,000 IU/m2 bleomycin) with electric pulses to enhance drug uptake, aiding in the destruction of the tumor cells. Studies evaluating reduced bleomycin dosage in ECT have shown positive outcomes. This study investigates whether halving the chemotherapy dosage during ECT for cutaneous tumors is as effective as standard ECT treatment.
Methods:
This double-blinded, randomized clinical trial will include 110 biopsy-verified cutaneous metastases of any cancer type from approximately 55 patients with incurable cancer. Patients will be randomly assigned (1:1 ratio) to receive either a full or half dose of bleomycin. The primary endpoint is overall tumor response after 3 months, assessed using the modified Response Evaluation Criteria in Solid Tumors. Biological samples during ECT will measure the distribution of bleomycin in normal tissue, tumors, and blood via liquid chromatography. In addition, the percentage of tumor cells will be correlated with bleomycin concentration. Side effects will be monitored, and qualitative interviews with a subset of patients before and after treatment will explore their experiences with cutaneous tumors and ECT. The study will be conducted at Zealand University Hospital, Roskilde, and Copenhagen University Hospital, Herlev Gentofte.
Discussion:
Advances in cancer treatment have led to increased survival rates, and the patients we treat with ECT are often fragile. If the reduced bleomycin dose proves as effective as the standard dosage, it could reduce side effects and thereby benefit elderly, frail, and kidney-compromised patients. The findings of this study may be relevant not only to the treatment of cutaneous metastases but also to deep-seated tumors treated with ECT.
Introduction
Cutaneous metastases may be of significant distress to patients due to symptoms such as pain, bleeding, oozing, and psychological stress.1,2 Between 0.7% and 10% of all cancer patients develop cutaneous metastases, most often in those with advanced-stage disease, particularly among individuals with breast cancer or melanoma. 3 Treating these cutaneous conditions in patients with incurable cancer remains a complex and challenging task, emphasizing the need for treatment approaches that balance oncological outcomes with patients’ quality of life (QoL).
Over the past 20 years, electrochemotherapy (ECT) has become a standard treatment for cutaneous malignancies in Europe.4,5 According to NICE (National Institute for Health and Care Excellence) guidelines, it has a role in the treatment of basal cell carcinoma, squamous cell carcinoma, and in the palliative treatment of skin tumors of nonskin origin and melanoma,6,7 and is endorsed by ESMO (European Society for Medical Oncology) guidelines for the treatment of melanoma metastases. 8 ECT enhances the local uptake of chemotherapeutic agents into tumor cells by administering the chemotherapy either intratumorally or intravenously, followed by application of brief electrical pulses to the tumor. This procedure temporarily increases cell membrane permeability, facilitating the entry of chemotherapy into the cells and leading to cell death.9,10 ECT is typically a one-time treatment, although retreatment is possible in cases of recurrence or new tumor development. 4
ECT treatment of cutaneous metastases has demonstrated consistently high response rates across various tumor histologies. A study including nearly 2500 cutaneous metastases of different histologies reported a complete response (CR) rate of 71% and an overall response rate of 86%. 11 Given the significant potential of this treatment, it is of great interest to investigate whether the chemotherapy dosage may actually be reduced. A lower dose could benefit frail and kidney-impaired patients while also expanding ECT’s potential for treating deep-seated tumors12–15 and for use in combination with immunotherapy. 16
Previous Research on Reduced Bleomycin and Rationale for 50% Reduction
ECT has utilized bleomycin at a standard intravenous dose of 15,000 IU/m2, based on its use in testicular cancer treatment. 17 Recent smaller studies have explored the feasibility of reducing bleomycin doses by up to 50% due to concerns about renal impairment, lung impairment, and patient age. These studies have indicated promising outcomes, suggesting that a reduction of up to 50% in bleomycin concentration could be as effective as the standard dose.18–21
Pharmacokinetics
Comprehensive pharmacokinetic studies are essential to confirm the mechanism and efficacy of reduced bleomycin in ECT. Previous research has highlighted age-related differences in bleomycin clearance, emphasizing the importance of individual dosing strategies based on age and renal function. 22 To evaluate the feasibility of reducing bleomycin for all patients, similar analyses will be conducted in this study. Furthermore, we aim to investigate whether bleomycin is taken up more readily in tumors than in normal skin, and whether the percentage of tumor cells within the tumor influences this uptake, as suggested by previous research. 23
Aim
This protocol outlines a double-blinded, randomized controlled clinical trial (RCT) to evaluate whether a 50% reduction in bleomycin dosage maintains efficacy in ECT for cutaneous metastases. Additionally, the study will include analysis of QoL and the distribution of bleomycin in skin, tumors, and blood for both dosage groups (Fig. 1).
