Abstract
Background
This study explores how gut metabolites, produced through bacterial metabolism in the gut, influence neurological conditions like Alzheimer's disease (AD). Key metabolites such as succinate and short-chain fatty acids signal through the autonomic nervous system and can cross the blood-brain barrier, impacting central nervous system functions.
Objective
The aim is to examine the role of the gut microbiota in compensating for metabolic deficiencies in AD. By analyzing wild-type (WT) and APP/PS1 mice, the study investigates how the microbiome affects key metabolic processes and whether it can slow AD progression.
Methods
High-throughput sequencing data from the gut microbiomes of APP/PS1 transgenic AD model mice and age-matched WT C57BL/6 male mice were analyzed for microbial and metabolite profiles.
Results
Alpha and beta diversity analyses showed differences in microbial composition between groups. Partial least squares discriminant analysis and Anosim confirmed distinct microbiome profiles in WT and APP/PS1 mice. At the genus level, Vescimonas was more abundant in WT mice, while Odoribacter, Lacrimispora, Helicobacter, Bacteroides, and Alloprevotella were more prevalent in APP/PS1 mice.
Conclusions
While taxonomic differences did not directly link specific microorganisms to AD, functional analysis identified key metabolites—acetyl-CoA, glucose, succinate, lipids, choline, and acetylcholine—that may alleviate energy deficits and synaptic dysfunction. This study suggests that the microbiome may help compensate for AD-related impairments, opening avenues for microbiome-based therapies.
Keywords
Introduction
Diagnosis and prognosis of human diseases can be greatly improved by identifying microbiota-derived molecules in tissues such as those in blood, urine, and feces. These microbiota exhibit distinct signatures in various body regions. However, gut microbiota dysbiosis can disrupt intestinal integrity, enabling microbial molecules to migrate to other tissues and organs, potentially serving as disease biomarkers. 1 These molecules can negatively impact the host immune system and overall health, contributing to disease onset or progression. Numerous clinical studies have identified disease-associated microbiota signatures useful for disease detection and monitoring. For instance, 3-(4-hydroxyphenyl)-lactate, phenylacetic acid, and succinate are associated with liver fibrosis, hepatic steatosis and/or fibrosis in nonalcoholic fatty liver disease.2–5
Conversely, certain metabolites are vital for disease prevention, and their deficiency can hasten disease onset or progression. The gut microbiome may supply these essential molecules to compensate for their insufficient levels. Therefore, targeting specific microbes, metabolites, or enzymes offers a promising strategy for therapeutic interventions to treat or slow the progression of various human diseases.
In the context of Alzheimer's disease (AD), its pathogenesis is influenced by multiple factors, including aging, lifestyle habits, inflammation, oxidative stress, gut microbiota dysbiosis, and imbalances in critical metabolites.6–10 AD is a neurodegenerative disorder characterized by a gradual deterioration of cognitive functions and memory loss. 11 It is recognized as the leading cause of dementia in the elderly population, 12 encompassing an irreversible decline in both episodic memory and general cognitive capacity. 13 The pathology of AD is marked by the presence of amyloid-β plaques and tau-containing neurofibrillary tangles. 14 The extent of cognitive impairment among AD patients varies considerably. Mild cognitive impairment denotes the earliest stage of symptomatic cognitive decline, 15 whereas severe cognitive impairment represents a profound level of cognitive dysfunction that diminishes independence and leads to the onset of dementia. 14
Essential metabolites include those that supply the necessary energy for the human body. Recent studies highlight the critical interrelationship between glucose metabolism, acetyl-coenzyme A (acetyl-CoA), and ATP production in maintaining neuronal health and function. 16 Recent reports suggest that disruptions in this metabolic network can contribute to the pathogenesis of AD. 11 Nevertheless, our understanding of the gut microbiota's role in disease prevention by providing metabolites that are inadequately synthesized internally or acquired externally remains limited. For example, early reductions in glucose uptake and metabolism can lead to decreased production of acetyl-CoA and ATP, setting the stage for energy deficits and synaptic dysfunction.11,17 In addition, impaired mitochondrial function elevates oxidative stress, contributing to the neurodegenerative processes characteristic of AD.18–20 Furthermore, reduced acetyl-CoA levels directly affect acetylcholine (ACh) synthesis, leading to cholinergic deficits that are characteristic of AD and contribute to cognitive decline. 21
To our knowledge, no previous studies have thoroughly investigated whether the gut microbiota can compensate for insufficient levels of metabolites that correlate with disease onset or progression. However, recent research suggests that the gut bacteriome contributes to the biosynthesis of lipopolysaccharide (LPS), which activates microglia in the brain through the gut-brain axis. 22 LPS also contributes to the biosynthesis of proinflammatory cytokines that are hallmarks of AD pathogenesis. Several enzymes are involved in LPS biosynthesis and subsequent events; however, the most crucial enzyme serving as the ultimate precursor for the inner core oligosaccharide is ADP-glyceromanno-heptose 6-epimerase. 23
We hypothesize that the progression and severity of AD may be linked to an insufficient supply of essential metabolites from the gut microbiota. To test this, we are conducting a functional analysis to predict the levels of these crucial molecules in the gut microbiota of wild-type (WT) mice and age-matched amyloid precursor protein/presenilin-1 (APP/PS1) mice, which carry the genetic mutation responsible for early-onset AD. Our study aims to elucidate the potential role of gut microbiota in mitigating or slowing the progression of AD.
Methods
This study utilized paired-end sequencing reads of the 16S rRNA (V3–V4 region) gene from the gut microbiomes of 8-month-old male transgenic AD model mice with the amyloid precursor protein Swedish mutation (APPswe)/PS1dE9 (APP/PS1) and age-matched WT C57BL/6 male mice. These sequencing datasets were originally generated by OE Biotech Co., Ltd (Shanghai, China), and obtained from the BioProject database at the National Center for Biotechnology Information under project number PRJNA687556, as previously described. 24 The original study reported that all mice were individually housed under standard pathogen-free conditions and acclimated to the laboratory environment for one week before fecal sample collection. During this period, the animals were provided a balanced diet with appropriate levels of carbohydrates, fiber, minerals, and vitamins. 25
Clean read datasets were further analyzed at the Beijing Genome Institute in Hong Kong, China. The sequencing data were processed using QIIME software version 1.7.0. 26 Generated tags were compared to the reference SILVA database (http://www.arb-silva.de/), and chimeric sequences were removed with the UCHIME algorithm. 27 Effective tags were analyzed with UPARSE software version 7.0.1090, 28 after which a representative sequence for each operational taxonomic unit (OTU) was selected, and taxa were annotated, 29 where taxonomic classification was performed at the phylum, genus, and species levels. Multiple sequence alignment was conducted using MUSCLE software version 3.8.31. 30 Then, partial least squares discriminant analysis (PLS-DA) was conducted using R software (version 2.15.3) to determine ecological distances among samples and groups. Alpha diversity was assessed using the number of observed species, Shannon, and Simpson indices to evaluate diversity within individual samples. A species accumulation curve (Specaccum) was generated to ensure saturation of taxa across the gut microbiome of these individuals. Based on OTUs clustering results, the OTU table was normalized, then, common (core microbes) and unique (cloud microbes) information for the two groups were introduced in a Venn diagram. As beta diversity measures, rarefaction curves were generated to determine the minimum number of reads commonly present in all samples, while Anosim was conducted using the Vegan package's anosim function in R software to justify sample grouping. Additionally, effect size measurements (LEfSe) and heatmaps of weighted UniFrac beta diversity, produced from the data matrices, were used to detect taxon abundance and relative abundance of microbes in the two ecological niches.
Functional prediction was performed using the Phylogenetic Investigation of Communities by Reconstruction of Unobserved States (PICRUSt) software package (version 1.1.4; http://picrust.github.io/picrust/). 31 We conducted a comparison of Kyoto Encyclopedia of Genes and Genomes (KEGG) enzymes between the two groups of gut microbiota. After identifying enriched enzymes in the gut microbiome of WT mice, we proceeded to analyze the associated pathways, subcategories, and categories. For taxonomic comparison of gut microbiomes, we established a threshold difference of ≥60% abundance between the groups. In terms of functional analysis, we utilized a threshold of 0.2-fold reduction in abundance in the APP/PS1 group to investigate functional profiles across the different functional levels.
Results
Statistics for the clean reads of gut microbiomes from 8-month-old male transgenic AD model mice (APPswe/PS1dE9) and age-matched WT C57BL/6 male mice are presented in Supplemental Table S1. The number of OTUs ranged from 1001 to 2007 in WT mice and from 871 to 1784 in APP/PS1 mice, indicating a higher species count in the WT group. Notably, the highest (83,548) and lowest (54,737) counts per sample were found in the APP/PS1 group. Alpha diversity measures, including observed species, Shannon index (emphasizing taxon richness), and Simpson index (emphasizing taxon evenness), revealed no significant differences between the two groups (Supplemental Figures S1 and S2 and Supplemental Table S2), demonstrating the consistency of the sequencing datasets. The species accumulation curve showed a continuous increase with the number of sequenced samples, leveling off at the end, indicating that the 28 samples were sufficient to cover the entire taxa of these mice gut microbiomes (Supplemental Figure S3).
To further validate the sequencing datasets, PLS-DA demonstrated complete separation between the samples of the two groups (Figure 1), indicating differential taxa occurrence due to the disease. The number of observed species, used as a rarefaction metric, showed that an optimal sequencing depth of ∼55,000 reads was necessary to encompass all samples (Figure 2 and Supplemental Table S3). This depth allowed for the assessment of taxonomic relative abundance between the gut microbiomes of WT and APP/PS1 groups. Although not statistically significant, the Anosim plot indicated greater variability between the groups compared to within each group (Supplemental Figure S4). The Venn diagram results revealed an average of 583 unique microbes per group, with 3409 shared microbes, highlighting the differential abundance in gut microbiomes between the two groups and underscoring the impact of host-microbe interactions in AD (Supplemental Figure S5 and Table S4).
