Abstract
Ground support technology in underground mines has evolved over time in response to challenges associated with mining at depth and poor ground conditions. The performance of individual ground support elements is defined by their reinforcement and surface support mechanisms, element configuration, and material properties. In response to industry demand, ground support manufacturers have prioritized the development of ground support elements that meet a set of acceptable, or desirable, mechanical properties under quasistatic or impact loading conditions. The degradation of ground support elements when exposed to corrosive environments has not received the same level of attention. This paper investigates the influence of low-carbon steel alloys on the susceptibility to corrosion of ground support elements. It draws from a database developed over several years on the electrochemical behaviour of ground support steel and its chemical composition in a simulated mine electrolyte as lixiviate. Two predictive statistical models were developed to interpret the impact of steel composition and the influence of specific alloying elements. The results of this study have significant implications for ground support elements exposed to corrosive underground environments.
Keywords
Introduction
A successful ground support system employs both reinforcement and surface support elements that work as a system to maintain the stability of an excavation under the anticipated load and ground conditions over the working life of an excavation. Reinforcement is the process where rock bolts and cable bolts are applied internally to the rock mass, while surface support is a technique in which elements such as shotcrete, steel mesh, and straps are applied externally to excavation surfaces (Hadjigeorgiou and Potvin, 2011).
Ground support technology in underground mines has evolved over time in response to challenges associated with mining at depth and poor ground conditions. The performance of individual ground support elements is defined by their reinforcement and surface support mechanisms, element configuration, and material properties. This has led to significant developments in rock reinforcement and surface support elements that have higher energy-absorbing capacity than conventional ground support units (Li et al., 2014; Potvin and Hadjigeorgiou, 2020).
The focus of suppliers to mine operations has been on developing ground support elements that meet a set of acceptable, or desirable, mechanical properties under quasistatic or impact loading conditions. Although establishing the mechanical behaviour of ground support elements is an important element of the design process, it does not address the long-term performance of a ground support system. In an underground mining environment, it is recognised that ground support may degrade over time and compromise the integrity of an excavation (Hadjigeorgiou, 2016). Degradation can be triggered by any one or a combination of these factors, including mine-induced seismicity; drive convergence resulting in loading of individual reinforcement or support elements; blast damage associated with explosive gases and flyrock; material quality and the presence of manufacturing flaws; installation issues, such as bolt orientation, grout quality, damage to protective coatings or galvanization; damage to reinforcement or support caused by equipment and corrosion of support systems.
The susceptibility to corrosion of any element of a ground support system, including reinforcement and surface support, may result in falls of ground (Figure 1). This is often referred to as the weakest link theory.

Corroded ground support resulting in a fall of ground.
Of interest is the impact of different alloy combinations on the resulting corrosion rate under electrochemical investigations. This paper investigates the influence of low-carbon steel alloys on the susceptibility to corrosion of ground support elements. It draws from a database developed over several years on the electrochemical behaviour of ground support steel and its chemical composition in a simulated mine electrolyte as lixiviate. Two predictive statistical models were developed to interpret the impact of steel composition and the influence of specific alloying elements. The results of this study have significant implications for ground support elements exposed to corrosive underground environments. The intent of the paper is to provide a working tool for suppliers in recognizing the potential for increased susceptibility to corrosion of certain steel combinations. This may be important at mine sites where ground support will be exposed during its working life to a corrosive environment.
Corrosion of ground support
The corrosion of ground support is a very complex process with multiple factors dictating the susceptibility to corrosion of ground support systems (Figure 2). The steel chemical composition is invariably the starting point of any investigation, but the material processing and manufacturing of ground support elements are significant factors. The ground control configuration has a significant impact. For example, given that friction rock stabilizers are hollow and do not employ cement or resin grout as part of the installation process, they are more susceptible to corrosion than other ground support elements (Dorion and Hadjigeorgiou, 2014). The use of corrosion inhibitors comes into play when it is necessary to prolong the effective life of a ground support element in a corrosive environment. Protective coatings and galvanization have been shown to be effective under certain conditions; however, there can be significant variations in performance between coatings. This was demonstrated by Hadjigeorgiou et al. (2019) in a long-term study of different coatings applied on expandable rock bolts in a corrosive environment. In addition, there can be significant variations in the level of corrosivity at different sites.
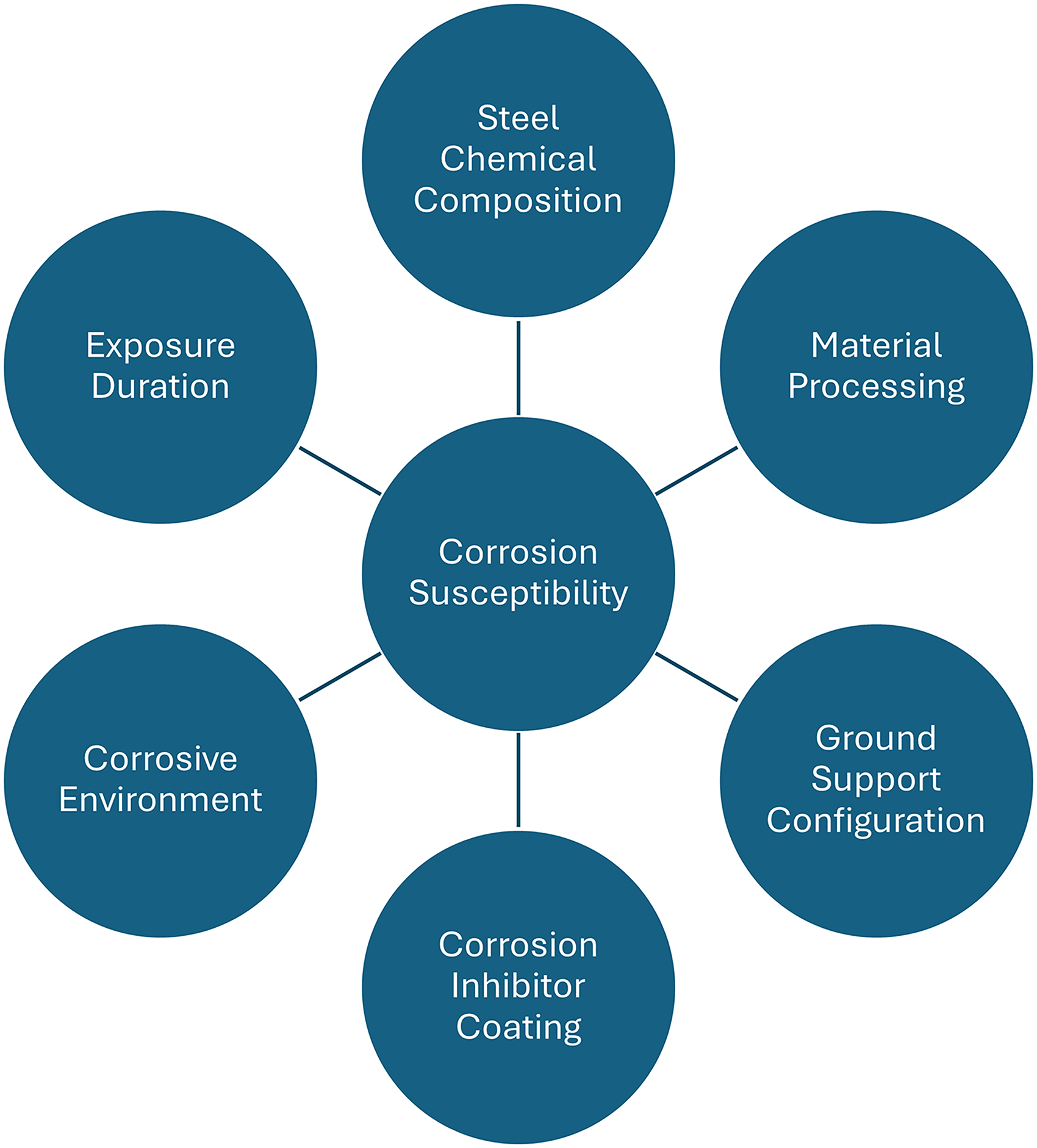
Factors that influence the susceptibility to corrosion of ground support elements.