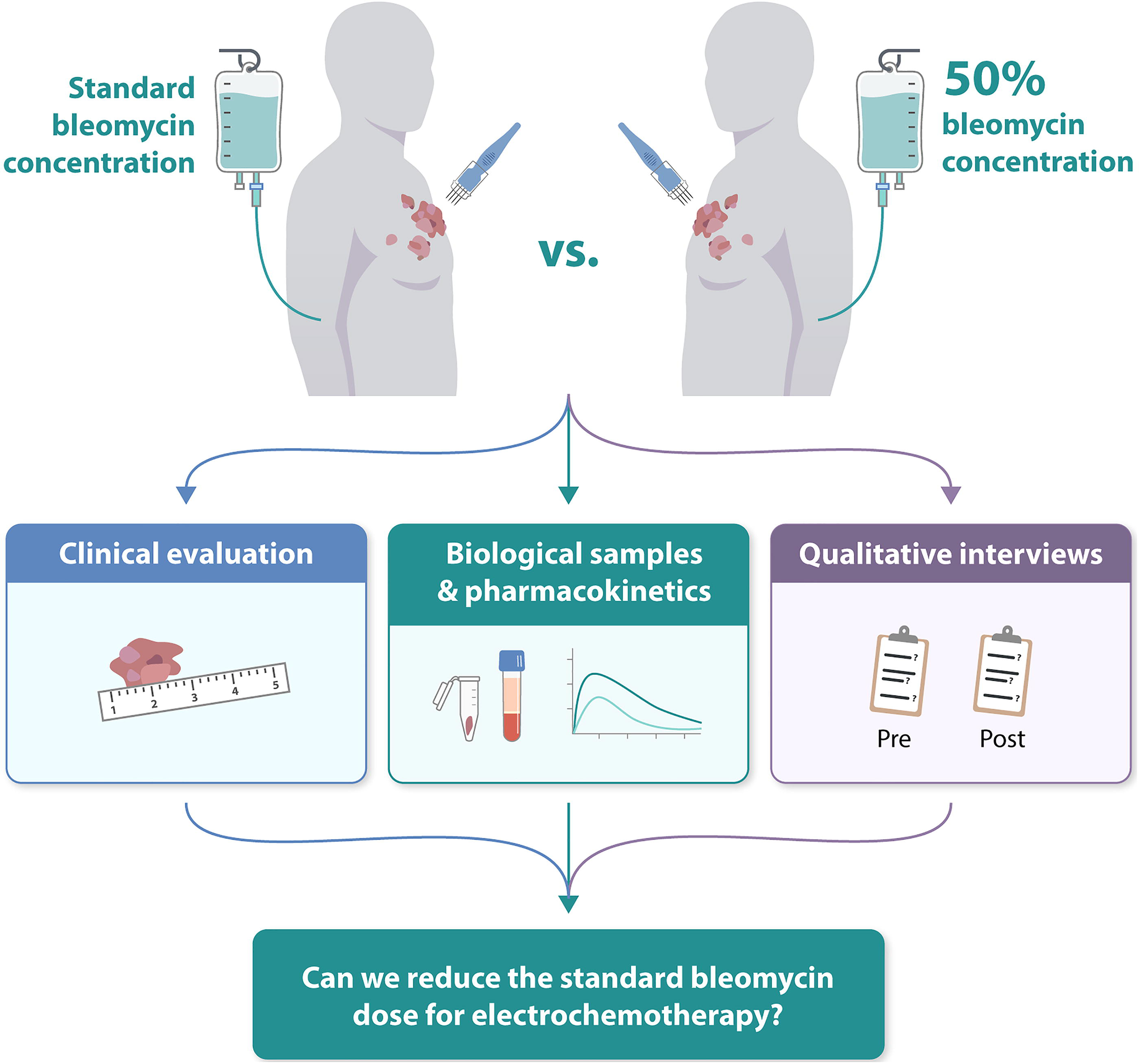
Graphic abstract illustrating the design and objectives of the BLESS Trial, which investigates whether the dose of bleomycin in electrochemotherapy for cutaneous metastases can be safely reduced. Patients are stratified by tumor size and randomized to receive either a full or a half dose of bleomycin. The primary endpoint is response measured 3 months posttreatment. Pharmacokinetic analyses assess bleomycin concentration in blood, tumor tissue, and normal tissue to understand the relation between time and concentrations and to determine whether the 8-minute interval from bleomycin infusion to pulse application is necessary. Bleomycin concentrations in tumor and normal tissue will be compared with assess distribution differences. Furthermore, concentrations in tumor biopsies will be correlated with the percentage of tumor cells in the biopsy. Qualitative interviews as well as questionnaires investigate the patient-reported outcomes in both treatment groups.