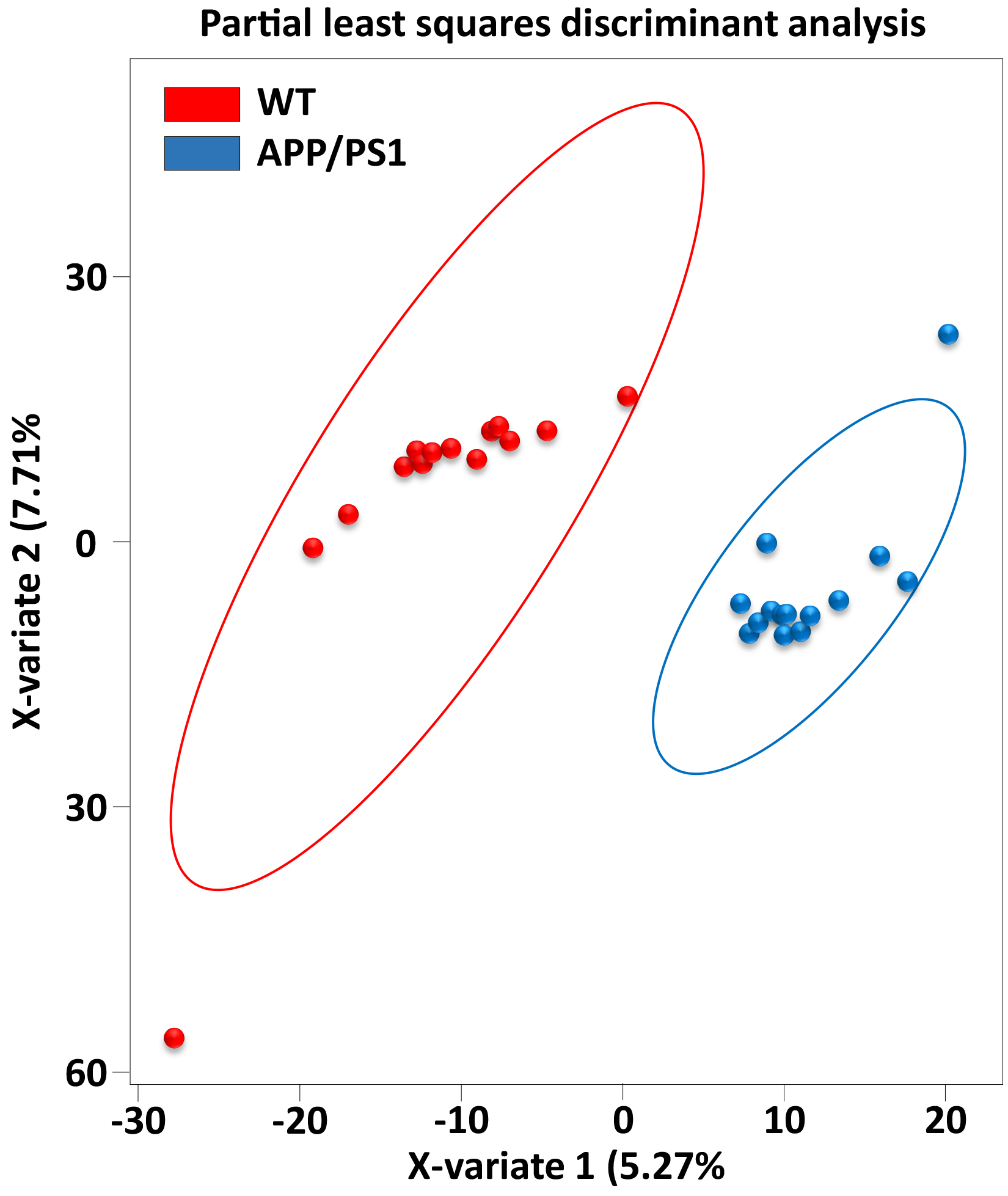
Partial least squares discriminant analysis (PLS-DA) of gut microbiomes of wild-type male mice (WT) and age-matched transgenic male littermates expressing amyloid precursor protein/presenilin-1 (APP/PS1), the causative factor for early onset AD. PLS-DA is a statistical technique used to identify and quantify the differences between groups based on predictive modeling.
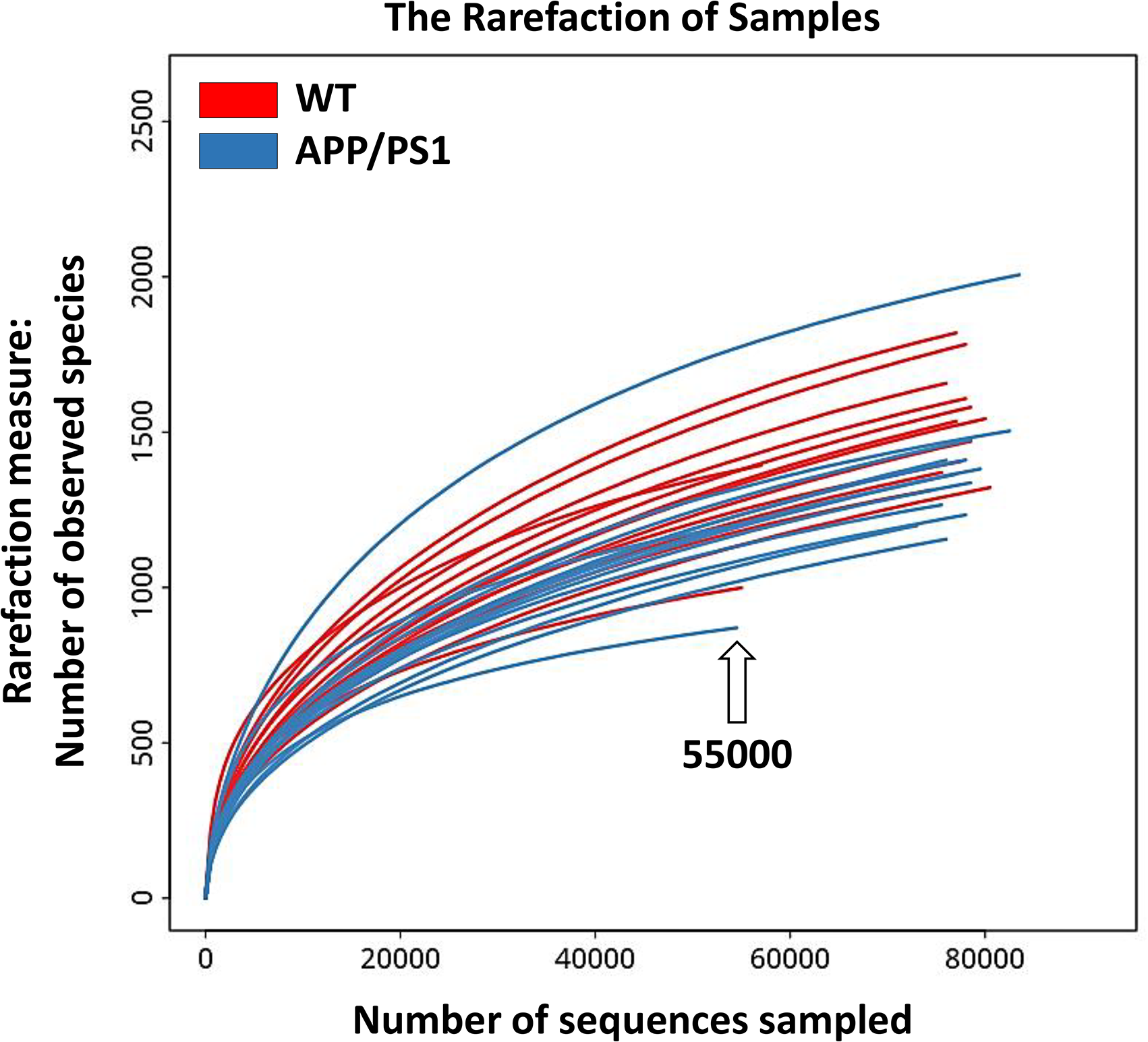
Number of observed species as a rarefaction metric, delineating the optimal sequencing depth (∼55,000 sequence reads) necessary to encompass all samples within the datasets, facilitating the assessment of taxonomic relative abundance as a metric of beta diversity across the gut microbiomes of wild-type (WT) male mice and age-matched transgenic male littermates expressing amyloid precursor protein/presenilin-1 (APP/PS1). Additional details can be referenced in Supplemental Table S3.
The annotation results, detailing the quantity of microbial queries in the gut microbiomes of wild-type male mice (WT) and age-matched transgenic male littermates expressing amyloid precursor protein/presenilin-1 (APP/PS1), are presented in Supplementary Table S5. Notably, members of the phylum Bacteroidota exhibited the highest records.
The results of microbe abundance at the phylum, genus, and species levels are detailed in Supplemental Figures S6–S11 and Supplemental Tables S6–S8. Across both groups, Bacteriodota (synonym Bacteroidetes) and Bacillota (synonym Firmicutes) were the most abundant phyla (Supplemental Figure S6a). However, the phyla Deferribacterota and Candidatus Saccaribacteria were more prevalent in the gut microbiome of WT mice, while Planctomycetota and Campylobacterota were more abundant in APP/PS1 mice (Figures S6(b) and S7). At the genus level, Duncaniella and Muribaculum were the most prevalent across both groups (Figure S8a).
In terms of relative abundance, Vescimonas was more prevalent in WT mice, while Odoribacter, Lacrimispora, Helicobacter, Bacteroides, and Alloprevotella were more prevalent in APP/PS1 mice (Supplemental Figures S8(b) and S9 and Supplemental Table S7). The results shown in the cladogram illustrating the linear discriminant analysis effect size (LEfSe) algorithm also indicate the high abundance of Alloprevotella in APP/PS1 mice (Figure 3). At the species level, Muribaculum intestinale, Duncaniella muris, and Paramuribaculum intestinale were the most common in both groups (Supplemental Figure S10a). Relative abundance analysis showed that Mucispirillum schaedleri was overrepresented in WT mice, while Bacteroides acidifaciens, Helicobacter ganmani, Duncaniella dubosii, and Alloprevotella rava were more abundant in APP/PS1 mice (Supplemental Figures S10b and S11 and Supplemental Table S8).
