Abstract
Background
Alzheimer's disease (AD) is a prevalent, incurable, and chronic neurodegenerative condition characterized by the accumulation of amyloid-β protein (Aβ), disrupting various bodily systems. Despite the lack of a cure, phenolic compounds like cannabidiol (CBD), a non-psychoactive component of cannabis, have emerged as potential therapeutic agents for AD.
Objective
This systematic review explores the impact of different types of cannabidiol on AD, unveiling their neuroprotective mechanisms.
Methods
The research used PubMed, Scopus, and Web of Science databases with keywords like “Alzheimer's disease” and “Cannabidiol.” Studies were evaluated based on title, abstract, and relevance to treating AD with CBD. No restrictions on research type or publication year. Excluded were hypothesis papers, reviews, books, unavailable articles, etc.
Results
Microsoft Excel identified 551 articles, with 92 included in the study, but only 22 were thoroughly evaluated. In-vivo and in-silico studies indicate that CBD may disrupt Aβ42, reduce pro-inflammatory molecule release, prevent reactive oxygen species formation, inhibit lipid oxidation, and counteract Aβ-induced increases in intracellular calcium, thereby protecting neurons from apoptosis.
Conclusions
In summary, the study indicates that CBD and its analogs reduce the production of Aβ42. Overall, these findings support the potential of CBD in alleviating the underlying pathology and symptoms associated with AD, underscoring the crucial need for further rigorous scientific investigation to elucidate the therapeutic applications and mechanisms of CBD in AD.
Introduction
Alzheimer's disease (AD), the leading source of dependency and disability among individuals aged 65 and older, stands as the predominant cause of dementia in the elderly. Although there is a lower age range that is afflicted, it affects women more often than males and adults over 65 more frequently. The World Alzheimer's Report of 2018 estimates that 50 million people worldwide are living with dementia, with AD affecting two-thirds of those individuals.1,2 According to estimates, the number of people with AD will reach 152 million by the year 2050 as life expectancy rises. 3 AD can be treated with cannabidiol (CBD), which has been shown to slow down the disease progression. 4 According to the World Health Organization, 60–80% of the estimated 43.8 million dementia sufferers worldwide are thought to have AD, an age-related neurodegenerative disorder. 5 Cannabis sativa plants, also known as Hemp, naturally contain cannabinoids called phytocannabinoids which contain more than 400 different chemicals, the most well-known of which are delta-9-tetrahydrocannabinol (9-THC) and CBD. 6 The US FDA has approved the use of Sativex, a new formulation of THC and CBD, to treat multiple sclerosis and the injection of Sativex decreased the amounts of amyloid-β (Aβ) oligomers, phosphorylated tau, and GSK3 expression in a mouse model with human tau. 7 Consequently, the elements of the endocannabinoid system are thought to be a possible target for phytocannabinoids as a defense against AD pathogenesis its capacity to play a crucial part in preserving the body's homeostasis, which includes the brain, endocrine, and immunological systems so, it is far more challenging to comprehend how phytocannabinoids affect endocannabinoid system regulation. However, CBD has no psychotropic side effects and is well tolerated. 8 It performs several neuroprotective actions by modulating different receptors (Figure 1). 9
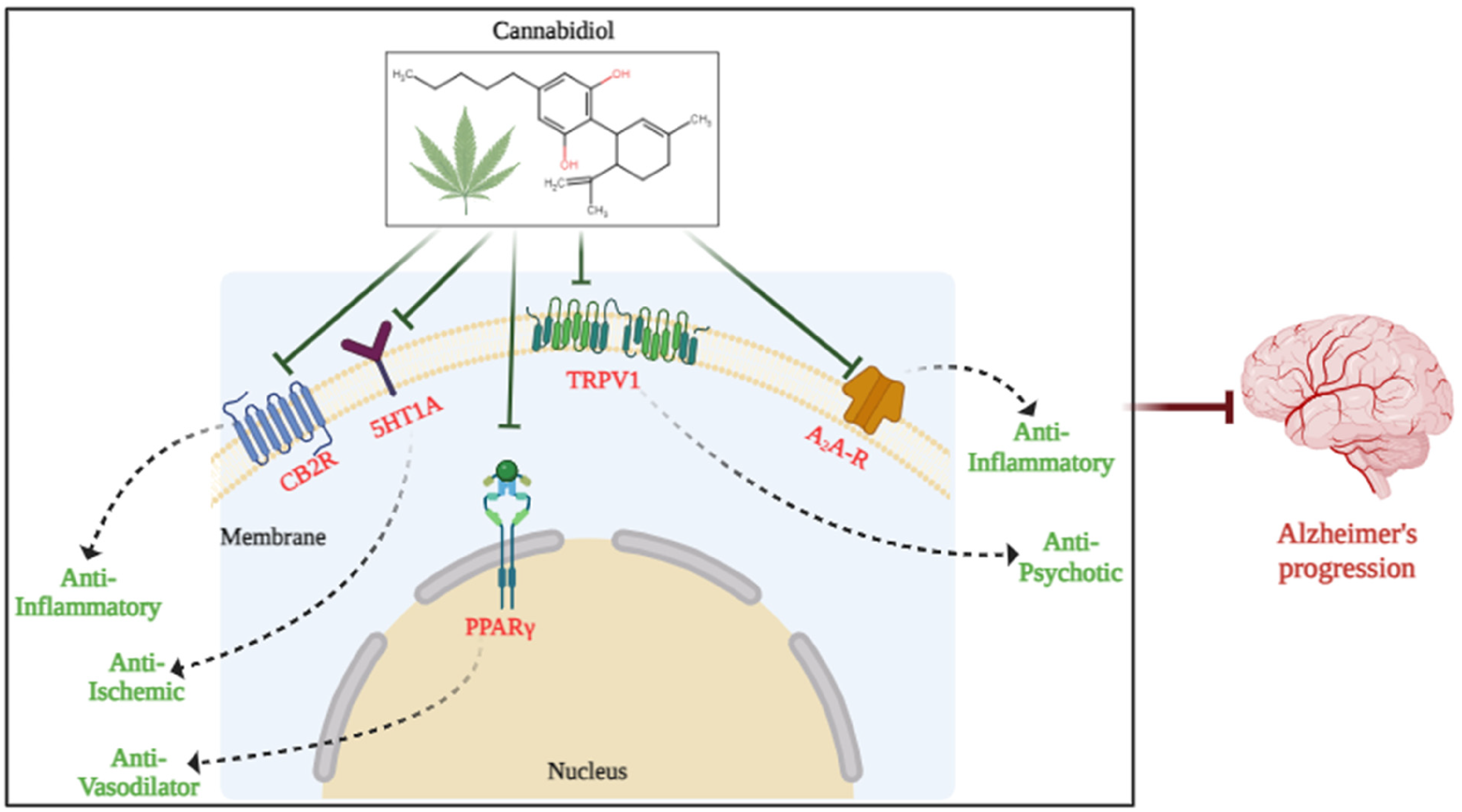
Potential biomarkers for CBD therapy of AD. Several potential biological targets of CBD involved in treating AD have been proposed, including CB2, 5-HT1A, TRPV1, PPARγ, and Adenosine receptors. These proposals based on the properties of each potential receptor target of CBD and well-established mechanism studies of CBD-mediated therapeutic functions.
