Abstract
The ammonium uranyl carbonate, (NH4)4UO2(CO3)3, is an important material used in UO2 and U3O8 ceramics production for the nuclear fuel fabrication. Thermal study and kinetic analysis of ammonium uranyl carbonate conversion under isothermal conditions has been studied in air atmosphere to obtain the tri-uranium octoxide (U3O8), using muffle furnace equipment, UV–visible spectrophotometer, gas adsorption, Hg porosimetry, laser granulometry, and optic spectroscopy. The textural properties (specific surface area, morphology, pore size, grain size, inter-particular porosity, and intra-particular porosity) and characteristics (uranium content and stoichiometry) of the prepared samples were estimated from the physical–chemical characterization. The kinetic parameters were estimated by a fitting of the experimental data. The activation energy
Introduction
This article is an extension of a previous paper where the ammonium uranyl carbonate (AUC) samples under non-isothermal conditions were decomposed and characterized. 1
Using modeling approach and under isothermal conditions, the kinetic parameters of the solid-state reactions (activation energy
The uranium oxide (UO2) is used as nuclear fuel in the nuclear fuel cycle. This choice is due to the chemical and physical properties of the UO2 at high temperature.
The characteristics of a UO2 and U3O8 powder influence the final properties of the UO2 sintered pellets.5–7
The irradiation behavior and the performance of the nuclear fuel are influenced by the characteristics and properties of the U3O8 powders and UO2 pellets.
The AUC is used in the production of UO2 powders destined for UO2 pellet fabrication of nuclear fuel. The ignition process of the AUC powder is one of the solid-state reaction mechanism involved in the nuclear fuel process.
The AUC powder precipitates from the uranyl nitrate solution or can be also precipitate in amine salt extraction from a uranyl sulfate solution. The UO2 powder resulting from these two ways will have different textural and structural properties.
In addition, the AUC decomposition conditions during the ignition process influence the U3O8 powder properties and the UO2 pressing and sintering processes.
The ignition process of the AUC occurs with a great evolution of gases (CO2 and NH3). The porosity and the pore structure of the products UO3 or U3O8 obtained after the decomposition of the AUC are conditioned by the form in which the gases CO2 and NH3 evolved. The porosity resulted from the AUC thermal decomposition will condition the sintering and the in-pile behaviors of UO2 pellets.
The AUC decomposition can be presented in the following reaction 1
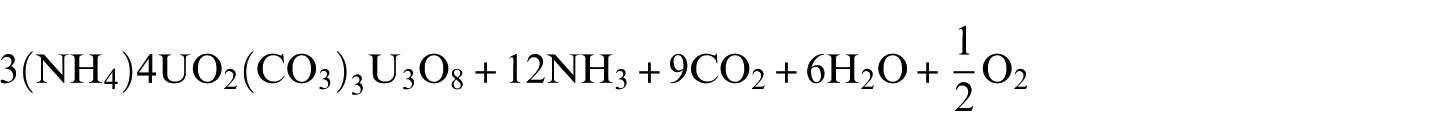
Several authors have studied the decomposition of uranyl carbonate or ammonium uranyl compounds. In our previous study, we gave a detailed description of previous work cited on literature on the isothermal and non-isothermal decomposition of AUC.8–14
In this work, isothermal decomposition of AUC compound has been examined using several characterization techniques. The purpose of this study is to carry out the uranium oxide powder characterization obtained from the isothermal decomposition of the AUC. Once the AUC is decomposed to U3O8 powder in air atmosphere, the textural properties of the intermediate and U3O8 phases have been studied by using UV–Visible spectrophotometer, gas adsorption (Barrett, Joyner and Halenda and Brunauer, Emmett and Teller), mercury porosimeter, laser granulometry, and optic spectroscopy methods. The effect of the ignition process is discussed in terms of the inter-granular porosity of the powder as measured by mercury intrusion porosimetry. The values of the kinetic parameters (activation energy
Kinetic modeling
Modeling approach
Kinetic studies of different uranyl compounds have been investigated by many researchers. Different models can be employed to study the kinetics of uranyl compound decomposition, which include nucleation models, geometrical contraction models, diffusion models, and reaction order models. These models of kinetic expressions have been employed to represent the kinetic of the solid-state reaction.3,4,5,15,16
Table 1 presents the summary of the various kinetic expressions used in our study, to treat the isothermal kinetics of the AUC decomposition.2,16
Kinetic model functions for treating the conversion

The resulting kinetic parameters of the AUC ignition process, from the application of various model equations, are used to interpret the reaction mechanism. The variation of activation energy
Isothermal kinetic methods
Two methods are used to analyze the isothermal kinetic data: the model-free (iso-conversional) methods and the model-fitting (conventional) methods.