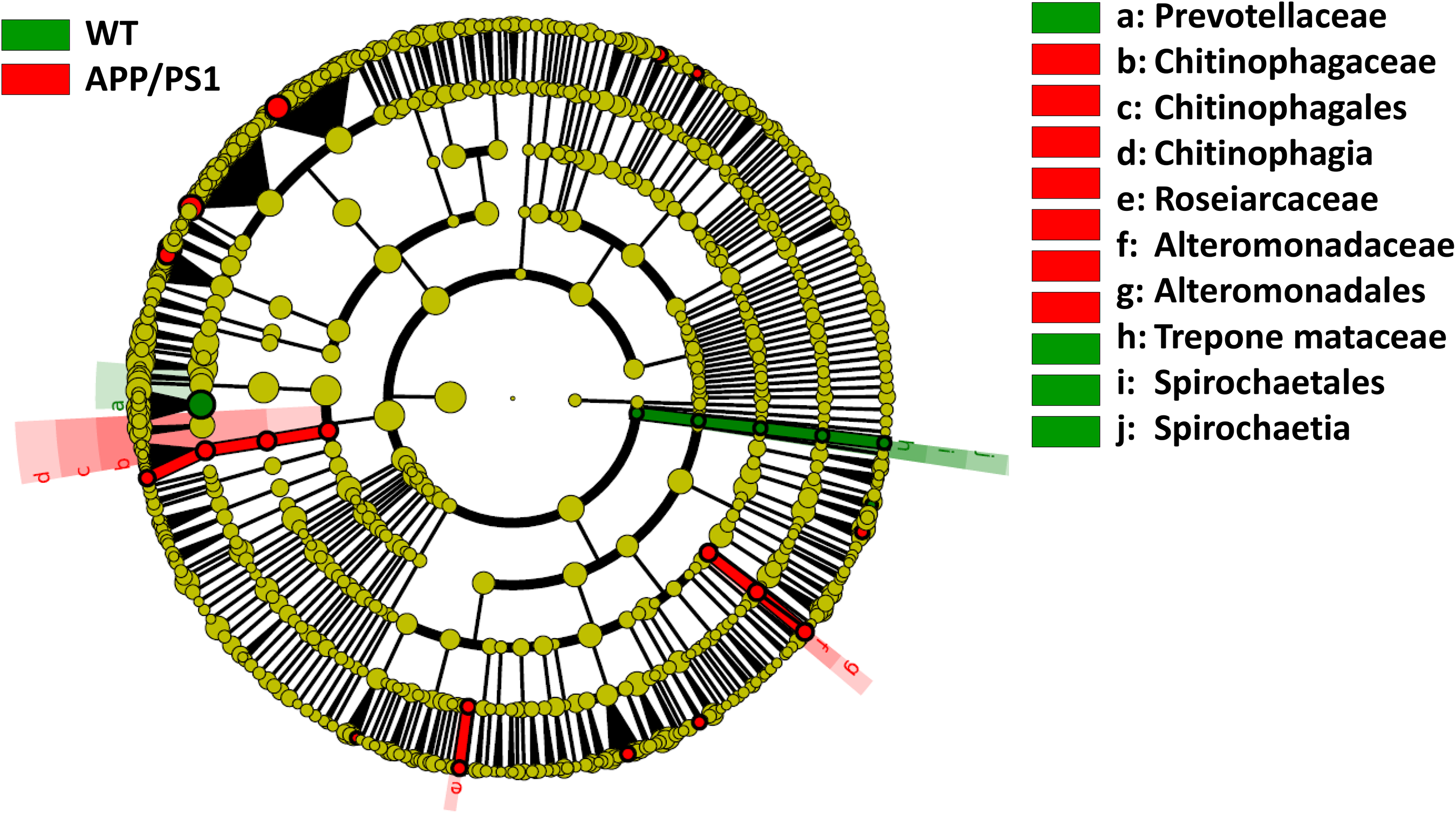
Cladogram illustrating the linear discriminant analysis effect size (LEfSe) algorithm performed utilizing the online platform Galaxy (version 1.0.0; https://huttenhower.sph.harvard.edu/galaxy/) to discern discriminant microbial taxa within the gut microbiomes of wild-type male mice (WT) and age-matched transgenic male littermates expressing amyloid precursor protein/presenilin-1 (APP/PS1).
At the functional level, 847 enzymes were highly abundant (>3000 queries) across the gut microbiomes of WT male mice and age-matched transgenic male littermates expressing APP/PS1 (Supplemental Table S9).
This number was reduced to 77 after selecting enzymes that showed a fold decrease in abundance of ≥0.2 in the APP/PS1 mice with AD (Figure 4). Among these, 65 enzymes present in 58 KEGG pathways were further analyzed (Figure 5 and Supplemental Tables S10). Abundance of these pathways is shown in Supplemental Table S11. These pathways are distributed across 13 subcategories and four categories, with “Metabolism” and its subcategories “Amino acid metabolism”, “Carbohydrate metabolism”, “Metabolism of cofactors and vitamins”, and “Metabolism of terpenoids and polyketides” being the most prominent (Supplemental Figures S12 and S13 and Supplemental Tables S12 and S13). The most abundant pathways include “Arginine biosynthesis”, “Alanine, aspartate and glutamate metabolism”, “Other glycan degradation”, “One carbon pool by folate”, and “Cholinergic synapse” (Figure 5). Except for the last three pathways in Supplemental Table S10, which respectively belong to “Organismal systems”, “Genetic Information Processing”, and “Environmental Information Processing”, all other pathways fall under the “Metabolism” category.
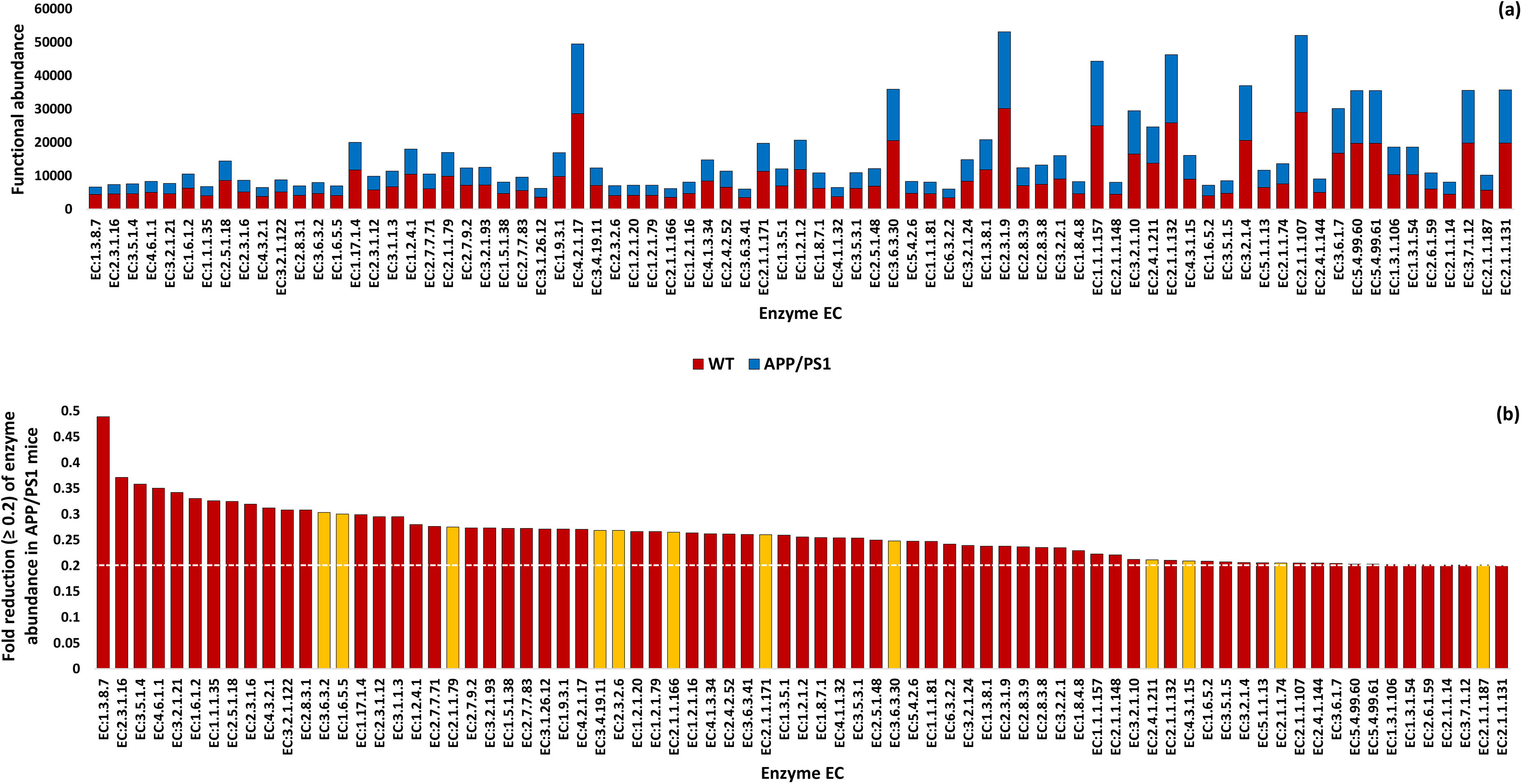
Functional abundance of enzymes exhibiting >3000 queries across the gut microbiomes of wild-type male mice (WT) and age-matched transgenic male littermates expressing amyloid precursor protein/presenilin-1 (APP/PS1) (a) and showed a fold decrease in abundance of ≥ 0.2 in the APP/PS1 mice (b). Dotted white line corresponds to the fold reduction threshold of ≥ 0.2 of KEGG enzyme abundance in APP/PS1 mice. Enzymes highlighted in orange do not participate in any KEGG pathways and were therefore excluded from further analysis. Additional details can be referenced in Supplemental Tables S9 and S10.
