Abstract
Background:
Alzheimer's disease (AD) is a growing public health problem in the aging population, with limited treatment options. We previously reported that Centella asiatica herb water extract (CAW) attenuates cognitive decline in murine models of AD and aging.
Objective:
To explore changes in the hippocampal metabolome associated with CAW's modulation of cognitive function and amyloid-β (Aβ) plaque load in aged Tg2576 and wild-type (WT) mice.
Methods:
We compared cognitive function, hippocampal Aβ plaque burden, and hippocampal metabolite profile in 20-month-old Tg2576 female mice and their WT littermates following 3–5 weeks treatment with CAW (0, 200, or 1000 mg/kg/d p.o.). Cognitive testing included contextual fear response (CFR) and novel object recognition task (NORT). Aβ plaque burden was measured via immunohistochemistry. Metabolomic profiles of mouse hippocampi were obtained using liquid chromatography coupled with high resolution tandem mass spectrometry.
Results:
CAW treatment resulted in dose-related improvements in CFR and NORT performance of Tg2576 and WT mice. However, while CFR correlated with neurosignaling and glycosylated ceramide levels, NORT performance correlated with lysophosphatidylcholines and oxidized metabolites, and Aβ accumulation was linked to elevated excitatory and suppressed inhibitory neurotransmission. Only a subset of the metabolite changes induced by CAW in Tg2576 mice represented a reversal of metabolite differences between Tg2576 and WT mice, suggesting the involvement of other pathways in CAW's cognitive effects.
Conclusions:
Mechanisms underlying CAW's cognitive effects extend beyond reversing metabolic effects of Aβ accumulation. The data support the potential use of CAW to manage memory challenges in aged individuals with or without AD.
Keywords
Introduction
Alzheimer's disease (AD), the most common cause of dementia worldwide, is characterized by brain pathological changes of extracellular plaques of amyloid-β peptides (Aβ), intracellular tangles of abnormally phosphorylated tau protein, neuronal death, and neuroinflammation. 1 Age is the strongest risk factor for AD and consequently societal burden due to AD is increasing along with collective aging of the world population. 2 Estimates predict 13.8 million Americans will be affected by AD by the year 2060. 1 Several transgenic mouse models have been developed to aid the discovery of new drugs for AD. 3 Tg2576 and 5XFAD mice are two transgenic models of Aβ accumulation that contain the human Aβ precursor protein gene (hAβPP) with the familial “Swedish” mutation which facilitates the cleavage of Aβ precursor protein (AβPP) to the pathogenic Aβ42. This results in widespread formation of the Aβ plaques reminiscent of AD in several brain regions of these mice including the hippocampus.3–6 Tg2576 mice, used in the present study, typically develop these Aβ plaques and age-dependent cognitive impairments starting from 10–11 months of age. 7
With an urgent need for novel disease-modifying therapies, 8 natural products are being investigated as possible sources of treatments to ameliorate AD progression, either as isolated compounds or complex extracts.9–11 In addition, plants with known ethnobotanical uses provide a greater potential for putative treatment discovery in screening assays than randomly chosen natural products.12,13 Here we continued our evaluation of the ethnobotanical plant Centella asiatica (L.) Urban; family Apiaceae (CA) for its potential to reduce cognitive decline associated with AD. CA is highly regarded in the Indian Ayurvedic tradition as “rasayana” or rejuvenating herb with the ability to improve memory, 14 demonstrating neurological and neuroprotective effects. 15 In clinical studies, CA herb or CA extracts improved cognitive function in healthy middle-aged adults, 16 elderly adults, 17 and elders with mild cognitive impairment (MCI). 18 A clinical trial examining safety and target engagement of CA in MCI and mild AD is currently underway (NCT05591027).
In preclinical studies, CA improves cognition in rodent models of oxidative stress,19–21 aging, 22 and AD.23–25 In our own earlier studies, oral administration of a CA water extract (CAW) at 200 mg per kg of body weight per day (mg/kg/d) for 5 weeks improved cognitive deficits in both the Tg2576 23 and 5XFAD24,25 transgenic mouse models of AD. 3 CAW (200 mg/kg/d) improved learning and memory of Tg2576 mice in the Morris water maze paradigm without altering Aβ levels in the cortex in vivo or inhibiting acetylcholinesterase enzyme in vitro. 23 Further exploration of the putative mechanisms of action of CAW demonstrated that treatment of neuroblastoma cells and/or primary neurons with CAW in vitro improves antioxidant response, ameliorates mitochondrial dysfunction, and increases dendritic arborization.26–28 Relevant pathways have also been observed in vivo, showing increased expression of antioxidant response element genes, mitochondrial electron transport genes, and synaptic genes in the brain following oral administration of CAW (200 mg/kg/d) in young and aged wild-type (WT) mice 22 and at this and higher doses (200, 500, and 1000 mg/kg/d) in 5XFAD mice.24,25
To validate the effectiveness of 1000 mg/kg/d CAW on cognition observed in 5XFAD mice in a second mouse AD model, and elucidate possible dosing effects, we utilized both the 200 mg/kg/d and 1000 mg/kg/d doses in the Tg2576 mouse model and their WT littermates in the present study. Effects on Aβ plaque burden were assessed by immunohistochemistry (IHC), rather than ELISA as used in our earlier study in this model, 23 to visualize Aβ plaque distribution in the cortex and hippocampus. Genotypic and CAW treatment effects on hippocampal-dependent contextual fear memory were evaluated using the conditioned fear response (CFR) paradigm.24,25 Using the novel object recognition task (NORT)24,29 we assessed object recognition memory for which both hippocampal and cortical inputs are implicated.
Metabolomic analyses have played an increasingly important role in the evaluation of metabolic pathway changes related to AD using biological samples from humans and rodent models,30,31 including Tg2576 mice.31–34 In humans, extensive metabolite changes were reported in plasma, cerebrospinal fluid (CSF), and brain tissue, which were linked to changes in amino acid, lipid, and nucleic acid metabolism, 35 consistent with the emerging concept that AD is a metabolic disorder of the brain. 36 We have previously reported 37 baseline differences in the cortical metabolome of WT and 5XFAD mice,24,25 as well as changes to the metabolome observed following treatment with CAW (200, 500, and 1000 mg/kg/d). We have also reported that 20 month old Tg2576 mice, which are known to have memory deficits associated with Aβ plaque formation, 7 have multiple metabolic challenges 34 compared to age-matched WT littermates. These changes include accumulation of oxidized metabolites, a shift in the ratio of metabolites toward more excitatory, and less inhibitory, neurosignaling metabolites, and shifts in phospholipid and ceramide metabolites, which may promote signaling, inflammation, and survival of neurons. The observed metabolic shifts in the hippocampal metabolites of Tg2576 mice suggest a potential role of hippocampal metabolism in their behavioral deficits. Additionally, processes giving rise to these metabolites may represent druggable targets to manage neurocognitive challenges.
The objective of the current study was to explore the cognitive effects of CAW at two doses (200 and 1000 mg/kg/d) in 20-month-old female Tg2576 mice and their WT littermates (previously only 200 mg/kg/d was explored in these groups). In addition, we intended to gain insights into hippocampal metabolic changes that may accompany the expected cognitive effects of CAW in mice of both genotypes. We targeted the hippocampus for exploration as it is a site for Aβ deposition38–40 and metabolic changes 34 in the Tg2576 model, and has an established role in contextual fear memory,41–44 as well as in novel object recognition memory. 45 We selected hippocampal tissue from untreated and 1000 mg/kg/d CAW-treated mice for comparative metabolomics analysis due to the greater effectiveness of the higher dose in cognitive function in the 5XFAD model. 24 Cognition and hippocampal metabolomics were also assessed in untreated and CAW-treated WT littermates in order to confirm cognitive deficits in the Tg2576 animals and to explore differential effects of CAW in these two genotypes. We hypothesized that CAW administration would result in a dose dependent decrease in hippocampal Aβ deposition and an improvement of cognitive function in Tg2576 mice, and that these effects would be associated with changes to the hippocampal metabolome, the nature of which would be evaluated.
Methods
Production of CAW
Oregon's Wild Harvest (Redmond, OR) supplied dry CA herb (Lot# GOT-03193c-OHQ01). Identity of plant species was validated in our laboratory using thin layer chromatography with comparisons to previously used lots22,23,46 and examples in the literature. 47 To produce CAW, dried CA leaves and stems (160 g) were boiled under reflux in distilled water (2 L) for 2 h, plant debris were filtered out, and the extract was freeze dried to yield ∼10–13% dry CAW, which was stored at −20 °C until use. Original plant material voucher samples were deposited at the Oregon State University Herbarium (OSC-V-258627) and at OHSU, in addition to voucher samples of the several CAW batches used for these experiments (CAW F-N). CAW was given ad libitum to Tg2576 and WT littermates in their water bottles. Concentrations of CAW 0, 2, 10 mg/mL were prepared in the drinking water to deliver approximately 0, 200, and 1000 mg/kg/d doses respectively, based on previously observed water consumption rates. 24
Animals and experimental design
These studies were conducted in accordance with NIH Guidelines for the Care and Use of Laboratory Animals and approved by the Institutional Animal Care and Use Committee of the Portland VA Medical Center (IACUC #: 3260–17).
A 3 × 2 factorial design was used that included two mouse models (WT and Tg2576 mice) and three CAW doses (0, 200, and 1000 mg/kg/d). These doses were based on previous studies where we had observed cognitive effects of CAW in WT or Tg2576 mice (200 mg/kg/d CAW) and 5XFAD mice (200, 500, and 1000 mg/kg/d).22–24
Mice used in this study were on a C57BL/6:SJL background, with alternate breeding to WT SJL or C57BL/6 dams for each generation. The female Tg2576 and WT littermates used in these studies were the result of a cross of Tg2576 heterozygous sires with SJL WT dams (Jackson Laboratory, Bar Harbor, ME). Transgene status was assessed using PCR of transgenic human AβPP (hAβPP; possessing the Swedish mutation – KM670/671NL) from tail sample DNA. Animals were maintained in a climate-controlled environment, on a 12-h lighting schedule. Female Tg2576 mice (19.0 ± 0.04 months of age) were given PicoLab Laboratory Rodent Diet 5L0D (LabDiet, St. Louis, MO) and water or CAW-supplemented water ad libitum for a total of 5 weeks. Changes in water level were noted each day to monitor water consumption; due to group housing this could only be noted on a per cage basis and not for each individual mouse. NORT was performed after 3 weeks of treatment, and subsequently CFR testing during week 5, and animals euthanized after 5 weeks of treatment. Right brain hemispheres were fixed in 4% formaldehyde for immunohistochemical analysis, while the left hippocampi and cortex were dissected and snap frozen for metabolomics analyses. The experimental design is summarized in Figure 1.
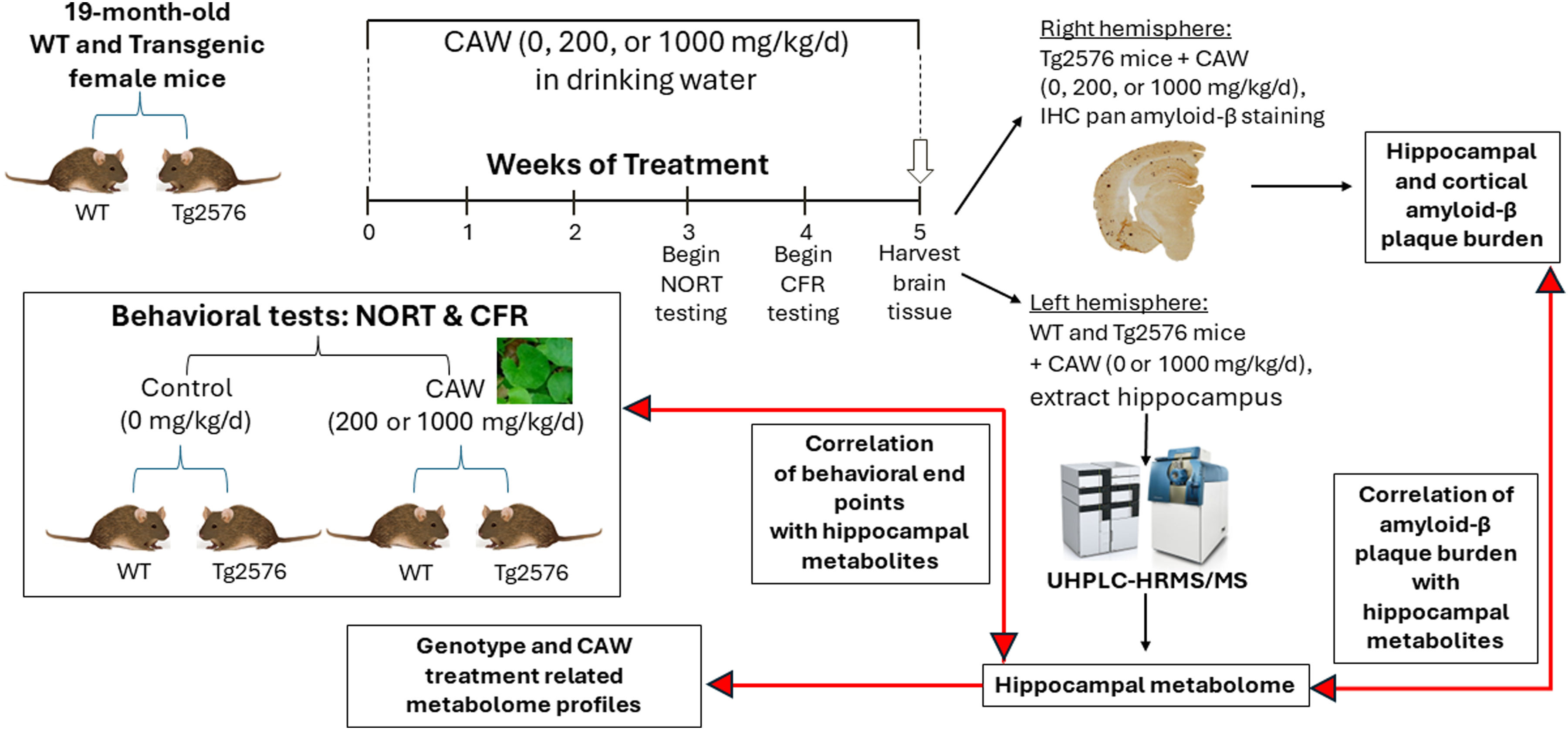
Experimental methodology workflow. Aged (19-month-old) female transgenic Tg2576 and WT littermate mice were treated with 0, 200, or 1000 mg/kg/d CAW for 5 weeks. Behavioral testing was performed on all treatment and genotype groups (inset). At 3 weeks of treatment, novel object recognition testing (NORT) was performed over 4 days. At 4 weeks of treatment, conditioned fear response (CFR) testing was performed over 2 days. After 5 weeks of treatment, brain tissues of all animals were harvested. Right brain hemispheres from transgenic mice were fixed and sectioned for immunohistochemistry (IHC) staining for pan amyloid-β. Left brain hemispheres of 0 and 1000 mg/kg/d CAW-treated WT and Tg2576 animals were dissected to extract the hippocampi for untargeted analysis by ultra-high performance liquid chromatography coupled with high resolution tandem mass spectrometry (UHPLC-HRMS/MS). The resulting metabolome profiles were independently compared to amyloid-β plaque burden, and NORT and CFR test results to elucidate any underlying correlations between the test results and changes in specific metabolites.