Methods
Study design and setting
This investigator-initiated study is a parallel, two-arm, double-blinded RCT in which patients are stratified based on the size of their largest cutaneous tumor and subsequently randomized (1:1) to receive either a 15,000 IU/m2 bleomycin or 7500 IU/m2 bleomycin. The study aims to include 110 tumors from approximately 55 patients with incurable cancer (Fig. 2).
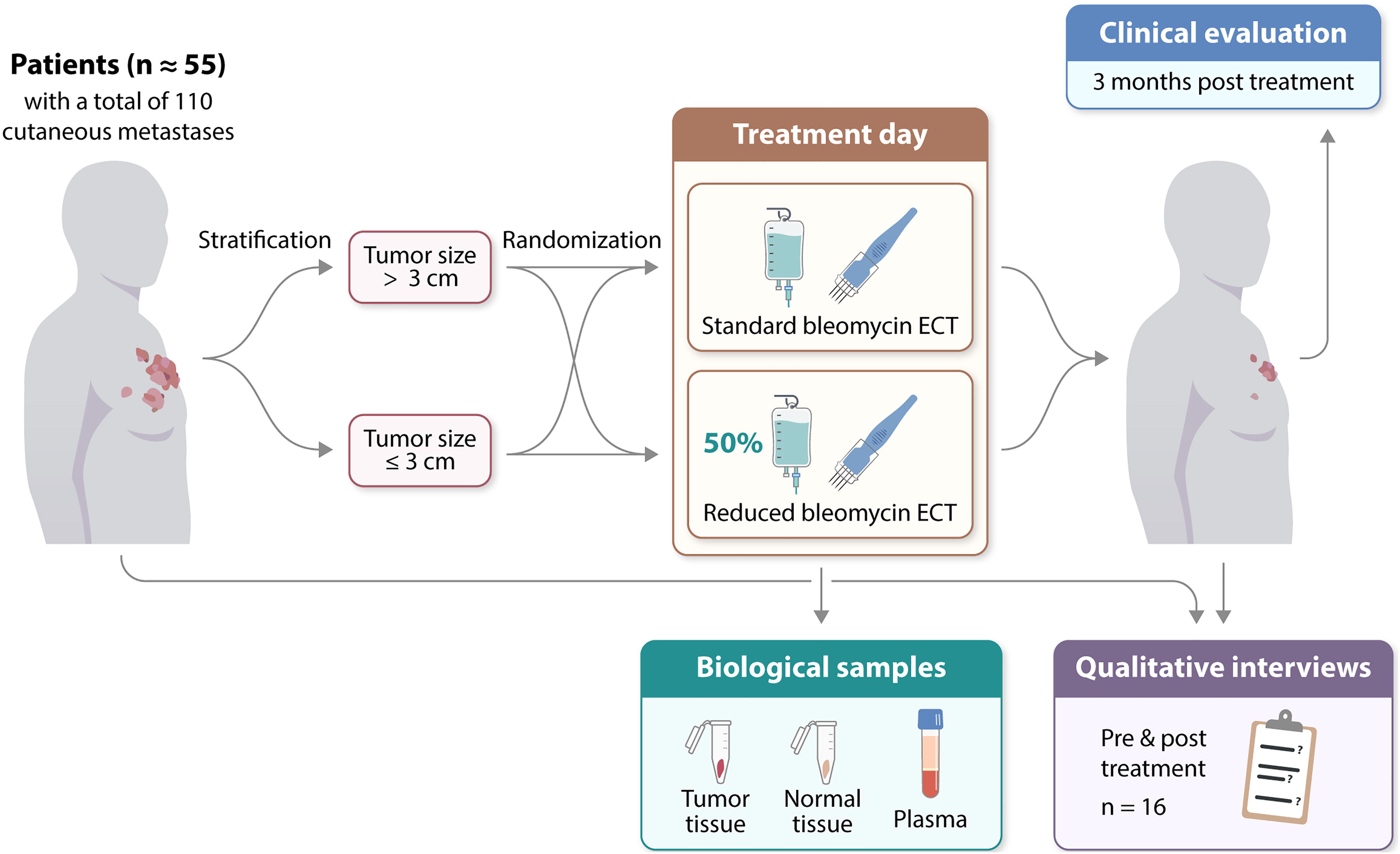
Study design of the BLESS Trial, a randomized controlled trial. A total of 110 tumors from approximately 55 palliative patients will be included. Patients will be stratified based on tumor size and then randomized to receive full dose or a 50% reduced dose of bleomycin. The primary endpoint is treatment response evaluation 3 months posttreatment. Qualitative interviews will be conducted with 16 patients both before and approximately 3 months after treatment. On the day of treatment, biological samples will be collected for pharmacokinetic analyses. ECT, electrochemotherapy.