Model-free/standard iso-conversional methods
Model-free methods calculate the reaction activation energy (
This method can be derived from equation (1)2,3,4

Linearization of equation (1) gives

Equation (2) can be reorganized to give

A plot of −ln
More details about the calculation method of conversion fraction (
Isothermal model-fitting methods (conventional method)
In order to analyze the isothermal kinetic data from the conventional method, two fits are involved:
The first determines the rate constant (

The second fit determines kinetic parameters

The slope from this second fit gives the reaction rate constant (
Materials and methods
The AUC commercial grade material powder was used in our study. To carry out the isothermal AUC decomposition experiments, a muffle furnace (carbolite) was used. The samples about 10 g were heated, in air atmosphere, in an open cylindrical platinum crucible of 30-mm diameter and 45-mm height. The temperatures ranging from 100°C to 900°C were selected for the experiments.
To investigate the textural properties of samples, the following methods were used: laser granulometer analyzer (Mastersizer) to measure the average and the distribution of the particle size; the BET method using ASAP surface area analyzer (Micromeritics) to measure the specific surface area; and the mercury porosimetry (Micromeritics) to measure the porosity. The method description to evaluate the N2 adsorption isotherm measurements, the evolution of porosity and specific surface area of the samples, porosity, and pore size distribution of samples has been shown elsewhere. 17 The UV–visible spectrophotometer was used to calculate the stoichiometry of the obtained uranium oxide powders (O/U ratio). 18 The measurements were performed in triplicate, and each result presents the mean ± standard deviation (SD).
Results and discussion
Characterization of the raw and treated AUC samples
The results of the chemical and physical characterization of the used AUC powder (Figure 1) have been given elsewhere. 1

Samples of AUC and U3O8 powders.
Optic microscope photographs of the AUC and U3O8 samples are shown in Figure 2. In this figure, it can be observed that the original AUC powder (Figure 2(a)) is presented in the agglomerates and heterogeneous form. After the isothermal decomposition (ignition process), the AUC powder becomes more homogeny with the spherical shape. We note that the particles of AUC powder are less agglomerated after ignition (Figure 2(b)), then the original AUC (Figure 2(a)).

(a) Original AUC sample and (b) U3O8 sample, AUC treated at 500°C.
The particle size and pore size distribution have an effect on the UO2 and U3O8 powder properties. Using the principle of laser diffraction, the mean diameter of AUC and U3O8 samples were found to be 44 ± 2 µm and 25 ± 2 µm, respectively.
The general shape of the sample’s N2 adsorption–desorption isotherms of AUC and U3O8 did not change, showing just an increase in the amount of nitrogen adsorbed for U3O8 sample. The dominant pore for AUC and U3O8 samples occurs in the diameter ranges from 3 to 6 nm and 4 to 25 nm, respectively. The AUC and U3O8 samples have pore volumes of 0.001 and 0.020 cm3 g−1, respectively. The AUC sample exhibits a narrower pore distribution; this shows that the isothermal decomposition makes the pore size distribution wider for U3O8 sample than AUC sample.