Ground support in an underground mine can be exposed to both atmospheric and aqueous environments. Atmospheric corrosion occurs when a ground support element is exposed to air and pollutants. Typically, water vapour will condense onto the surface of the ground support and initiate a corrosion reaction. The resulting corrosion rate is accelerated by an elevated temperature, high relative humidity, and the presence of pollutants. Aqueous corrosion is associated with the presence of wet environments or groundwater. Due to a constant and/or periodic exposure to the solution, the aqueous environment is more corrosive to the ground support than the atmospheric environment.
Villaescusa et al. (2008) identified significant variations in susceptibility to corrosion in Western Australian mines compared to Eastern Australian mines. Similarly, the work of Vandermaat et al. (2016) provided examples of corrosive environments in Australian coal mines. Hadjigeorgiou et al. (2008) and Dorion and Hadjigeorgiou (2014) characterized corrosive environments in Canadian hard rock mines. In all cases, long-term exposure in a corrosive environment will result in degradation of ground support. In major infrastructure, such as a ramp, this may require excessive rehabilitation, and the susceptibility to corrosion should dictate the choice of reinforcement and surface support.
Low-carbon steel for ground support
Most ground support elements (rock bolts, mesh and straps) are made of low-alloy steel, containing between 1.0% and 5.0% alloying elements by mass. The alloying elements control the mechanical properties of the steel, manufactured through a hot-rolling process (Hadjigeorgiou, 2024). The chemical composition of steel controls its strength, hardenability, formability, weldability, ductility and susceptibility to corrosion (Hadjigeorgiou et al., 2020, 2023) (Table 1). Furthermore, the processing (including melting, cooling, and heat treatment) can change the grain structure and volume fraction of phases of the metal and influence its tensile strength, hardness and brittleness.
Advantages and limitations of specific elements in the chemical composition of low-carbon steel used for manufacturing rock bolts (Hadjigeorgiou et al., 2020).

Ground support manufacturers recognize that multiple combinations of alloying elements can be used to attain predefined mechanical performance requirements for steel components. It is therefore common to undertake trade-offs in the elements used to favour a particular mechanical property, for example, higher load capacity or elongation. The selection of a particular steel composition is often driven by supply chain constraints and efforts to minimize costs. The susceptibility to corrosion has not received the same level of scrutiny as establishing the mechanical properties of steel used to manufacture ground support elements.
Although steel used in ground support complies with some national and steel standard, e.g., ASTM International (ASTM; formerly American Society for Testing and Materials), European Standard (EN), National Standard of the People's Republic of China (GB), South African National Standard (SANS) and Canadian Standards Association (CSA), there are significant variations in these standards requiring strict diligent controls (Hadjigeorgiou, 2024).
Influence of alloying elements on corrosion
The addition of alloying elements to steel influences the corrosion performance of the steel material. A higher C content in steel produces more cementite precipitation, leading to microgalvanic corrosion with the ferrite phase (Hayden et al., 2022; Kadowaki et al., 2019). For example, the addition of Mn concentrations up to 5% to low-carbon steel resulted in a more active corrosion potential and a higher corrosion rate after 30 min of exposure to an acidic solution (Kim and Kim, 2015). Increasing P content from 0.01% to 0.10% by weight to low-carbon steel was associated with enhanced cathodic reaction kinetics and, consequently, an increase in the anodic corrosion current density (Kim et al., 2010). When the mass fraction of S increases, there will be a higher probability of having MnS inclusion, potentially leading to localized corrosion. The presence of chemical elements such as silicon, copper, nickel, aluminium and cobalt in the steel composition was reported to improve the corrosion resistance attributed to a more protective oxide layer that inhibited corrosion reactions on the surface of the steel (Jang et al., 2009; Jones, 1996; Kim et al., 2011; Sato et al., 2020). All these studies are examples of how individual chemical elements impact the corrosion performance of steel. However, the corrosion resistance of steel is attributable to the simultaneous combination of all chemical elements that influence the corrosion behaviour. It is also recognized that different corrosive environments will result in a variation in performance in steel of diverse chemical compositions.
Underground corrosive environments
As illustrated in Figure 2, the corrosion of ground support is a complex phenomenon, and several factors can influence the corrosion rate of ground support. This paper focuses on the influence of the chemical composition of ground support steel when exposed to an aggressive corrosive environment.
In general, the corrosion behaviour of ground support is controlled by three factors: the corrosion resistance of the ground support element, the corrosivity of the environment, and exposure time. The first factor is attributed to any characteristics inherent in the ground support element controlling its corrosion resistance and level of exposure to the environment. These include the chemical composition of steel, surface coating, heat treatment, and configuration or mechanical design. The corrosivity of the environment is characterized by the type of underground corrosive mine environments, temperature, pH, total dissolved solids (TDS), conductivity, presence of bacteria accelerating corrosion, etc. The exposure time represents the duration the ground support element is exposed to the underground mine environment. A longer exposure time will lead to more extensive corrosion on the ground support element.
Database
The authors have been investigating the corrosion of ground support for many years, including fracture analyses of failed elements (Hadjigeorgiou and Thorpe, 2020), the effectiveness of different coatings to mitigate corrosion (Hadjigeorgiou et al., 2019) and susceptibility to corrosion of different steel types (Hadjigeorgiou et al., 2020, 2023, 2024).
The results of accelerated corrosion tests on ground support steel were used to build the dataset for the statistical analysis presented in this paper. The ground support elements covered in this analysis include expandable bolts, friction rock stabilizers, meshes, mesh straps and mechanical hybrid rock bolts. The chemical composition of each element in the database was determined using inductively coupled plasma atomic emission spectroscopy (ICP-AES) and carbon and sulphur analyses in accordance with ASTM E1479-16 (2016) and ASTM E1019-11 (2011).
All accelerated corrosion tests were undertaken using the same ASTM procedures and using the same aggressive electrolyte. In addition, the chemical composition of all ground support elements was established in accordance with the ASTM standards. The chemical composition of all samples in the database was determined by the authors as opposed to using typical values provided by suppliers.
Accelerated corrosion
All accelerated corrosion tests were consistently performed using the same electrochemical techniques and lixiviates. The replicates were prepared using similar procedures. The chemical composition of the lixiviate for every test is summarized in Table 2, where the final pH was adjusted to 2.7 by using diluted sulphuric acid (H2SO4). Consequently, any variation in the electrochemical properties of ground support steel was attributed to the chemical composition of the steel components.