As a result, the medicinal potential of CBD, particularly for AD, has drawn growing attention to its neuroprotective and non-psychoactive qualities, because the majority of preclinical research has shown that CBD has largely beneficial neurological effects. As far back as the late 1800s, medical professionals in the UK and the United States noted that extracts from cannabis were associated with a decrease in the occurrence of epileptic seizures. 10 In comparison to conventional antiepileptic medications, CBD exhibits greater effectiveness as an anticonvulsant, offering increased specificity and fewer neurotoxic side effects. 11 Additionally, CBD has great selectivity, no excitability in the central nervous system, and can decrease the duration and amplitude of the post-discharge cAMP response element-binding protein (CREB). The intracellular protein CREB controls the expression of genes that are crucial for dopaminergic neurons. Through G protein-coupled receptors, dopamine modulates CREB's phosphorylation. 12 The creation of efficient treatments for AD continues to be of utmost importance given its high prevalence and significant morbidity and mortality, since the identification of the neurofibrillary tangles and cerebral Aβ plaques, which are the pathological manifestations of AD and contain aggregates of hyperphosphorylated tau protein. On PC12 neuronal cells, research-employing CBD in vitro was carried out which findings demonstrated that CBD stimulated the Wnt/β-catenin pathway to activate the peroxisome proliferator-activated receptor (PPARγ) by exhibiting neuroprotection. 13 As a result, lowered reactive oxygen species (ROS) generation, lipid peroxidation, and proteins such as tau hyperphosphorylation, and suppressed acetylcholinesterase (AChE) while safeguarding PC12 cells from neurotoxicity and oxidative stress. 14 According to a study done on SH-SY5YAPP+ cells, it decreased the amounts of amyloid protein precursor (APP) full-length protein and Aβ synthesis.15–17 AD is a neurodegenerative disorder that causes permanent memory loss and cognitive decline because of a little disruption in the cerebral cortex and hippocampus functioning. 18 In PC12 cells exposed to A, CBD reduced the formation of Aβ plaques, stopped the creation of ROS and the oxidation of lipids, restrained the amount of neuronal apoptosis caused by caspase 3 decrease, and counteracted increases in intracellular Ca+2 caused by Aβ. CBD was anti-inflammatory in an in vivo model. 19 CBD's act as an anti-inflammatory compound by reducing inducible nitric oxide synthase (iNOS) and interleukin (IL)-1β protein expression. Additionally, it prevented the hyperphosphorylation of tau protein in PC12 neurons triggered by Aβ. The mode of action of CBD was later demonstrated to be dosage-dependently antagonistic of pro-inflammatory nitric oxide (NO), tumor necrosis factor-alpha (TNF-α), and IL-1β via PPARγ. 20 By specifically inhibiting PPARγ-related NF-κB, GW9662 (a PPARγ antagonist) reduced reactive gliosis and prevented that impact. Following exposure to Aβ, neurogenesis was increased by activity of anandamide and CBD. Furthermore, recent preclinical studies have demonstrated various beneficial effects of CBD, such as its anticonvulsant, anti-nausea, and analgesic properties.
Moreover, these studies have shown that CBD provides superior protection for neurons against free radicals compared to ascorbate or tocopherol. 21 THC is equally cytotoxic as it is cytoprotective, protecting neurons and healthy cells but not many cancer cells. Various conditions, including multiple sclerosis, Parkinson's disease, AD, epilepsy, and other systemic disorders resulting from nervous system degeneration or anomalies, can be effectively treated with CBD, thanks to its neuroprotective advantages, as supported by both preclinical and clinical research. 22 CBD is a frequent subject of research in the context of neurodegenerative diseases and their associated patho-mechanisms. 23
Materials and methods
Study protocol
The present systematic review was performed according to the Preferred Reporting Items for a Systematic Review and Meta-analysis (PRISMA) guidelines.24,25
Literature search strategy
Data were retrieved from PubMed, Scopus, and Web of Science (WoS) databases 26 from 7 May 2023 using the following keywords “Cannabidiol” and “Alzheimer's disease”. There were only English-language and freely available (open-access) literatures were chosen.
Eligibility criteria
We evaluated each study's title and abstract before including, all eligible studies, which included studies that corresponded to our objectives for the treatment of AD with CBD, studies with sufficient and comprehensive data, studies without restrictions from in vitro, in vivo, or in silico research, and studies without limitations on publication year. Hypothesis, communication, systematic reviews, books and documents, meta-analyses, randomized controlled trials, posters, conferences, current opinions, letters to the editor, and unavailable articles were all omitted from the exclusion criteria. 27
Collection of data
Two reviewers (N.P., K.P.) independently screened the titles and abstracts of papers using the aforementioned keywords after the duplicate articles had been removed. 28 After that, depending on the inclusion and exclusion criteria, relevant full-text articles were independently assessed. 29 Reviewer M.J. decided to exclude the publications that did not adhere to the study's objective.
Data synthesis and statistical analysis
Data were pooled using a random effects model when there was variability among the papers to give a more conservative assessment of the range of cannabinoids impact on AD. Heterogeneity was assessed using the I2 index of 50% and the chi-squared test with a p-value of 0.05, both of which are used to determine a substantial level of heterogeneity. 30 Heterogeneity is the variance between distinct study findings that has not happened by mistake. 31 Reviewers M.J. & N.P. used Microsoft Excel (Windows XPC) for the statistical analysis.
Results
Screening and searching of literature
A thorough scan of the electronic databases up to May 2023 turned up 551 items. 371 publications were checked for titles and abstracts after duplicates were eliminated, and 180 of them were excluded. 220 articles met the requirements for inclusion and exclusion, allowing for a more thorough evaluation of their entire texts. The systematic review could comprise 91 papers in total. Figure 2 depicts the procedure of the literature search and screening process. Out of 91 papers, 21 articles were carried further for molecular mechanism elucidation in AD with CBD or its analogs. These 21 articles were mainly focused on in silico, in vivo, and in vitro approaches.