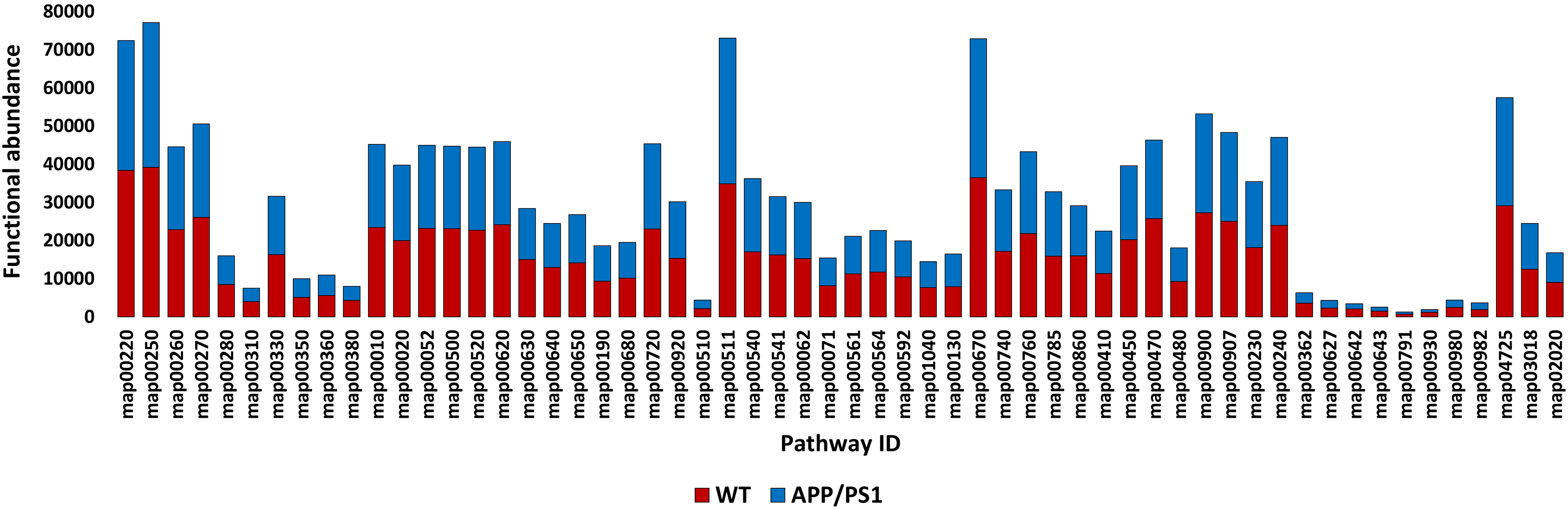
Functional abundance of KEGG pathways that host the selected enzymes for further analysis within the gut microbiomes of wild-type male mice (WT) and age-matched transgenic male littermates expressing amyloid precursor protein/presenilin-1 (APP/PS1). Additional details can be referenced in Supplemental Tables S10 and S11.
Functional analysis in this study underscores the possible influence of the gut microbiome on AD in a mouse model. Key metabolites in AD pathogenesis, such as glucose, acetyl-CoA, and ATP, which are critical for energy production and defense against the disease's progression, influence a series of upstream and downstream events. Our findings indicate that these metabolites are produced more abundantly by enzymes that are highly enriched in the gut microbiome of WT individuals compared to that of AD mice (Figures 6–8 and Supplemental Table S9).
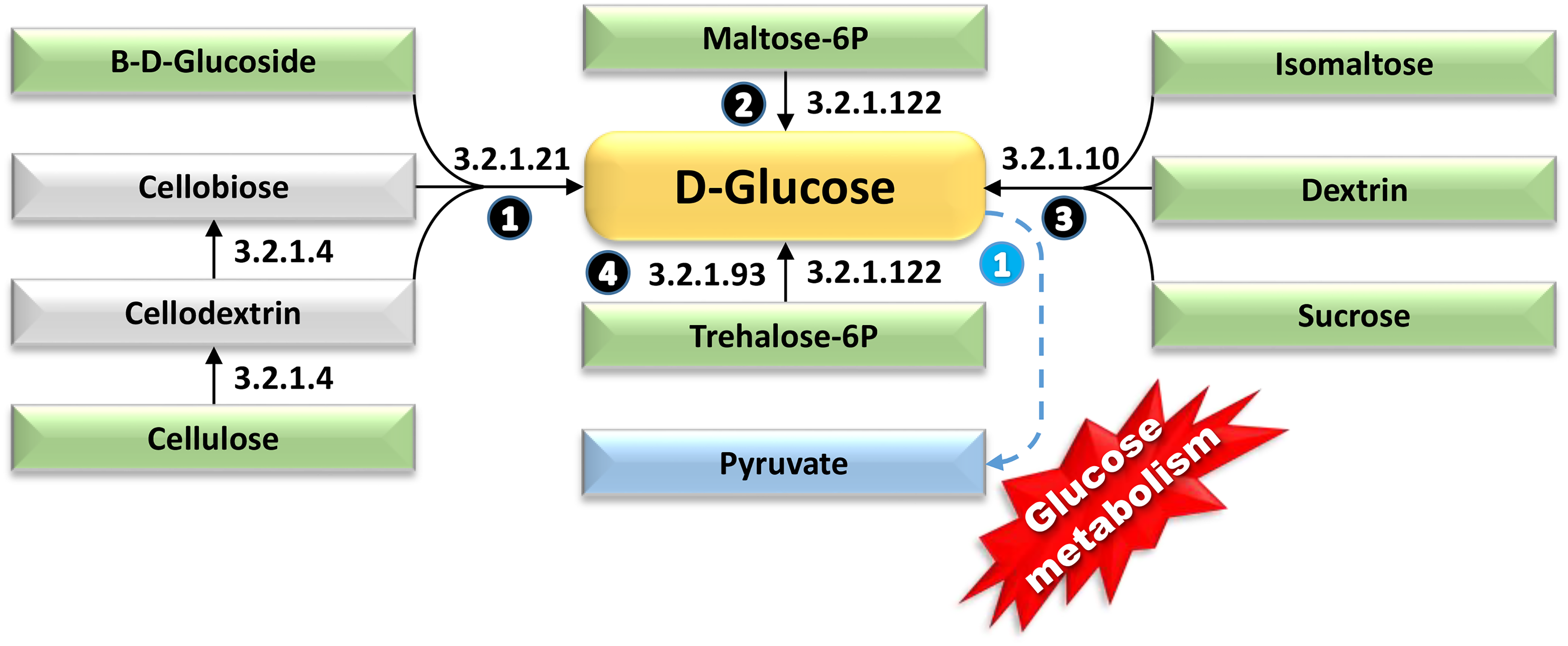
Elucidation of the four metabolic avenues with highly enriched enzymes in the gut microbiomes of wild-type male mice (WT), culminating in glucose as the ultimate product molecule that eventually broke down to produce pyruvate via glucose metabolism or glycolysis. Pyruvate is a central molecule in cellular respiration, serving as a gateway between glycolysis and the citric acid cycle. These avenues demonstrate a significant decrease in abundance (≥ 0.2-fold) in age-matched transgenic male littermates expressing amyloid precursor protein/presenilin-1 (APP/PS1), intricately interconnecting diverse KEGG pathways. Metabolites initiating the various pathways are enclosed within green boxes, while the final products of each pathway are outlined within blue boxes. Solid arrows depict singular metabolic reactions, whereas dotted arrows indicate involvement of multiple metabolic processes. Numbers within black circles denote upstream reactions of the core metabolite, e.g., glucose, whereas numbers within blue circles indicate downstream reactions. Enzymes highlighted by black arrows indicate instances of contrasting enrichment between the two genotypes, while those annotated with red arrows signify enzymes with consistently sustained levels of enrichment. EC:5.4.2.6 = Beta-phosphoglucomutase, EC:3.2.1.122 = Maltose-6′-phosphate glucosidase, EC:3.2.1.93 = Alpha,alpha-phosphotrehalase, EC:3.2.1.10 = Oligo-1,6-glucosidase, EC:3.2.1.4 = Cellulase. Further information is available in Supplemental Figures S26 and S27.