Chemical composition of the lixiviate for the accelerated corrosion test.

Methodology
Given the complex nature of corrosion, a statistical approach often serves as a useful tool for understanding the corrosion behaviour of steel material. Legault and Leckie (1974), using a stepwise regression analysis, proposed three multiple linear regression equations for three different atmospheric corrosive environments (industrial, semirural and marine) to explain the effects of the alloying elements in steel, consisting of copper, nickel, phosphorus and silicon, on the corrosion rate of low-alloy steel after 15.5 years of exposure. The proposed models showed a different role of each alloying element in determining the corrosion rate associated with a range of environments. The statistical analyses used experiment data on 270 low-alloy steel samples reported by Larrabee and Coburn (1962). Other statistical relationships between alloy composition in steel and corrosion loss associated with atmospheric corrosion were reported by Townsend (2001), in which the corrosion loss (μm) showed an exponential dependence with time for three different atmospheric environments. Different sets of alloying elements were found to be statistically significant in different environments.
This paper presents a statistical analysis of the relationship between the chemical composition of ground support steel and the electrochemical properties due to exposure to an acidic aqueous environment with a high concentration of salts. The proposed statistical relationships provide a better understanding of the role of each alloying element in the corrosion resistance of ground support steel. From a practical point of view, this will assist in a more proactive approach in selecting ground support products to be installed in a corrosive underground mine environment.
The range of specific chemical elements is explicitly stated in some standards for manufacturing a particular rock reinforcement. For example, ASTM A1011/A1011M-23 (2023) specifies the compliance requirements for the chemical composition of Grade 60 Class 2 steel used for friction rock stabilizers, pertaining to maximum carbon, sulphur, manganese, phosphorus, copper, nickel and chromium concentrations and minimum titanium content as a mass fraction.
While the effects of individual chemical elements on the corrosion performance of steel have been investigated, the corrosion resistance of steel is attributable to the simultaneous combination of all the chemical elements that influence the corrosion behaviour. Different characteristics of a corrosive environment will result in different corrosion performance between different steels of varying chemistry.
Statistical relationship between steel composition and electrochemical behaviour
Multiple linear regression aims to investigate the nature and degree of a relationship between a response variable (dependent variable) and multiple predictor variables (independent variables). The statistical relationship between variables is established by determining the coefficient of intercept (
In the undertaken statistical analysis, the predictor variables were the chemical composition of ground support steel determined using ICP-AES and carbon and sulphur analyses in accordance with ASTM E1479-16 and ASTM E1019-11, respectively. The response variable was selected from the electrochemical properties of ground support steel measured from accelerated corrosion testing.
In total, 15 datasets were gathered for the statistical analysis. Figure 3 shows the distributions of data values for the predictor variables, which consist of the mass fractions of the alloying elements summarized in box-and-whisker plots. In addition to the variability in the data and the average value, the plots contain information about the quartiles, outliers, skewness of the distribution, and maximum and minimum values that are not outliers. The data distribution of C, Mn, P, Al and Cr mass fractions is positively skewed, while the distribution of Si and Cu is skewed to the left. Some outliers were identified in Mn, S, Cu, Ni, Cr, V, Mo, Co and Ti datasets. The box-and-whisker plots for Mo, Co and Ti indicate that these elements were not found or below the detection limits for most rock bolt and mesh steel samples.
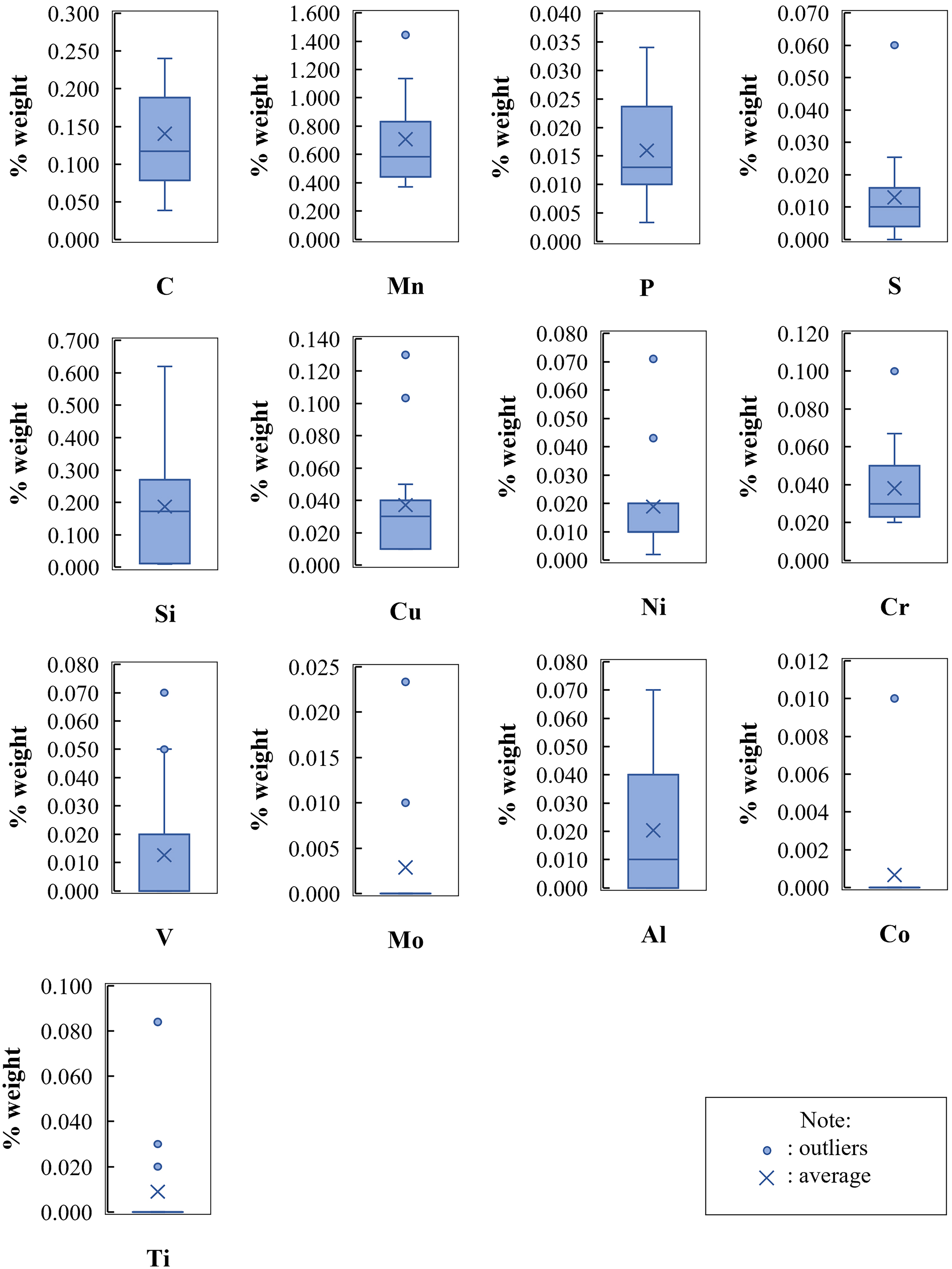
Distribution of chemical compositions in the datasets for the predictor variables.