Novel object recognition task (NORT)
To test recognition memory, we used the NORT, which takes place over four days (two days open field, two days NORT testing). During the two-day open field habituation, each mouse participated in a 10-min per day acclimatization period in the empty testing apparatus (38 cm×38 cm×64 cm high). On day three, two identical objects (A1 and A2) were placed inside the apparatus in the NW and SW corners, 10 cm away from the walls. Objects were about 6 cm by 6 cm and 10 cm high and made from non-porous materials (plastic, metal, glass) for ease of cleaning/deodorizing. Mice were allowed to explore the objects for three 10-min trials, separated by 1 h. Two hours after their last habituation trial, one object was replaced with a previously unexplored object (B1) in an identical position. Mice were allowed to explore the familiar and novel objects for 10 min. Twenty-four hours after their last habituation period, object B1 was replaced with a new previously unexplored object (B2) in an identical position and mice were allowed to explore these objects for 10 min. The white acrylonitrile butadiene styrene apparatus and objects were wiped down with 70% ethanol in between each animal to disinfect and remove scent cues. Exploration behavior was recorded using Any-Maze Software (Erie, PA) and defined as a front paw or nose within 2 cm without climbing on the object. All videos were manually evaluated, and object exploration times recorded by one trained technician, blinded to experimental conditions. Recognition memory was measured as the difference in exploration between novel and familiar objects in seconds. We utilized the NORT to assess deficits in object recognition in Tg2576 female mice versus WT female littermates and evaluate any potential benefit received from CAW treatment (0, 200, 1000 mg/kg/d CAW: WT – n = 31, 13, 14; Tg2576 – n = 27, 17, 13).
To avoid an undue effect of extreme responses on evaluations in NORT, novel and familiar object responses above 30 s were set at 30 s and novel object preferences above 20 s were set at 20 s. We did not evaluate the novel object discrimination ratio because the responses were not normally distributed.
Conditioned fear response (CFR)
We utilized a contextual fear paradigm to test fear memory, associating an unpleasant experience (i.e., electrical shock) with a specific environment (i.e., testing cage). To evaluate baseline freezing behavior in the test cage, a habituation period of 5 min for each mouse was implemented and freezing time was recorded using Any-Maze tracking software. Immediately following habituation, the contextual fear was established with three consecutive 1-min periods in which a mild electric shock of 0.5 mA for 1 s was randomly administered through the cage floor once in each period. The shock stimulus timing randomness was generated by Fusion v6.0 for SuperFlex Edition Software (Omnitech Electronics Inc., Columbus, OH). Twenty-four hours post-habituation, each mouse was reintroduced into the same testing cage environment and freezing was measured using Any-Maze tracking software over a 5-min interval without any shock stimulus. Contextual fear response was calculated as the difference between the time freezing (in seconds) 24 h post-shock (raw contextual freezing) and baseline habituation freezing (pre-shock) for each animal. The CFR testing cage was wiped down with 70% ethanol in between animals to disinfect and remove scent cues. To evaluate hAβPP transgene and CAW effects on fear-based memory, we characterized the CFR of untreated and CAW-treated, WT and Tg2576 mice (WT: untreated, n = 25; 200 mg/kg/d CAW, n = 13; 1000 mg/kg/d, n = 14; Tg2576: untreated, n = 21; 200 mg/kg/d, n = 9; 1000 mg/kg/d, n = 12).
Immunohistochemistry (IHC) of Aβ plaque burden
At sacrifice, the right brain hemispheres were surgically removed and fixed in 4% formaldehyde/phosphate-buffered saline overnight. Samples were then subjected to increasingly concentrated solutions of sucrose (0–30%) and stored at −80°C until required. Hemispheres were coronally sectioned in 20 µm thick sections via a cryo-histomat and subsequently processed in Netwell inserts in 6-well plates. Sections of similar depths were incubated in a quenching solution of 30% methanol and 0.3% hydrogen peroxide in 1X tris-buffered saline (TBS) to reduce endogenous catalase activity. Sections were then blocked using 10% horse serum, 2% bovine serum albumin, and 0.5% triton in 1X TBS. Sections were stained using a pan-Aβ antibody (44–136, Invitrogen, Carlsbad, CA) at a concentration of 1:1000 overnight at room temperature. A secondary antibody (anti-rabbit; 1:200; Vector Laboratories, Burlingame, CA) was applied to visualize staining. A diaminobenzidine counterstain (Sigma Fast 3,3-Diaminobenzidine Tablet Set, D-4418; Sigma-Aldrich Corp, St. Louis, MO) was applied before sections were mounted on slides and scanned with PrimeHisto XE (Pacific Image Electronics, Torrance, CA). Images collected were quantified using FIJI software. Images were grayscale converted and threshold was set to zero to remove background staining for untreated WT negative control sections. These settings were applied consistently throughout each staining group by one technician blinded to treatment conditions. Staining in the hippocampus and cortex of each sample was measured as area stained (% of total area) within the defined region or as particles per area (cm2), with a particle being defined as a distinct region of pan-Aβ staining as designated by FIJI software.
Metabolite extraction
Methanol and water (liquid chromatography–mass spectrometry (LC-MS)-grade) were purchased from EMD Millipore (Burlington, MA, USA). Formic acid (certified ACS reagent) was from Fisher Chemicals (Suwanee, GA, USA). L-Methionine-(methyl-d3), was used as internal standard (Sigma Aldrich; St. Louis, MO, USA). For metabolomic analysis, hippocampal tissue was analyzed from untreated WT (n = 9) and Tg2576 (n = 12) mice and CAW-treated (1000 mg/kg/d) WT (n = 10) and Tg2576 (n = 12) mice. The extraction protocol was as previously reported 48 with some modifications. Briefly, the samples were homogenized using a PrecellysTM 24 bead ruptor homogenizer (Bertin Technologies, USA). The whole frozen hippocampus was accurately weighed and placed in a 2 mL homogenization tube prefilled with 1.4 mm ceramic beads. Methanol/ethanol (50/50 v/v) containing methionine-(methyl-d3) (1.0 µg/mL) as internal standard was added at 4°C (10 µL of solvent/mg of tissue). The spiked samples were homogenized for 20 s at 5000 rpm three times with 30 s of cooldown in between. Samples were placed at −20°C for 1 h and spun (15,000 g, 10 min, 4°C) to collect the supernatant. The resultant supernatant from each sample was transferred to HPLC vials (Microsolv, Leland, NC, USA) for LC-MS analysis. Aliquots (20 µL) from each sample were mixed to generate a quality control (QC) sample.
Liquid chromatography coupled with high resolution tandem mass spectrometry (LC–HRMS/MS) analysis
Untargeted LC–HRMS/MS analysis was carried out using a method previously described48–50 with some modifications. Briefly, data-dependent acquisition in the positive ion mode was conducted on an AB SCIEX TripleTOF® 5600 mass spectrometer (AB SCIEX, Concord, Canada) coupled to Shimadzu Nexera UHPLC system. Chromatographic separation was performed on an Inertsil Phenyl-3 column (4.6 × 150 mm, 100 Å, 5 µm; GL Sciences, Rolling Hills Estates, CA, USA) held at 50°C. A gradient with two mobile phases was used: Mobile phase A was water (LC-MS grade) with 0.1% v/v formic acid and B, methanol (LC-MS grade) with 0.1% v/v formic acid. After 1 min at 5% B, the linear elution gradient was as follows: 1 min, 5% B; 11 min, 30% B; 20 min, 100% B; 25 min, 100% B; 30 min, 5% B; and 35 min, 5% B. The injection volume was 5 μL with a flow rate of 0.4 mL/min. Samples were randomized before injections. A QC sample was analyzed every five liquid chromatography (LC) runs. The IonSpray voltage was set at 4500 V and the source temperature was 500°C. Period cycle time was 950 ms; accumulation time 100 ms; m/z scan range 100–1400; and collision energy 35 V with collision energy spread of 15 V. Mass calibration of the time-of-flight analyzer was performed automatically after every fifth LC run.
Metabolomics data processing
LC-HRMS/MS data processing was enabled using Progenesis QITM software V2.0 (NonLinear Dynamics, United Kingdom) and used for peak picking, retention time correction, peak alignment, and metabolite identifications/annotations. The metabolite annotation workflow was as described by us previously.49–51 Progenesis supports our in-house metabolite library (based on the Mass Spectrometry Metabolite Library of Standards (MSMLS) which is a collection of >600 small molecule metabolites (IROA Technologies, Bolton, MA, USA)) that allowed Level 1 (L1) annotations. Additional metabolite annotation was received by searching online databases such as METLIN MS/MS library (plugin V1.0.6499.51447), HMDB and LipidBlast, and these annotations are referred to as Level 2 (L2) annotations (See Supplemental Table 1). Annotation confidence was achieved in accordance with reporting criteria for chemical analysis suggested by the Metabolomics Standards Initiative (MSI).52,53 Tentative annotations were made based on accurate mass (error <10 ppm), fragment ion spectral pattern (library score > 50), and isotope pattern (library score > 50). Ions generated from QC samples were retained for annotation and included in the dataset if the coefficient of variation of their abundance did not exceed 30%. Relative quantities of metabolites were determined by calculating their corresponding peak areas.
To account for drift during the metabolomics run, the annotated metabolites were normalized by support vector regression (SVR), a non-parametric machine learning-based algorithm, using MetNormalizer package in R software (version 1.2.5042). The SVR normalization method uses QC samples as a training dataset to build a regression model based on the drift in intensity of each metabolite. Signal changes of the QC samples are used to evaluate drifts in the intensity of metabolites which represent run-to-run fluctuations due to drifts in platform performance. The SVR algorithm predicts and corrects peak intensities of the same metabolite across the samples. In this method, similar to maximal margin algorithm in support vector machine, a non-linear kernel function optimizes and maximizes the margin using supervised learning models for regression. In order to remove unwanted drifts in the measurements of metabolite peaks during data acquisition (intra- and inter-batch), a model for each peak in QC samples (with consideration of injection order and peak intensity) was built using SVR normalization and used to predict the peak intensities of the samples. Finally, each peak intensity in the samples were divided by the predictive peak intensity which were calculated using SVR model to give normalized abundances of each metabolite. 54 Relative standard deviations (RSDs) of peaks were used to evaluate the SVR normalization method using Rfast package in R software (version 1.2.5042).
Statistical analyses
Statistical analyses were performed using SAS version 9.4 software (SAS Institute, Cary, NC). The effects of CAW treatment on CFR, NORT, hippocampal plaque burden, and hippocampal metabolite profile were evaluated as described in Table 1. In Models A-D, the effect of CAW treatment was evaluated using a priori, orthogonal contrasts (using the CONTRAST statement): 200 mg/kg/d CAW-treated versus untreated mice and 1000 mg/kg/d CAW-treated versus untreated mice within each genotype separately and combined. In Model E, the effect of CAW treatment was evaluated using a priori, orthogonal contrasts: 1000 mg/kg/d CAW-treated versus untreated mice within each genotype separately. In Model F, to avoid undue influence of extreme values in hippocampal metabolite measures, Spearman rank correlation coefficient was used to determine the association between hippocampal metabolites with CFR, NORT, and hippocampal plaque burden.
Parameters evaluated, statistical analyses used, and factors investigated in statistical analyses.