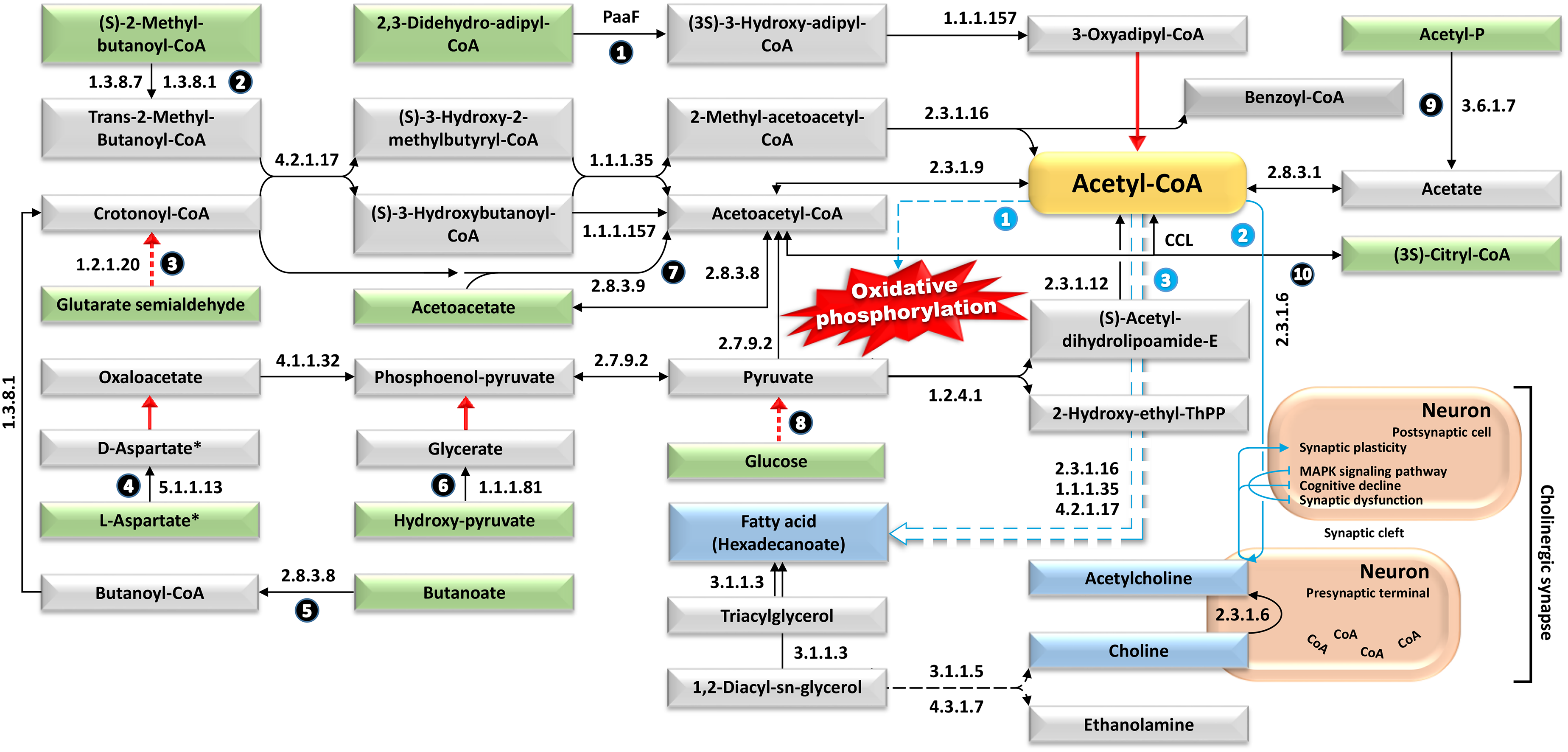
Illustration of the metabolic avenues with highly enriched enzymes in the gut microbiomes of wild-type male mice (WT), with acetyl-CoA emerges as a pivotal metabolite orchestrating these avenues. Such avenues exhibit a substantial decrease in abundance (≥ 0.2-fold) in age-matched transgenic male littermates expressing amyloid precursor protein/presenilin-1 (APP/PS1), intricately linking diverse KEGG pathways and ultimately facilitating the synthesis of hexadecanoate fatty acid. Precursor molecules initiating the various avenues are represented by metabolites enclosed within green boxes, the end products of each avenue are depicted within blue boxes, while acetyl-CoA that acts as both a precursor and an end product is shown in orange box. Solid arrows portray individual metabolic reactions, while dotted arrows indicate participation of multiple metabolic processes. Numbers within black circles denote upstream reactions of the core metabolite, e.g., Acetyl-CoA, whereas numbers within blue circles indicate downstream reactions. Enzymes highlighted by black arrows denote instances of divergent enrichment between the two genotypes, whereas those marked by red arrows indicate enzymes with consistently maintained levels of enrichment. EC:4.1.1.32 = Phosphoenolpyruvate carboxykinase (GTP), EC:4.1.3.34 = Citryl-CoA lyase (CCL), EC:1.1.1.35 = 3-hydroxyacyl-CoA dehydrogenase, EC:1.1.1.81 = Hydroxypyruvate reductase, EC:1.1.1.157 = 3-hydroxybutyryl-CoA dehydrogenase, EC:1.2.4.1 = Pyruvate dehydrogenase (acetyl-transferring), EC:1.3.8.1 = Short-chain acyl-CoA dehydrogenase, EC:2.8.3.1 = Propionate CoA-transferase, EC:2.7.9.2 = Pyruvate, water dikinase, EC:3.1.1.3 = Triacylglycerol lipase, EC:1.3.8.7 = Medium-chain acyl-CoA dehydrogenase, EC:3.6.1.7 = Acylphosphatase, EC:2.8.3.8 = Acetate CoA-transferase, EC:1.17.1.9 = Formate dehydrogenase, EC:2.3.1.9 = Acetyl-CoA C-acetyltransferase, EC:2.8.3.9 =Butyrate–acetoacetate CoA-transferase, EC:2.3.1.12 = Dihydrolipoyllysine-residue acetyltransferase, EC:5.1.1.13 = Aspartate racemase, EC:2.3.1.16 = Acetyl-CoA C-acyltransferase, EC:2.3.1.6 = Choline O-acetyltransferase, EC:4.2.1.17 = Enoyl-CoA hydratase (PaaF), EC:1.2.1.20 = Glutarate-semialdehyde dehydrogenase. Asterisks denote the presence of metabolites as illustrated in Figure 7. Additional details can be found in Supplemental Figures S14, S16, S18, S19, S22-S25, S29, S31, S32, S34, S35, S41-S44, S51, S53, S55, S57, S61-S63, and S69.
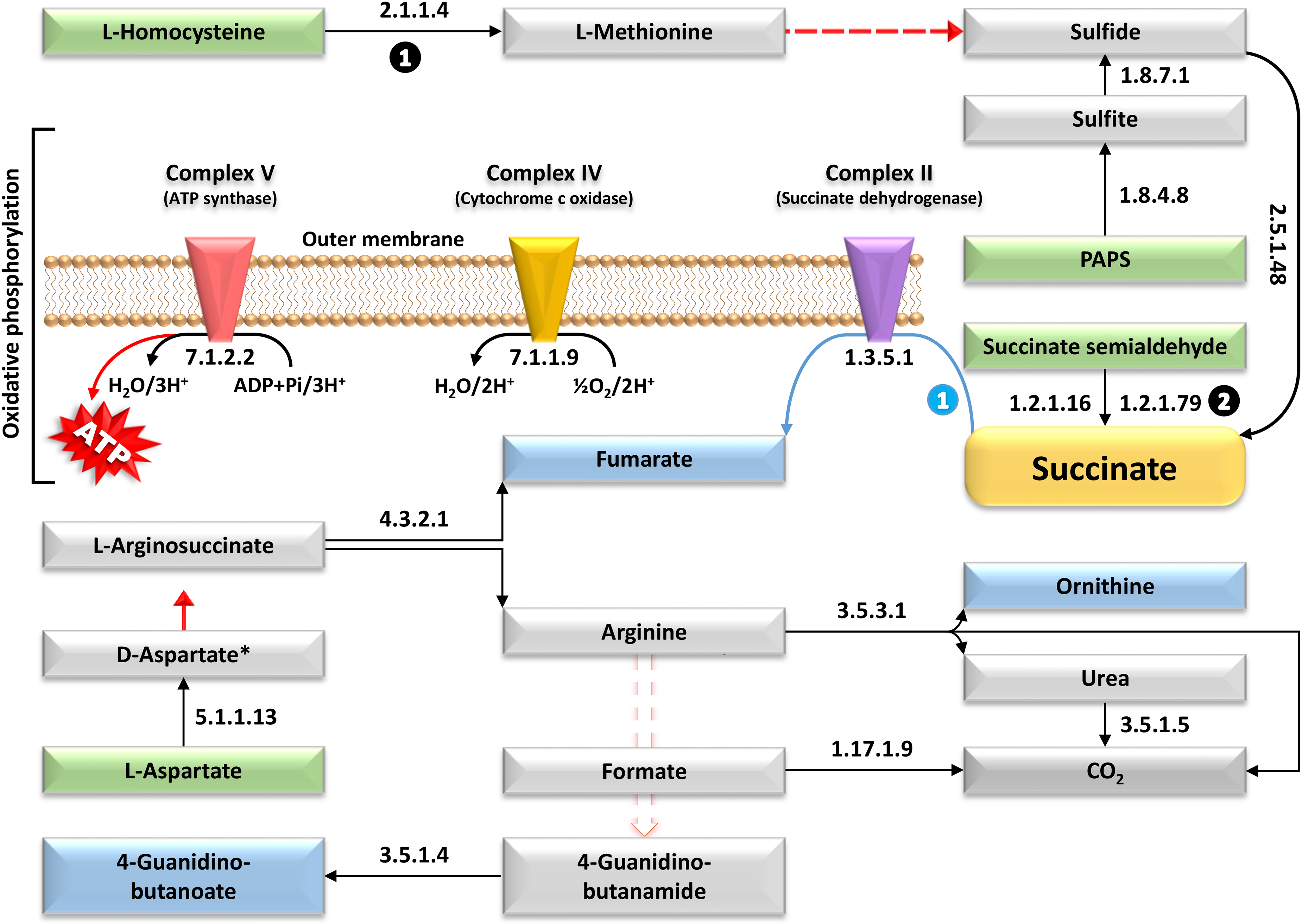
Elucidation of the metabolic avenues with highly enriched enzymes in the gut microbiomes of wild-type male mice (WT), where ATP emerges as the ultimate end product molecule. These avenues exhibit a notable decrease in abundance (≥ 0.2-fold) in age-matched transgenic male littermates expressing amyloid precursor protein/presenilin-1 (APP/PS1), intricately intertwining diverse KEGG pathways and ultimately facilitating the synthesis of several crucial metabolites. Precursor molecules initiating the various pathways are represented by metabolites enclosed within green boxes, while the end products of each pathway are delineated within blue boxes. Solid arrows depict individual metabolic reactions, whereas dotted arrows signify involvement in multiple metabolic processes. Numbers within black circles denote upstream reactions of the core metabolite, e.g., succinate, whereas numbers within blue circles indicate downstream reactions. Enzymes highlighted by black arrows denote instances of divergent enrichment between the two genotypes, while those marked by red arrows indicate enzymes with consistently maintained levels of enrichment. EC: 7.1.2.2 = Heme-transporting ATPase, EC:2.5.1.48 = Cystathionine gamma-synthase (CGS), EC:1.2.1.79 = Succinate-semialdehyde dehydrogenase (NADP(+)), EC:1.1.1.81 = Hydroxypyruvate reductase, EC:1.3.5.1 = Succinate dehydrogenase (quinone), EC:1.3.8.1 = Short-chain acyl-CoA dehydrogenase, EC:3.5.3.1 = Arginase, EC:1.8.7.1 = Assimilatory sulfite reductase (ferredoxin) (Sir), EC:7.1.1.9 = Cytochrome-c oxidase, EC:1.17.1.9 = Formate dehydrogenase, EC:7.1.1.1 = NAD(P)(+) transhydrogenase (Re/Si-specific), EC:3.5.1.4 = Amidase, EC:1.8.4.8 = Phosphoadenylyl-sulfate reductase (thioredoxin/CysH), EC:5.1.1.13 = Aspartate racemase, EC:2.1.1.14 = 5-methyltetrahydropteroyltriglutamate–homocysteine S-methyltransferase (MET), EC:1.2.1.16 = Succinate-semialdehyde dehydrogenase (NAD(P)(+)), EC:4.3.2.1 = Argininosuccinate lyase. Asterisks denote the presence of metabolites as illustrated in Figure 6. Additional details can be found in Supplemental Figures S15, S17, S20, S21, S30, S32-S34, S36, S50, S54, S55, and S65.