The study is conducted at two centers in Denmark: (1) the Department of Clinical Oncology and Palliative Care, Zealand University Hospital, Roskilde and (2) the Department of Oncology, Copenhagen University Hospital, Herlev Gentofte Hospital. The planned 2-year inclusion period commenced in October 2024.
Participants
Patients are eligible if they meet the inclusion and exclusion criteria (Table 1). Due to the biopsy requirements, their largest tumor must have a size sufficient to perform the listed biopsies. All patients receive both written and oral information about the study and are offered a minimum of 24 hours for consideration. Inclusion requires written informed consent. Additionally, patients may choose to take part in qualitative interviews and undergo a biopsy in the treated area after 12 months.
Inclusion and Exclusion Criteria

ECT, electrochemotherapy.
Primary endpoint
The primary endpoint is to evaluate and compare the overall tumor response between tumors treated with the standard dose of bleomycin and those treated with half the dose. Response will be measured at 3 months and compared with baseline.
Treatment response will be evaluated at the tumor level using the modified Response Evaluation Criteria in Solid Tumors,
24
defined as follows:
Complete Response (CR): disappearance of the tumor. Partial response (PR): a decrease of at least 30% decrease in the tumor’s largest diameter. Progressive disease: an increase of at least 20% in the tumor’s largest diameter. Stable disease: neither 30% decrease nor 20% increase of the largest diameter of the tumor.
A maximum of seven tumors per patient will be evaluated. The response rate will be calculated as the number of responding tumors relative to the total number of treated tumors. Tumor response will be evaluated through clinical examination, including ruler measurements and photographic documentation.
Secondary endpoints
All secondary endpoints will be analyzed in the entire study group as well as for the two dose groups separately (Table 2).
Secondary Endpoints

mRECIST, modified Response Evaluation Criteria in Solid Tumors; POSAS, Patient and Observer Scar Assessment Scale; EORTC QLQ-C30, European Organisation for Research and Treatment of Cancer – Quality of Life Questionnaire, Core 30; eGFR, estimated glumerular filtration rate; BSA, Body Surface Area.
Ethics, safety, monitoring, and dissemination
This study has been approved by the Danish Medicines Agency and the Medical Ethics Committee, CTIS-number 2024-513360-25-00. It adheres to the Declaration of Helsinki and Good Clinical Practice (GCP) and is monitored by the GCP Unit in Copenhagen.
Participants are patients who would otherwise be eligible for ECT, ensuring no additional treatment-related risks.
Informed consent is required, and participants are insured under the Danish Patient Compensation Scheme. The study may be terminated early if severe side effects make continuation unacceptable, or enrollment is too low to complete the trial, or investigators deem patient safety to be at risk.
The study is registered at ClinicalTrials.gov, with the first patient included on October 29, 2024. Findings, whether positive, negative, or inconclusive, will be submitted for publication in international peer-reviewed scientific journals and presented at relevant conferences. Reporting will follow the CONSORT 25 and TIDieR 26 guidelines.
Patient involvement
The protocol was reviewed during its design phase on January 19th, 2022, in a meeting with the Patient Panel for Cancer Research at the Department of Clinical Oncology and Palliative Care, Zealand University Hospital. The panel included nine members, all cancer patients or caregivers. Members responded positively to the proposed study and found randomized reduced-dose bleomycin acceptable. Additionally, the panel proposed minimizing mandatory follow-ups to offer patients a more flexible schedule.
Randomization and blinding
Independent of the study researchers, randomization numbers were assigned to either half or standard dose of bleomycin using blocks of 10 to ensure balanced group allocation. The process was facilitated by the open-source program Sealed Envelope (Sealed Envelope | Randomization [randomization] and online databases for clinical trials). Randomization envelopes are securely stored in two locations at each site: the hospital pharmacy and the oncology department’s medication room. This dual storage arrangement is designed to enable emergency unblinding if required.
At the pharmacy, medication will be prepared according to the randomization envelopes. Treatment bags will be labeled with the patient ID (randomization number) but will not reveal the concentration of bleomycin, ensuring blinding for all personnel except the pharmacist.
Recruitment procedure and pretreatment assessment
Eligible patients will be identified during appointments in the plastic surgery or oncological outpatient clinics. Included patients will be stratified based on the size of their largest cutaneous tumor (≤3 cm or >3 cm) and subsequently receive a randomization number. During the pretreatment assessment, data will be collected on medical history, comorbidities, tumor histology and TNM classification, previous and current antineoplastic treatment, surgical history, pain score (Numeric Rating Scale 27 ), physical status, and baseline symptoms related to the tumors and overall cancer status. Additionally, up to seven cutaneous metastases will be identified and numbered, and their localization, size, availability for electroporation, and previous radiation/electroporation will be registered. Photographs, including a ruler for scale, will be taken of each tumor (Table 3). Data will be entered into an electronic Case Report Form using REDCap (Research Electronic Data Capture).
