Abstract
Blast furnace (BF) operation depends on the smooth upward movement of gas and its pressure drop. The slag properties like viscosity affects the gas flow inside the furnace. It is well-known that Al2O3 contributes to the increase of slag viscosity. For smooth BF operation, the maximum limit of Al2O3 in the Indian BF slag is around 18.5%. Higher Al2O3 contents make tapping of the BF slag difficult. In the present work, FACTSAGE calculations were carried out to estimate the viscosity and melting temperature of 20% Al2O3 slag varying MgO% between 7 and 13 and basicity (B2, defined as wt% CaO/wt % SiO2) between 0.9 and 1.2 to find an optimum MgO% and basicity for easy tapping of the slag. The results of the calculations provide three combinations of MgO (12,12.5,13%) and B2 (0.95) of slag for operating 20% Al2O3 in slag which were validated through experimental studies involves the inclined plane test, hot stage microscopy and X-ray diffraction.
Introduction
Over the last decade, hot metal production in India has increased from 39 to 73 Mt, and consequently, the consumption of iron ore has increased by 86%. 1 The increased production of hot metal has forced steelmakers to resort to low-quality iron ore, with a higher alumina (Al2O3) content. As a result, alumina input to the blast furnace (BF) has increased from 52 to 62 kg/thm. 2 Higher Al2O3 content in the BF slag increases the viscosity and liquidus temperature, which, in turn, would cause difficulty during the taping of BF slag.
The physical properties of BF slag, that is, viscosity, surface tension etc., and chemical properties like liquidus temperature (melting temperature of slag) determine the slag flow which would affect the smooth BF operation. The viscosity and liquidus temperature of the slag affect the upward flow of gas. Reduced permeability of solid bed due to higher viscosity of the slag would result in increased resistance to flow of gas and liquid inside the furnace.3,4 The temperature at which molten slag forms is another important factor to be considered. It is imperative that the slag should reach the melting point as early as possible for easy flowability inside the furnace. Higher melting point would imply more coke consumption in melting the slag in the lower part of the furnace shaft. To compensate for these effects, the coke rate and flux rate need to be increased to lower the viscosity and increase the flowability of the high alumina slag which would lead to a drop in the overall productivity of the BF. The term “Flowability” is defined in the present work as the ease of flow of a slag under fast cooling. This is considered different from the fluidity which is defined as the reciprocal of viscosity of a Newtonian liquid.
Substantial research has been carried out to mitigate the negative effects of excess alumina in BF with the inclusion of MgO in the BF slag.5–8 Earlier studies by Zhang et al. show that the maximum permissible amount of Al2O3 for BF is approximately 19% with MgO/Al2O3 ratio in the range of 0.65–0.7 and MgO (12.35–13.3%). 9 However, Shiau et Al. have suggested maintaining of MgO content of 5.4% in the slag for 15% Al2O3 for attaining lower slag viscosity and liquidus temperature. 10 Increase of MgO content to 9–11% has been reported to diminish the detrimental effect of high alumina (15–20%) in BF. 11 A subsequent increase of MgO content (11–12%) leads to an increase in the primary spinel phase formation in higher alumina slag thus raising the viscosity. 12
Several viscosity models 13 have been developed to predict the viscosity of high alumina slag in BF. Experimental studies have also been carried out to explore the inclusion of other additives to reduce the viscosity of a high Alumina slag.14,15 It is generally known that an increase in both MgO and basicity results in depolymerisation of the structure of aluminosilicate thereby reducing the viscosity of BF slag. Indian BF operation is carried out with a slag composition of Al2O3: 18.5%, B2[CaO /SiO2]:1 and MgO: 9.5%. Further addition of MgO would lead to an increase in the unit cost of hot metal production. One way to reduce the viscosity of the high alumina slag is to maintain a stable MgO composition in the existing slag. It is necessary to optimise the slag composition so that the slag properties of the existing slag are retained with minimum addition of MgO. However, the existing literature/research are somewhat unclear regarding the combined effect of basicity(B2) and MgO on the liquidus temperature and viscosity of slag containing 20% Al2O3.
In this context, the objective of this work is to determine an optimum range of MgO and B2 so that the physiochemical properties of the slag with increased Al2O3 to 20% would be comparable to that of the slag containing 18.5% Al2O3. The effects of MgO and B2 on the liquidus temperature and viscosity of a slag system (CaO-SiO2-MgO-20% Al2O3) have been studied. It is to be noted that, in the current work, basicity is defined as wt%CaO/wt% SiO2. The choice of this definition was to enable studying the effects of MgO and Al2O3, so that the effects are not superimposed if these components are included in the definition of basicity.
Methodology
In the current study, physicochemical properties such as melting, viscosity, and phase analysis of the BF slag were examined. Thermodynamic calculation using FACTSAGE were performed to estimate the chemical properties and viscosity. Flowability of the slag was measured using inclined plane test. Hot stage microscopy and X-ray diffraction (XRD) were used to validate the FACTSAGE results.