As an additional statistical test for the effect of CAW and hAβPP transgene status on CFR and NORT, we grouped the responses in thirds and compared the categorical responses using Fisher's exact test. We also examined correlations of hippocampal plaque burden to CFR and NORT responses (Table 1, Model G). All tests were two-sided. Significance of group differences was determined to be at p ≤ 0.05. We did not adjust for multiple comparisons.
Results
LC-HRMS/MS analysis of hippocampal tissue from CAW-treated and untreated Tg2576 or WT mice
A total of 182 metabolites were unambiguously annotated in hippocampal samples of CAW-treated and untreated Tg2576 and WT mice of which 21 were ceramides (Supplemental Tables 1 and 2). CAW (1000 mg/kg/d) treatment significantly altered the levels of 20 metabolites in Tg2576 mice (10 upregulated and 10 downregulated) and 18 metabolites in WT mice (all but one upregulated) as shown in Table 2.
Annotated hippocampal metabolites significantly altered by the Tg2576 transgene 34 or treatment with CAW (1000 mg/kg/d) for 5 weeks.

Metabolite abundance of CAW-treated and untreated Tg2576 and WT mice metabolites were compared to determine the effects of Tg2576 transgene and CAW treatment. Green and red symbolizes a relative increase and decrease in metabolite levels respectively. Correlations between these significantly altered metabolites and behavioral outcomes and Aβ burden are also shown (positive correlations in green and negative correlations in red). Hippocampal metabolites are classified according to potential biological functions relevant to cognition and brain health.
Cer: ceramide; DG: diglyceride; dUMP: deoxyuridine monophosphate; GMP: guanosine monophosphate; MG: monoglyceride; PA: phosphatidic acid; PC: phosphatidylcholine; PE: phosphatidylethanolamine; PI: phosphatidylinositol; PS: phosphatidylserine; TG: triglyceride.
CAW treatment of aged Tg2576 mice reversed the hAβPP-linked deficit in recognition memory
To evaluate the genotype and CAW treatment effects on recognition memory, we implemented the NORT, which consisted of a training session with two identical objects, then two testing sessions, one session with one replacement novel object at 2 h after habituation, and another session at 24 h after habituation, this time with a novel replacement of the preceding novel object.
Two hours after habituation, untreated Tg2576 mice spent less time exploring each object than untreated WT mice (familiar object: p = 0.05, Supplemental Figure 1A; novel object: p = 0.0002, Supplemental Figure 1B). Twenty-four hours after habituation, untreated Tg2576 mice spent similar time exploring each object than untreated WT mice with no difference at 24 h post-training (familiar object: p = 0.99, Supplemental Figure 1C; novel object: p = 0.50; Supplemental Figure 1D), because fewer untreated WT mice spent time exploring novel objects than 2 h post-habituation (p = 0.006).
Two hours after habituation, CAW treatment decreased exploration of the familiar object at 200 mg/kg/d (p = 0.002) but not at 1000 mg/kg/d (p = 0.80). No effect of CAW treatments was observed on familiar object exploration in both genotypes combined at 200 mg/kg/d (p = 0.62) and at 1000 mg/kg/d (p = 0.59; p = 0.96 for both treatment groups combined). When both time points were combined, CAW treatment increased exploration of the familiar object at 200 mg/kg/d (p = 0.03) but not at 1000 mg/kg/d (p = 0.86).
Two hours after habituation, CAW treatment increased exploration of the novel object in Tg2576 mice at 1000 mg/kg/d (p = 0.03) and half as much at 200 mg/kg/d (p = 0.19; p = 0.03 for both treatment groups combined; Supplemental Figure 1). Twenty-four hours after habituation, CAW treatment also increased exploration of the novel object in Tg2576 mice at 200 mg/kg/d (p = 0.03) and at 1000 mg/kg/d (p = 0.11; p = 0.02 for both treatment groups combined). Upon combining testing times for each mouse, CAW treatment increased exploration of the novel object in Tg2576 mice at 200 mg/kg/d (p = 0.03) and at 1000 mg/kg/d (p = 0.02; p = 0.003 for both treatment groups combined) to exploration times similar to WT mice. CAW treatments did not alter novel object exploration in WT mice at 200 mg/kg/d (p = 0.86) nor at 1000 mg/kg/d (p = 0.98; p = 0.90 for both treatment groups combined).
To evaluate preference for a novel object 2 and 24 h post-habituation, we calculated the difference between novel and familiar object exploration times and named it novel object preference (Figure 2). Two hours after habituation, untreated WT mice but not untreated Tg2576 mice showed a preference for the novel object. The (least-squared means ± standard error of mean for WT Control was 4.19 ± 0.69 s and for TG control was 1.39 ± 0.73 s (p = 0.03 for genotype differences; Figure 2A). CAW treatment of Tg2576 mice at 200 mg/kg/d (p = 0.003) and 1000 mg/kg/d (p = 0.02; p = 0.0007 for both treatment groups combined) reversed the hAβPP-linked decline in novel object preference. No CAW treatment effects on novel object preference were observed in WT mice (200 mg/kg/d: p = 0.13; 1000 mg/kg/d: p = 0.34). Comparing least-squared means ± standard error of mean, WT control at 4.19 ± 0.69 s did not differ significantly from WT + 1000 mg/kg CAW at 4.59 ± 1.21 s (p = 0.26). However, Tg2576 mice + 1000 mg/kg CAW at 5.07 ± 1.26 s showed significantly higher recognition memory than Tg2576 controls at 1.39 ± 0.73 s (p = 0.009).
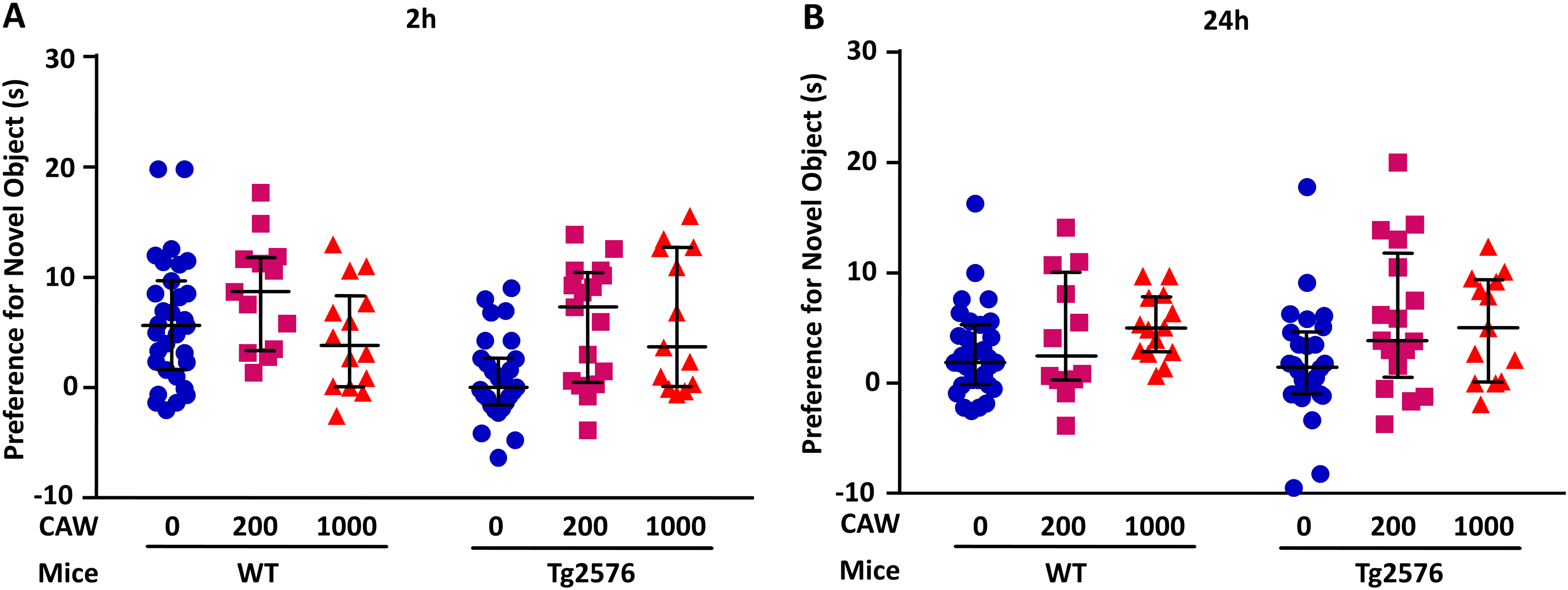
CAW treatment of aged Tg2576 mice reversed the hAβPP-linked deficit in recognition memory. Novel object recognition memory changes were measured with and without hAβPP transgene and three-week daily treatment of 20-month-old female WT and Tg2576 mice with 0, 200, and 1000 mg CAW per kg of body weight per day (mg/kg/d). (A) Two hours after habituation, untreated WT mice but not untreated Tg2576 mice showed a preference for the novel object (p = 0.03 for genotype differences calculated as difference between novel and familiar object exploration times). CAW treatment of Tg2576 mice at 200 mg/kg/d (p = 0.003) and 1000 mg/kg/d (p = 0.02; p = 0.0007 for both treatment groups combined) reversed the hAβPP-linked decline in novel object preference. No CAW treatment effects on novel object preference were observed in WT mice (200 mg/kg/d: p = 0.13; 1000 mg/kg/d: p = 0.34). (B) Twenty-four hours after habituation, untreated WT mice and untreated Tg2576 mice did not differ in preference for the novel object (p = 0.03 for genotype differences; Figure 2B), because some untreated WT control mice had lost interest in the novel object. CAW treatment of Tg2576 mice at 200 mg/kg/d (p = 0.005) and 1000 mg/kg/d (p = 0.09; p = 0.005 for both treatment groups combined) increased novel object preference. No CAW treatment effects on novel object preference were observed in WT mice (200 mg/kg/d: p = 0.151; 1000 mg/kg/d: p = 0.16). Upon combining testing times, CAW treatment increased preference for the novel object in Tg2576 mice at 200 mg/kg/d (p = 0.0002) and at 1000 mg/kg/d (p = 0.009; p < 0.0001 for both treatment groups combined) to preferences similar to WT mice. CAW treatments did not alter preference for the novel object in WT mice at 200 mg/kg/d (p = 0.16) nor at 1000 mg/kg/d (p = 0.77; p = 0.26 for both treatment groups combined). Data for individual animals are shown. The horizontal lines indicate median and interquartile range for each treatment group.
Twenty-four hours after habituation, untreated WT mice and untreated Tg2576 mice did not differ in preference for the novel object (p = 0.03 for genotype differences; Figure 2B), because some untreated WT control mice had lost interest in the novel object. CAW treatment of Tg2576 mice at 200 mg/kg/d (p = 0.005) and 1000 mg/kg/d (p = 0.09; p = 0.005 for both treatment groups combined) increased novel object preference. No CAW treatment effects on novel object preference were observed in WT mice (200 mg/kg/d: p = 0.151; 1000 mg/kg/d: p = 0.16).
Upon combining testing times, CAW treatment increased preference for the novel object in Tg2576 mice at 200 mg/kg/d (p = 0.0002) and at 1000 mg/kg/d (p = 0.009; p < 0.0001 for both treatment groups combined) to preferences similar to WT mice. CAW treatments did not alter preference for the novel object in WT mice at 200 mg/kg/d (p = 0.16) nor at 1000 mg/kg/d (p = 0.77; p = 0.26 for both treatment groups combined).
To compare performance of animals in the different treatment groups, we applied the Fisher's exact test. For the NORT, we combined both time points (2 h and 24 h) and grouped preferential novel object exploration length into three recognition memory groups: low (<5 s preference for novel object exploration), intermediate (5–10 s greater preference for novel object), and high (>10 s greater preference for novel object). Over two-thirds of the untreated mice displayed low recognition memory (41 of 58 tested mice), with a greater proportion from the Tg2576 group (89% – 24 of 27) versus the WT controls (55% – 17 of 31) (p = 0.008). Three untreated Tg2576 mice and two untreated WT mice did not engage with either object. CAW treatment of Tg2576 mice correlated with a decrease in the proportion of mice with low recognition memory to 35% (6 of 17) for 200 mg/kg/d (p = 0.0005) and 38% (5 of 13) for 1000 mg/kg/d (p = 0.002). In addition, CAW treatment of Tg2576 mice was associated with improvement in recognition memory group. More Tg2576 mice treated with 200 mg/kg/d CAW (4 of 17; p = 0.02) and 1000 mg/kg/d CAW (3 of 13; p = 0.03) showed high recognition memory than untreated Tg2576 mice (0 of 27). In comparison, 3 of 31 untreated WT mice were in the high recognition memory group.
To gain a better understanding of the relation between hippocampal metabolism and novel object preference, we averaged the responses of both testing sessions for each mouse and correlated them with their hippocampal metabolite levels (Figure 3). Stronger recognition memory was linked to equal numbers of increased and decreased levels of metabolites. Most striking was the positive link between novel object preference and accumulation of fatty acid ethyl esters: All three annotated fatty acid ethyl esters were positively correlated with a preference for novel object exploration at (all r > 0.27 and p ≤ 0.08). Noteworthy was the inverse link between novel object preference and accumulation of lyso-phosphatidylcholines (LPC): all seven annotated LPC were inversely linked to recognition memory overall and within Tg2576 mice. Also of note was the inverse link between novel object preference and accumulation of oxidized amino acids and peptides: both annotated oxidized amino acids, oxidized glutathione (L-Cysteine-glutathione disulfide (CySSG)) and methionine sulfoxide, were significantly inversely linked to novel object preference (Figure 3).