Trial Procedures
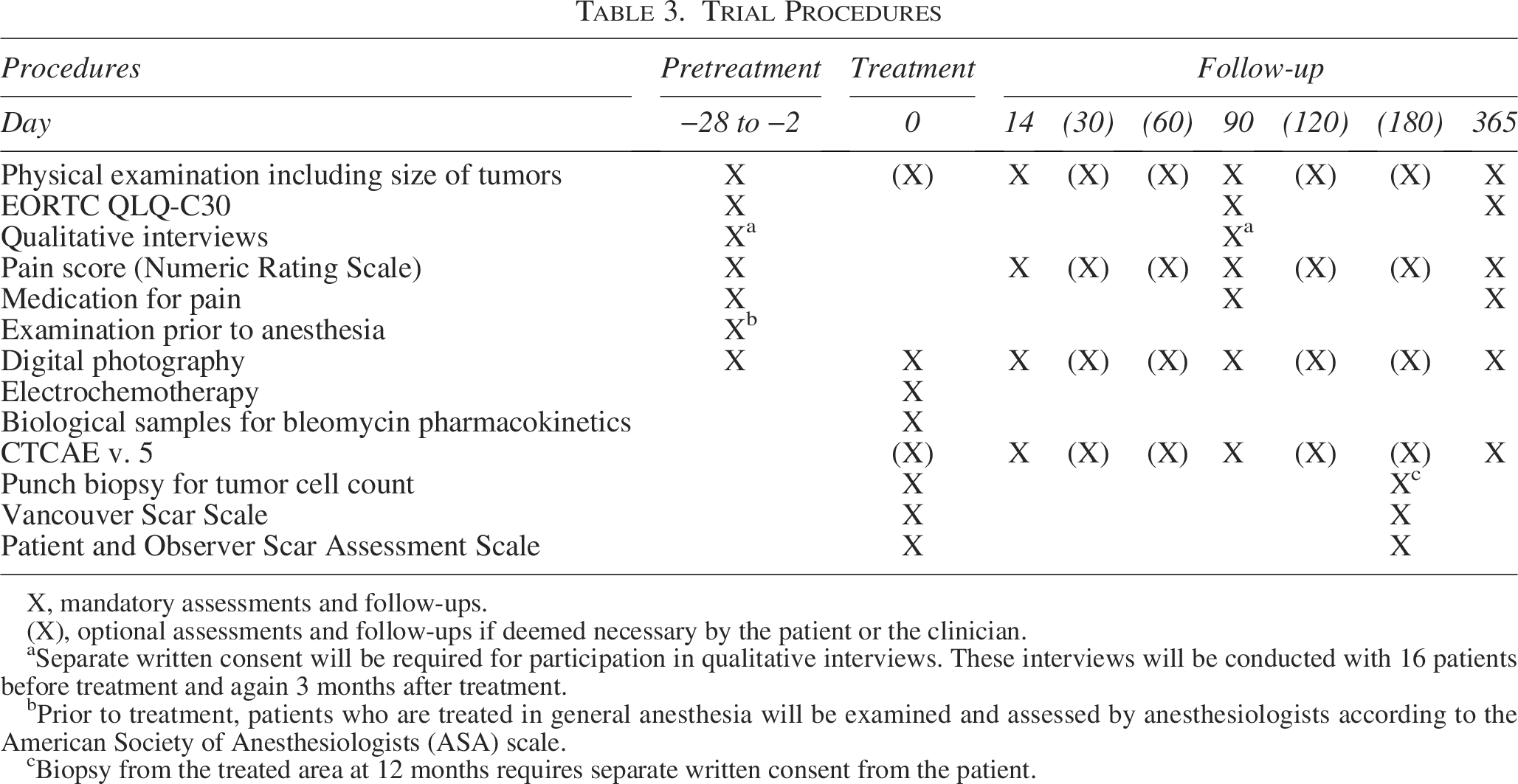
X, mandatory assessments and follow-ups.
(X), optional assessments and follow-ups if deemed necessary by the patient or the clinician.
Separate written consent will be required for participation in qualitative interviews. These interviews will be conducted with 16 patients before treatment and again 3 months after treatment.
Prior to treatment, patients who are treated in general anesthesia will be examined and assessed by anesthesiologists according to the American Society of Anesthesiologists (ASA) scale.
Biopsy from the treated area at 12 months requires separate written consent from the patient.