Simulation
FACTSAGE Software Version 8.1 was used to calculate the liquidus temperature, viscosity, and equilibrium phase analysis. FT oxide database was used for all the calculations. Various calculations were performed to optimise the BF slag properties. In Indian Steel industry, the average BF slag composition observed is 18.5% Al2O3, 9.5% MgO, and CaO/SiO2 =1; this composition is considered as reference composition for the current work. The major components in the BF slag are CaO, SiO2, Al2O3, MgO and the summation of these four components around 97% of BF slag. The FeO content is however quite low, 0.2–0.45% and thus may not have any significant impact on the slag properties. Since the properties of the reference composition are well suitable for the BF operation, the properties of the synthetic slags were studied in the present work with reference slag as the benchmark.
The aim of the present study is to investigate the impact of MgO and basicity on the slag properties affected by the adverse effect of alumina increase in the slag. The MgO content was varied between 7% and 13% and the basicity was toggled between 0.9 and 1.2. In the mentioned range of the MgO and basicity values, compositions were identified that have the same properties as reference composition (slag containing 18.5% Al2O3, MgO: 9.5%, and basicity 1). In the present study, a case has been presented where the Al2O3 in the slag is 20% and, at the same time, an optimum combination of the MgO and basicity has been obtained for similar performance in BF through thermodynamic calculation.
Experimental
For BF operation, the flowability (as defined in the present work) of the slag is important. Therefore, considering the practical importance, inclined plane test method was adopted to measure this parameter. Standard dimensions and experiment conditions were followed for the flow measurement as mentioned in the Slag Atlas.16,17 Hot stage microscope technique was used to study the melting behaviour of the slag.
The experiments were performed with synthetically prepared slags; 100g of each slag was prepared using laboratory grade reagents: CaO, Al2O3, MgO, SiO2. The compositions of the slags studied are listed in Table 1. The minor constituents in the reference slag,
Chemical composition of slag based on 97% constituent of BF slag (in wt.%).

The reagents were weighed (according to Table 1), mixed properly and normalised to 100% as stated in Table 2. The samples were heated using graphite crucibles in a muffle furnace to 1550 °C and held at that temperature for a time duration of 45 min in order to ensure complete melting and homogenisation of the slag. The flowability of the slag was measured using Inclined Plane Test Arrangement.12,13 The experimental set-up for measuring the flow length of the slag using this unit is shown in Figure 1.

Chemical composition of slag normalised to 100% of BF slag (in wt.%).

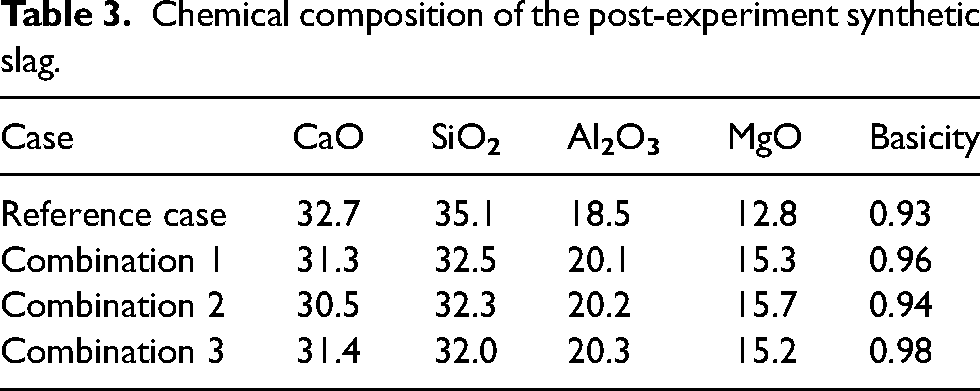
This arrangement allows the molten slag samples to be poured onto an inclined plane in air at atmospheric pressure. In view of the human factor involved in the pouring of the slag on to the inclined plane, efforts were taken to repeat the experiments in an identical manner. The pouring method of the slag was standardised,16,17 strictly following the procedure given in Slag Atlas16,17 so that the time taken to pour on the inclined plane was the almost same in each experiment. The slag flowed along the length of plane and the solidified length of the slag was measured. The length of the solidified slag was correlated with the flowability of the slag, which could be related to slag viscosity as viscous slag will flow at slower rate. The inclined plane test was carried out six times for each composition to ensure repeatability of the results. The post-experiment slag was pulverised to powder size of less than 200 μm to determine the phases. X-ray diffractometer of XPERT-PRO, PANalytical system was used. The chemical composition was determined using XRF spectroscopy of ThermoFischer Scientific and are presented in Table 3. The melting point of slag was measured using a Hot-stage Microscope with Automatic Image Analyzer of Hesse Instruments.
Results and discussion
Chemical composition of the post-experiment synthetic slag.