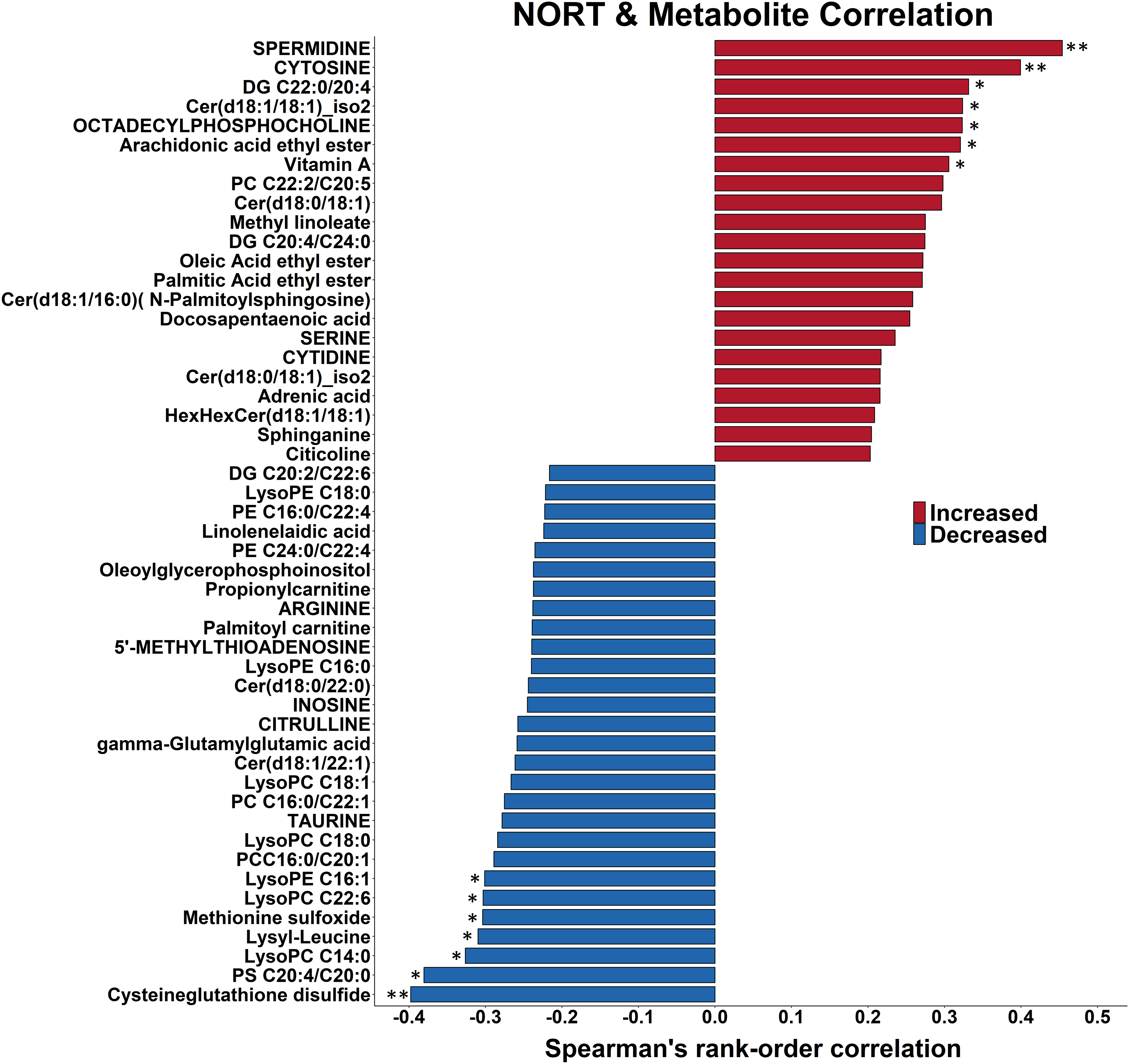
Novel object preference is correlated with higher levels of fatty acid ethyl esters and lower levels of lysophosphatidyl cholines and oxidized amino acids. The 50 strongest Spearman rank correlations between novel object preference and hippocampal metabolite levels in 20-month-old female Tg2576 mice and their WT littermates that received 0 or 1000 mg/kg/d CAW (Centella asiatica water extract) are shown. Correlations are organized by direction and strength. The strongest 2 positive correlations and the two strongest negative correlation are significant at p ≤ 0.01 (two asterisks) while the next 5 strongest positive correlations and the next 6 strongest negative correlations are significant at 0.01 ≤ p ≤ 0.05 (one asterisk). DG: diacylglycerol; Cer: ceramide; PC: phosphatidylcholine; PE: phosphatidylethanolamine; PS: phosphatidylserine.
Deficits in recognition memory were linked to presence of hAβPP transgene (Figure 2). Seven of 15 metabolites that were significantly correlated with novel object preference were altered in the opposite direction (at p < 0.08) with the presence of the hAβPP transgene (Supplemental Table 3). Novel object preference and absence of the hAβPP transgene were linked to lower levels of both oxidized amino acids (CySSG and methionine sulfoxide) and higher levels of vitamin A.
CAW treatment was linked to better recognition memory (Figure 2); four of the 7 positively changed metabolites (vitamin A, spermidine, ceramide Cer d18:1/C18:1, and diacylglycerol DG C20:4/C22:0) were also increased by CAW treatment at p < 0.10 (Figure 3).
Novel object preference was not correlated with fear memory extinction (data not shown), as also indicated by the fact that none of the hippocampal metabolites were significantly correlated with both contextual fear and preference for a novel object (Supplemental Table 3).
CAW treatment of aged mice shortened fear responses to a novel environment and delayed fear memory extinction
To evaluate the effect of hAβPP transgene and/or CAW treatment (200 and 1000 mg/kg/d) on fear memory, we utilized a CFR paradigm with untreated or CAW-treated WT and Tg2576 mice. All but one mouse showed fear responses to a novel environment (i.e., habitual freezing), which ranged from 12 to 115.5 s. CAW treatment shortened fear responses to a novel environment at 1000 mg/kg/d (habitual freezing: p = 0.04; Supplemental Figure 2A) but not at 200 mg/kg/d (p = 0.32). Transgene status did not alter habitual freezing (p = 0.30) nor did it modify the effect of CAW treatment on habitual freezing (p = 0.63).
CAW treatment strengthened fear memory retention 24 h after shock-induced classical fear conditioning in the same environment at 1000 mg/kg/d (post-shock freezing: p = 0.008; Supplemental Figure 2B) and at 200 mg/kg/d (p = 0.004). Transgene status did not alter fear memory retention (p = 0.84) nor did it modify the effect of CAW treatment on fear memory retention (p = 0.30).
To adjust for differences in habitual freezing, we calculated the difference between post-shock and habitual freezing and named it contextual freezing (Figure 4). All mice showed classical conditioning responses with strong fear memory retention and had at least 26 s of contextual freezing until fear memory extinction. At 1000 mg/kg/d, CAW treatment increased contextual freezing (p = 0.007). Only half of the increase was observed at 200 mg/kg/d CAW treatment (p = 0.14). Transgene status did not affect contextual freezing (p = 0.39) nor did it modify the effect of CAW treatment on contextual freezing (p = 0.29).
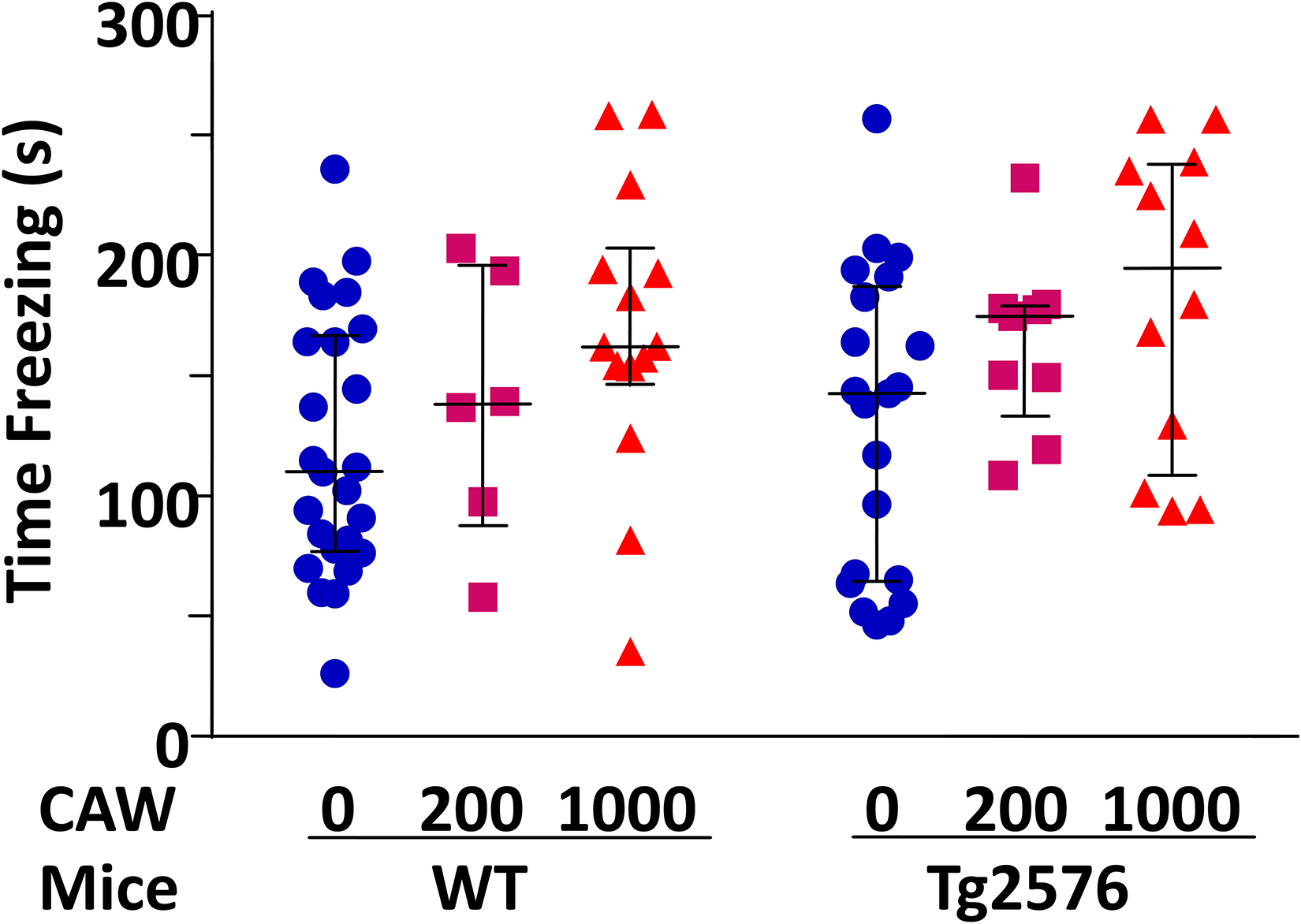
CAW treatment at 1000 mg CAW (mg/kg/d) improved contextual fear response in WT and Tg2576 mice. Contextual fear memory changes were measured with and without hAβPP transgene and five-week daily treatment of 20-month-old female WT and Tg2576 mice with 0, 200, and 1000 mg CAW per kg of body weight per day (mg/kg/d). At 1000 mg/kg/d, CAW treatment increased contextual freezing (p = 0.007). Only half of this increase was observed at 200 mg/kg/d CAW treatment (p = 0.14). Transgene status did not affect contextual freezing (p = 0.39) nor did it modify the effect of CAW treatment on contextual freezing (p = 0.29). Data for individual animals are shown. The horizontal lines indicate median and interquartile range for each treatment group.
To compare performance of animals in the different treatment groups, we applied the Fisher's exact test. All mice showed strong fear conditioning responses but differed in the time length until mice realized the previously painful environment was now safe (i.e., memory extinction). Contextual freezing was grouped into: fast fear memory extinction (<100 s adjusted-contextual freezing), medium fear memory extinction (100–200 s adjusted-contextual freezing), and slow fear memory extinction (>200 s adjusted-contextual freezing). Dividing the contextual freezing time range into thirds, a higher proportion of untreated mice were in the lower third (fast fear memory extinction; 0–100 s; 21 of 46 mice) than 200 mg/kg/d CAW-treated (2 of 15 mice; p = 0.03) or 1000 mg/kg/d CAW-treated mice (4 of 26 mice; p = 0.01; Figure 4). Transgene status did not affect grouping in the lower third (p = 0.17). A lower proportion of untreated mice were in the upper third (slow fear memory extinction; >200 s; 3 of 46 mice) than 1000 mg/kg/d CAW-treated mice (9 of 26 mice; p = 0.006) but not 200 mg/kg/d CAW-treated mice (2 of 15 mice; p = 0.59), which were primarily in the middle third (100–200 s; 11 of 15 mice). Transgene status did not affect grouping in the upper third (p = 0.25).
To gain insight into the relation between hippocampal metabolism and fear memory extinction, we correlated hippocampal metabolite abundances with contextual freezing responses (Figure 5). Fear memory retention was linked to elevated levels of hippocampal metabolites: 48 of the 50 metabolites with the strongest correlations were positively correlated to fear memory retention; these include 11 hippocampal metabolites (3 amino acids, 9 lipids), which were positively correlated at p < 0.05.