Quality of life
QoL will be assessed using the EORTC QLQ-C30 questionnaires 28 at three time points: before treatment, 3 months after treatment, and 12 months after treatment. At inclusion, patients will also be invited to participate in interviews conducted before treatment and again 3 months after treatment. The first 16 consenting patients will be interviewed face to face by a nurse who is otherwise not involved in the study. Each interview will last 1–2 hours, following a semistructured interview guide focused on QoL, patients’ experiences with cutaneous metastases, and ECT.
Treatment
All patients will be treated according to the standard procedure for ECT. 4 Patients are expected to undergo general anesthesia, as this study involves intravenous bleomycin, which is typically used for larger or multiple tumors. However, in case local anesthesia is indicated, this is allowed in the study. Following anesthesia, bleomycin will be administered intravenously over a few minutes. An 8-minute interval will follow before applying the electric pulses to the tumor using needle electrodes. At both sites, the Cliniporator (IGEA, Capri, Italy) is being used, and linear or hexagonal electrodes will be selected based on the tumor size and physician’s choice. 4 The choice of electrode is recorded and will be reported.
Biological samples at treatment day
The biological samples collected on the day of treatment include venous blood samples and 3 mm punch biopsies from both tumor tissue and normal skin (Fig. 3). All samples, except the biopsy used to analyze tumor cell percentage, are immediately placed on ice and transferred to a −80°C freezer. Tumor biopsies are collected from the same tumor.
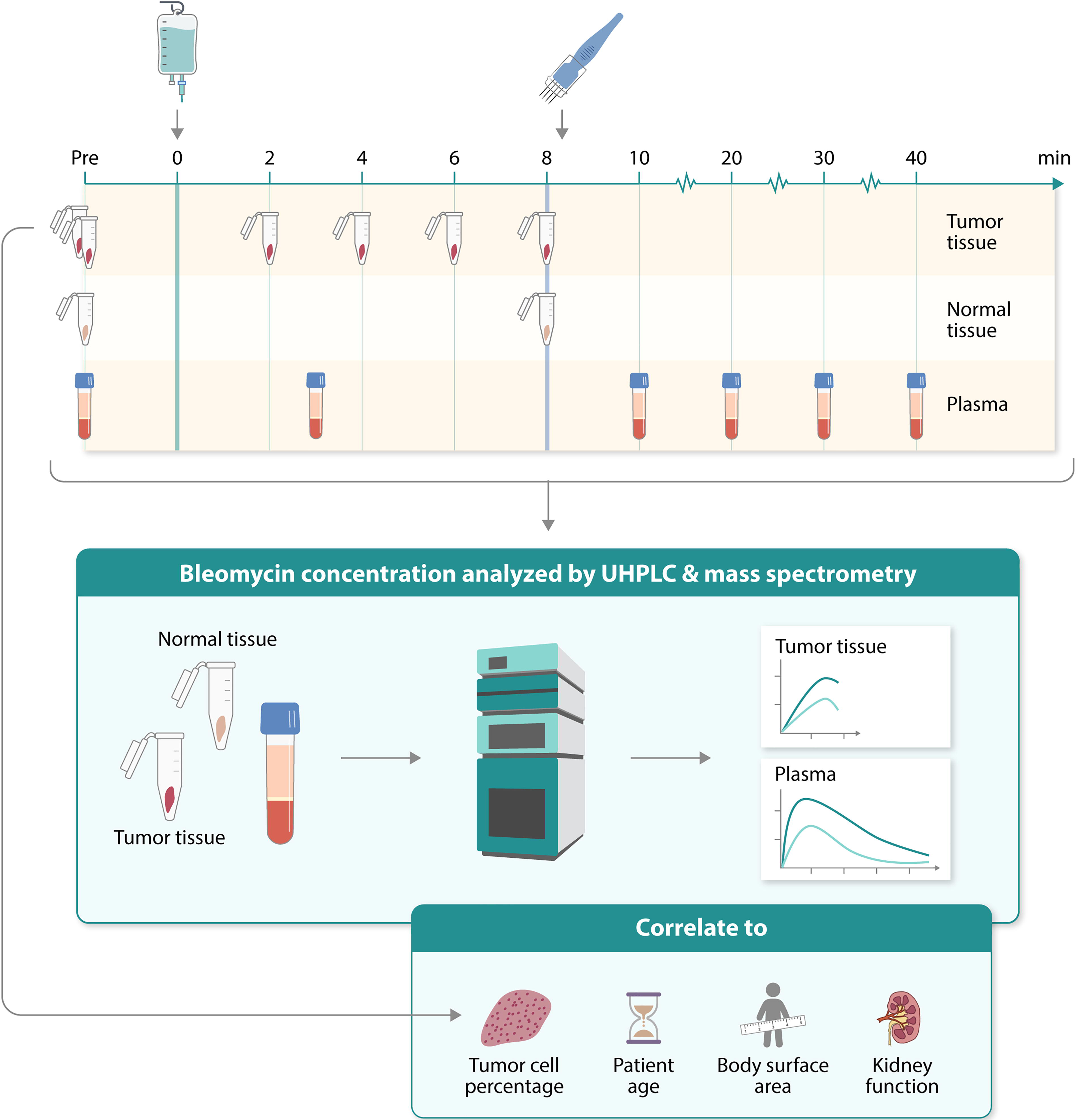
Illustrates the time points of biological sample collection on the treatment day. Plasma and tissue samples will be analyzed using ultra high performance liquid chromatography and mass spectrometry, and bleomycin concentration will be correlated with tumor cell percentage, patient age, body surface area, and kidney function.