A regression analysis was performed to derive a statistical relationship between the steel composition and the parameters from the accelerated corrosion test. This is summarized in Figure 4. Based on the results of the accelerated corrosion tests, the open-circuit potential (EOCP) and the polarization resistance (RP) were selected as the response variables. During the tests, both these properties were measured directly by the potentiostat and were not dependent on any formula or the concentration of the alloying elements. Moreover, as these two properties were measured in the first two sequences within the accelerated corrosion testing, they were not affected by the sequence of the electrochemical technique or the applied potential range to the electrode surface. Individual models for each response variable were fitted separately and assessed as to whether the statistical relationship was significant and whether the model had predictive power.
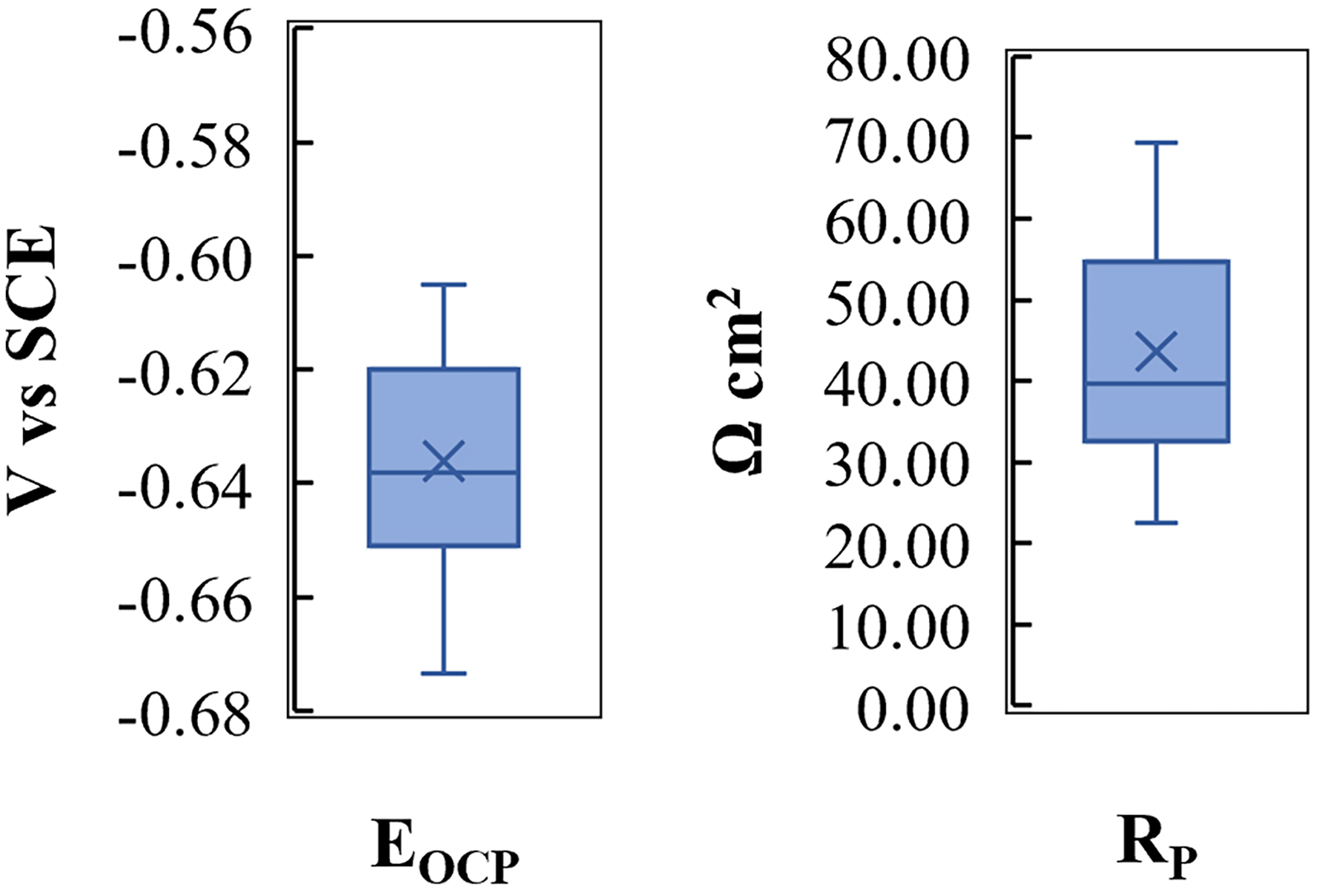
Distribution of the results of accelerated corrosion tests for candidates of the response variables.
Statistical modelling: forward stepwise selection method
Given the large number of predictor variables relative to the number of datasets, a stepwise regression using the forward selection method was used to fit the model. Instead of fitting the full model to the datasets, the forward selection method sequentially adds one predictor variable to the model by evaluating its statistical significance. The evaluation was focused on comparing the
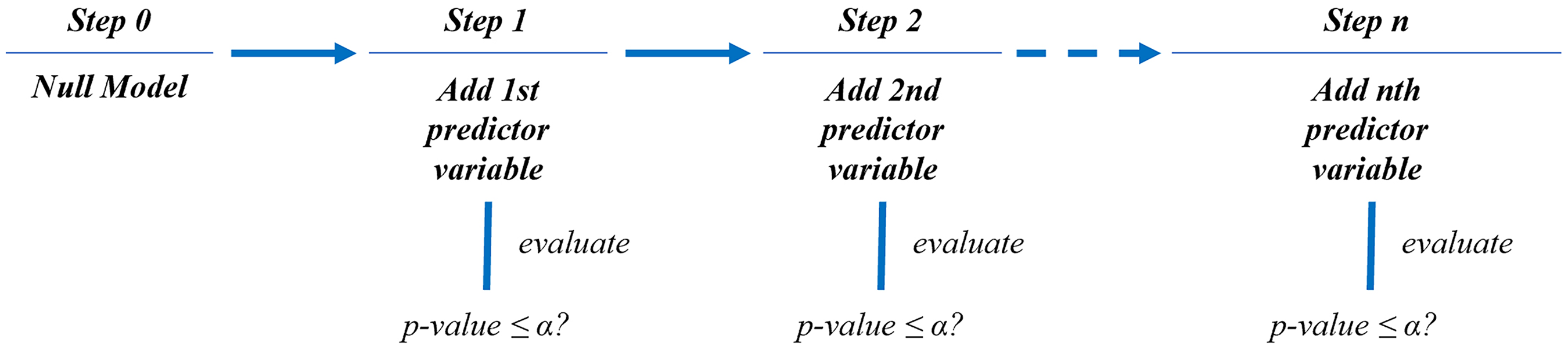
Simplified workflow for fitting a regression model using stepwise regression–forward selection method.