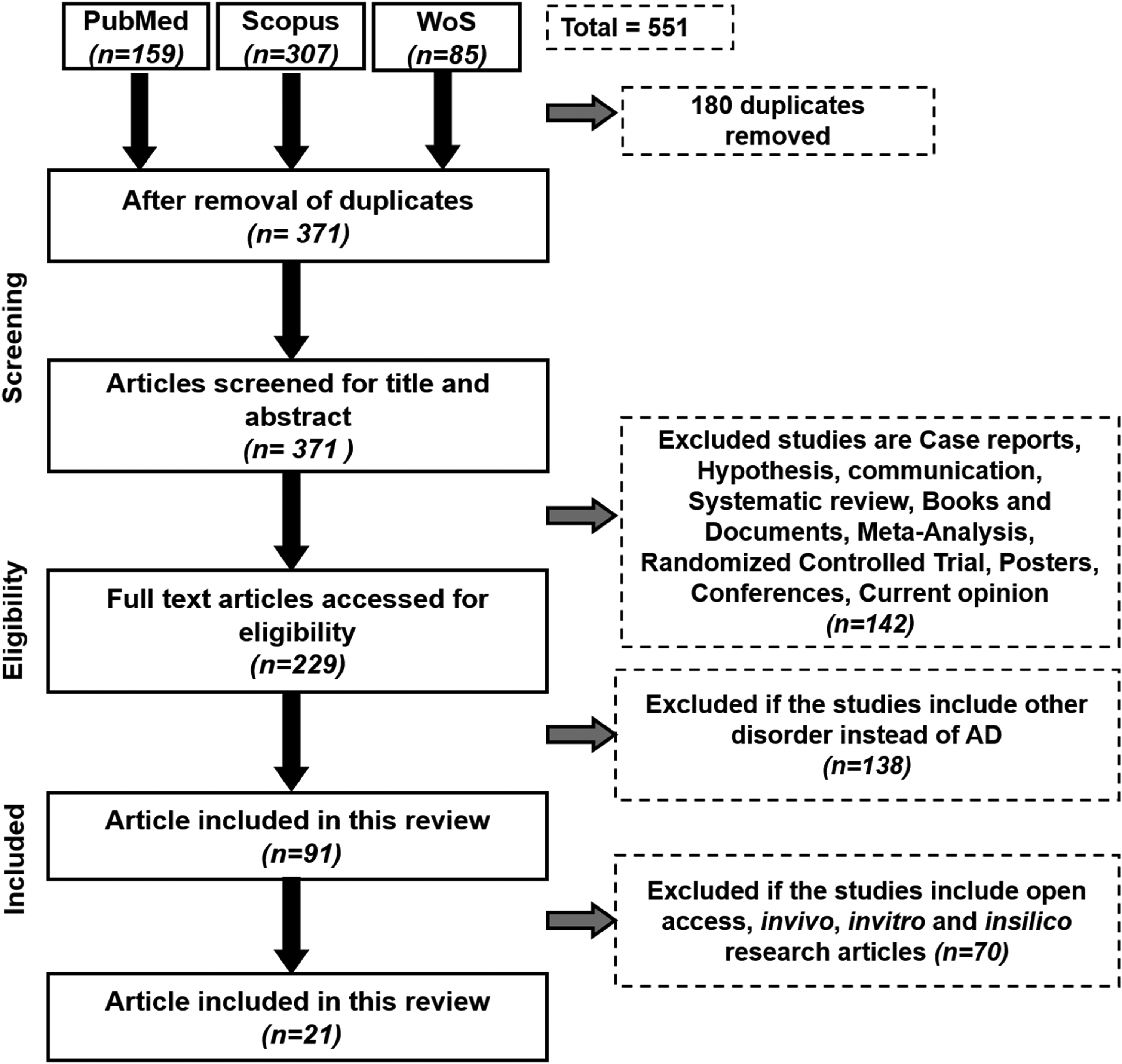
Schematic representation of the workflow.
Data extraction
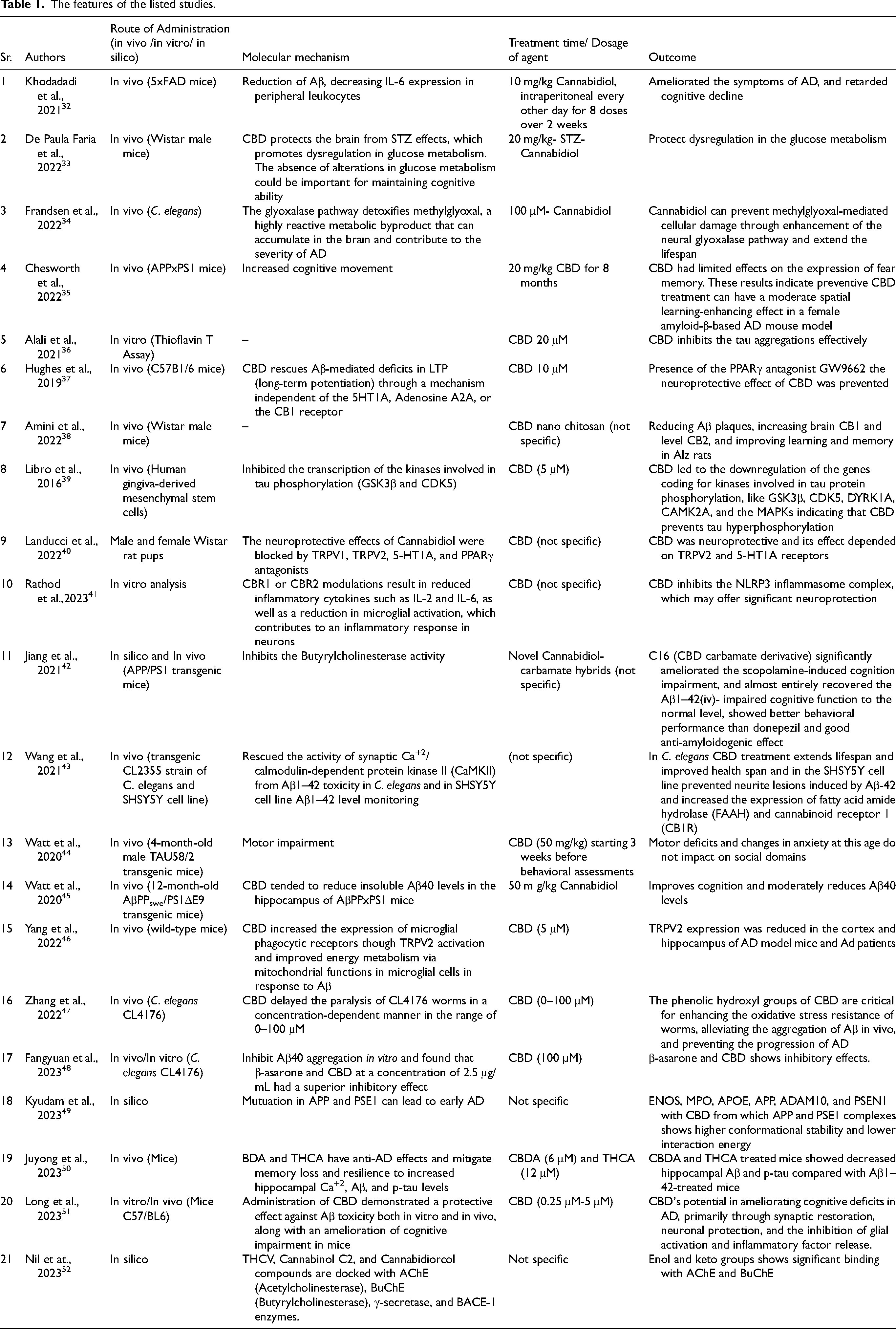
Authors NP & KP extracted the data from each article, which they subsequently compiled in Table 1, and author MJ checked it under the following headings: author information, route of administration (in vivo, in vitro, and in silico), molecular mechanism, treatment duration/agent dosage, and the effects of co-administering CBD and its equivalents on AD.