Simulation results
FACTSAGE 8.1 was used to calculate the liquidus temperatures and viscosities of all the slags corresponding to pre-experiment compositions. For all thermodynamic calculations, FT oxide database was used. The optimum value of the MgO and basicity to operate with 20% Al2O3 in the slag having the liquidus temperature and viscosity which are less than or equal to the corresponding property of the reference composition was calculated and presented in Table 4.
Optimum amount of MgO and basicity in slag studied as compared to reference slag.

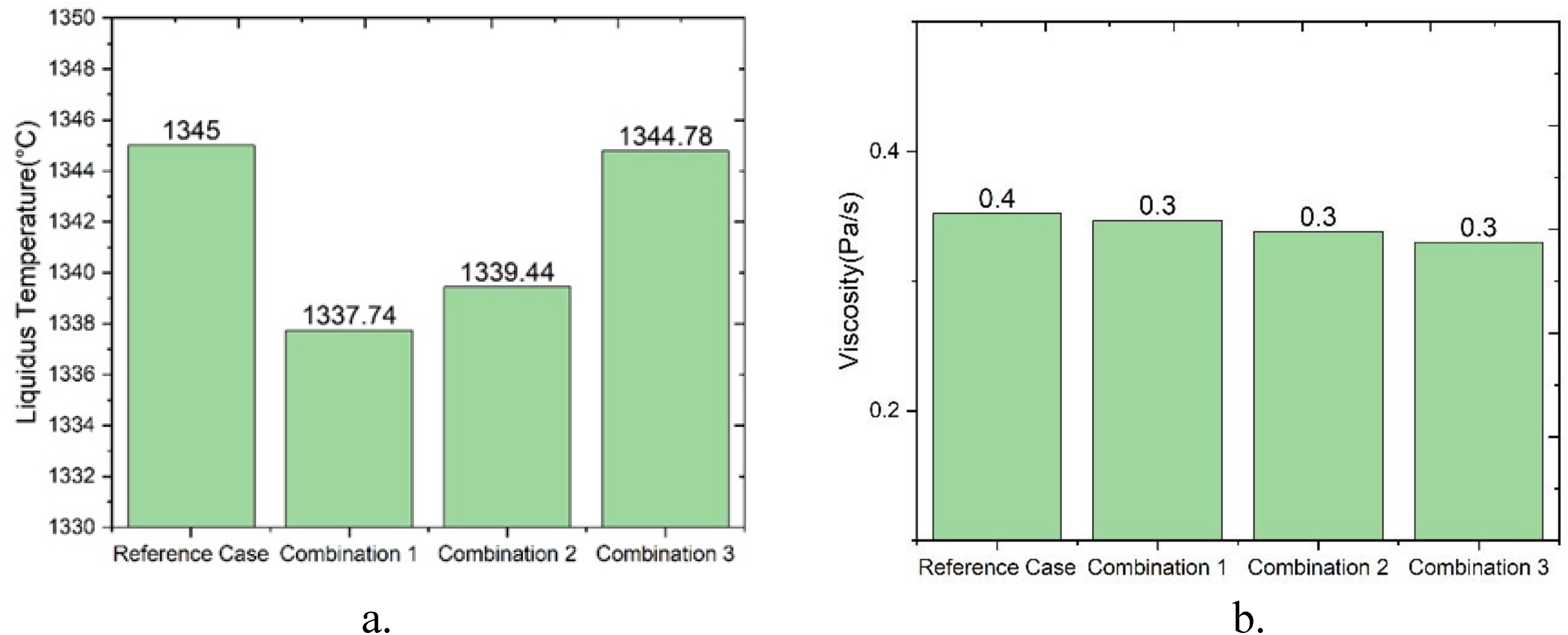
It is evident from Table 4 that there are three possible combinations of MgO and basicity, for slag having 20% Al2O3, with the liquidous temperature and viscosity in the same range as the reference case. In all the three combinations, the MgO varies between 12% and 13%, whereas the B2 is 0.95. The effect of MgO on the liquidus temperature and viscosity for the slag 20% Al2O3 is illustrated in Figure 2. The viscosity value calculated at 1550 °C from FACTSAGE shows very little drop in viscosity (0.017 Pa/s) as compared to the reference slag. The liquidus temperature shows a decrease of about 7 °C for combination 1 increasing to nearly the same value as the reference slag with combination 3.