Biopsies
To determine the tumor cell percentage, one biopsy will be taken before bleomycin infusion.
To quantify bleomycin levels in the tumor, five biopsies will be taken: one before bleomycin infusion and four at 2, 4, 6, and 8 minutes after bleomycin infusion.
To quantify bleomycin levels in normal skin, two biopsies will be collected: one before bleomycin infusion and another at 8 minutes after bleomycin infusion.
Blood tests
Venous blood samples will be collected before bleomycin infusion and at 5, 10, 20, and 40 minutes after bleomycin infusion. Samples will be centrifuged at 2000 rpm for 10 minutes at room temperature. Subsequently, 2 mL plasma will be transferred to tubes and stored at −80°C until analysis.
Analysis for bleomycin content in biopsies and plasma
Biological samples will be prepared and analyzed for bleomycin content at Roskilde University’s Department of Science and Environment, following previously established protocols.29,30
Bleomycin separation and quantification will be performed using ultra-high-performance liquid chromatography coupled with quadrupole mass spectrometry. 30
Biopsy sample preparation will include grinding into a fine powder and suspension in 0.1% formic acid (HCOOH) before sonication at 35 kHz. The samples will then be centrifuged at 8600 g, and the supernatant will be filtered. Solid-phase extraction will be performed using Phree phospholipid removal cartridge, with acetonitrile as an eluent. 29
Plasma samples will be prepared by adding CuSO4 and BLM-A5 as an internal standard. Protein, phosphate, and particulate matter will be removed using Phree 96-well plate with cold acetonitrile. The resulting solution will be filtered before further analysis. 30
Patient follow-up
The follow-up schedule and the different interventions include mandatory follow-ups will take place at 2 weeks, 3 months, and 12 months posttreatment. Optional follow-ups may be arranged at 1, 2, 4, and 6 months if deemed necessary by the patient or the clinician. At all follow-up appointments, physical evaluations will be conducted, including assessments of tumor size, pain scores, and documentation of adverse events.
During the appointments at 3- and 12-month follow-up visits, the appearance and characteristics of the treated area will be evaluated by Vancouver Scar Scale 31 and the Patient and Observer Scar Assessment Scale (POSAS). 32 In addition, patients will be asked to complete the EORTC questionnaire, and any current medication will be documented. For patients who provided consent, a biopsy of the treated area will be conducted 12 months posttreatment (Table 3).
Statistical methods
Power calculation
This noninferiority study aims to compare the overall response at the tumor level 3 months after ECT using two different doses of bleomycin. The sample size was calculated to ensure sufficient statistical power to determine whether halving the bleomycin dose is noninferior to the standard dose. The formula for noninferiority study with continuous data was used. 33 The standard deviation was derived from 55 studies from 1996 to 2021, which investigated ECT with intravenous bleomycin across various tumor histologies. 21 An acceptable noninferiority margin was defined as a 10% difference in response rates. The significance level was set at 0.05 with 80% power. Based on prior ECT studies reporting drop-out rates around 16%,11,34–37 a conservatively drop-out rate of 20% was applied. As a result, the total number of tumors required was calculated to be 110. A prior study involving 987 patients with cutaneous malignancies of different histology reported an average of 2.5 tumors per patient. 11 Based on this, we anticipate an average of at least 2 tumors per patient and therefore plan to enroll approximately 55 patients.
Primary and secondary endpoints
Tumor response will be analyzed using a χ2 test, comparing the number of responding tumors with the total number of treated tumors 3 months posttreatment. For the Vancouver Scar Scale and POSAS, nonparallel statistical analyses will be used to compare esthetic outcome with their scales in the two groups. Descriptive statistics will be used when analyzing the histological samples, all-cause mortality, and adverse events. The EORTC QLQ-C30 generates both ordinal and nominal data. Raw data will be transformed to a linear scale (0–100) according to the EORTC guidelines. The ordinal data will be subgroup compared using one-way analysis of variance test and the nominal using Fisher’s exact test.