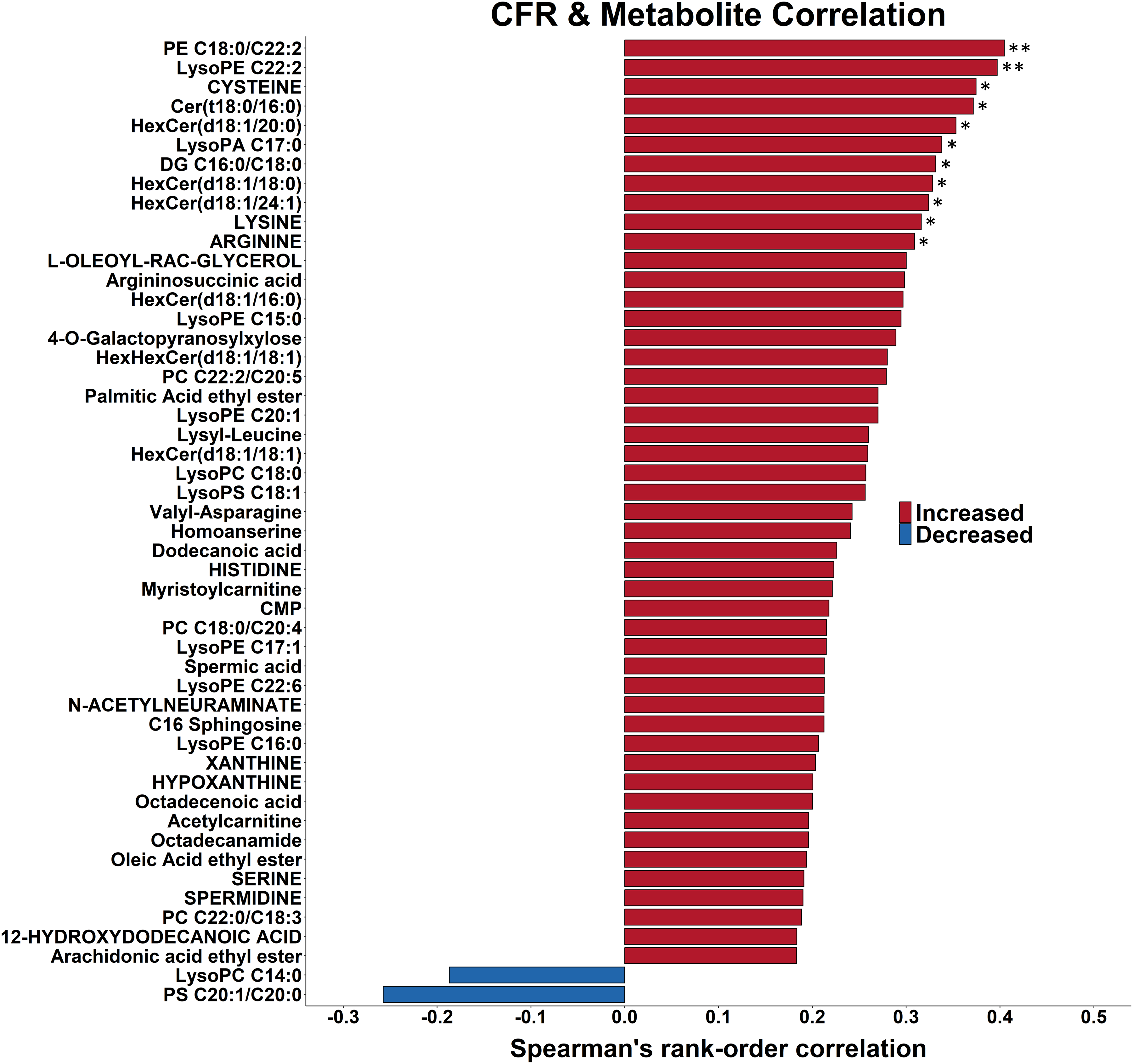
Fear memory retention is correlated with elevated levels of hippocampal metabolites. The 50 strongest Spearman rank correlations between contextual fear response and hippocampal metabolite levels in 20-month-old female Tg2576 mice and their WT littermates that received either 0 or 1000 mg/kg/d of Centella asiatica water extract for 5 weeks are shown. Correlations are organized by direction and strength. The highest two positive correlations are significant at p ≤ 0.01 (two asterisk) and the next 10 are significant at 0.01 ≤ p ≤ 0.05 (one asterisk). PA: phosphatidic acid; DG: diacylglycerol; Cer: ceramide; PC: phosphatidylcholine; PE: phosphatidylethanolamine; PS: phosphatidylserine.
Fear memory retention was linked with higher levels of glycosylated ceramides (Figure 5): all seven annotated glycosylated ceramides were positively correlated with contextual freezing responses; 3, 3, and 1 were positively correlated at p < 0.05, 0.05 < p ≤ 0.11, and p = 0.29, respectively. Three of the eight annotated protein-encoding amino acids involved in neurosignaling (cysteine, lysine, and arginine) were positively correlated with fear memory retention (p < 0.05).
Among the 11 hippocampal metabolites that were positively correlated to fear memory retention (Figure 5), two metabolites (cysteine and lysoPE C22:0) were also increased by CAW treatment and two metabolites (lysoPE C22:2 and hexosylCer d18:1/18:0) were also increased by hAβPP transgene status (all p < 0.05; Table 2).
CAW treatment of aged Tg 2576 mice resulted in a non-significant downward shift of Aβ accumulation
To evaluate CAW treatment on Aβ accumulation, we measured Aβ area and particle number per area in the hippocampus and cortex of untreated or CAW-treated Tg2576 mice using IHC (n = 5–6, Figure 6). Due to the low number of mice per group, we present descriptive statistics to increase the statistical power (Figure 7). In untreated Tg2576 mice, the median Aβ area in the hippocampus was 0.51% (range: 0.21–0.58%) and 0.60% in the cortex (range: 0.11–0.66%). CAW treatment resulted in a non-significant downward shift of Aβ accumulation at 1000 mg/kg/d: hippocampus Aβ particle number per area (−31%; p = 0.42); hippocampus Aβ area (−65%; p = 0.18); cortex Aβ particle number per area (−19%; p = 0.17); cortex Aβ area (−22%; p = 0.23). Four of six 1000 mg/kg/d CAW-treated mice had a smaller combined Aβ area (hippocampus and cortex) than any of the untreated mice. After 200 mg/kg/d CAW treatment, the response was half that of 1000 mg/kg/d CAW treatment for Aβ area: hippocampus Aβ particle number per area (0%; p = 0.84); hippocampus Aβ area (−32%; p = 0.68); cortex Aβ particle number per area (+12%; p = 0.92); cortex Aβ area (−29%; p = 0.27).
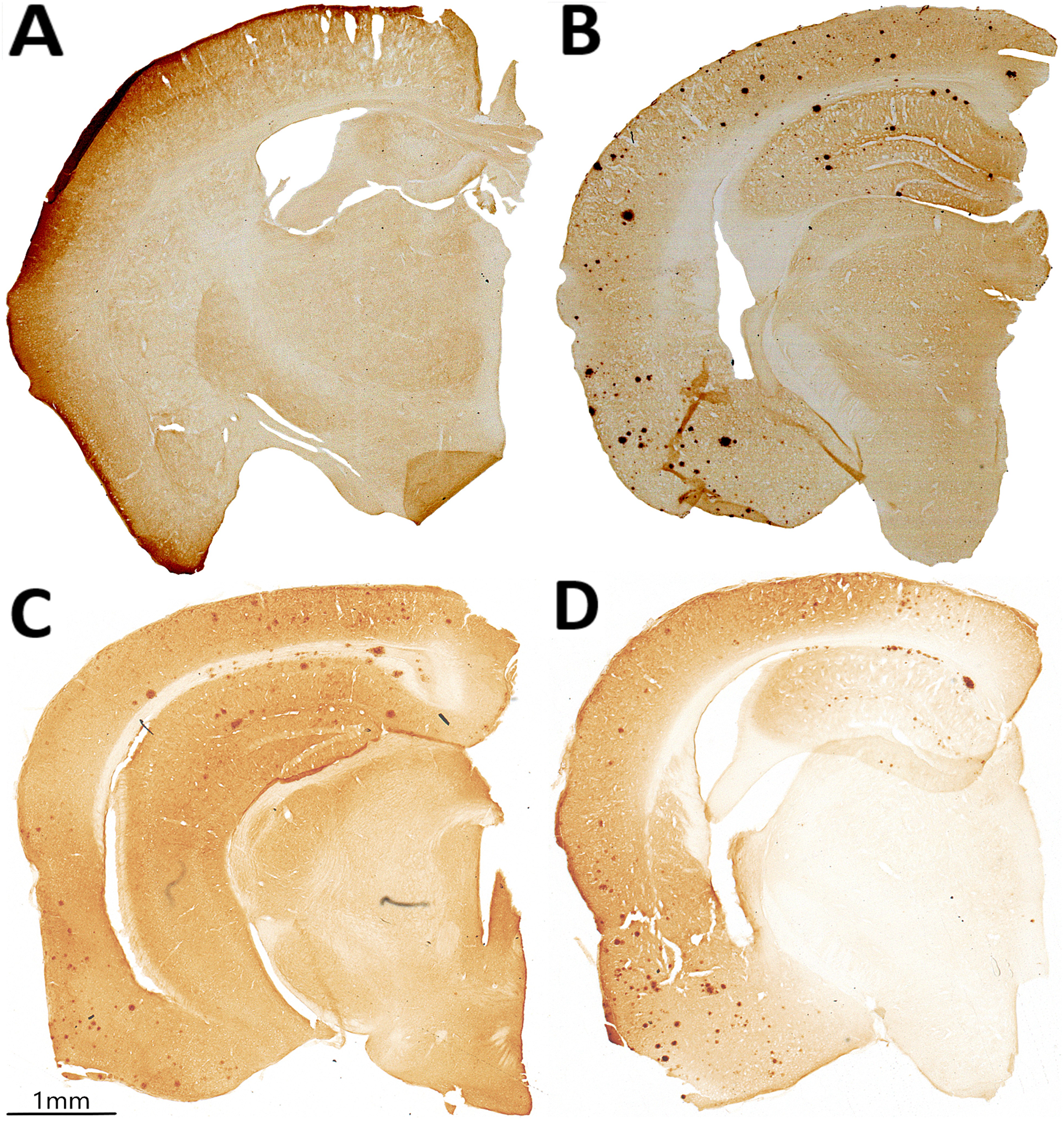
Immunohistochemical staining for pan-Aβ on coronally-sliced brain sections of 20-month-old untreated WT and Tg2576 mice and Tg2576 mice treated daily with 200, or 1000 mg/kg/d CAW (Centella asiatica water extract) for 5 weeks. Representative coronal brain sections from (A) untreated WT mice, (B) untreated Tg2576 mice, (C) 200 mg/kg/d CAW-treated Tg2576 mice, and (D) 1000 mg/kg/d CAW-treated Tg2576 mice.