The features of the listed studies.
Discussion
Effects of CBD on AD pathology
The activation of microglia-induced neuroinflammation arises from multiple molecular pathways. In a 2020 study by dos-Santos-Pereira et al., CBD (1–10 µM) was found to inhibit the release of TNF-α, IL-1, and glutamate in LPS-induced microglial cell cultures. This effect was attributed to CBD's anti-inflammatory and antioxidant properties, which were evident through the downregulation of IkappaB (IκB) kinase-2 and NADPH oxidase apocynin. Notably, the anti-inflammatory impact of IB kinase-2 was synergized by its antioxidant capacity, as confirmed by the subsequent reduction in NF-κB-mediated signaling events. 53 Numerous studies have shown that the NF-κB pathway is essential for CBD's ability to inhibit neuroinflammation in TLR4-mediated pathways. When stimulated by extracellular inducers such as LPS, IRAK-1, and IκB undergo ubiquitination, leading to NF-κB activation and inflammation. CBD (1, 5, or 10 µM) partially inhibits IRAK-1 degradation, preserves IκB, and reduces NF-κBp65 phosphorylation, thus contributing to the suppression of inflammatory gene expression in LPS-activated BV-2 microglia via the IκB-NF-κB pathway reversal. 54 CBD's anti-inflammatory effects are mediated through the IFN-STAT pathway, which activates genes for CXCL10, CCL5, and CCL2. STAT family members express pro- and anti-inflammatory genes, with STAT 1 homodimer having pro-inflammatory effects. Increased IL-10 production or binding to STAT3- causes anti-inflammatory effects in different IL-10-inducible genes’ consensus sequences. CBD dose-dependently decreased proinflammatory STAT1 activation and enhanced STAT3 activation, resulting in anti-inflammatory effects and inhibition of inflammation, linked to STAT1 and STAT3 expression control (Figure 1). 55 Additionally, CBD may reduce fatty acid amide hydrolase (FAAHs) enzymatic activity and boost the activity of anandamide and 2-AG concentrations, which would lower AD pathology, and excessive glutamatergic transmission.23,56 The G protein-coupled receptor 55 (GPR55), which does not share any amino acid sequences with the conventional CB1 and CB2 receptors, is regarded as a third cannabinoid receptor.57,58 In contrast to the Gi-coupled CB1 and CB2 receptors, the G-coupled receptor GPR55 is unique. Intracellular calcium and neuronal excitability are elevated when GPR55 is activated, which in turn affects cellular migration and proliferation and glutamate release in the hippocampus formation, which may be related to inflammation and cognitive functions.59,60 CBD's ability to interact with diverse receptors, encompassing serotonin, vanilloid, adenosine, peroxisome proliferator-activated, opioid, and dopamine receptors.51,61,62 Therefore, in addition to the presumed effects of cannabinoid receptors, CBD offers intriguing medicinal potential. Due to its antidepressant or anxiolytic effects 61 and its function as a neurotransmitter in the brain, it has been demonstrated that the serotonin 1A receptor (5-HT1A) receptors are primarily responsible for TRPV1 and are a crucial biological target for CBD treatment in numerous forms of pain and inflammation. 62 However, in the context of inflammation, all of these receptors may collaborate rather than function separately. For instance, a recent study revealed that blocking A2A receptors might enhance CB2 receptor-mediated signaling in the context of an A2A-CB2 receptor heteromer. Numerous target genes and signaling cascades are regulated by PPARγs, which also start distinct physiological reactions such as cell division, proliferation, and anti-inflammatory activities.63,64 Fatty acid-binding proteins can transport CBD to the nucleus, where it subsequently binds to PPARγ directly, activating it and causing a variety of PPARγ-mediated alterations. 65 PPARγ has been frequently found to be a crucial regulator of the neuroprotective impact of CBD against AD progression. 66 Furthermore, CBD has been referred to as an allosteric opioid receptor modulator. 67 There were three types of CBD studies possible in AD in silico, in vitro, and in vivo.
In silico, in vitro, and in vivo study of CBD in AD
The Achilles server's in silico study tools were used to analyze the interaction between CBD and tau protein through best-fit docking. Donepezil, rivastigmine, and galantamine with binding affinities of −8.77, −7.0, and −7.97 kcal/mol were employed as reference AChE receptor analogs for comparison.68,69 Using memantine as a reference with a binding affinity of approximately 6.83 kcal/mol, THCV, Cannabinol C2, and Cannabidiorcol from cannabis demonstrated superior inhibition of AChE and butyrylcholinesterase (BuChE). Computational analysis suggests that THCV, Cannabinol C2, and Cannabidiorcol are the most effective compounds for inhibiting these enzymes associated with AD.70,71 Through both biochemical and in silico methods, it was shown that CBD interacts with tau protein, inhibiting its amyloid fibril development via hydrogen bonds and hydrophobic forces. CBD also promotes the transformation of tau protein from a random coil shape to a more rigid structure, reducing the formation of tau clumps. 72