To understand the mechanism underlying the similar properties of slag containing 18.5% Al2O3 and 20% Al2O3, equilibrium module of FACT SAGE was used for the calculation of phases formed by increasing Al2O3 18.5% to 20%. The liquid slag in the reference case (slag containing 18.5% Al2O3) is formed from melilite, spinel and anorthite phases as calculated at 1200 °C and presented in Figure 3. Similarly, the liquid slag for 20% Al2O3 is also formed from melilite phase, spinel and anorthite phases. In addition to the above-mentioned phases, clinopyroxene is yet another phase that forms before the slag is completely molten. The observed phase-details including the crystal structure are given in Table 5. It is observed from Figure 3 that there is decrease of melilite phase with rise of MgO content when the reference case is compared to the combinations. This is supported by the slag atlas 18 for quaternary system of CaO-SiO2-Al2O3-MgO which shows that increase of MgO content reduces the region of melilite and anorthite phases. The decrease in viscosity of the experimental slag may be attributed to the formation of clinopyroxene phase, and reduction of both melilite and anorthite phase results in the depolymerisation of the silicate structure.
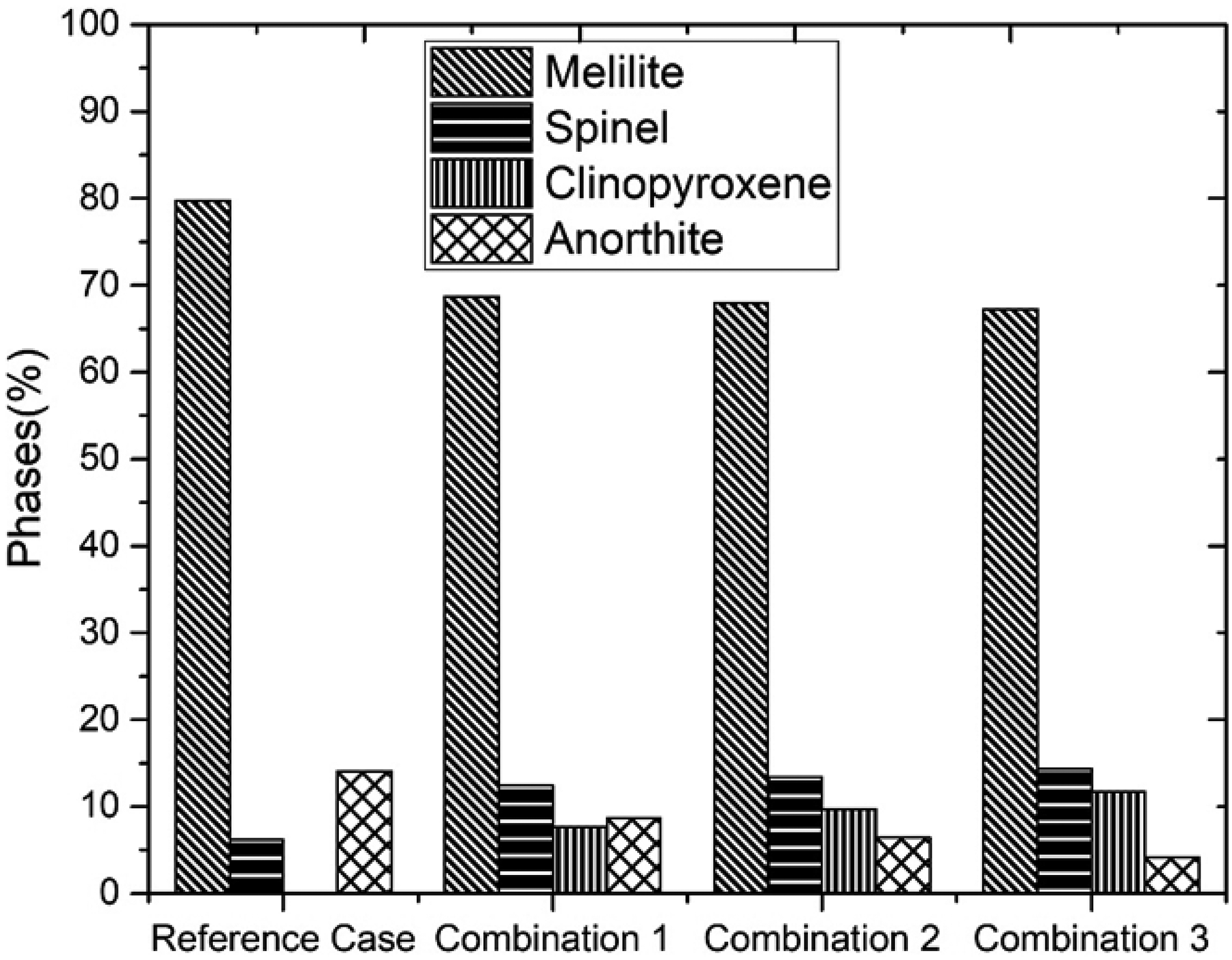
Variation of phases with different combination.
Phases detected and their chemical composition.