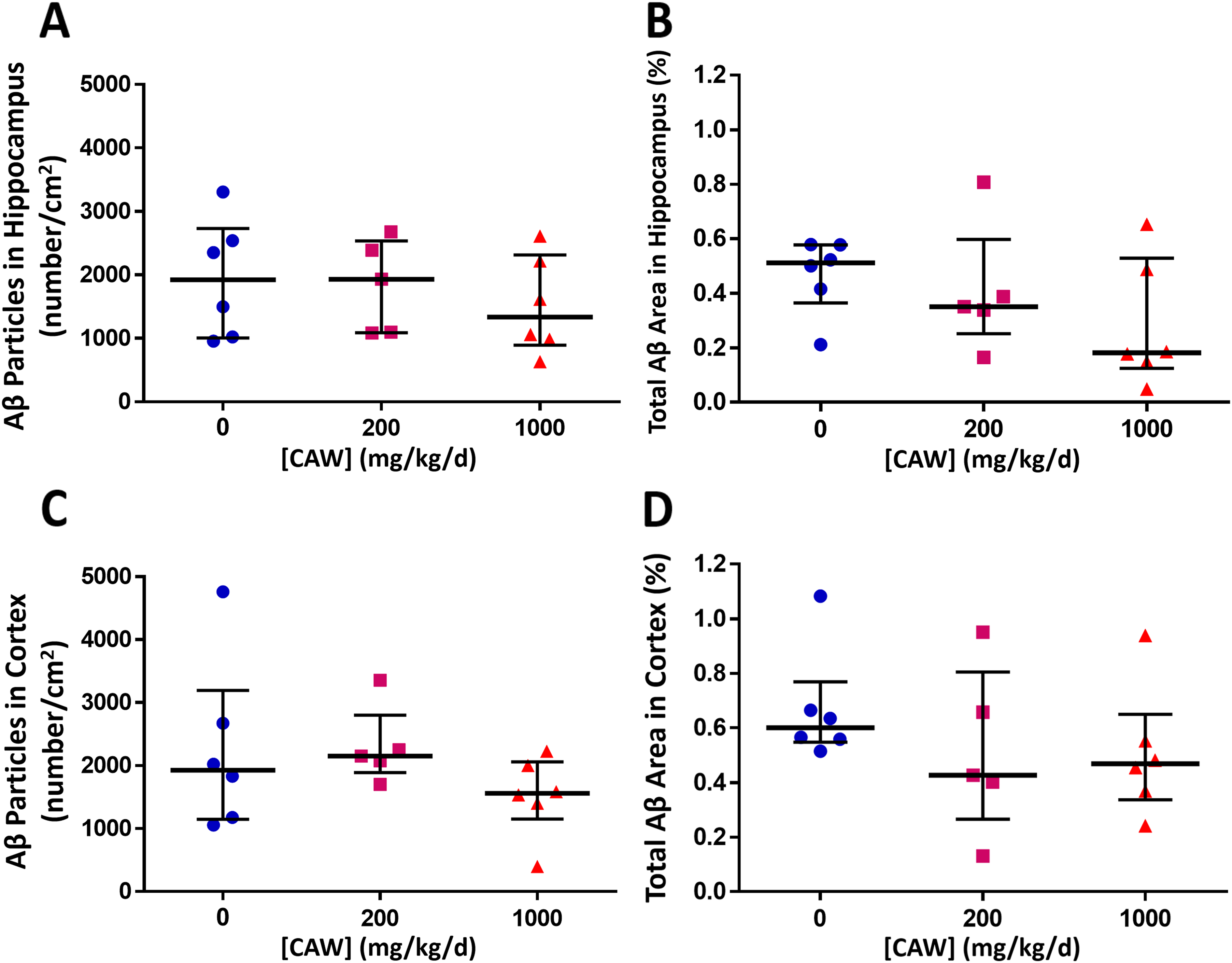
CAW treatment of aged female Tg2576 mice resulted in a non-significant downward shift of Aβ accumulation. The effect of five-week daily treatment of 20-month-old female Tg2576 mice with 0, 200, and 1000 mg Centella asiatica water extract (CAW)/kg/d on Aβ aggregation measured as density (number/cm2) and % area of Aβ particles. After 5-week treatment, (A) Aβ particles in hippocampus (median values) were lower in Tg2576 mice with 1000 mg/kg/d CAW treatment (200 mg/kg/d: 0% versus untreated mice, p = 0.84; 1000 mg/kg/d: −31% versus untreated mice, p = 0.42). (B) Total Aβ area (median value) in hippocampus was lower in Tg2576 mice with CAW treatment (200 mg/kg/d: −32% versus untreated mice, p = 0.68; 1000 mg/kg/d: −65% versus untreated mice, p = 0.18). (C) Aβ particles in cortex (median values) were lower in Tg2576 mice with 1000 mg/kg/d CAW treatment (200 mg/kg/d: + 12% versus untreated mice, p = 0.92; 1000 mg/kg/d: −19% versus untreated mice, p = 0.17). (D) Total Aβ area in cortex (median values) were lower in Tg2576 mice with CAW treatment (200 mg/kg/d: −29% versus untreated mice, p = 0.27; 1000 mg/kg/d: −22% versus untreated mice, p = 0.23). Data for individual animals are shown. The horizontal lines indicate median and interquartile range for each treatment group.
To gain insight into the link between hippocampal metabolism and Aβ plaque accumulation, we correlated in the hippocampus levels of annotated metabolites with Aβ area and with particle number per area (Figure 8). The strongest correlated metabolite with both was citicoline (CDP choline) with positive correlations of r = +0.64 (p = 0.03) with Aβ area (Figure 8A) and r = +0.55 (p = 0.07) with Aβ particle number per area (Figure 8B). Noteworthy was that Aβ plaque accumulation was linked to lower ceramide levels (8 of 9 C18 and C20 ceramides were negatively correlated with Aβ area and 8 of 9 C18 and C20 ceramides were positively correlated with Aβ particle number per area). Aβ plaque accumulation was also linked to lower levels of C22:6-containing lipids (5 of 7 C22:6-containing lipids were negatively correlated with Aβ area and 6 of 7 C22:6-containing lipids were negatively correlated with Aβ particle number per area). No metabolite was significantly correlated with both Aβ area and Aβ particle number in the hippocampus. Nor were hippocampal Aβ area and Aβ particle number per area correlated (r = +0.11; p = 0.66)
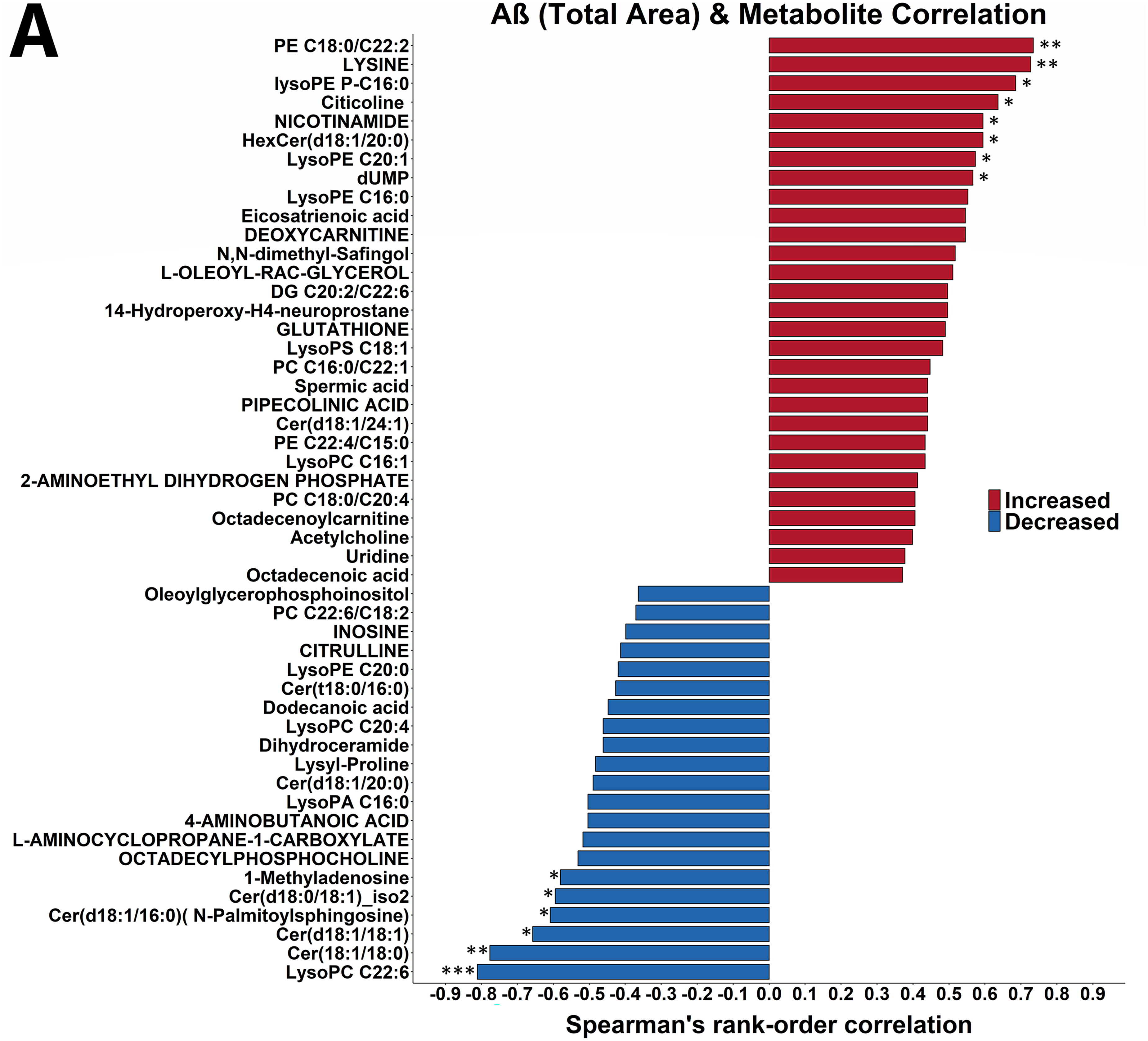
Hippocampal Aβ plaque accumulation is correlated with lower levels of hippocampal ceramides, higher levels of fatty acid ethyl esters, and lower levels of C22:6 containing lipids. The 50 strongest Spearman rank correlations between hippocampal Aβ aggregation and hippocampal metabolite levels in 20-month-old untreated mice, and Tg2576 mice treated for 5 weeks at 1000 mg/kg/d CAW are shown. Correlations are organized by direction and strength. (A) Correlation between Aβ area and hippocampal metabolite levels. The two strongest positive and two strongest negative correlations between Aβ area and hippocampal metabolites are significant at p < 0.01 (two asterisk; for p < 0.001 three asterisk) and the next six strongest positive and the next four strongest negative correlations are significant at 0.01 ≤ p ≤ 0.05 (one asterisk). (B) Correlation between Aβ particle number per area and hippocampal metabolites. The two strongest positive correlations and the two strongest negative correlations between Aβ particle number and hippocampal metabolites are significant at p < 0.001 (three asterisks) or p ≤ 0.01 (two asterisks) and the next four negative correlations are significant at 0.01 ≤ p ≤ 0.05 (one asterisk). DG: diacylglycerol; Cer: ceramide; PC: phosphatidylcholine; PE: phosphatidylethanolamine; PS: phosphatidylserine.
The correlations between hippocampal metabolism and Aβ aggregation were much stronger than those with recognition-based or fear-based memory (Figure 8 versus Figures 3 and 5). The three strongest correlations between hippocampal metabolites and Aβ area (Figure 9) were with LPC C22:6 (r = -0.81, p = 0.001), ceramide dC18:1/C18:0 (r = -0.78, p = 0.003), and lysine (r = +0.73, p = 0.007). The strongest impact was on phospholipid metabolism. Eight of 9 annotated C18 and C20 ceramides were inversely correlated with Aβ area, whereas 4 of the 6 annotated glycosylated C18 and C20 ceramides were positively correlated with Aβ area. In addition, 3 of the 5 annotated plasmalogens were positively correlated with Aβ area at r ≥ + 0.40. Phospholipid precursors and lysine metabolites were both positively and inversely correlated with Aβ area. Four of the 6 annotated lysine-affiliated molecules were strongly correlated with Aβ area at r ≥ |0.35|, both of the lysine-containing dipeptides were negatively correlated with Aβ area whereas lysine and its metabolites trimethyllysine and pipecolic acid were positively correlated at r ≥ + 0.36. Of the 7 annotated glycerophospholipid and sphingolipid precursors each, 6 glycerophospholipid precursors were strongly correlated with Aβ area at r ≥ |0.36| and 4 sphingolipid precursors were strongly correlated with Aβ area at r ≥ |0.41|.
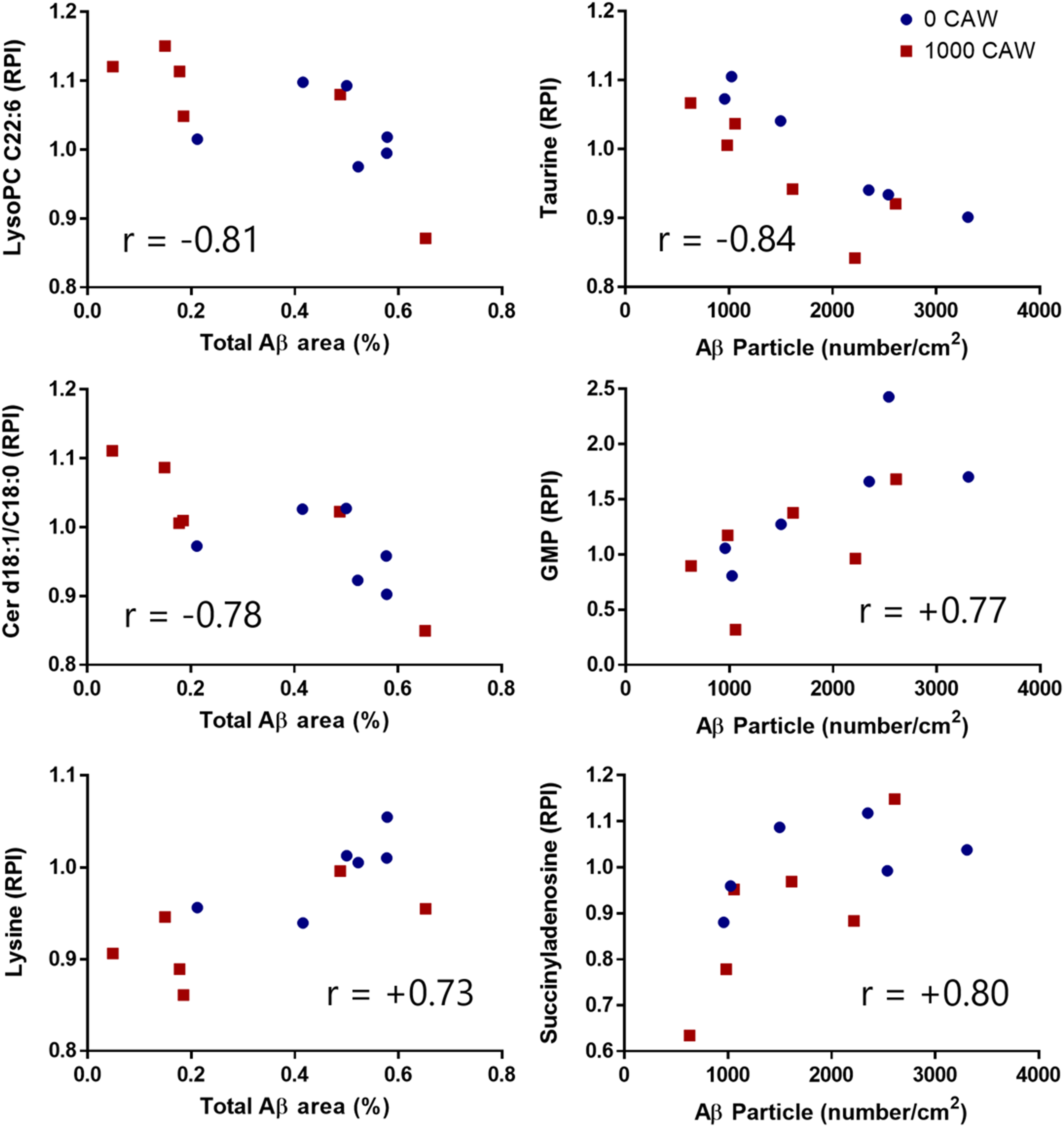
Several metabolites show strong correlations (r > 0.7) to hippocampal Aβ load. The three strongest Spearman rank correlations are shown between hippocampal metabolite levels and either hippocampal Aβ area (left column) or Aβ particle number (right column) in 20-month-old female Tg2576 mice either untreated, or treated with 1000 mg/kg/d CAW for 5 weeks. Data points refer to individual mice untreated (blue circle) or CAW-treated (red square). GMP: guanosine monophosphate.