As there is no universally agreed
The regression analysis was used to obtain the ‘best’ model that can explain the variance in the response variable based on the variance in the selected predictor variables and has high predictive power. With the number of predictor variables available to fit the model, there is a higher chance that the model will be overfitting the data, especially at a high
Results
Based on the parametric study, the predicted
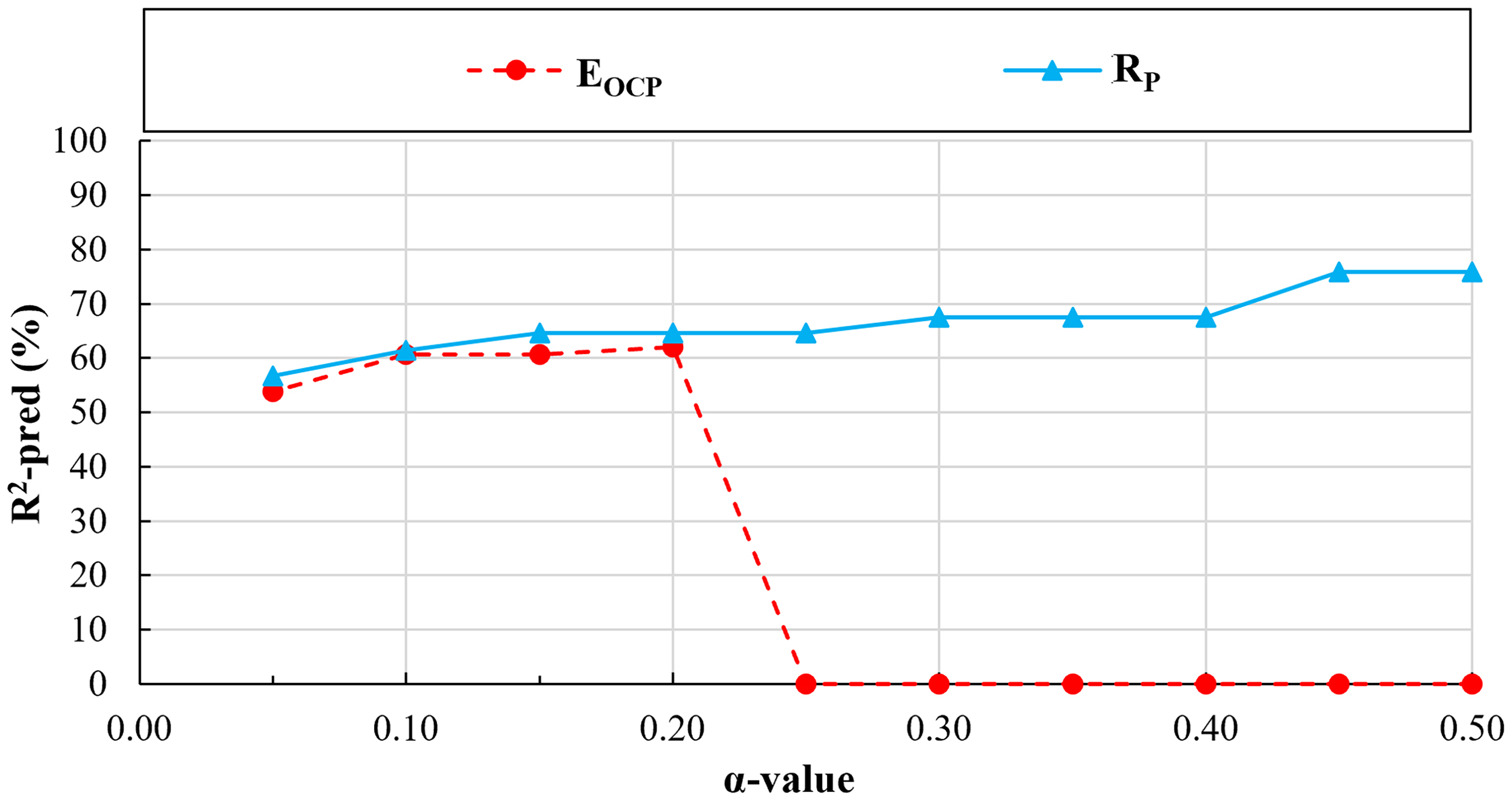
The results of parametric studies: predicted
The models for EOCP and RP are listed below in equations (1) and (2), respectively. The equation for EOCP includes six terms, two of which are interaction terms between Mn and V and between Mn and Ti. On the other hand, the model for RP includes only individual chemical elements without interaction terms. The quadratic term is not statistically significant in both models; thus, it is not included. The parameters indicating the performance of both models are summarized in Table 3. High coefficient-of-determination (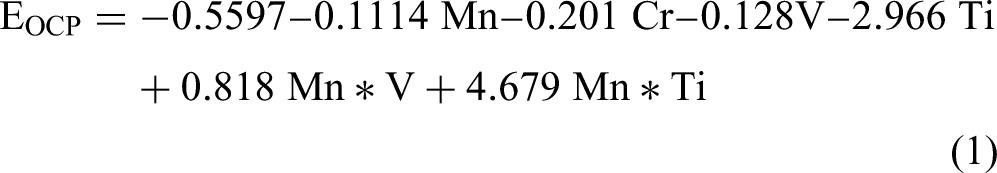
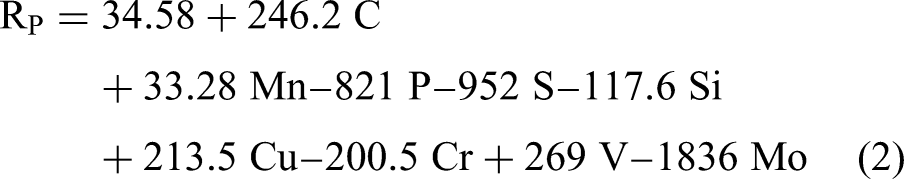
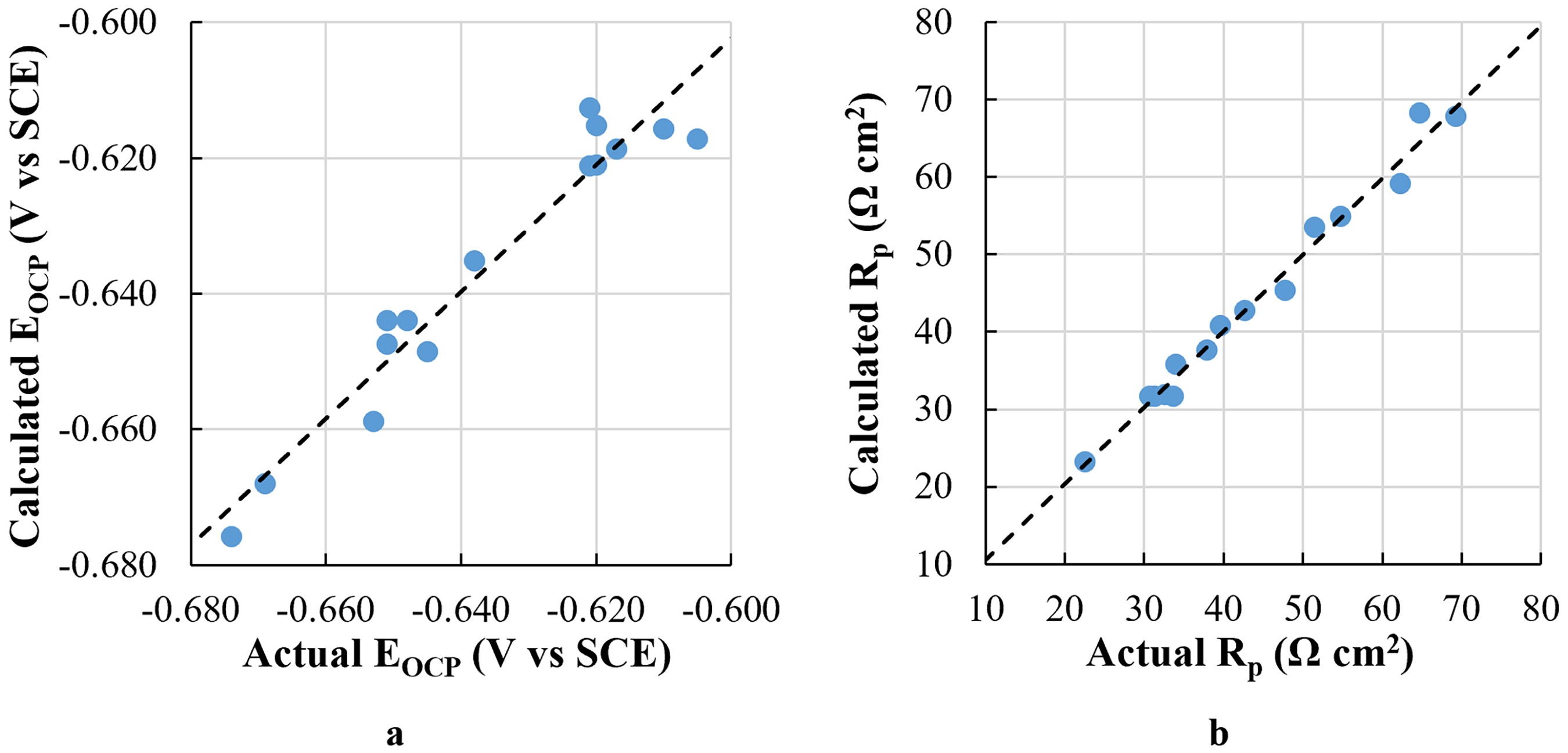
Correlation plots comparing the actual and calculated values using the regression equation: (a) EOCP and (b) RP.