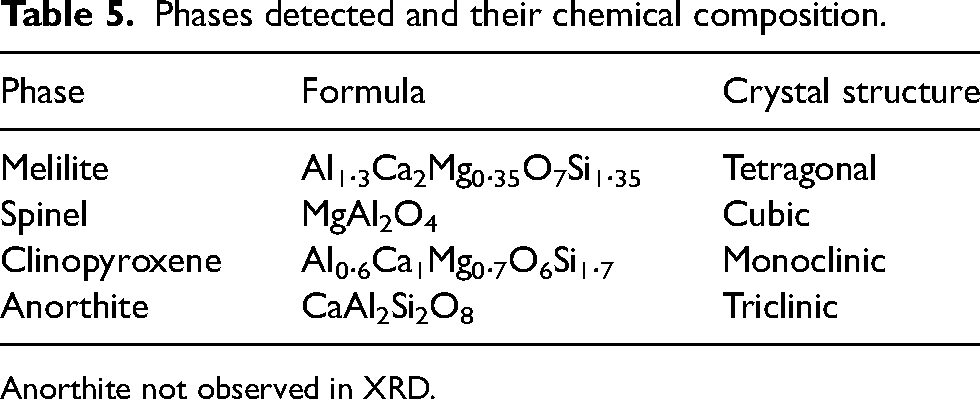
Anorthite not observed in XRD.
Experimental results
Incline plane test
The viscosity obtained from FACTSAGE for the slag containing 20% Al2O3 is shown in Figure 2(b). As the viscosity is a measure of the resistance of the liquid to flow, flowability was measured to understand the change in viscosity of the slag. The average flow length for the three combinations was same as that of the reference case as evidenced by the results of incline plane test. Very little scatter was observed in the results even though the experiments were repeated multiple times. The average flow length and the scatter are shown by the error bars in Figure. 4. It is seen in this figure that the average flow length for the slag increases from 12 to 12.5% MgO and remains constant from 12.5 to 13% MgO content in the slag. The flow length shows a polynomial correlation with viscosity as shown in Figure 5 having
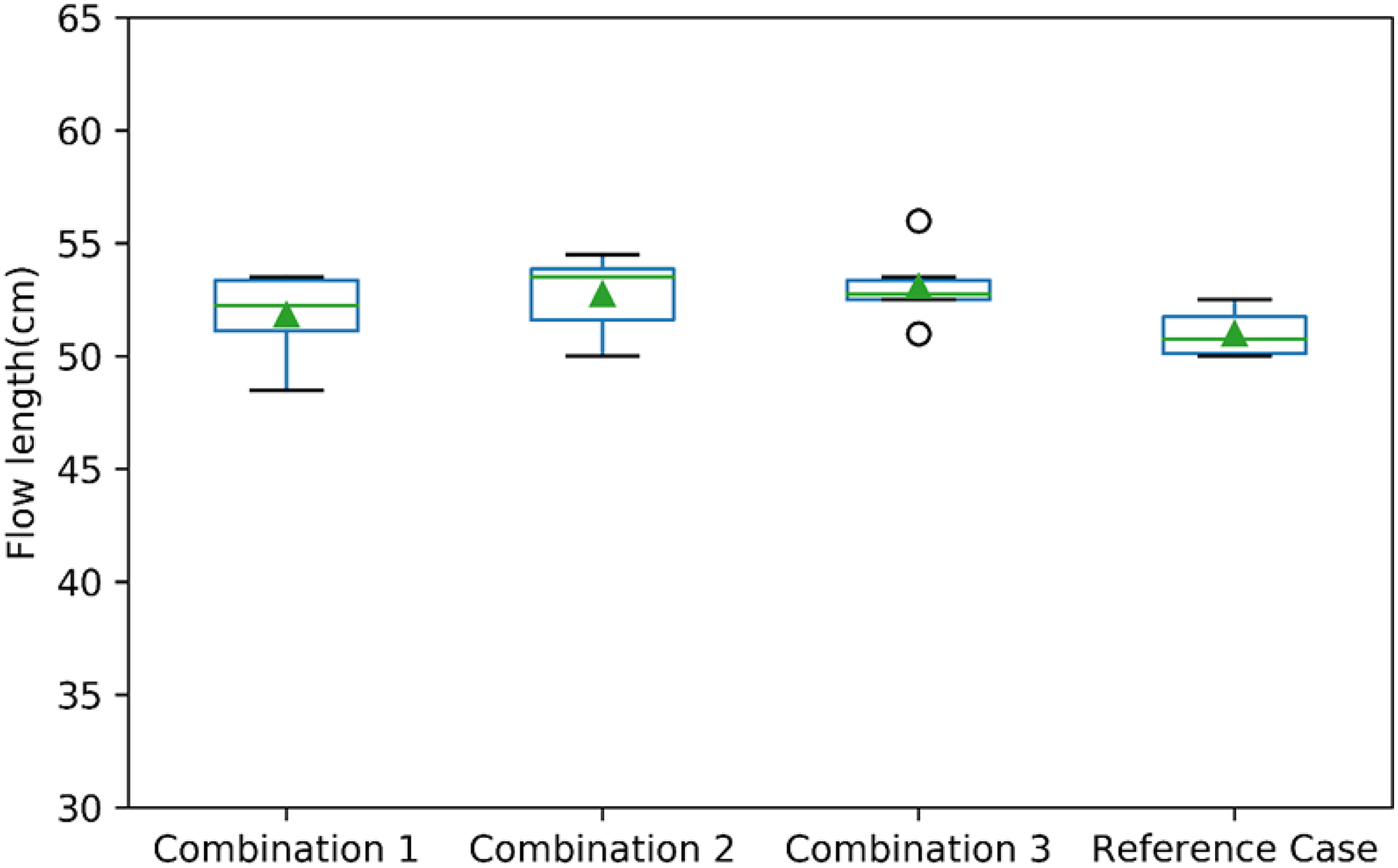
Flow length of different combinations and reference case.