The three strongest correlations between hippocampal metabolites and Aβ particle number per area (Figure 9) were with taurine (r = -0.84, p = 0.0006), succinyladenosine (r = +0.80, p = 0.002), and GMP (r = +0.77, p = 0.003). The strongest impact was on the annotated oxidized metabolites (all 7 positively correlated), fatty acyl carnitines (all 6 negatively correlated), sulfur amino acid-affiliated molecules (8 except taurine positively correlated), and pyrimidines (all 8 correlated at r ≥ |0.23|).
Three metabolites (phosphatidyl ethanolamine C18:0/C22:2, hexosylCer d18:1/20:0, and lysine) were significantly correlated with both fear-based memory retention and Aβ area (Supplemental Table 3). No metabolites were significantly correlated with both fear-based memory retention and Aβ particle number. Only one metabolite (spermidine) was significantly linked to Aβ particle number as well as novel object preference. Only one metabolite (LPC C22:6) was linked to Aβ area as well as to novel object preference. Neither Aβ particle number nor Aβ area were correlated significantly with contextual freezing or with novel object preference (data not shown).
Two metabolites (ceramide d18:1/18:1 and N-palmitoylsphingosine) were significantly altered in the same direction with presence of the hAβPP transgene and Aβ area. Three metabolites (C14:0 carnitine, N-acetyl-aspartic acid, and guanosine monophosphate (GMP)) were significantly altered in the same direction with presence of the hAβPP transgene and Aβ particle number.
Two metabolites (N-palmitoylsphingosine and plasmalogen LPE C16:0) were significantly altered in opposite directions with 1000 mg/kg/d treatment (plasmalogen LPE C16:0 only in Tg2576 mice) and increasing Aβ area. Three metabolites (spermidine, succinyladenosine, and GMP) were significantly altered in opposite directions with 1000 mg/kg/d treatment (GMP only in Tg2576 mice) and increasing Aβ particle number per area.
Discussion
The incidence of AD is increasing with an aging population, and preferentially impacts women. 55 Increasing evidence using omics techniques indicates that AD may be a metabolic disorder of the brain. 36 With limited effective treatments for AD, CAW has emerged as a promising therapeutic for managing neurocognitive challenges 15 ; however, its mode of action is still under investigation. The objective of the current study was to gain insights into changes in hippocampal metabolism that may be relevant to the cognitive improvement observed with CAW in aged female Tg2576 23 and WT mice. 22
CAW treatment reverses only a limited number of hippocampal metabolite changes induced by introduction of the hAβPP transgene
We have previously reported the metabolomic changes observed in the hippocampus of female Tg2576 mice in comparison to WT littermates. 34 CAW treatment of Tg2576 mice significantly altered the abundance of 20 hippocampal metabolites (10 increased and 10 decreased) (Table 2, Supplemental Table 4), only eight of these changes were reversals of the changes resulting from the introduction of the hAβPP transgene (Table 2). These included the neurotransmitters acetylcholine (increased by hAβPP transgene, decreased by CAW), 5-aminopentanoate (decreased by hAβPP transgene, increased by CAW), and phenacylamine (decreased by hAβPP transgene, increased by CAW), three lipids, i.e., phosphatidic acid PAi24:0/C21:0 (decreased by hAβPP transgene, increased by CAW), phospholipid PC C22:2/C20:5 (decreased by hAβPP transgene, increased by CAW) and lysophospholipid LysoPC C16:1 (increased by hAβPP transgene, decreased by CAW), the purine GMP (increased by hAβPP transgene, decreased by CAW) and the phytocompound jasmonate (decreased by hAβPP transgene, increased by CAW). However, none of these compounds also correlated with performance in CFR or NORT, and only GMP levels were positively corelated with the number of Aβ particles. This suggests that direct reversal of hAβPP induced metabolic changes are not the underlying mechanism for improvement in cognitive function by CAW. This finding is also in agreement with the observed improvement in cognitive function in both Tg2576 and WT animals following CAW treatment. Our earlier studies have implicated amelioration of oxidative stress and mitochondrial dysfunction as important targets for CAW, that are not unique to AD.
A previous study by our group 37 examined the effects of CAW (200, 500, and 1000 mg/kg/d) on the cortical metabolome of 8-month-old, male and female WT and 5XFAD mice (a mouse model of AD that develops Aβ plaques at a younger age. 5 Although the direction and magnitude of the effects of CAW varied with genotype and sex, CAW was noted to affect purine, nicotinate, nicotinamide and glycerophospholipid metabolism. One common finding was lowered GMP levels in response to CAW treatment in the cortex of female 5XFAD mice (p = 0.095) mice 37 and female Tg2576 mice in the present study (Table 2). A unique finding of the current study was an effect of CAW on neurosignaling molecules including ceramides in female Tg2576 mice which had not been noted in 5XFAD mice.
CAW contains a wide range of compounds, 56 of which triterpenoids (asiatic acid, madecassic acid, and glycosides) and caffeoylquinic acids are those most likely to be associated with its cognitive effects. 15 To our knowledge the effects of these individual compounds on the murine hippocampal metabolome have not been evaluated. However, in other metabolomic studies, a CA extract enriched with triterpenes increased serum levels of the endogenous metabolites O-succinyl-L-homoserine, homocarnosine, choline, and citrulline in humans. 57 The triterpene asiatic acid significantly altered hepatic sphingolipid and glycerophospholipid metabolites in a mouse model of rifampicin and isoniazid induced liver injury. 58 Chlorogenic acid (a monocaffeoylquinic acid found in CA) significantly activated purine and thiamine metabolism in SH-SY5Y neuroblastoma cells. 59 Further studies are required to determine the role of individual constituents on the hippocampal metabolomic changes induced by CAW that we have observed in Tg2576 and WT mice (Table 2, Supplemental Tables 4 and 5).
Presence of the hAβPP transgene increases Aβ accumulation in hippocampus and cortex of aged female mice and decreases novel object exploration but not fear memory extinction
The hippocampus is thought to be involved in the acquisition and consolidation of contextual fear memory. 60 Using the well-established CFR model, we examined contextual fear-based memory in aged Tg2576 female mice (20-months old) and their WT female littermates. Previous studies in Tg2576 mice reported mixed CFR responses, indicating fear memory deficits in aged Tg2576 mice in some studies61–67 but not in others.68–70 The aged Tg2576 mice in this study, had a similar contextual fear response compared to WT mice. A potential explanation is that fear is one of the strongest inducers of acquisition and consolidation of memories. A longer time between fear stimulus and test (24 h in the present study) or a lighter shock (0.5 mA in the present study) may aid in detecting changes in contextual fear-based memory responses due to the Tg2576 genotype.
Evidence for hippocampal involvement in novel object recognition memory is more tenuous than for contextual fear memory. 45 Using the well-established NORT model, we examined genotypic and CAW treatment effects on novel object interest and preference in aged Tg2576 female mice (20-months old) and their WT female littermates. Previous Tg2576 studies reported mixed results from NORT evaluation71,72 but with an overall consensus that aged Tg2576 mice show deficits in recognition memory.73–76 In the present study, a large proportion of untreated aging mice (71%) demonstrated poor recognition memory, including 5 mice that were not interested in either object. This is in stark contrast to the robust contextual fear-based memory observed in all tested mice. These contrasting results in the two behavioral tests are consistent with observations of different parts of the brain being involved in different types of memory.
Novel object interest and preference appeared more impaired in untreated Tg2576 mice (24 of 27 total) than untreated WT mice (17 of 31 total). Untreated WT mice showed more interest in the novel object than did the untreated Tg2576 mice at 2 h but not at 24 h post-training. Due to their advanced age, it is possible that the untreated WT mice may lose interest in a novel object at 24 h post-training and this may have muted any differences to Tg2576 animals.77–79
The primary hallmark of AD is aberrant Aβ/AβPP metabolism, and hAβPP catabolite aggregation plays a role in AD-related neurodegeneration and cognitive impairment.80,81 Cognitive function and memory decline have been reported with accumulation of both soluble82–85 and insoluble86,87 Aβ in the hippocampus of Tg2576 mice. In the present study, the extent of Aβ accumulation observed in Tg2576 mice was not linked to cognitive function (i.e., CFR and NORT); however, the negative relationship between plaque burden and cognitive function is less straightforward than as originally suggested in the amyloid hypothesis.88,89 Moreover, the Aβ-afflicted area in the hippocampus and cortex was below 1% in all tested Tg2576 mice in this study, slightly under the previously reported 2% total plaque area in the prefrontal cortex in 16 month old Tg2576 mice, making it less likely to impact cognitive function. 90 We also observed that only a limited number of hippocampal metabolites were related to both Aβ plaques and memory (Supplemental Table 3).
CAW treatment increased fear memory retention and novel object exploration and non-significantly lowered Aβ accumulation in aged female mice
In this study, we did not compare the cognitive function of these aged mice directly to younger animals of the same genotypes. However we anticipated some degree of cognitive impairment as age-related deficiencies in novel object recognition have been reported for both WT 91 and Tg2576 mice, 75 as have age-related impairments in contextual fear response in WT 92 and Tg2576 mice. 63
Previous studies in our laboratory showed that CAW treatment at 200 mg/kg/d strengthened contextual fear memory in the 5XFAD mouse model of AD. 24 Here we observed a non-significant improvement in contextual fear response with 200 mg/kg/d CAW treatment, however we did show a significant positive CAW effect with a higher CAW dose (1000 mg/kg/d) in combined genotypes. In addition, 1000 mg/kg/d CAW treatment correlated with a decrease in habitual freezing in a novel environment during CFR training, possibly due to CA's known anti-anxiety effects.93–96
Improvements in novel object exploration after CAW treatment in rodent models has been previously reported by us and other groups,24,29,97,98 including our study of 200 mg/kg/d CAW treatment in 20 month old WT mice. 29 In the present study, CAW treatment was associated with improvement of novel object interest and preference 2 h after training in aged Tg2576 mice (but not in aged WT mice, contrary to our previous study 29 ), mitigating the memory deficit observed in untreated Tg2576 mice compared to untreated WT controls. This effect was similar at both doses of CAW (200 mg/kg/d and 1000 mg/kg/d). These results validate previous studies demonstrating CAW's positive impact on recognition memory. However, when we looked at the CAW effect on exploration of the novel object separately, we observed a reduced effect at 200 mg/kg/d versus 1000 mg/kg/d CAW, indicating a dose-dependent effect of CAW on novel object recognition memory.
We previously reported 200 mg/kg/d CAW treatment was insufficient to alter Aβ hippocampal plaque formation in female 5XFAD mice, another Aβ-linked AD mouse model 24 or soluble or insoluble Aβ40 or Aβ42 levels in the hippocampus of female Tg2576 mice. 23 Similarly in the present study, 200 mg/kg/d CAW treatment for 5 weeks did not significantly alter Aβ plaque formation in the hippocampus (32% reduction) or the cortex (12% increase) of aged Tg2576 mice. At the 1000 mg/kg/d dosage, we observed a 65% decrease in hippocampal Aβ plaque area, which was not significant due to two outliers in the treatment group and a 19% decrease in Aβ plaque area in the cortex. It is possible that a lower than expected Aβ plaque burden 90 in our colony produced conditions not robust enough to show an impact of CAW treatment as the Aβ plaque area in the cortex was <1%, even at 20 months of age. However, it remains possible that Aβ plaque formation may be impacted by higher doses and/or longer treatment regimens of CAW in Tg2576 mice. In summary, CAW treatment at 1000 mg/kg/d may partially reduce Aβ plaque formation; more importantly, the cognitive benefits of aged Tg2576 mice are apparent in response to short-term (i.e., 5 weeks) CAW treatment despite lack of significant effects on Aβ burden.
Metabolic shifts in the aged female hippocampus linked to neurocognitive function and Aβ accumulation
The hippocampus in conjunction with amygdala and ventromedial prefrontal, entorhinal, posterior parietal, and perirhinal cortices participates in threat recognition and fear-based responses.41,99 Metabolomics analysis of hippocampal tissue of untreated and 1000 mg/kg/d CAW-treated mice of both genotypes allowed us to better understand metabolic changes associated with fear-based memories. Strength of contextual fear-based memories was linked to 1) an upregulation of hippocampal metabolism; 48 of the 50 strongest correlations with metabolites were positive and 2) upregulation of two metabolite clusters involved in neurosignaling: (a) glycosylated ceramides, which play an important role in sphingolipid metabolism, and (b) amino acids involved in neurosignaling (cysteine, lysine, and the nitrous oxide precursor arginine). However, our metabolomics analysis results did not allow us to better understand the mechanisms behind the beneficial effects of CAW treatment on contextual fear memory, as only two metabolites (cysteine and lysoPE C22:2) were linked positively to both CAW treatment and CFR.