Concerning in vitro studies, CBD has several properties that are directly related to its therapeutic effectiveness for AD 73 found that CBD reduced glycogen synthase kinase-3 (GSK-3; the enzyme that causes tau hyperphosphorylation). Importantly, CBD increased PPARγ-induced APP ubiquitination in APP-transfected human neuroblastoma cells, which resulted in decreased APP expression 74 and decreased Aβ peptide expression. In addition, CBD decreased the expression of the genes that code for the secretases involved in the enzymatic production of Aβ, such as BACE1, PS1, and PS2. CBD has also been found to weakly, but fascinatingly; inhibit direct secretase. 75 In addition, hemp seed oil (80% CBD) protected MC65 cells, an AD neuron-like cell model, from amyloid toxicity. 76 CBD also exhibits the potential to mitigate mitochondrial dysfunction, a significant aspect of AD. Moreover, CBD reduces oxidative stress by lowering ROS and NOX isoforms, including NOX, known for ROS production. 77 In vitro data suggest that CBD can rescue the Wnt/-catenin pathway, linked to tau hyperphosphorylation, and reduce iNOS protein synthesis and NO release. 78 Additionally, CBD's ability to reduce reactive gliosis may be associated with its inverse agonist role at the cannabinoid receptor 2 (CB2), a factor implicated in the condition. 79 Male 5xFAD transgenic mice aged 9 to 12 months were employed. Mice in the treatment group received eight intraperitoneal injections of CBD isolate at a level of 10 mg/kg every other day for two weeks. Mice receiving a placebo were given it on the same schedule as receiving CBD. With reduced amyloid levels in brain tissue and enhanced TREM2 expression, CBD may help with amyloid clearance or prevent amyloid formation. Much like its enhancement of contextual memory in an AD mouse model, CBD increased the expression of IL-33 in astrocytes, a cytokine that stimulated microglial amyloid phagocytosis. Consequently, CBD could potentially serve as a safe and effective disease-modifying treatment to delay neurocognitive decline, partially by altering the function of glial cells. 32 Wistar male rats were administered 3 mg/kg of STZ, with [18F] FDG PET scans taken before and seven days after the injection. The animals were treated with either saline (STZ-saline) or 20 mg/kg of cannabidiol for a week. Novel object recognition assesses short- and long-term memory. The STZ-saline group exhibited significant reductions in [18F] FDG uptake in the whole brain, particularly in a hypometabolism cluster near the lateral ventricle. STZ-cannabidiol rats had a smaller cluster, and specific regions like the motor cortex, hippocampus, and thalamus showed less hypometabolism compared to STZ-saline animals. Memory performance was unchanged in the STZ-cannabidiol group. This suggests that cannabidiol may hold promise as an early treatment for the neurodegenerative processes observed in AD. 33
Concerning in vivo studies to define the mechanism by which CBD reduces AD, the model organism C. elegans was selected. Transgenic C. elegans models have been created for several diseases, including AD. The advantages of using C. elegans over rat animal models include its short lifespan, low cost, ease of manipulation, and accessibility to a large number of multicolor reporter structures. The APP and tau genes share 38% of their genome sequences with C. elegans, according to Consortium and Elegans Sequencing (1998). Our understanding of the etiology and pathophysiology of AD as well as the molecular mechanisms behind treatment action has further advanced thanks to the discovery of transgenic C. elegans worms that express the human Aβ or tau protein. This study found that CBD prevented Aβ aggregation in vivo, which substantially reduced the course of AD. Importantly, it was found that CBD's phenolic hydroxyl groups are essential for slowing AD's progression. This study suggests CBD's anti-inflammatory qualities give it its anti-AD therapeutic effects. 34 Therapeutic benefits of CBD have been observed in APPxPS1 mice at different doses (ranging from 5 to 50 mg/kg and using CBD-enriched extract). Chronic CBD treatment reduces astrocytic response and the expression of the cell surface adhesion molecule CCL4 in the APPxPS1 microenvironment. However, its impact on Aβ levels in the hippocampus is only modest. Nevertheless, there is currently no evidence demonstrating a significant effect of therapeutic CBD treatment on other AD-related receptors and molecules in APPxPS1 mice, including brain-derived neurotrophic factor, PPARγ, ionized calcium-binding adaptor molecule 1 (IBA1), and various cytokines. 35
CBD receptors present in the brain
CBD's lipophilic features led to the assumption that they cause various physiological functions via inhibiting cell membranes in an unstructured way. 80 Type 1 and type 2 cannabinoid receptors, respectively, make up the endocannabinoid system CB1 receptors, which are G protein-coupled receptors (GPCRs) linked to the Gi/o protein (G protein alpha subunits), inhibit intracellular adenylyl cyclase and N-type calcium channels when activated. 81 CB1 receptors have not been found in human microglia; however, they are found in the CNS, predominantly on neurons. 82 In addition, direct CB1 activation is said to be the most common GPCR in the mammalian brain and contains some adverse effects on the CNS which may be severe. 83 While inhibiting FAAH typically elevates anandamide levels and consequently boosts CB1 activation, it also leads to an increase in the levels of other endogenous FAAH substrates that activate various receptors. These receptors include the transient receptor potential cation channel subfamily V part 1 (TRPV1), the PPAR, the orphan GPCR 119 (GPR119), and the orphan GPCR 55 (GPR55). 84 Peripheral immune cells primarily contain the CB2 receptor. 85 CB2 is mostly present in microglia cells and only weakly expressed in the brain under normal circumstances. 86 Additionally, it was possible to distinguish a modest level of CB2 expression on neurons, where its activation controls neuronal excitability. 87 CB2 expression is elevated on microglia when there is an inflammatory response, as well as on T-cells, macrophages, or astrocytes. However, the inflammatory environment influences expression levels. Following an attempt with LPS or IFN-γ, microglia treated with CB2 agonists had a decreased pro-inflammatory response.88–90 Recently, a study unveiled the structural changes of CB1R upon agonist binding, elucidating the conformational mechanism responsible for the diverse structures and signaling biases of CB1R agonists. 