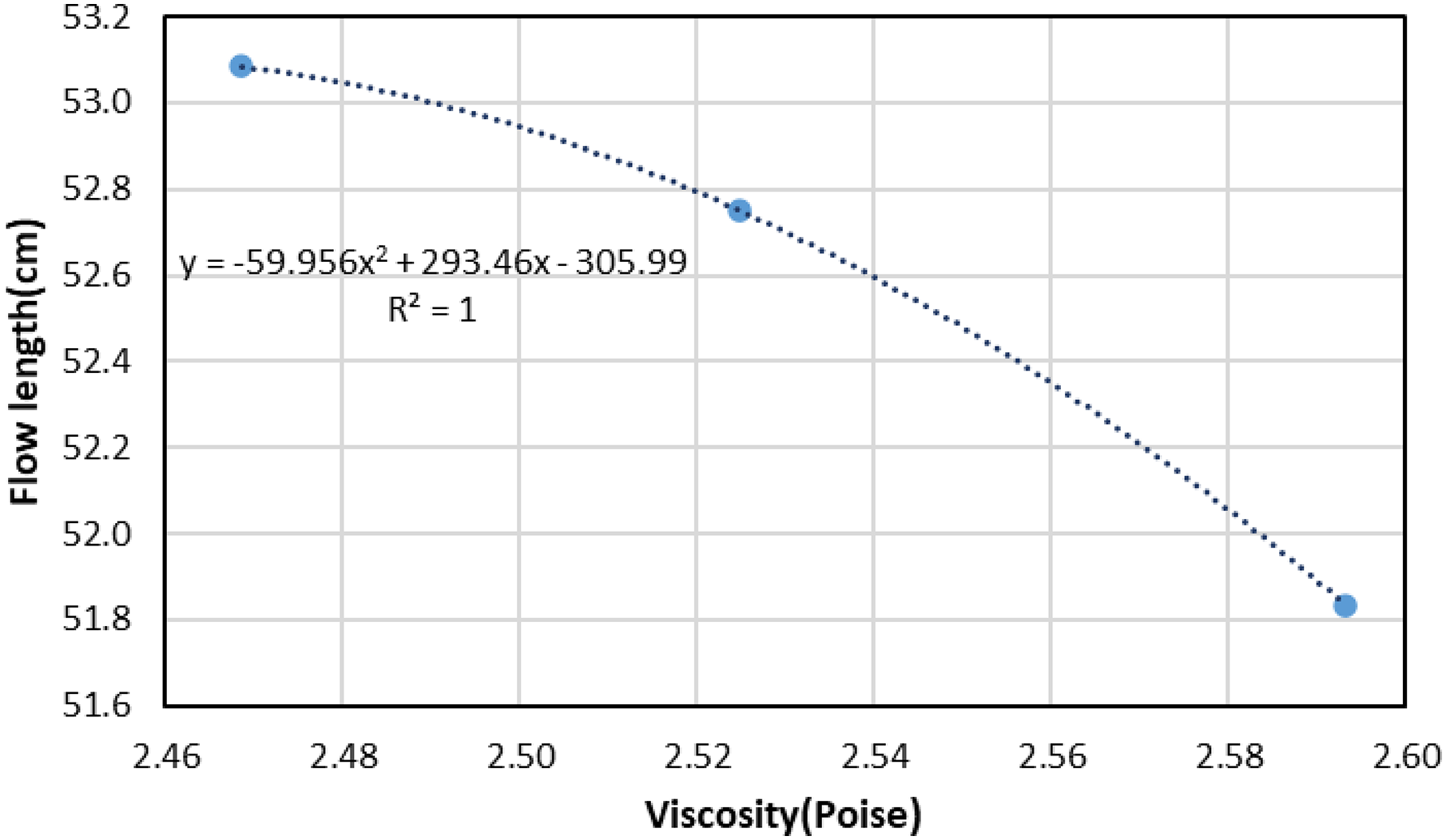
Correlation between flow length and viscosity.