Summary of the model's performance.

Tables 4 and 5 summarize the results of hypothesis testing (
Summary of hypothesis testing for regression coefficients in the EOCP model.

Summary of hypothesis testing for regression coefficients in the RP model.

The goodness of fit of both models was examined by evaluating the residual plots to determine whether the least-squares regression assumptions were met. The residual plots for both models (EOCP and RP) are shown in Figures 8 and 9, respectively. Regarding the assumption that the residuals are normally distributed, the quantile–quantile (Q–Q) plots for both models showed linear relationships between the normal theoretical quantiles and the data quantiles in terms of
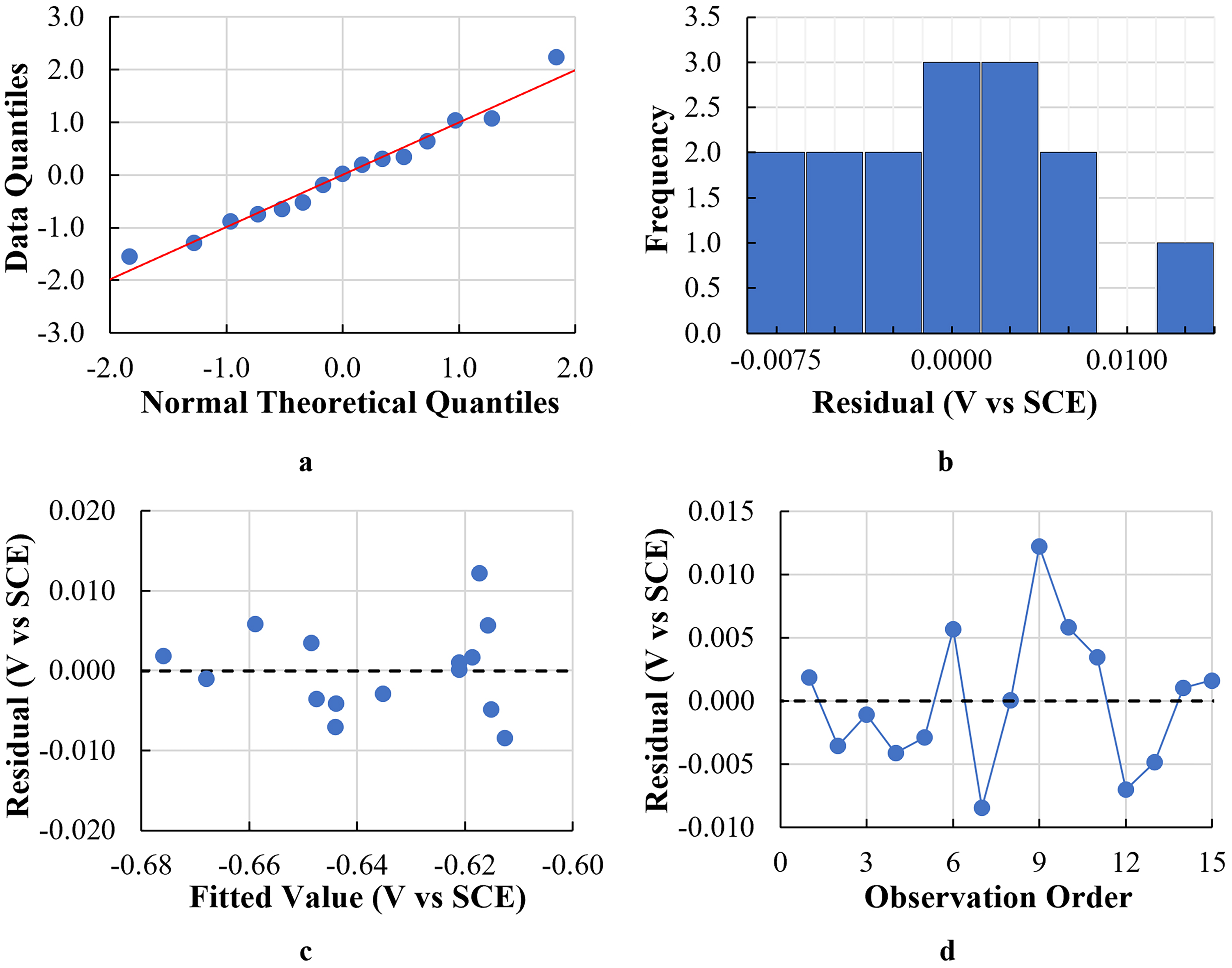
Residual plots for the EOCP model: (a) Q–Q plot. (b) Histogram of residuals (c) versus the fit plot and (d) versus the order plot.