91 CBD is believed to act as a negative allosteric modulator (NAM) for both CB1R and CB2R. CB1R certainly serves as an NAM, regulating CB1 signaling when CBD are present. The most prevalent endogenous cannabinoid signaling neuronal model is CB1 NAMs, which are found in autaptic hippocampal neurons. 92 CBD's capacity to inhibit the CB2R active state (NAM activity) is a result of the pentyl chain's insertion into the hydrophobic pocket between TMs 2, 3, and 7. The activation of CB2R with various-length CBD analogs and in addition to the canonical long-form.93,94 Two other CB1R isoforms with short N-termini that also result from alternative splicing have been discovered. Current investigations in non-human models found inconsistencies thus the biological 95 and pharmacological features of all the dual splice variants are still to be investigated. In comparison to the CB1R, the CB2R exhibits more species variations between humans and rats, with amino acid sequence similarity of just over 80% among humans and rodents. Two isoforms of CB2R are recognized in humans: one predominantly in the testis and minimally in brain reward regions, and another mainly in the spleen with limited brain presence. Their roles in endocannabinoid-mediated synaptic transmission are unclear.96–98 CB1R is the most widely expressed GPCR in the brain, with highest levels in the olfactory bulb, cerebellum, basal ganglia, and hippocampus, and moderate expression in areas like the hypothalamus, amygdala, and cerebral cortex. 89 Mitochondrial CB1R is strongly linked to neuroprotection following cerebral ischemia/reperfusion. 99 CB2R, initially found in macrophages in the spleen, is prominent in immune cells and moderately expressed in peripheral tissues.100,101 Both CB1R and CB2R are expressed in the CNS, with CB2R regulating brain activity via Ca+2-activated channels.102,103 The role of CB1 receptors (CB1R) in modulating physiological functions through the endocannabinoid system. CB1R, found in high numbers throughout the human body, 104 each with a range of physiological functions influenced by the CB1R and the endocannabinoid system. CB1R expression varies dramatically in a variety of clinical situations, 105 highlighting its vital function in a wide range of biological processes, influences neurotransmission by regulating the release of GABA and glutamate.106,107 This receptor protects neurons from excitotoxicity, a harmful process in neurological conditions.108,109 by reducing NO production, zinc mobilization, and enhancing brain-derived neurotrophic factor expression. It also inhibits calcium influx and glutamate release, highlighting its neuroprotective properties.110,111
In numerous neurodegenerative disorders, abnormal activation of the CB1R and other parts of the whole endocannabinoid system has been found.112,113 Considering the foregoing, it is possible to speculate that Aβ deposition triggers the release of endocannabinoids in the neurons and glia, which stimulate CB1-mediated neuroprotective mechanisms and control the release of inflammatory mediators into microglia into CB2 receptors. Future research may explore the therapeutic potential of CB1/CB2 agonists and FAAH antagonists in AD. Recent pilot studies indicate that daily treatment with dronabinol for two weeks significantly improved overall Neuropsychiatric Inventory scores, including agitation, aberrant motor behavior 114 and nighttime behaviors in late-stage dementia patients (including those with AD and vascular dementia). Elevated CB1 receptor expression and endocannabinoid system activity suggest a possible pathway for compensating dopaminergic neuron degradation in the substantia nigra. Preclinical studies in AD animal models have also shown benefits from CB1R modulation. 115 The protective effects of Arachidonyl-2'-chloroethyl amide (ACEA) upon AE-induced memory impairment were eliminated when CB1 receptor blockade by AM251 was applied. Rats co-treated with AE + WIN + AM251 with AE + ACEA + AM251 did not display any memory-protecting effects. 116
The CB2R plays a crucial role in immune function within the CNS and peripheral areas. Microglia, a persistent community of cells similar to macrophages, play a major role in this function. In pathological situations, microglia act as immune effector/accessory cells, moving and multiplying during damage and inflammation. They release cytokines like interleukin-1, IL-6, and tumor necrosis factor-α, which express MHC class I and II antigens and the CD11/CD18 complex complementing receptor. 117 Additionally, phagocytic, microglia can digest antigens and perform cytolytic actions when activated. Contrarily, these cells have been linked to nervous system disorders such as MD, AD, Parkinson's disease, and even acquired immune deficiency syndrome (AIDS) dementia in addition to their involvement in host defense and tissue regeneration in the CNS. 118
Molecular mechanisms of CBD in AD (all the mechanisms)
Due to its anti-inflammatory and antioxidant effects, CBD's neuroprotective benefits are aimed at modulating receptors and channels implicated in neurodegenerative disorders. 117 G protein-coupled receptors, TRPV1, and PPARγ are just a few of the non-endocannabinoid signaling systems that CBD interacts with According to 113 ; the GABAergic modulation of CBD may also contribute to its therapeutic effects Figure 3.
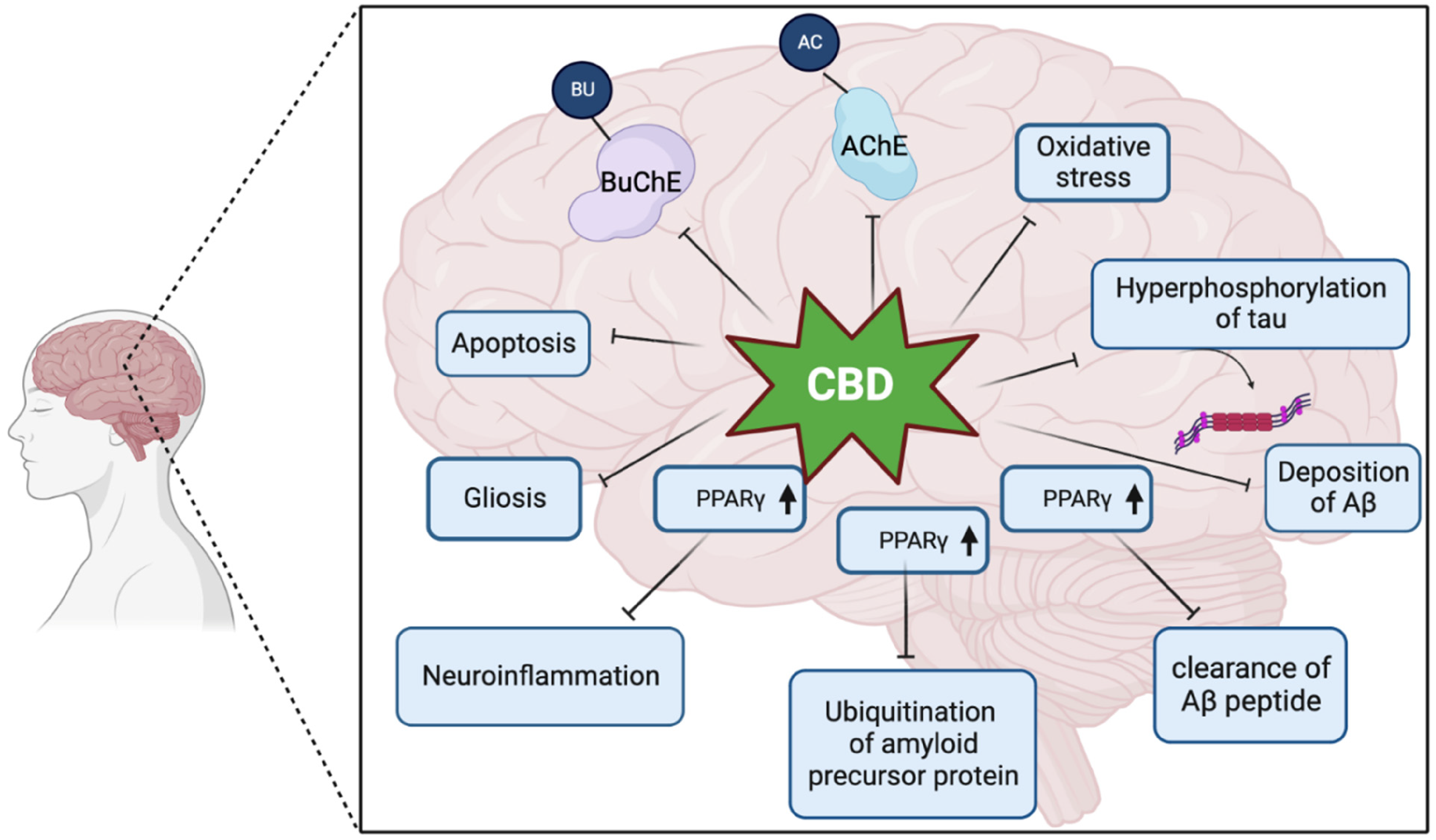
Different molecular mechanism that is affect by CBD in AD brain. BuChE, butyrylcholinesterase; AChE, acetylcholinesterase; PPARγ, peroxisome proliferator-activated receptor.