In glucose biosynthesis, four metabolic avenues involving five highly enriched enzymes in gut microbiome of WT mice, namely, beta-phosphoglucomutase (EC:5.4.2.6), maltose-6′-phosphate glucosidase (EC:3.2.1.122), alpha,alpha-phosphotrehalase (EC:3.2.1.93), oligo-1,6-glucosidase (EC:3.2.1.10), and cellulase (EC:3.2.1.4) took place (Figure 6). As a result of glycolysis, the breakdown of glucose to pyruvate, a cascade of events was initiated, leading to the production of acetyl-CoA, a core metabolite and precursor for energy production and neuronal synaptic plasticity. Ten avenues, supported by numerous upstream enzymes that are highly enriched in gut microbiome of WT mice, produce acetyl-CoA more intensively compared to that of AD mice (Figure 7). Similarly, downstream avenues from acetyl-CoA, such as “Cholinergic synapse” and “Oxidative phosphorylation”, are promoted by enzymes enriched in gut microbiome of WT mice. The cholinergic pathway, which includes choline and ACh production, is particularly active in the WT gut microbiome. Acetyl-CoA, central to the tricarboxylic acid (TCA) cycle, directly supports “Oxidative phosphorylation” and ATP biosynthesis (Figure 7). Additionally, three highly enriched enzymes downstream of acetyl-CoA in gut microbiome of WT mice support fatty acid (hexadecanoate) biosynthesis, crucial for energy generation.
Succinate, another core metabolite, is produced more intensively by enzymes in WT gut microbiomes, further promoting oxidative phosphorylation and ATP synthesis (Figure 8). Other metabolites, such as ornithine and 4-Guanidino-butanoate, are also produced more abundantly in the WT gut microbiome. Avenues for these metabolites utilize arginine as a substrate (Figure 8). Further details on the pathways and metabolic avenues are provided in Supplemental Figures S14–S71.
Discussion
Researchers often prefer male mice in amyloid precursor protein/presenilin-1 (AP) transgenic studies to avoid the hormonal fluctuations of female mice, reduce variability linked to sex-specific disease differences, and maintain consistency with existing data that predominantly involves male subjects. 32
Microbes and Alzheimer's disease
Differential abundance of gut microbiomes of the 8-month-old male transgenic AD model mice (APPswe/PS1dE9) and age-matched WT C57BL/6 male mice resulted in the increase of genus Vescimonas for WT mice, while Odoribacter, Lacrimispora, Helicobacter, Bacteroides, and Alloprevotella for the AD mice (Supplemental Figures S8(b) and S9 and Supplemental Table S7). Genera Odoribacter, Lacrimispora, Vescimonas, and Alloprevotella are predominantly commensal members of the gut microbiota and are not traditionally considered pathogenic. Their role in health and disease primarily revolves around their metabolic activities and interactions within the gut ecosystem. 33 Vescimonas is an anaerobic bacteria found in the gut microbiota of various animals that play a role in fermenting complex polysaccharides, a good characteristics for the hosting organisms. 34 While specific studies on Vescimonas are limited, its presence in gut microbiome datasets indicates its role in microbial community dynamics and metabolic functions. 34 The most well-known pathogen is Helicobacter pylori, which colonizes the stomach and is associated with gastritis, peptic ulcers, and an increased risk of gastric cancer. 35 Pathogenic Bacteroides species produce enzymes and toxins that aid in tissue invasion and evasion of host immune responses. 36 Although the genus Alloprevotella is typically a common component of the normal microbiota, certain conditions can render it pathogenic, particularly in diseases such as inflammatory bowel disease. 37 There is evidence that gut Alloprevotella is capable of producing SCFAs like acetate, propionate, and butyrate through the fermentation of dietary fibers. 38 In turn, SCFAs can cross the blood-brain barrier (BBB) and exert neuroprotective effects, modulating neuroinflammation and promoting neuronal health.39–41 Therefore, a reduced presence of this microbe could potentially impact the development of AD. However, further research is needed to elucidate the specific mechanisms by which Alloprevotella contributes to disease processes and to identify potential therapeutic targets. In addition, a recent report exemplifies how fecal microbiota transplantation elucidates the relationship between gut microbiota and AD. 42 In this study, fecal microbiota from AD model mice was transplanted into germ-free mice. The recipients exhibited cognitive impairments and increased amyloid-beta plaque deposition, suggesting that the microbiota from AD mice can influence AD-related phenotypes. The study indicated that alterations in the microbial community can directly impact disease progression through mechanisms such as inflammation and amyloid-beta accumulation.
The microbes involved in the biosynthesis of the most prevalent enzymes in the pathways “Arginine biosynthesis,” “Alanine, aspartate and glutamate metabolism,” “Other glycan degradation,” “One carbon pool by folate,” and “Cholinergic synapse”, as illustrated in Figure 5, are as follows. Escherichia coli and Bacillus subtilis are well known to contribute to the first pathway; Escherichia coli and Corynebacterium glutamicum are involved in the second; Clostridium and Ruminococcus participate in the third; Escherichia coli and Streptococcus pneumoniae are linked to the fourth; and Pseudomonas aeruginosa is associated with the fifth pathway.43–47
Overall, while the aforementioned genera have been studied in the context of gut microbiota composition and various diseases, including metabolic disorders and inflammatory conditions, their specific roles or implications in AD are not well defined. Further research focusing on the gut-brain axis and microbiota-brain interactions may provide more insights into whether and how these genera could influence AD progression or mitigation strategies. Therefore, we propose that taxonomic studies may not definitively establish a direct link between a particular microorganism and the development of AD. Consequently, our investigation has prioritized the examination of functional profiles in male WT mice and age-matched transgenic AD model male mice using the PICRUSt.
Metabolites and Alzheimer's disease
Extensive prior research has established that advancing age is the most significant risk factor for AD and related dementias. 48 As previously highlighted in AD patients, metabolic perturbations and pathogenesis encompass disruptions in glucose metabolism and ACh biosynthesis, accompanied by reductions in acetyl-CoA, succinate, ATP, hexadecanoate fatty acid levels, and alterations in the glycosylation process.49–51
The interplay among these factors is essential for sustaining neuronal health and functionality. 16 This pathological process starts with a decline in glucose biosynthesis and metabolism, leading to reduced production of acetyl-CoA and ATP. Choline O-acetyltransferase (EC:2.3.1.6) uses these compounds to generate ACh within neurons to ensure synaptic plasticity (Supplemental Figures S44 and S69). However, impaired mitochondrial function results in lower availability of acetyl-CoA and ATP, which directly diminishes ACh synthesis rates.
This cholinergic deficit, a characteristic of AD, contributes to cognitive decline. 21 Under these severe conditions, even if choline is present in sufficient amounts, neurons cannot support the biosynthesis of ACh required for transport to postsynaptic cells. This insufficiency exacerbates energy deficits, synaptic dysfunction, and triggers a cascade of mitogen-activated protein kinase (MAPK) events. These interconnected dysfunctions collectively accelerate the progression of AD (Figure 7). Within this context, several metabolites implicated in preventing cognitive decline and the progression of AD are biosynthesized in greater abundance in the gut microbiome of WT mice compared to APP/PS1 mice as detailed below.
Glucose
Glucose is recognized as the primary energy source necessary for ATP synthesis in the brain, while acetyl-CoA is primarily viewed as a fundamental energy precursor that supports brain cell viability. 11 Glucose plays a crucial role in preventing neurotoxic damage and exerts neuroprotective effects in various brain pathologies.52–54 Clinical studies have shown that a reduction in cerebral glucose metabolism (hypometabolism) precedes the onset of AD pathology and symptoms and is more pronounced than the changes observed in normal aging.55–57 Since energy production from glucose is essential for most brain activities and for maintaining cellular homeostasis, an inadequate energy supply to brain cells is likely to contribute to the neuropathological processes in both aging and age-related dementias such as AD. 58
The majority of energy derived from glucose oxidation is generated in mitochondria, and numerous mitochondrial-dependent functions are found to be impaired during aging and in AD.58–60 Consequently, brain mitochondrial dysfunction is hypothesized to play a crucial role in the pathological hallmarks of AD.59,61
Glucose is the brain's primary energy source, and its metabolism is vital for maintaining proper neuronal function. 54 In AD, there is significant evidence of impaired glucose metabolism, commonly referred to as cerebral hypometabolism. Studies have demonstrated that individuals with AD show decreased glucose uptake and utilization in key brain regions, particularly the hippocampus and cerebral cortex. 62 This reduction in glucose metabolism is evident even in the early, preclinical stages of AD, suggesting a potential role in the initial pathophysiological changes. When glucose metabolism is compromised, ketone bodies can serve as an alternative energy source (Figure 6). This metabolic adaptability involves converting ketone bodies into acetoacetyl-CoA and subsequently into acetyl-CoA, which then enters the TCA cycle to produce ATP. 63 Research indicates that ketone metabolism may compensate for the impaired glucose metabolism in AD, offering an alternative energy source to maintain neuronal function. 64 In the present study, both glucose and ketone metabolic pathways appear to be disrupted in the gut microbiome of APP/PS1 mice with AD.
Moreover, extensive evidence suggests the existence of brain insulin resistance in AD.65,66 Insulin signaling is vital for the uptake of glucose in the brain, and impairments in this pathway can lead to reduced glucose utilization. These disruptions set off a series of events that eventually culminate in cognitive decline. 67 In this study, glucose metabolism was shown to be impaired in APP/PS1 mice, which serve as a model for early onset AD, when compared to age-matched WT mice (Figure 8, and Supplemental Figures S26 and S27). This impairment was observed across seven metabolic pathways within two interacting pathways, utilizing precursors such as sucrose, dextrin, isomaltose, maltose-6P, trehalose-6P, D-B-glucoside, and cellulose (Figure 8). Functional analyses of the microbiomes from both groups indicated a reduction in enzyme activities driving these pathways by at least 0.2-fold in APP/PS1 mice (Supplemental Table S9).