Hot stage microscopy test
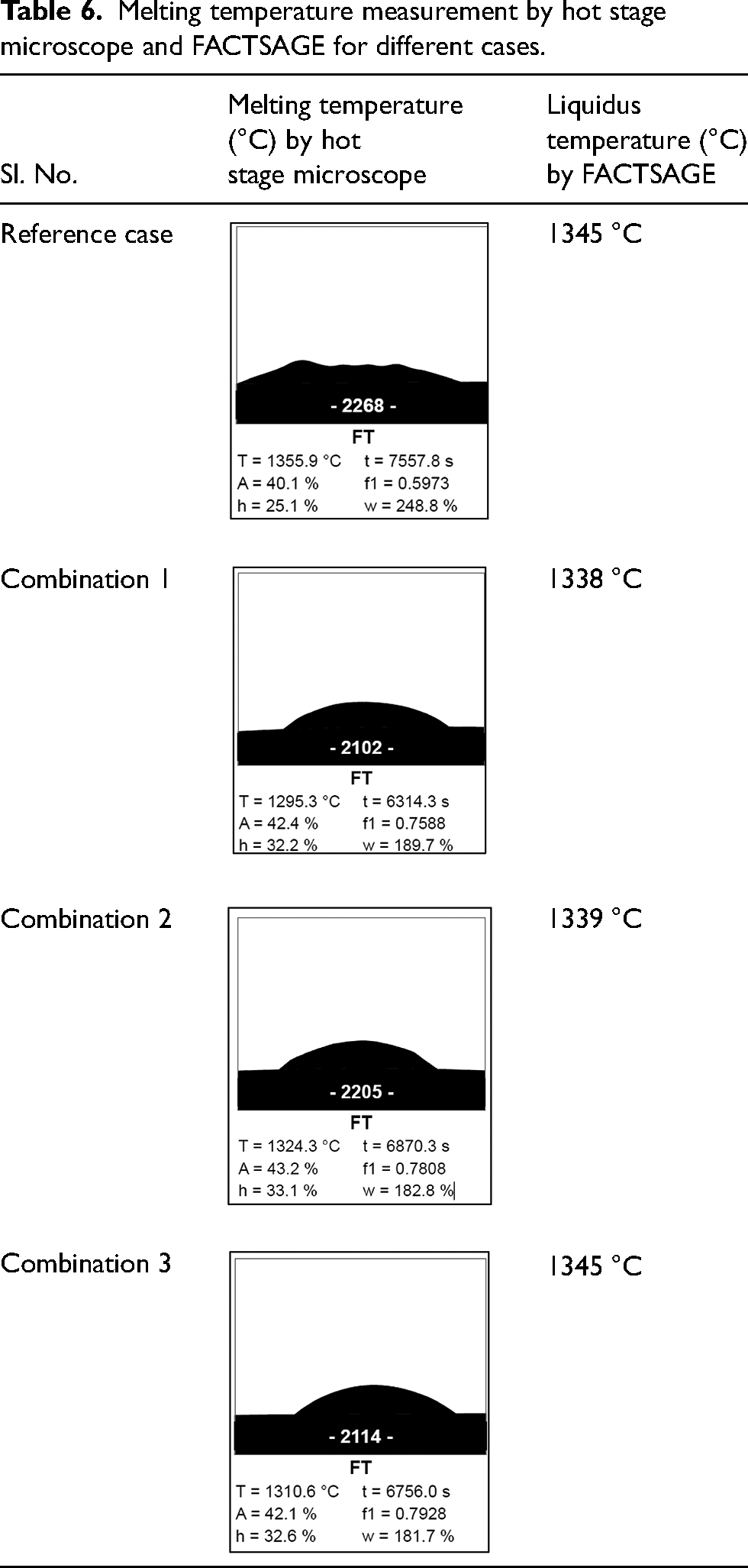
The liquidus temperatures obtained from FACTSAGE were further validated through the hot stage microscopy test. The melting temperature was measured and found to be 1295 °C, 1324 °C, 1310 °C for the three combinations respectively. The melting temperature from the microscope test represents the temperature at which liquid slag has lost the shape completely. For all the three combinations, the melting temperature is less than that of the reference case as mentioned in Table 6, thereby reaffirming that the range of 12–13% MgO and basicity 0.95 is suitable for operating with a slag containing 20% Al2O3.
Melting temperature measurement by hot stage microscope and FACTSAGE for different cases.
XRD analysis
The XRD patterns of the various combinations of slag cooled to room temperature are shown in Figure 6. The phases observed through XRD analysis of the reference case are melilite and spinel. The phases (melilite, spinel, and clinopyroxene) detected for all the three combinations through XRD agree with those obtained through FACTSAGE. However, the latter indicates a decrease in the amount of anorthite phase with increase of MgO content. On the other hand, the peak of anorthite is missing in the XRD analysis results. This may indicate that the amount of anorthite present is likely to be below the detection limits of XRD. Thus, the viscosity and liquidus temperature of 20% Al2O3 are in the same range of 18.5% of Al2O3 due to increase of MgO content from 9.5 to 12–13% and reducing basicity 1 to 0.95.