Rheostats for adenosine
ARs (A1R, A2AR, A2BR, and A3R) are endogenous adenosine-induced GPCRs that play a part in a variety of physiologic processes. Adenosine stimulation of A1R and A3R ends in the stimulation of repressive G (Gi/o) proteins, 119 which inhibits adenylate cyclase alongside reducing cyclic adenosine monophosphate (cAMP) within the cells. A2AR and A2BR, on another side, induce G protein activity and, as a result, an increase in cAMP. ARs stimulation includes the alteration of secondary messengers and supplementary signaling channels like that of phospholipase C, a Ca+2-dependent protein kinase associated with cell interaction; phosphoinositide 3-kinases signaling convoluted during cell differentiation, 120 proliferation and growth; and ion channel initiation and Ca+2 modulation. Although CBD appears to work through a variety of signaling pathways, 121 it appears that A2AR is involved in its anti-inflammatory actions. CBD's anti-inflammatory impact is mediated directly through A2AR, whose activation causes the immune response to be regulated and pro-inflammatory cytokines to be reduced. In healthy conditions, the striatal neurons have a high level of A2AR expression compared to glial cells and neurons outside of the striatum. Additionally, this receptor subtype is found in the olfactory tubercle, caudate putamen, and nucleus accumbens. It is important to note that brain injuries cause microglia to express more A2AR, which results in signal transductions that do not happen in cells that express the receptors normally, 122 such as promoting the release of cytokines. Strong support was offered for an A2AR-CB1R interaction taking place in the hippocampus by merging in vivo and complementing molecular approaches. The A2AR/ CB1R oligomer appears to be important for how well CBD can counteract the cognitive impairment brought on by the psychoactive cannabis constituent 9-THC in a declarative and spatial memory exercise. 123 The pre-treatment with the selectively pre- and postsynaptic A2AR antagonist SCH442416 as well as KW-6002, respectively, revealed for the first time that this CBD action was primarily reliant on presynaptic A2AR receptor activation. 124 Microglial activation results in an increase in CB2 and A2A receptor expression. 125 The goal was to identify microglial heteromer 126 and any variations in A2A-CB2Hets expression between dormant and active microglial cells. The outcomes from signal transduction assessments in resting microglia were consistent with those observed in co-transfected HEK-293 T cells. Through evaluations of cytosolic cAMP levels, ERK1/2 phosphorylation, and DMR, it was revealed that the A2AR and CB2R agonists generated a distinct and non-additive signal when applied together, resembling the signal achieved with CGS21680. 126 CBD tends to improve adenosine signaling,, 127 as a result, there is a rise in extracellular adenosine as well as a decrease in neuroinflammation. In cultured N13 cells and native microglial cells, CBD reduced ATP-activated intra-cellular Ca+2 rise. Over N13 microglial cells including basic rat microglia, the application of ZM241385. CB2 appears to be more crucial in controlling128,129 the activation of microglia after stroke. Targeting CB1 is limited in therapeutic potential due to its widespread presence in the brain among both neurons and glia, the psychotropic effects of agonists like phytocannabinoids or synthetic CB1R agonists, and the adverse effects associated with the use of rimonabant, a selective CB1R ligand, in humans. Recently, paeoniflorin, a prominent constituent of Paeonia lactiflora, has been found to activate CB2R, which plays a role in regulating the polarization of microglia and macrophages in the CA1 area of the hippocampus in vascular dementia models.130,131 In vivo, CBD with quite a dosage of 20 mg/kg was in a position to intercept amyloid-β-induced consequent disfigurement. CBD did lower the assertion of the gene-producing IL-6, a pro-inflammatory cytokine, following three weeks following intraperitoneal treatment. As a result of these findings, CBD that interacts with A2AR might be a promising treatment option. 132 In conclusion, CBD has anti-inflammatory effects in AD models via A2AR activation. In cultured N13 and primary microglial cells, CBD-suppressed ATP-induced intracellular Ca+2 rise, along with A2AR receptors can be implicated during this process. CBD inhibited the production associated with pro-inflammatory cytokine IL-6 in vivo, 133 which reversed the cognitive impairment caused by Aβ.
GPR3, GPR6, and GPR12
GPR3, GPR6, and GPR12 being known as forsaken receptors since their inland agonist is unknown. Before being cloned from human genomic libraries by numerous independent researchers,134–137 GPR3 was first isolated from a mouse cDNA library in 1993. 138 It was also recently cloned from a library of porcine cDNA 139 GPR3 was located on chromosome 1p35–1p36.1 by Marchese et al. 130 This receptor's location was determined by Song et al. 134 and Iismaa et al. 138 to be 1p34.3-p36.1. Neurons across several brain areas, including the cortex, thalamus, hypothalamus, amygdala, hippocampus, pituitary, and cerebellum, express GPR3 mRNA widely.136–140 Notably, postmortem brain tissue slices from people with AD show that the GPR3 protein is overexpressed in neurons. 140 Along with other tissues, the eye, lung, kidney, liver, testes, and ovary all express GPR3 mRNA.136–140 In 1994, a rat cDNA library was used to clone GPR6, which was then given the name rCNL3. 141 Its cloning from human genomic DNA came next in 1995. 134 GPR6's chromosomal location was determined to be 6q21–22.1 by Heiber et al. 142 and Song et al., 143 respectively. The brain's neurons, notably those in the striatum (caudate, putamen, nucleus accumbens, and olfactory tubercle), are the primary sites of GPR6 mRNA expression. It can also be found, though to a lower extent, in the frontal cortex, retrosplenial cortex, hippocampus, amygdala, and hypothalamus.140,141 GPR6 expression seems to be minimal in peripheral tissues. In a study by Song et al., 142 GPR6 mRNA was not detected in various peripheral organs such as the heart, spleen, lung, liver, muscle, kidney, and testis in rats. GPR12, originally known as R334 in rats 144 and later as GPCR01 in mice, 145 was eventually cloned from human genomic DNA in 1995 and mapped to chromosomal region 13q12. 134 GPR12 mRNA was initially identified in the pituitary gland. In the brain, this transcript is present in neurons located in regions like the frontal cortex, piriform cortex, thalamus, hypothalamus, hippocampus, amygdala, and olfactory bulb. Peripherally, GPR12 mRNA can be found in the testis and oocytes.140,144–146 However, the transmembrane domains of these types of forsaken receptors have 35% amino acid sequence similarity 147 with both CB1R and CB2R. It was only recently discovered that GPR3, GPR6, and GPR12 constitute new CBD markers. While CBD has numerous known markers, the latest finding in GPR3, GPR6, and GPR12 148 as additional CBD markers offer a fresh choice on the drug's therapeutic potential and benefits that calls for more research. In the β–arrestin 2 assignment experiment, 148 CBD was not the powerful target for GPR3 (EC501 M) or GPR6 (EC50100 nM). It also is not a very potential target for GPR12-directed cAMP buildup (EC5010 M). CBD, on the other hand, still serves as a molecular framework for the development of new, possibly more powerful, 149 and efficient GPR3, GPR6, and GPR12 agonists. C1q, a protein that binds to Aβ plaques and assists in plaque clearing while simultaneously generating a harmful inflammatory response, 150 has been discovered in people with AD. GPR6 is elevated in AD animal models via a C1q-mediated pathway, in accordance to Benoit et al., 2013. In the mice model, it was discovered that GPR6 can require C1q-mediated neuroprotection with odds to Aβ -induced neurotoxicity. As a result, GPR6 may perform a powerful role in the neuroprotection of AD. 151