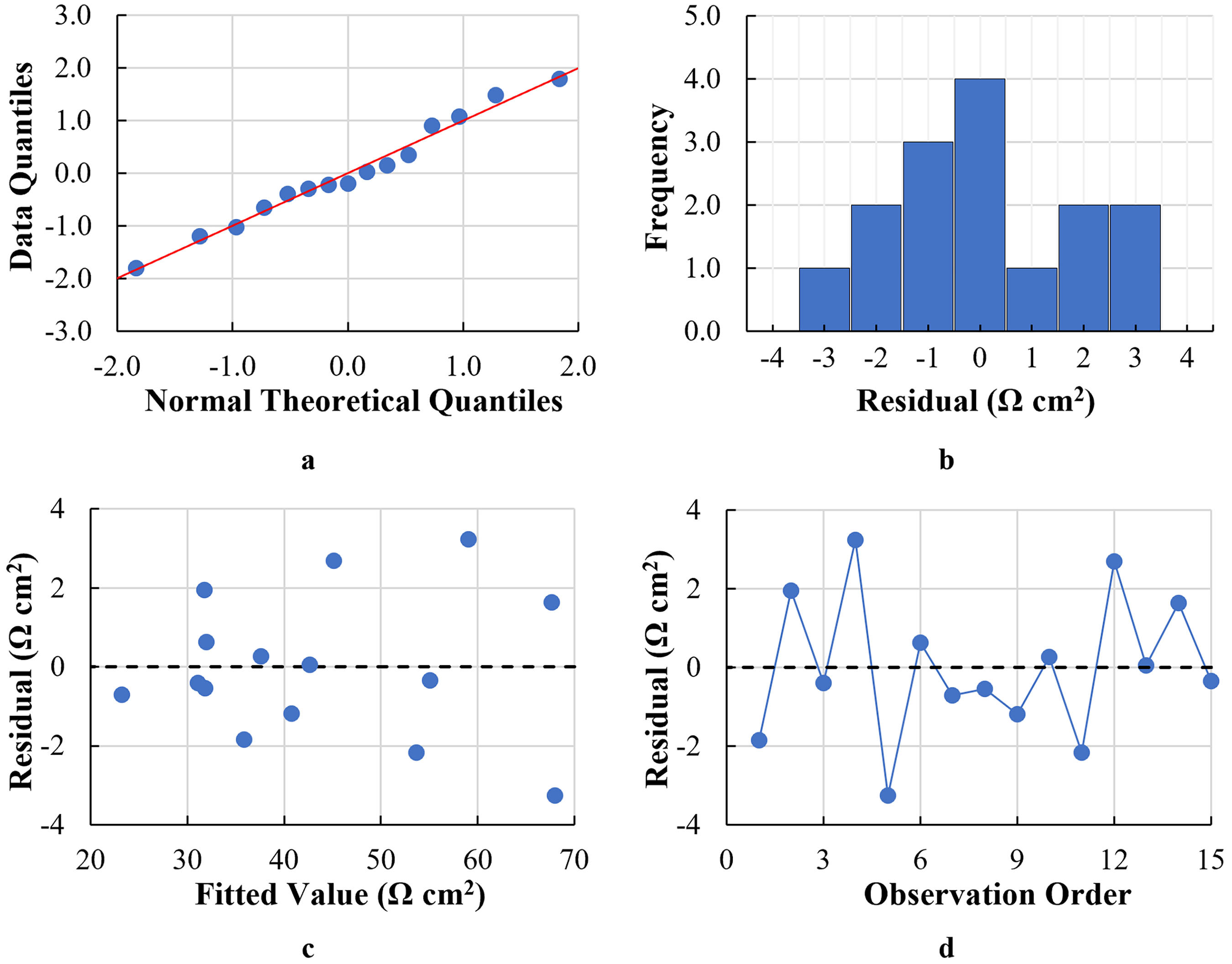
Residual plots for the RP model: (a) Q–Q plot. (b) Histogram of residuals (c) versus the fit plot and (d) versus the order plot.
Discussion
Two statistical models relating the open-circuit potential (EOCP) and the polarization resistance (RP) to the chemical compositions of ground support steel were obtained through multiple regression analysis using the forward stepwise selection method. Both models show a reasonable goodness of fit and predictive power, indicated by the
The corrosion process can be examined from both thermodynamic and kinetic perspectives that focus consecutively on the spontaneity of the corrosion reaction and the reaction rate. The open-circuit potential (EOCP) indicates the reactivity of the metal/alloy exposed to a corrosive environment. More noble materials with a high magnitude of EOCP are more corrosion resistant to specific environments than active materials with low or more negative EOCP. The polarization resistance is inversely related to the corrosion rate of metal/alloy. A material with a higher polarization resistance can be interpreted as a material with a lower corrosion rate near the EOCP.
The effects of chemical elements which are statistically significant on the EOCP and RP are presented in Figure 10. If there is no V and Ti in the steel, the presence and addition of Mn and Cr will make the steel more active with a more negative EOCP. This can be associated with a lower standard electrode potential (E0) of Mn and Cr relative to Fe, leading to microgalvanic couplings with the Fe. For the RP value, as the model suggested that there is no interaction among chemical elements, two groups of chemical elements with different effects on the RP can be clearly distinguished. Carbon, manganese, copper and vanadium have beneficial effects on corrosion performance. Conversely, the presence of phosphorus, sulphur and silicon is shown to be detrimental to the polarization resistance, leading to an increased corrosion rate. Although chromium and molybdenum are typically beneficial if partitioned into the film to convey passivity, in this work, they are tied up as alloy carbides that promote local galvanic corrosion cells.
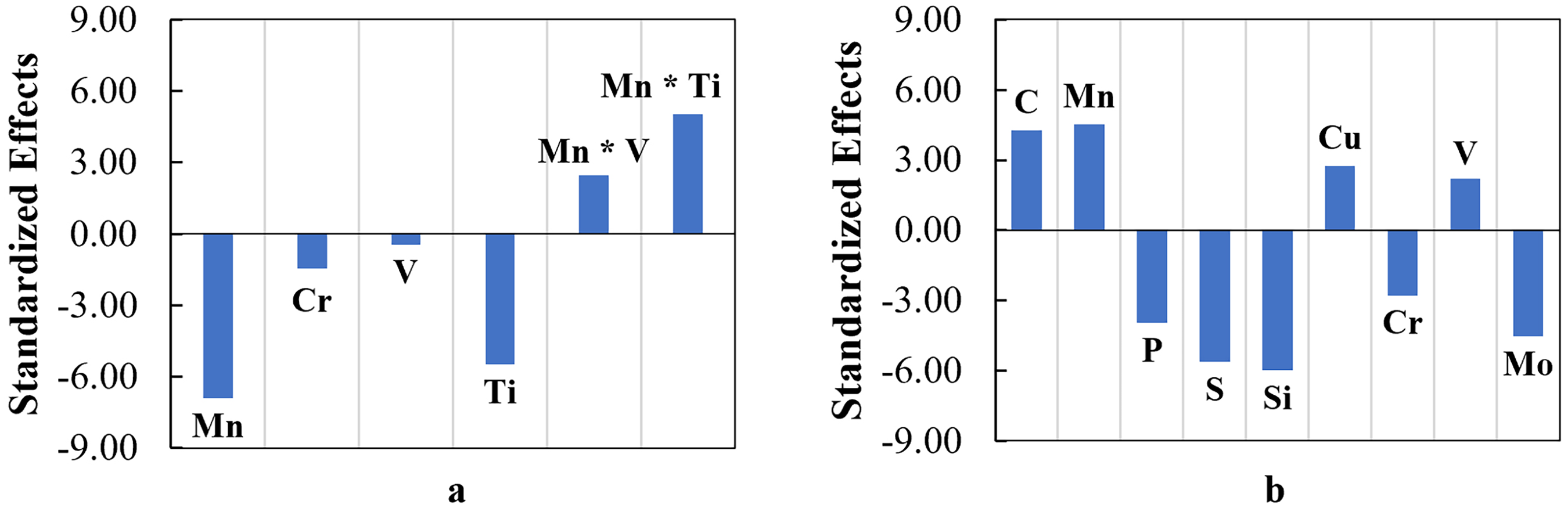
Standardized effects of terms representing chemical elements and/or interactions between mechanical elements on response variables: (a) EOCP and (b) RP.