Results of the UV–visible spectrophotometry analysis of the final oxide confirmed that the end product of AUC ignition process under isothermal conditions at temperature value
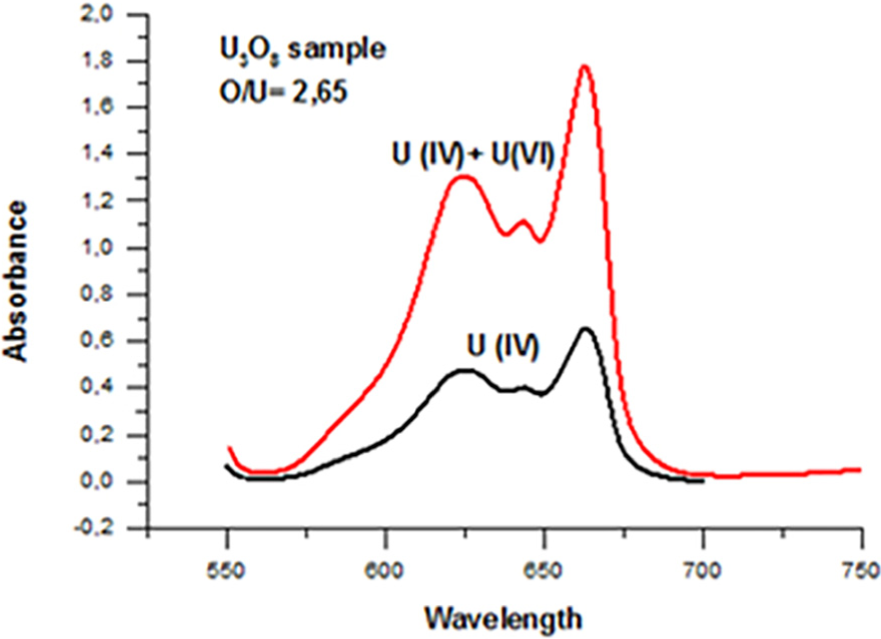
O/U ratio results of the various uranium oxides obtained by UV–visible spectrophotometry.
The specific surface area and pore distribution of AUC and tri-uranium octoxide samples obtained from the AUC decomposition as a function of temperature have been studied. The specific surface area, pore volume and pore diameter evolution of uranium oxide (intermediate phase), and tri-uranium octoxide as a function of temperature have also been studied. The dependence of these parameters on the isothermal decomposition temperature can be seen.
The plots of mercury penetration volume against absolute Hg intrusion pressure for the AUC and U3O8 samples show that the pressure up to 10 atm. represents the filling of macrovoids in the studied samples. It can be seen that the cumulative mercury intrusion volume of the AUC sample is higher than the U3O8 sample, indicating that isothermal treatment decreases the pore volume.
The porosity distribution results of the AUC and U3O8 powders, as obtained with intrusion mercury porosimetry, show the dominant pore size of 5–40 µm for AUC sample and 5–12 µm and 0.1–1 µm for U3O8 sample. The AUC powder is mono-modal, and the U3O8 powder is bi-modals. In the case of U3O8 powder, the first step is due to the inter-particular pores (range from 5 to 12 µm of pore size) and the second to the intra-particular pores (range from 0.1 to 1 µm of pore size). This porous structure observed during the ignition process of the AUC particle is produced by the gases generated during its decomposition.
Comparison of pore size distributions by mercury immersion between the AUC and the U3O8 powders shows that the intra-particular porosity appears in the U3O8 oxide (region from 0.1 to 1 µm). The porosity distribution of U3O8 is displaced to lower pore size and has lower range, indicating that the low porosity detected after decomposition of AUC sample is due to the internal porosity of the U3O8 powder. The observed shift of porosity distribution toward lower pore size, in the case of the U3O8 sample, is produced by the breaking of the intra-particular pores of the AUC during the ignition process.