Analysis of interviews
Qualitative interviews will be audiorecorded, transcribed, and analyzed using thematic analysis, as recommended by Braun and Clarke. 38 To support the analysis process, NVivoR for Windows (QSR International) will be used.
Discussion
This study combines a double-blinded RCT, QoL assessments, and pharmacokinetic analyses, offering a comprehensive approach to advancing the understanding of the mechanisms and application of ECT.
Study design
This trial compares standard-dose bleomycin with a 50% reduced dose. Although exploring additional dosing levels might improve the chances of identifying the optimal dose, such an expansion would require a larger study and exceed available resources and timeframe. A previous study demonstrated encouraging results with a 50% dose reduction, 18 and by focusing exclusively on this reduction, we aim to collect meaningful data without overextending the trial capacity.
The primary endpoint is overall tumor response 3 months after treatment. As tumors in this study will be treated with intravenously bleomycin, they are typically larger and may require more time to exhibit full therapeutic response. 39 Earlier response assessments, as conducted in other studies,12,18,35,40 may not fully capture the therapeutic effects on such lesions. Given the palliative context, follow-up is limited to 12 months.
Tumor-level response evaluation
Evaluating ECT response at the patient level might seem simpler and more intuitive, as all tumors within a patient receive either the half or standard dose of bleomycin. However, tumors within the same patient can vary significantly in size, prior irradiation, and recurrence status—all factors that influence their response to ECT.11,35,40 Given that ECT is a local ablative treatment, despite the systemic administration of bleomycin, we have chosen to assess response at the tumor level as a primary endpoint, consistent with standard practice. 4 As a secondary endpoint, we will evaluate complete and partiel response at the patient level.
Pharmacokinetics of bleomycin
The concentration of bleomycin in blood during ECT with intravenous administration has previously been studied.22,41 These studies found that the optimal window for applying electric pulses, based on plasma bleomycin concentration, is 5–15 minutes for patients under 65 and 8–40 minutes for those over 65, due to slower drug clearance in the latter group. 22 Currently, ECT with intravenously bleomycin follows a standard protocol recommending an 8-minute wait after bleomycin infusion before applying electric pulses. 4
Our study aims to refine the timing of pulse delivery by analyzing bleomycin levels in both tumor biopsies and venous blood samples, while simultaneously evaluating clinical response. To our knowledge, this integrated approach has not been applied in this scale before. Optimizing pulse timing could potentially reduce anesthesia duration for patients. While our biopsies will not extend beyond the 8-minute mark, we aim to identify trends in bleomycin concentration over time.
In addition to tumor biopsies, we will analyze bleomycin levels in normal skin to investigate whether drug preferentially accumulates in tumor tissue. Previous research suggests that the disaccharide component of bleomycin may enhance its uptake in tumor cells relative to normal skin cells. 23 To explore this further, tumor cell percentage will be correlated with the measured bleomycin concentration in the tumor biopsies, shedding light on its distribution dynamics.
Quality of life
This study focuses on patients with incurable cancer, aiming to improve their quality of life. To gain a deeper understanding of the complexities surrounding patient experiences and QoL, qualitative interviews are essential, as these aspects cannot fully be captured by EORTC questionnaires. 42 The number of patients selected for interviews has been carefully considered to strike an appropriate balance—sufficient to provide a nuanced and comprehensive perspective on the research questions, yet limited enough to allow for in-depth analysis without the material becoming unmanageable or overly broad.43,44
Limitations
While this study is designed to address several important questions, it is not without limitations. The inclusion of patients with any type of biopsy-verified cutaneous metastasis means that we may have a limited ability to distinguish responses based on specific pathologies. Additionally, our correlation of bleomycin concentration in tumors and kidney function will be constrained, as we are including only patients with a minimum creatinine clearance above 50 mL/minute. This area requires further investigation, given the critical role of renal excretion in bleomycin clearance and the challenges posed by patients with impaired kidney function.
Perspectives
This dose-de-escalating study holds promise for improving ECT treatment, particularly for elderly and multimorbid cancer patients with cutaneous metastases. Additionally, the study will provide valuable insights into bleomycin pharmacokinetics, potentially optimizing ECT in deep-seated tumors.
Authors’ Contributions
F.A.B., A.B., and J.G. made the first draft of the trial protocol. C.K.L., E.E.R., M.P.M., T.A.M.A., L.R.H., N.K., B.M., and M.A.T. contributed to the design of the protocol. M.T. wrote the first draft of the article and made the first draft of figures and tables. All authors contributed to revisions, read. and approved the final version of the article.
Footnotes
Author Disclosure Statement
None of the authors declare any conflicts of interests.
Funding Information
This work was supported by The Danish Cancer Society grant number R325-A18717. The study also received funding from Region Sjællands Sundhedsvidenskabelige Forsknings Fond grant number R46-A2242.