Although the correlation between the alloying elements and the open-circuit potential (EOCP) is significant, it serves limited practical purposes as the range of the EOCP magnitudes among steel samples is only within −0.674 to −0.605 V(SCE). Considering the standard electrode potential of Fe is −0.440 V(SHE) or approximately −0.681 V(SCE) and by assuming that the cathodic reaction is only the hydrogen evolution reaction, the effect of alloying elements on the EOCP is negligible. The opposite condition was observed in the polarization resistance (RP). Even though the total mass fraction of the alloying elements is less than 5%, the variance in the RP value is significantly higher than that of the EOCP. It can be seen from the ratio of the highest to the lowest RP, which can reach up to 3. For this reason, an interpretation of the mechanisms associated with the alloying elements shown in Figure 10, leading to the reduction or increase of electrochemical properties, is focused on the polarization resistance (RP).
The beneficial effects of adding alloying elements such as Mn, Cu, and V to the corrosion resistance of steel exposed to an acidic solution were also reported by Kim and Kim (2015), Jang et al. (2009) and Yun et al. (2024). When Mn and Cu are added to steel, they can form a solid solution with Fe and, when oxidized, form a more protective layer on the corroded surface, leading to a reduction in the corrosion rate. Although the effect of increasing C contents on the RP seemingly implied that the relationship between these two variables is solely for curve-fitting purposes, the beneficial effect of C in lowering the corrosion rate agrees with the interpretation of the statistical relationship between the alloying elements and the corrosion rate by Townsend (2001). The addition of vanadium will reduce the precipitation of carbides from other stronger carbide-forming elements, thereby increasing the corrosion resistance of steel.
However, since the mass fraction of V is low compared to that of Cr and Mo, the chromium and molybdenum carbides are still present, especially when the C content is higher than that of Cr and Mo. Consequently, a microgalvanic coupling between these carbides and the matrix phase can lead to an elevated corrosion rate as reflected in Figure 10. Moreover, in a specific acidic corrosive environment, adding Cr and Mo to the steel can lead to a porous corrosion product, which is detrimental to the corrosion resistance of the steel (Kondo et al., 2023). The other negative influence of the addition or increase in the concentration of P, S and Si interpreted from the statistical analysis aligned with the findings reported by Kim et al. (2010) and Stern (1955). An increase in the mass fraction of P results in an elevated kinetics of the hydrogen evolution reaction, causing an increased corrosion rate of steel. The lowered polarization resistance (RP) due to the addition of sulphur content is related to the presence of sulphide inclusion in the matrix. Cleary and Greene (1967) also reported an increasing trend in the corrosion rate due to increased Si content in low-carbon steel.
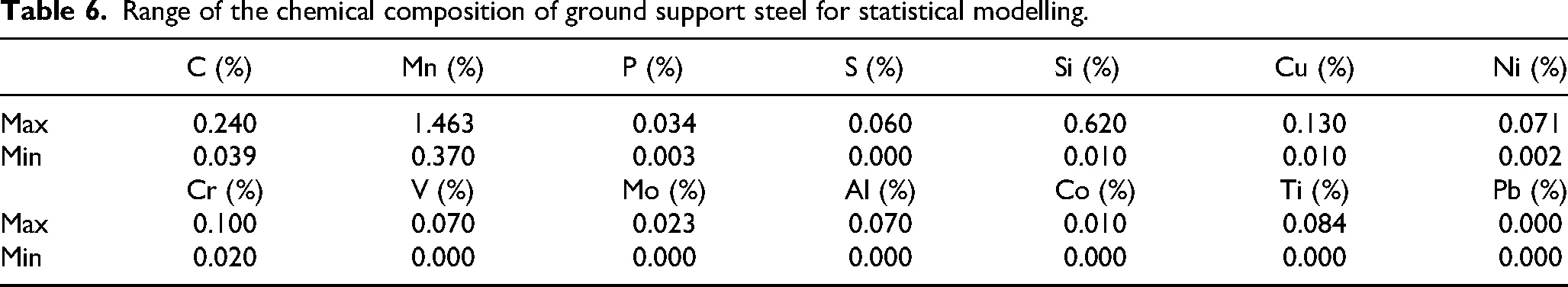
When the proposed regression equations for EOCP and RP are used to predict a new observation, an emphasis should be placed on the fundamental limitations attributed to the models/equations. First, the model was derived from a specific range of chemical compositions of steel, where the heat treatment process is not considered. The maximum and minimum mass fractions of each alloying element of ground support steels considered in the statistical modelling are summarized in Table 6. Second, the models are for the electrochemical properties of ground support steel measured under specific environmental (electrolyte) conditions. In this case, the steel was exposed to an acidic solution with a defined chemical composition. Moreover, the polarization resistance was determined near the open-circuit potential.
Range of the chemical composition of ground support steel for statistical modelling.
Conclusions
Understanding the effect of chemical composition on the corrosion performance of steel is critical when ground support is installed in a corrosive mine environment. Corrosion can cause degradation of ground support, potentially leading to a premature failure due to loss of dimensional properties of its component.
However, it is typically not feasible to predict the corrosion performance using steel's chemical composition due to the complexity associated with the effects of various alloying elements. It is exacerbated by the fact that different characteristics of corrosive environments, along with the exposure time, may cause different corrosion performances of ‘similar’ steel.
Statistical modelling was performed using a database of ground support steel's chemical compositions and associated electrochemical properties measured by the same test set-up, preparation and lixiviate. Ground support elements in this database included expandable rock bolts, friction rock stabilizers, mechanical hybrid rock bolts (steel tendon and friction unit), meshes and mesh straps. Two statistical models were derived to describe the relationship between the chemical composition of ground support steel and the corrosion performance represented by EOCP and RP. Both models can satisfactorily explain the variance in the datasets and have a reasonable predictive power.
In addition to the statistical analysis, the influence of specific chemical elements on corrosion performance was also discussed. Manganese and chromium will make the steel more active with a lower EOCP. On the other hand, C, Mn, Cu and V have beneficial effects on the polarization resistance (Rp), reducing the corrosion rate. In contrast, the detrimental effect on the Rp was associated with the presence of P, S, Si, Cr and Mo. Despite the predictive capability of both models in predicting EOCP and RP by using the chemical composition of steel that is not originally from the database, caution should be exercised due to the limitations of both models associated with the range of chemical composition in the database and the conditions and set-up used to measure the EOCP and RP.
This paper is based on the analysis of results from accelerated corrosion tests on steel used for rock bolts, mesh and mesh straps. Access to these unique test data has allowed us to explore the relations between material properties. The significance of this research is to provide guidelines to ground support suppliers in selecting appropriate steel that would mitigate corrosion-related degradation in performance. The results of this investigation can complement the current focus on selecting steel properties that would potentially meet a series of mechanical properties.
Footnotes
Acknowledgements
The accelerated corrosion database was constructed based on work at the University of Toronto over several years. The authors would like to acknowledge the mining companies and ground support suppliers who financed past and ongoing studies on the corrosion of ground support at the University of Toronto.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Natural Science and Engineering Research Council of Canada.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