Isothermal kinetic study
The plots of
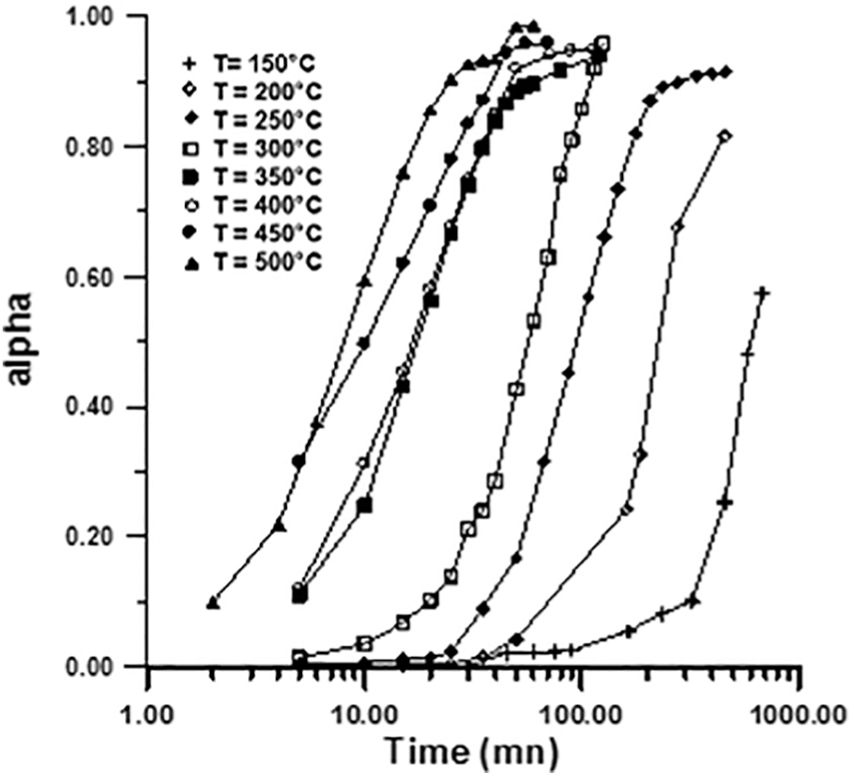
Fraction reacted (
Reaction rate of AUC isothermal decomposition
Figure 5 presents the reaction rate evolution for the isothermal decomposition of AUC powder versus the conversion rate
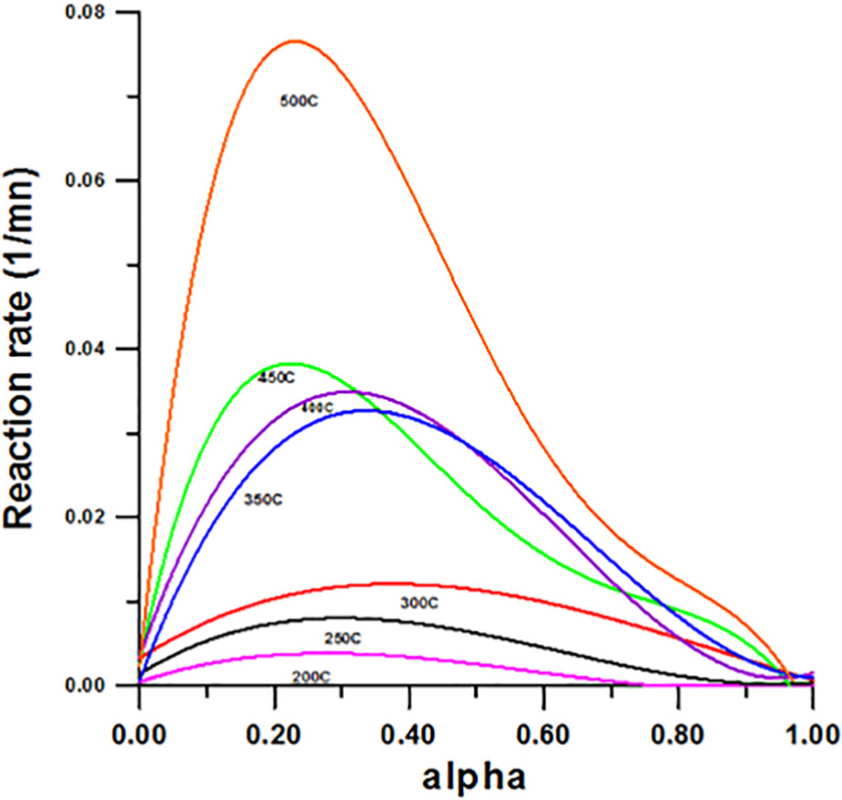
Reaction rate (d
Evaluation of kinetic parameters using iso-conversional and conventional methods
The kinetic parameters were determined from the isothermal curves, by using the conventional and iso-conversional (standard) methods.