The hippocampus and adjacent cortical regions in the medial temporal lobe 100 participate in novel object recognition memory.101–106 Metabolomics analysis of hippocampal tissue of untreated and 1000 mg/kg/d CAW-treated mice of both genotypes allowed us to better understand this type of memory. Lack of novel object preference may be linked to accumulation of LPC and oxidized amino acids and peptides, which were also linked to the presence of hAβPP transgene. 34 Improvements in novel object preference may be linked to fatty acid ethyl esters, which are non-oxidative ethanol metabolites and involved in plasmalogen synthesis, and metabolite changes in response to 1000 mg/kg/d CAW treatment, which spanned a variety of metabolite groups. Although both novel object preference and fear memory extinction were linked to metabolite changes in the hippocampus; those changes involved distinctly different metabolite groups. Fear-based memory retention were linked to heightened neurosignaling and accumulation of ceramides in hippocampal tissues. Ceramides accumulate in AD patients by both degradation of sphingolipids and glycolipids as well as ceramide synthesis. 107 In contrast, lack of novel object preference was linked to accumulation of LPC and oxidized metabolites in hippocampal tissues associated with hAβPP transgene. 34 Treatment with 1000 mg/kg/d CAW improved fear memory retention and novel object preference. Those changes are linked to a variety of metabolite groups, suggesting that CAW induces a variety of molecular targets and involves multiple metabolic pathways. This is not surprising, as CAW consists of a mixture of bioactive compounds, each of which may have a different molecular target.
Although cognitive measures did not correlate with hippocampal Aβ accumulation, Aβ accumulation was associated with changes in hippocampal metabolism in the present study. Correlations with hippocampal metabolites were stronger for Aβ accumulation than for cognitive measures, indicating the impact of Aβ accumulation on hippocampal metabolism. Hippocampal Aβ area and particle number per area were not correlated with each other, suggesting that Aβ plaque formation is not a linear process, in which first an area is affected and then Aβ particles accumulate. In support, there was limited overlap between hippocampal metabolites linked to hippocampal Aβ area and to Aβ particle number, suggesting there may be differences in metabolic processes involved in initial formation of Aβ particles and their accumulation. The strongest common metabolic theme linked to both hippocampal Aβ area and particle number was depletion of C22:6-containing lipids, indicative of the established link between Aβ plaque formation and oxidative stress and neuroinflammation.108–110 This is also supported by our observation of the link between increased Aβ plaque formation and accumulation of oxidized metabolites.
Among individual metabolites, the strongest link between hippocampal Aβ area and particle number per area was with citicoline (the full name cytidine-diphosphate choline). Citicoline provides the cytidine group for pyrimidine synthesis and most pyrimidines were strongly correlated with hippocampal Aβ area and particle number. Citicoline provides the choline group for phosphocholines and the glycerophosphocholine precursors glycerophosphocholine and octadecylphosphocholine were positively correlated with both hippocampal Aβ area and particle number (the latter being the exception for hippocampal Aβ area). Most importantly, citicoline provides the choline group for metacholine and the excitatory neurotransmitter acetylcholine, which were also correlated positively with both hippocampal Aβ area and particle number, indicating a link between Aβ plaque formation and increased phospholipid metabolism and neurotransmission.
We observed a link between Aβ plaque formation and elevated excitatory neurotransmission and suppressed inhibitory neurotransmission. Besides citicoline, the excitatory neurotransmitter N-acetyl aspartic acid was positively linked to Aβ particle number. In contrast, spermidine, which inhibits N-acetyl aspartic acid synthesis, was negatively linked to Aβ particle number as was taurine, an important inhibitory neurotransmitter. Depletion of taurine and spermidine have been linked to aging-associated tissue damage, as taurine and spermidine have a variety of neuroprotective functions, including protection against Aβ-mediated neurotoxicity, and have been successfully used to treat symptoms of neurodegenerative disease in preclinical models.111–113 In addition, our metabolomics results indicated elevated lysine and sulfur amino acid metabolism with Aβ plaque formation, both of which can be used as precursors for neurotransmitters.
We observed a link between Aβ plaque formation and increased phospholipid metabolism. 34 Levels of many glycerophospholipid precursors and sphingolipid precursors were linked to Aβ particle number. Another dominant phospholipid fraction in brain, plasmalogens, were elevated with increased Aβ area. Plasmalogens play a critical role in AD due to their function in neurosignaling.114,115 The third line of evidence was changes in ceramide metabolism. In an AD-mediated positive feedback loop, Aβ plaques increase sphingomyelinase activity, promoting the formation of ceramides, 116 while ceramides stabilize beta-secretase 1, promoting its AβPP processing and plaque formation. 117 Several studies reported a link between accumulation of pro-apoptotic C18:0 ceramide and sphingomyelins in human AD patients, senile plaques, and in normal aging mice.116,118–121 In contrast, no changes in hippocampal levels were observed in AD model mice lacking neuronal loss (AβPPSL, AβPPsw, and AβPPV717F).122,123 Ceramides are important components of the lipid membrane bilayer and lipid rafts that act in cell signaling and are key regulators of neuronal cell death in AD. Apoptosis is mainly induced by saturated long-chain ceramides (Cer C16:0 and C18:0), whereas very long-chain ceramides may interfere with the pro-apoptotic and ROS-generating effect of saturated long-chain FA.124–126 In contrast, monounsaturated ceramides do not have apoptotic properties 127 and glycosylated ceramides may inhibit apoptosis.128,129 In the present study, levels of C18 and C20 ceramides decreased and levels of C18 and C20 glycosylated ceramides increased with increased Aβ area, which would promote survival of Aβ-afflicted cells via increased ceramide glycosylation. In support, inhibition of glycosylceramide synthesis can decrease aggregation of tau in the hippocampus, 130 indicating a potential molecular mechanism for our results.
We also observed a strong positive correlation with succinyladenosine (r = 0.80) and GMP (r = +0.77) with Aβ plaque particle number. Accumulation of succinyladenosine is observed with adenosylosuccinate lyase ADSL deficiency, a congenital disease known to cause extensive neurotoxicity. 131 GMP is the degradation product of cyclic GMP (cGMP) by cyclic nucleotide phosphodiesterase, inhibitors of which are promising pharmacological target for treatment of neurodegenerative diseases including AD. 132 Levels of cGMP indicate both presence and severity of AD, as patients afflicted with AD have less cGMP in their CSF than normal aging adults and both Aβ plaque burden and cognitive function are inversely linked to cGMP levels in CSF of patients afflicted with AD.133,134 Involved in learning and memory processes, cGMP acts as a secondary messenger in neuronal signal transduction. 135 In the present study elevated GMP levels in Tg2576 mice were decreased by 1000 mg/kg/d CAW treatment, supporting the potential role of cyclic nucleotide phosphodiesterase inhibitors for treatment of neurocognitive diseases such as AD and as potential molecular target of CAW.
Limitations of the study
One limitation of the present study was that it was conducted only in female WT and Tg 2576 animals. The non-inclusion of male animals was due to practical considerations. Male Tg2576 mice are aggressive and have to be housed individually, raising costs particularly when they need to be aged to 20 months. For this reason, only female mice (which can be group housed) were used in the present study. The earlier study in younger WT and 5XFAD mice 37 indicated differences in CAW-induced metabolomic changes in male and female mice. A second limitation was that metabolite analysis was performed using only positive ionization mode in LC-MS, whereas the use of both positive and negative ionization would have yielded a larger number of metabolites for comparison. A single ionization mode had to be chosen due to resource limitations, and positive ion mode was selected as it works well for the wide range of metabolite classes that we obtained using a mixture of methanol and ethanol (1:1v/v) as monophasic extraction solvent which allows extraction of a wide range of polar and non-polar metabolites in combination with the applied chromatographic method based on a Phenyl-3 column. The combination of comprehensive extraction off metabolite with a well-established untargeted and short metabolomics screening method (in positive ionization mode) allowed for obtaining a comprehensive metabolomic dataset within the resources available and in a reasonable amount of time.
Conclusion
The goal of the present study was to better understand the mechanisms underlying the previously observed cognitive benefits of CAW in aged WT and Tg2576 mice through examination of changes in the hippocampal metabolome. In a novel approach, we correlated performance in memory tests and Aβ burden with the levels of metabolites. Short-term daily CAW treatment of aged mice at 1000 mg/kg/d improved both contextual fear retention and novel object recognition memory, and decreased habitual freezing potentially due to anxiolytic effects, whereas the lower dosage of 200 mg/kg/d CAW had a reduced but still positive effect on memory. Although both novel object preference and fear memory extinction were linked to metabolite changes in the hippocampus; those changes involved distinctly different metabolite groups. While contextual fear memory was linked to heightened neurosignaling and higher glycosylated ceramide levels, better novel object preference was linked to reductions of LPC and oxidized metabolites. Interestingly, poor performance in either cognitive measure did not correlate with hippocampal Aβ accumulation, although this accumulation was associated with changes in hippocampal metabolism. Only a subset of the metabolite changes induced by CAW in Tg2576 mice represented a reversal of the metabolite differences between Tg2576 and WT mice, suggesting the involvement of other pathways in CAW's effects on cognition. Given that CAW contains multiple bioactive compounds, a variety of hippocampal metabolites were linked to CAW treatment and behavioral and/or immunochemical measures. One of the most promising potential molecular targets was cyclic nucleotide phosphodiesterase, which converts cyclic GMP to GMP; the latter was elevated overall with increased Aβ plaque formation in Tg2576 mice and decreased with 1000 mg/kg/d CAW treatment. Overall, the results in the present study support CAW treatment as a potential tool for managing memory challenges in aged individuals with or without AD. The mechanisms underlying CAW's beneficial effects extend beyond reversing the effects of Aβ accumulation on the hippocampal metabolome. This study provides further evidence for the potential cognitive enhancing effects of CAW in individuals with AD, accompanied by changes to the hippocampal metabolome. A next step towards clinical translation could involve exploring corresponding, or related, changes in the plasma metabolome or through brain magnetic resonance spectral imaging (MRSI) in animals following CAW treatment. These plasma or MRSI metabolite changes could then be readily confirmed as evidence of target engagement by CAW treatment in human Phase I studies, prior to initiating more costly Phase II efficacy trials.
Supplemental Material
sj-docx-1-alr-10.1177_25424823241296740 - Supplemental material for Centella asiatica improves cognitive function and alters the hippocampal metabolome of aged Tg2576 and wild-type mice
Supplemental material, sj-docx-1-alr-10.1177_25424823241296740 for Centella asiatica improves cognitive function and alters the hippocampal metabolome of aged Tg2576 and wild-type mice by Donald G Matthews, Mona Khorani, Gerd Bobe, Maya Caruso, Armando Alcazar Magana, Nora E Gray, Joseph F Quinn, Jan F Stevens, Claudia S Maier and Amala Soumyanath in Journal of Alzheimer's Disease Reports
Footnotes
Acknowledgments
We thank Dr Kirsten Wright, OHSU Department of Neurology for the preparation of CAW used in this study, and Dr Jeff Morre of Oregon State University Mass Spectrometry Center for LC-MS of the metabolomics samples and Madeleine Waldram for assistance with ![]() . We specifically acknowledge our day-to-day animal care technicians and residing veterinarian at the Portland VA facilities as vital components of our dedication to maintaining high standards for all our animals’ health and well-being.
. We specifically acknowledge our day-to-day animal care technicians and residing veterinarian at the Portland VA facilities as vital components of our dedication to maintaining high standards for all our animals’ health and well-being.
Author contributions
Donald Grant Matthews (Data curation; Formal analysis; Investigation; Methodology; Writing – original draft; Writing – review & editing); Mona Khorani (Data curation; Formal analysis; Investigation; Writing – original draft; Writing – review & editing); Gerd Bobe (Conceptualization; Data curation; Formal analysis; Methodology; Writing – original draft; Writing – review & editing); Maya Caruso (Data curation; Investigation); Armando Alcazar Magana (Data curation; Formal analysis; Investigation; Writing – review & editing); Nora E Gray (Conceptualization; Investigation; Methodology; Resources; Writing – review & editing); Joseph F Quinn (Conceptualization; Methodology; Resources; Supervision; Writing – review & editing); Jan F Stevens (Conceptualization; Funding acquisition; Resources; Writing – review & editing); Claudia S Maier (Conceptualization; Data curation; Formal analysis; Methodology; Resources; Supervision; Writing – original draft; Writing – review & editing); Amala Soumyanath (Conceptualization; Funding acquisition; Methodology; Project administration; Resources; Supervision; Writing – original draft; Writing – review & editing).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: These experiments were financially supported by NIH-NCCIH grants R01AT008099 (Soumyanath), T32AT002688 (Matthews, Wright), and R00AT008831 (Gray), by the Department of Veterans Affairs Merit Review Grant IO1BX003440 (Quinn), and by the NIH instrumentation grant S10RR027878 (Stevens). Manuscript preparation was supported by NIH-NCCIH grant U19AT010829 (Soumyanath).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this study are available on request from the corresponding authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