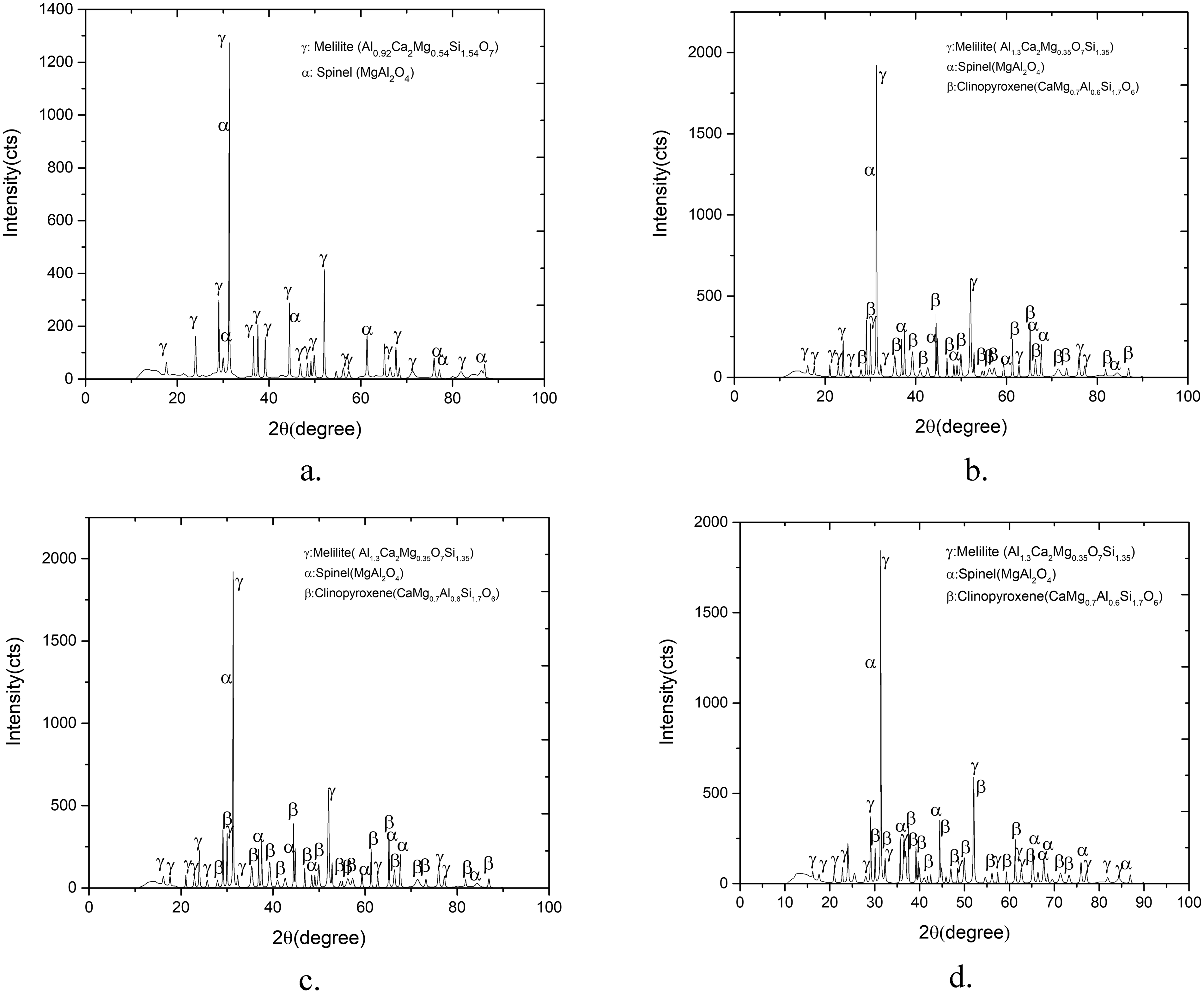
XRD pattern of samples; (a) reference case, (b) combination 1, (c) combination 2, (d) combination 3.
Conclusion
The operating regime of MgO and basicity for slag containing 20% Al2O3, which has similar liquidus temperature and viscosity as slag containing 18.5% Al2O3 was found to be MgO: 12–13% and basicity: 0.95. The simulation results are validated by experimentation and found to be matching. The phases observed through FACTSAGE and XRD are melilite phase, spinel and clinopyroxene for slag containing 20% Al2O3.The formation of clinopyroxene and reduction of anorthite during the pre-melting may be indicative of types of silicate units at the melting stage. This may result in the decrease in the viscosity as well as liquidus temperature of 20%Al2O3 slag.
Footnotes
Acknowledgements
We are grateful to TATA Steel Ltd for permitting us to publish the research. The valuable help by Mr Kanuri Manohar for helping us in preparation of synthetic slag in Muffle Furnace is gratefully acknowledged.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