Iso-conversional method
The activation energies
The
Activation energy (kJ mol−1) values obtained by iso-conversional method at various
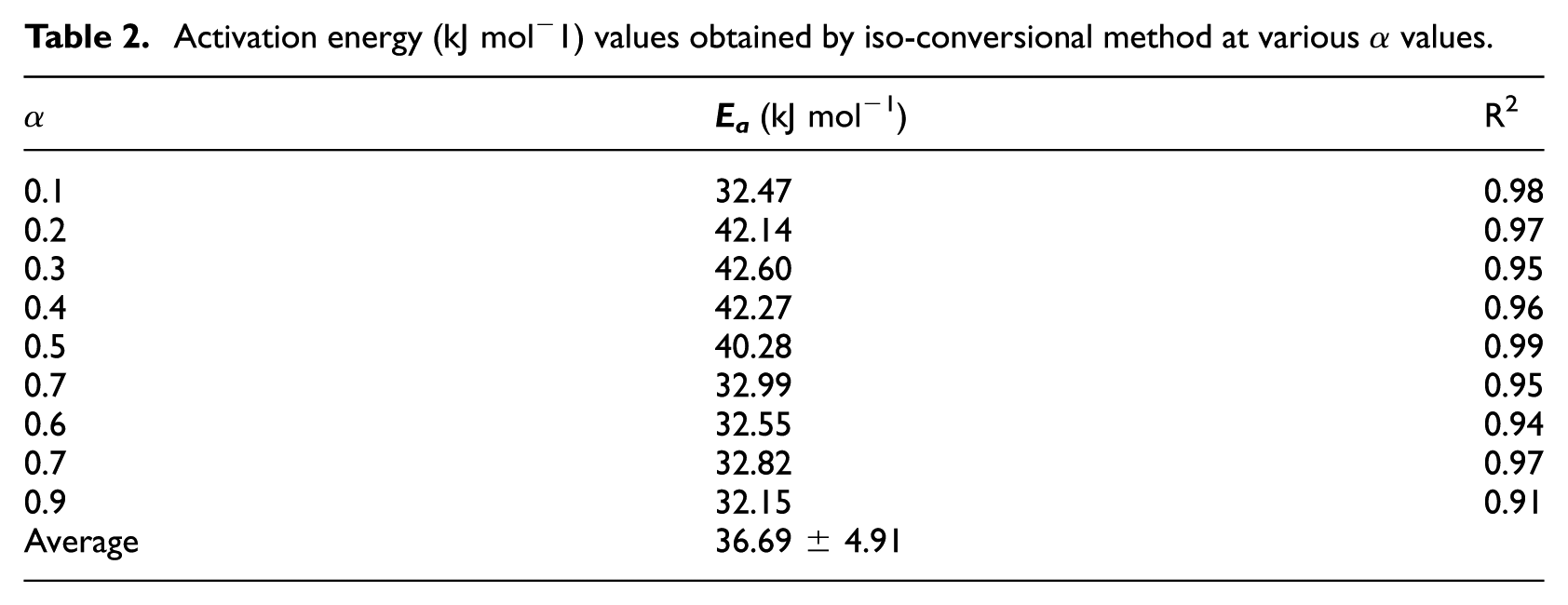
From Table 2, it can be seen that the activation energy obtained with the iso-conversional method varies from 32.15 kJ mol−1 (
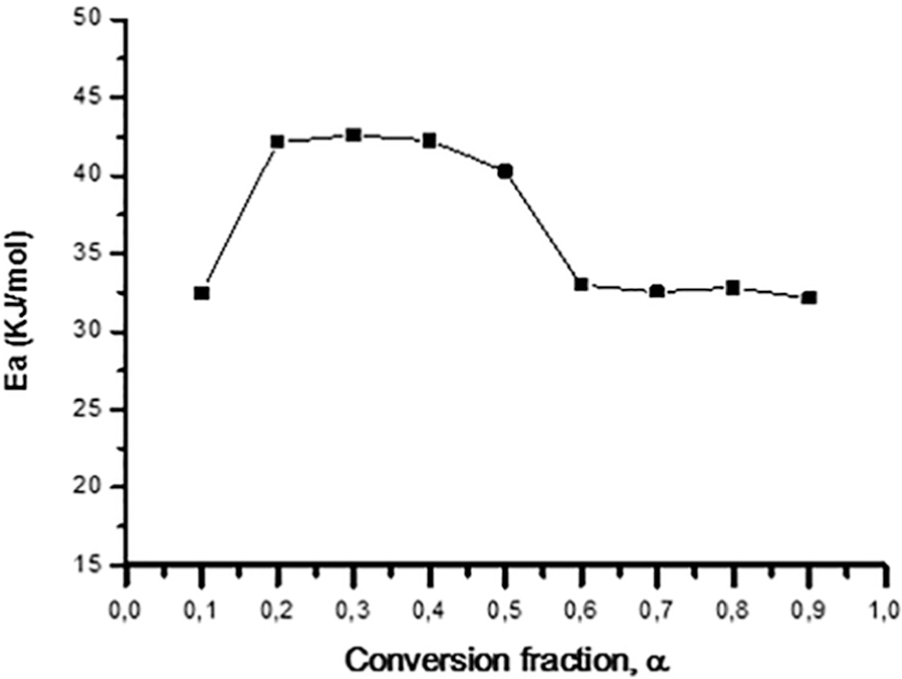
Activation energy as a function of conversion fraction for AUC decomposition usingiso-conversional method.