Acetyl-CoA
Acetyl-CoA is a critical central metabolic molecule involved in numerous cellular reactions, including energy production. 68 It serves as a fundamental precursor in the citric acid cycle (TCA cycle), contributing to ATP biosynthesis through the “Oxidative phosphorylation” pathway, which converts the energy harnessed from acetyl-CoA into ATP. 69 Any abnormalities in this pathway or during fatty acid β-oxidation can result in mitochondrial diseases. 70 In the context of AD, disruptions in glucose and pyruvate metabolism compromise acetyl-CoA levels, leading to decreased ATP synthesis and subsequent neuronal dysfunction. 71 Acetyl-CoA is produced from glucose via glycolysis and the pyruvate dehydrogenase complex, or from fatty acids through β-oxidation. 69
Energy deficits are particularly detrimental to neurons, which have high energy requirements. 50 The acetyl-CoA pool is essential for maintaining the brain's balance between anabolic and catabolic processes. 72 Preserving this pool is vital for neuronal survival, facilitated by glycolytic glucose metabolism. Thus, acetyl-CoA acts as a central molecule in various interconnected pathways, serving as a primary energy precursor. Additionally, acetyl-CoA is involved in the biosynthesis of fatty acids, cholesterol, and phospholipids, which are crucial for neuronal myelination, cell growth, remodeling, and maintaining cellular membrane integrity.72,73 Acetyl-CoA also serves as a precursor for synthesizing ACh, a neurotransmitter critical for learning and memory. Deficits in cholinergic function are characteristic of AD, and reduced availability of acetyl-CoA can exacerbate these deficits. 21 Recent research has highlighted a connection between excessive microglial activation, brain inflammation, and neuronal death. 74 Microglia, are a type of neuroglia (glial cell) that host the brain and spinal cord and considered as the brain's resident immune cells, which play a crucial role in maintaining brain health by clearing harmful debris. 75 Interestingly, the enzyme D-glycero-alpha-D-manno-heptose 1-phosphate guanylyltransferase (EC:2.7.7.71, HddC) involved in the ‘Lipopolysaccharide biosynthesis’ pathway is overproduced in WT mice. This enzyme is crucial in the synthesis of LPS in Gram-negative bacteria. 76 However, the enzyme directly responsible for synthesizing the precursor of the inner core oligosaccharide is ADP-glyceromanno-heptose 6-epimerase (AGME) (Supplemental Figure S39). 23 We hypothesize that the overproduction of HddC may lead to reduced production of AGME, thereby decreasing LPS synthesis. This reduction in LPS production could potentially lower microglial activation, which might slow the progression of AD. This finding was evidenced in research where the administration of choline to the APP/PS1 AD mouse model successfully halted cognitive decline. 77 Considering that the body's natural synthesis of choline may occasionally prove inadequate to meet physiological requirements, augmenting its production by the gut microbiome appears to delay the onset of AD, as indicated by findings from this study. Choline, resembling a B-vitamin, is primarily synthesized by the human body, but levels often do not satisfy the brain's nutritional requirements. 74 It also contributes to cell membrane integrity and plays a pivotal role in regulating gene expression. Emerging evidence suggests that the current recommended daily intake of choline may not adequately support optimal brain health and cognition. 78 Extensive research over several decades has shown that supplementing the maternal diet (during gestation and lactation) with choline profoundly benefits offspring brain health and cognition. 79
Succinate
Succinate plays a critical role in the TCA cycle, where it is involved in ATP production through oxidative phosphorylation. As organisms age, there is a decline in mitochondrial function, including the TCA cycle efficiency, which impacts energy production. Succinate levels can fluctuate under conditions of mitochondrial dysfunction, potentially contributing to age-related decline in cellular energy metabolism. 80 In AD, there is accumulating evidence suggesting alterations in metabolic pathways, including the TCA cycle. Succinate levels have been found to be dysregulated in the brains of individuals with AD, possibly contributing to mitochondrial dysfunction and oxidative stress observed in these patients. 81 Mitochondrial dysfunction is a hallmark of many neurodegenerative diseases, including AD. Succinate accumulation or dysregulation can impair mitochondrial function, leading to neuronal damage and cognitive decline observed in AD. 82 Targeting metabolic pathways involving succinate has emerged as a potential therapeutic strategy for neurodegenerative diseases. Modulating succinate levels or its downstream signaling pathways could potentially mitigate mitochondrial dysfunction and inflammation, offering new avenues for treatment. 83
Succinate dehydrogenase is an enzyme involved in the citric acid cycle (Krebs cycle), which participates in the electron transport chain and catalyzes the oxidation of succinate to fumarate, coupled with the reduction of ubiquinone to ubiquinol contributing to the generation of a proton gradient across the inner mitochondrial membrane. 80 This gradient is essential for ATP synthesis via ATP synthase (Complex V). Recent reports suggest that fumarate can act as an antioxidant by scavenging free radicals and protecting against oxidative damage. 84 While ornithine's role in the urea cycle and ammonia detoxification is well-established, its direct involvement in AD remains unclear. 85 Research continues to explore the intricate metabolic pathways and amino acid metabolism in the context of aging and neurodegenerative diseases. For a comprehensive understanding, future studies may elucidate whether ornithine or its metabolic pathways play specific roles in AD pathology or if they are merely part of broader metabolic changes associated with aging and neurodegeneration. In summary, while ornithine's primary role is in ammonia detoxification rather than directly in AD, understanding its metabolic context and potential indirect effects on brain health remains an area of ongoing research. We can claim that succinate is intricately involved in energy metabolism, oxidative stress, inflammation, and mitochondrial function, all of which play crucial roles in aging and neurodegenerative diseases like AD. However, further research into the specific mechanisms linking succinate dysregulation to these conditions could lead to novel therapeutic interventions.
Adenosine triphosphate (ATP)
ATP serves as the primary cellular energy carrier, indispensable for a multitude of cellular functions, particularly in neurons. 50 Neurons necessitate substantial ATP for maintaining ion gradients, neurotransmitter release, and synaptic plasticity. 17 In AD, mitochondrial impairment results in diminished ATP synthesis, which contributes to neuronal dysfunction and synaptic failure. 86 Furthermore, compromised mitochondrial function and reduced ATP levels can escalate oxidative stress, a pivotal contributor to AD pathology. 18 Reactive oxygen species generated during mitochondrial dysfunction have the potential to inflict damage on cellular constituents such as lipids, proteins, and DNA, thereby exacerbating neuronal injury and facilitating the progression of AD. 19 Considering the pivotal role of ATP as an energy source in addressing AD, an experiment involving two neuroprotective compounds, J147 and CMS121, was conducted. 68 J147 has also been studied as an anti-aging treatment in C. elegans. 87 These compounds primarily target ATP synthase, eliciting neuroprotective responses that include activating AMP-activated protein kinase (AMPK). 88 The principal KEGG pathway affected by aging and the administration of these compounds is ‘Oxidative phosphorylation’. In line with these findings, both substances have been shown to influence the expression of genes related to brain mitochondria across four of the five complexes of the electron transport chain (ETC) within the ‘Oxidative phosphorylation’ pathway. 89 These genes encompass subunits of complex IV, such as MT-CO1, MT-CO2, and MT-CO3 encoding cytochrome c oxidase (EC:7.1.1.9), as well as subunits of complex V, including MT-ATP6 and MT-ATP8, which encode ATP synthase (EC:7.1.2.2). In this study, there was a reduced abundance of these enzymes in the gut microbiome of AD mice (Supplemental Figure S33 and Supplemental Table S9). This suggests a plausible role for components of the gut microbiome in upregulating these enzymes by promoting the expression of their corresponding genes. Another possibility is that the gut microbiome directly supplies these enzymes to enhance this pathway and mitigate the onset of AD. Notably, J147 and CMS121 share additional similarities, as they enhance cognitive function in SAMP8 mice 68 and maintain elevated levels of acetyl-CoA by inhibiting acetyl-CoA carboxylase 1 (ACC1). 90 This process enhances histone acetylation specifically in histone H3 of cultured neurons and SAMP8 mice, which is pivotal for the formation of memories.
Choline and acetylcholine
Choline is essential for the synthesis of ACh, a neurotransmitter crucial for memory and learning functions. 51 The deficiency of ACh is a well-established pathological hallmark of AD, initially proposed as the cholinergic hypothesis. 51 AD is characterized by cognitive decline and has long been associated with dysregulation of the cholinergic system. The levels of ACh are influenced by the central cholinergic nervous system, which regulates its synthesis and release in neurons (postsynaptic cells) 91 Dementia severity correlates with synaptic loss between the basal forebrain and target tissues of the hippocampus and cortex, which is a primary contributor to memory decline. 92 Recent clinical evidence reveals significant neurodegeneration in AD brains, accompanied by reduced cholinergic neurons and severe ACh deficiency, with diminished activity of ACh transferase (EC:2.3.1.6). 93 In this study, a decrease in the abundance of this enzyme was observed in APP/PS1 mice, suggesting a potential role of this enzyme from the gut microbiome in the development of AD (Figure 6 and Supplemental Figure S69, and Supplemental Table S9).