Baldea et al.
19
have studied the thermal decomposition of ammonium uranium fluoride in the temperature range of 250°C–500°C using the Arrhenius relation. The activation energy value 43.67 kJ mol−1 was obtained; this value is very close to the
Conventional model-fitting method
The
The choice of the kinetic equation which describes the reaction mechanism of the AUC isothermal decomposition is done after the verification of several possible kinetic equations (Table 1). The kinetic parameter data have been calculated using the conventional model-fitting method. In order to estimate the pre-exponential factor, the kinetic equation function nucleation models (P2, P3, P4, P3/2, A2, A3, and A4), geometrical contraction models (R2 and R3), diffusion models (D1, D3 and D4), and reaction order models (F1, F2, F3, and F4) were tested to simulate the isothermal decomposition mechanism of AUC (Tab. 1).
The reaction rate constants
Reaction rate constants (−ln
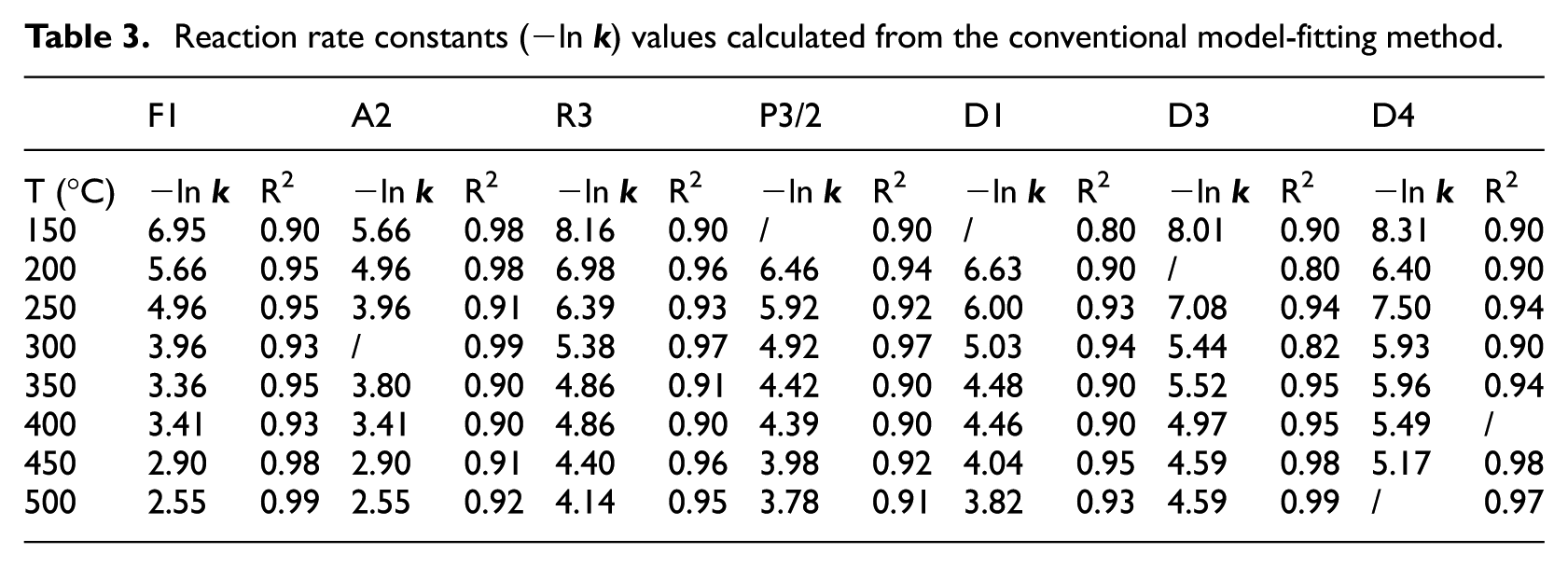
R2 values calculated according to the experimental values from plotting revealed that the decomposition process is an Avrami-Erofeev (A2) kinetic (R2 = 0.999).