Other metabolites/enzymes
The metabolite 4-Guanidinobutanoate (GBB) is involved in the metabolism of guanidino compounds, particularly in the context of creatine synthesis and metabolism. 94 While direct research linking GBB to AD or aging-related neurodegeneration is limited, we can say that GBB is an intermediate in the biosynthesis of creatine, a molecule critical for cellular energy metabolism, especially in tissues with high energy demands such as the brain. Note that creatine is involved in ATP buffering, neuroprotection, and maintenance of cellular energy homeostasis in the brain. 95 Creatine has neuroprotective properties and may mitigate oxidative stress and mitochondrial dysfunction, which are implicated in the pathogenesis of neurodegenerative diseases like AD.94,95 Moreover, fatty acids, specifically hexadecanoate, are crucial lipids essential for optimal brain function. Aging and AD generally alter neuronal structure, function, and signaling of which lipids play essential roles in forming cell membranes, insulating nerve fibers, and acting as precursors for signaling molecules, e.g., neurotransmitters.49,50 As aging and AD exhibit metabolic features such as heightened oxidative stress and disrupted metabolic function, we see these discrepancies coincide with changes in lipid structure and contribute to cognitive decline. On the other hand, nutraceuticals, particularly those influencing brain lipid metabolism through synergistic nutrient interactions, show potential for alleviating AD symptoms. These lipid-based therapies aim to restore a healthy brain lipid profile. The gut microbiota are also recognized as a source of beneficial lipids, suggesting another therapeutic approach for reversing AD symptoms via influencing systemic lipid metabolism and potentially impact brain lipid profiles through the gut-brain axis.96–99 Based on the results of the present study, we suggest that this strategy could involve enzymes and metabolites within the “Lipid metabolism” subcategory and related subcategories such as “Energy metabolism”, “Carbohydrate metabolism”, “Amino acid metabolism”, and “Nervous system”. Modulating these lipid-based pathways may balance nutrient and energy levels, thereby enhancing cognitive function and alleviating symptoms of AD (Supplemental Table S10).
Additional enzymes of interest include UDP-N-acetylgalactosamine diphosphorylase (AGX1) (EC:2.7.7.83) within the “Amino sugar and nucleotide sugar metabolism” pathway, and Beta-1,4-mannosyl-glycoprotein 4-beta-N-acetylglucosaminyltransferase (MGAT3) (EC:2.4.1.144) involved in “N-Glycan biosynthesis” (Supplemental Figures S28 and S37, respectively). These enzymes play critical roles in glycosylation processes, potentially implicating them in AD pathology by influencing glycosylation patterns of proteins such as amyloid-beta protein precursor and tau in the nervous system.20,100 Research has implicated dysregulated glycosylation in neurodegenerative diseases that contributes to the formation of neurotoxic amyloid-beta plaques and tau tangles, key pathological features of AD.101,102 These two enzymes are hypothesized to influence AD progression by affecting protein aggregation, synaptic function, and neuroinflammation.103,104 Furthermore, alpha-mannosidase (MAN2B1) (EC:3.2.1.24) of pathway “Other glycan degradation” (Supplemental Figure S38) is involved in the hydrolysis of terminal alpha-linked mannose residues from oligosaccharides and glycoproteins. It participates in the breakdown of glycoproteins, influencing cellular processes critical in neurodegenerative diseases. 20 Deficiencies in MAN2B1 can lead to lysosomal storage disorders, 105 impacting cellular homeostasis and potentially contributing to neurodegeneration observed in AD. Dysregulation of MAN2B1 activity may affect protein folding and aggregation processes linked to AD pathogenesis. 106 NAD(P)H dehydrogenase (quinone) (NQO1) (EC:1.6.5.2) of pathway “Ubiquinone and other terpenoid-quinone biosynthesis” (Supplemental Figure S47) has been implicated in AD as it is involved in protecting cells from oxidative stress by detoxifying reactive oxygen species and maintaining cellular redox balance. 107 Oxidative stress is a hallmark of AD pathology, contributing to neuronal damage and cognitive decline. Like the two other enzymes AGX1 and MGAT3, NQO1 modulates the production and clearance of amyloid-beta toxicity, affecting disease progression. 108 NQO1 also has anti-inflammatory properties and may regulate neuroinflammatory responses implicated in AD. 107 Although NAD(P)(+) transhydrogenase (EC:7.1.1.1) of pathway “Nicotinate and nicotinamide metabolism” (Supplemental Figure S50) was reported to play a role in maintaining cellular redox balance and energy metabolism, there are no prior reports that directly link this enzyme to AD pathology. In terms of the enzymes with low abundance in APP/PS1 mice in categories “Genetic Information Processing” and “Environmental Information Processing”, they showed no established link with AD based on current scientific literature.
Microbial metabolites, such as specific enzymes and compounds like succinate, are increasingly recognized for their potential effects on the brain, particularly through interactions with the BBB.109–111 Some microbial metabolites, including short-chain fatty acids (SCFAs), can cross the BBB and influence brain activity, which could be significant for neurodegenerative conditions like AD.40,41 Although the exact pathways by which these metabolites traverse the BBB and impact the central nervous system (CNS) are still being explored, current research indicates that they may play a role in modulating neuroinflammation and neurodegeneration. 41 This is believed to occur through interactions with brain-resident immune cells, such as microglia. Furthermore, metabolites like succinate may alter the BBB's integrity, potentially allowing other substances or inflammatory mediators to enter the brain and exacerbate disease processes.109,112
While it is acknowledged that the functional interplay between the gut microbiome and CNS functions remains an evolving field, recent reports have elucidated potential pathways through which the above mentioned metabolites may cross the BBB and influence neuronal health. 113 Mechanistic studies that delineate the transport mechanisms of some of these metabolites across the BBB elucidated the presence of various transport systems, such as the sodium-dependent glucose transporter (SGLT1) and the L-type amino acid transporter (LAT1), which facilitate the passage of glucose and other metabolites from the bloodstream into the CNS. 114 By integrating these insights, we can claim a more comprehensive overview of the gut-brain axis and its implications for neurodegenerative diseases. For example, SCFAs, derived from gut microbiota fermentation, were highlighted in recent investigations to cross the BBB via monocarboxylate transporters (MCTs), specifically MCT1 and MCT2.39,115–117 These transporters facilitate the uptake of SCFAs into astrocytes and neurons, where they can modulate neuroinflammatory pathways and energy metabolism. 39 The metabolite butyrate was also shown to enhance mitochondrial function and promote neuroprotection, suggesting a direct link between gut-derived metabolites and CNS health. 118 Several other studies have indicated that certain gut bacteria, such as those from the genera Faecalibacterium and Roseburia, can produce SCFAs that can influence brain health through the gut-brain axis. 119 As gut microbiome was documented to contribute to the pool of available acetyl-CoA, it produces SCFAs like acetate, which can be converted to acetyl-CoA. Recent studies indicate that certain transporters may facilitate crossing of acetyl-CoA to the BBB. 120 In terms of ACh, its role has been well documented in the context of cognitive function, where its receptors are expressed in various brain regions implicated in learning and memory, thus reinforcing the claim that gut microbiota may indirectly influence CNS processes through cholinergic signaling pathways. 121 While the connections between gut microbiota-derived metabolites and CNS functions remain an area ripe for exploration, these emerging evidence might support the plausibility of these interactions.
A very recent study has shown that mitochondrial function can vary significantly between males and females, influencing energy metabolism, oxidative stress responses, and potential susceptibility to neurodegenerative diseases. 122 The study indicated that females often exhibit more efficient mitochondrial respiration and greater resilience to oxidative stress compared to males. This disparity may affect how each sex responds to changes in gut microbiota and their metabolites, potentially influencing the progression of AD.
Conclusion and recommendation
In conclusion, our study underscores the critical role of Acetyl-CoA and other upstream and downstream metabolites in avoiding the impact of energy deficits, synaptic dysfunctions, and acceleration of the onset and progression of AD. 123 Acetyl-CoA serves as a vital metabolic hub connecting glycolysis to oxidative phosphorylation, facilitating the conversion of carbohydrates into ATP 69 Moreover, it plays a crucial role in ACh production, essential for neuronal synaptic plasticity, long-term memory consolidation, and the prevention of MAPK signaling cascades linked to cognitive decline.80,124,125 Our findings further validated previous hypotheses regarding the intricate dysregulation of metabolic pathways involving glucose, acetyl-CoA, succinate, ATP, ACh, and lipids, as well as glycosylation processes implicated in AD pathogenesis. Central to AD pathology is the compromised glucose metabolism observed in APP/PS1 mice, characterized by reduced enzyme activities across multiple downstream metabolic pathways.
Given the common impairments in energy production pathways such as glycolysis, the TCA cycle, and oxidative phosphorylation in AD, our study suggests that the gut microbiome significantly influences these metabolic processes to prevent AD onset and progression. Recent findings support our hypothesis, suggesting that the gut microbiome may compensate for these metabolic deficiencies, potentially reducing the risk or slowing the progression of neurological disorders such as AD.126,127 However, our study emphasized comprehensive metabolic processes that put gut microbiome as a major influencer of the disease. This insight opens avenues for developing novel therapeutic strategies aimed at restoring metabolic function and potentially slowing AD onset and progression through the administration of gut microbiome-derived enzymes and metabolites. A comprehensive understanding of these intricate interdependencies is critical for advancing effective therapeutic approaches targeting AD.
Supplemental Material
sj-docx-1-alr-10.1177_25424823241309024 - Supplemental material for Gut microbiome synthesizes important core metabolites to prevent cognitive decline and mitigate onset and progression of Alzheimer's disease
Supplemental material, sj-docx-1-alr-10.1177_25424823241309024 for Gut microbiome synthesizes important core metabolites to prevent cognitive decline and mitigate onset and progression of Alzheimer's disease by Nouf S Al-Abbas and Nehad A Shaer in Journal of Alzheimer's Disease Reports
Footnotes
Author contributions
Nouf S Al-Abbas (Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Software; Supervision; Validation; Visualization; Writing – review & editing); Nehad Shaer (Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Software; Supervision; Validation; Visualization; Writing – original draft; Writing – review & editing).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this study were originally generated by OE Biotech Co., Ltd (Shanghai, China) and are openly available in the BioProject database of the National Center for Biotechnology Information (NCBI), under project number PRJNA687556 (![]() ) as cited in the article: Zhang Z, Tan X, Sun X, et al. Isoorientin affects markers of Alzheimer's disease via effects on the oral and gut microbiota in APP/PS1 mice. J Nutr 2022; 152: 140–152.
) as cited in the article: Zhang Z, Tan X, Sun X, et al. Isoorientin affects markers of Alzheimer's disease via effects on the oral and gut microbiota in APP/PS1 mice. J Nutr 2022; 152: 140–152.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