The pre-exponential factor
Kinetics parameters for isothermal data using model fitting.
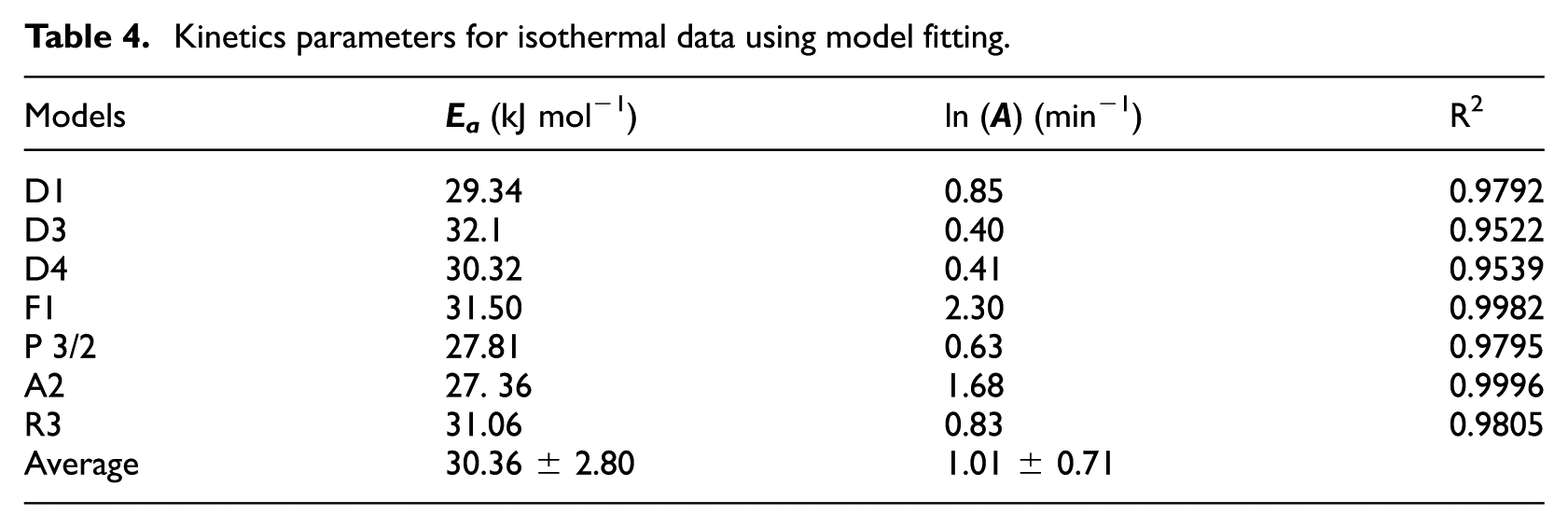
Model-fitting results agreed with those obtained from the Avrami-Erofeev A2 model, compared to the others models (F1, A2, R3, P 3/2, D1, D3, and D4). As Table 4 shows, all the models of reaction have a coefficient of correlation higher than 0.95. Using Avrami-Erofeev A2 model, the activation energy
Monshi et al.
20
have investigated the isothermal decomposition of un-irradiated and pre-γ-irradiated dehydrated uranyl acetate at different temperatures between 573 and 593 K. The activation energy values were found to be
Conclusion
This study confirms that AUC is decomposed to the tri-uranium octoxide (U3O8) at
Using model-free (iso-conversional) method, kinetic parameters were estimated at different conversion fractions (
Using model-fitting (conventional) method, we noticed the Avrami-Erofeev A2 model that correlates well with our experimental data. We conclude that the Avrami-Erofeev A2 model characterizes and explains the kinetics of AUC isothermal ignition process reaction. The kinetic parameters were estimated at different conversion fractions (
Using the model-fitting method, the nucleation and growth model (Avrami-Erofeev A2) can be successfully applied to the kinetics of AUC isothermal decomposition reaction, and the activation energy is found to be 27.36 kJ mol−1, using the AUC powder with 44-µm mean size fraction.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