PPARγ
PPARγ is a nuclear receptor that is also a ligand-activated transcription factor that controls overall levels of expression for genes mediators of inflammation, trophic factor synthesis, redox balance, glucose, and lipid metabolism. A ubiquitin E3 ligase is also known as PPARγ. with many lysine residues, notably Lysine48 which is a type of ubiquitin chain that is linked by lysine 48 (Lys48) and serves as the most efficient trigger of protein degradation. It is synthesized by the ubiquitin-conjugating enzyme Ube2 K (also known as E2–25 K and Huntington Interacting Protein 2) that can form polyubiquitin chains. Indeed, PPARγ activation prevents the NF-κB signaling pathway by inhibiting their pro-inflammatory genes transcription, 152 such as TNF-α, IL-6, and IL-1β. It was demonstrated that the pro-neuroprotective effect was reliant on PPARγ receptor activation. 153 Significantly, CBD did not promote neuronal differentiation when a PPARγ antagonist GW9662 (2 µM) was also delivered, which hindered its neuroprotective effects. 154 It is known that GW9662 binds to PPARγ permanently and possesses an IC50 with in nM range. 155 Even if these data are insightful, they require further validation given the numerous protective effects of PPAR receptor activation alone. 149 Neurogenesis and hippocampal function are also essential for the symptomatology of diseases associated with anxiety, depression, and AD. 156 Since the endocannabinoid (eCB) system plays a crucial role in the regulation of neuronal production and lifespan, Kempermann's team first investigated whether a cannabinoid like CBD would increase the longevity of dentate gyrus (DG) newborn neurons in mice.157,158 An innovative study using NeuN as a marker discovered that a diet rich in CBD significantly increased the co-localized immunoreactivity of 5-bromo-20-deoxyuridine (BrdU) and neuronal nuclei, suggesting pro-neurogenic effects. Notably, an intriguing counterargument was presented against THC's impact on this measure. Demirakca et al. (2010) later supported these pro-neurogenic effects of CBD in humans. 159 The endocannabinoid (ECB) system emerges as a crucial regulator of neonatal neuron formation, functional integration, survival, and maturation in the adult hippocampus. Endocannabinoid signaling plays a role in the development of neural progenitor cell progeny, which possess a functional ECB system. 160 According to Andrés Mach et al., 161 CB1 agonists cause neuronal growth and differentiation in the DG, in CB1 /- mice, which are also diminished. 162 Similar evidence has been presented for CB2 receptors, albeit in a more complicated way.163,164 In other words, CB1 receptors help to maintain adult neurogenesis while CB2 receptors appear to aid the recovery from allos9otatic neurogenic conditions. 165 Most of the fundamental mechanisms behind memory loss are unclear. Epistolary memory, spatial identification, and visual and semantic orientation are all affected by early memory problems in AD. 166 The DG within the hippocampus serves as a neurogenic niche where neural stem cells differentiate into new neurons that become part of the granular cell layer. 167 These newly generated DG cells in the adult brain integrate into neural networks and contribute to hippocampal function. 168 The DG mediates the learning, extinction, and retrieval phases of contextual fear conditioning, with the latter two exhibiting distinct neural patterns. 169 In the erasure of memories, contextual discrimination, and hippocampal plasticity, immature neurons play an important role.170,171 It has also been shown that immature neurons are drawn into networks for spatial memory. 172 Increasing neurogenesis in 5XFAD animals with Nestin-driven CreERT2 predicate Bax deletion restored the density of dendritic spines, increased the number of new neurons chosen into memory engrams, and changed the alterations made to both immature and mature neurons, eventually resulting in the recovery of contextual and spatial memory.156,173 As a result, PPARγ agonists like CBD, by decreasing downstream gene transcription controlled at NF-κB, 174 may have a type of anti-inflammatory effect via the molecular process monitored by GSK3-β. Using SH-SY5YAPP + cells, as a model of AD 145 investigated this method of action. CBD (10−9–10−6 M) was given to SH-SY5YAPP + cells during 24 h. CBD incorporation reduced the levels of expression and ubiquitination of those in the AβPP protein, resulting in a decrease in Aβ and neuronal death. To determine the specific role of PPARγ in mediating CBD's effects in SH-SY5YAPP + cells, CBD was manipulated using MK886 (3 μM) and GW9662 (9 nM), which are specific agonists of PPARα and PPARγ, respectively. Treatment with GW9662 (9 nM) led to a significant reduction in the effectiveness of CBD, indicating that the neuroprotective actions of CBD are mediated through PPARγ receptors. This suggests that CBD may have potential for AD treatment, partly due to its interaction with PPARγ. In an experiment using C57/black 6 mice, CBD was administered to hippocampus sections 30 min before soluble oligomer Aβ1–42 injection. CBD treatment enhanced synaptic potentiation and transmission over time, safeguarding against cognitive deficits induced by Aβ1–42. To better understand CBD's mechanisms, the perfusate was pre-treated with WAY 100635 (300 nM; 5-HT1A antagonist), ZM241385 (100 nM; A2AR antagonist), AM 251 (2 mM; CB1 inverse agonist), or GW9662 (2 mM; PPAR antagonist). 145 It was discovered that only GW9662 therapy reduced CBD's neuroprotective properties. 154 CBD is the cannabinoid with less affinity for PPARγ receptors from THCA, THC, CBGA, CBG, CBDA, and CBD 175 and this opens the door to new possible therapeutic options using acidic cannabinoids targeting PPARγ receptors at smaller dosages and in a more effective form.
Transient receptor potential channels (TRP)
TRP comprises a group of ion channels that are mostly found on cell membranes present in animal cells. CBD may bind with TRP, 176 hence reducing neuroinflammation. TRPV1, a member of the Vanilloid subfamily of TRP channels, acts as a robust antagonist of CBD. CBD stimulates TRPV1, which subsequently triggers the PI3 K/Akt signaling pathway, leading to the inhibition of glycogen synthase kinase 3. The inhibition of GSK-3 results in increased activity of the Wnt/β-catenin pathway, 177 which has a neuroprotective effect concerning neurotoxicity and oxidative stress that causes AD. CBD therapy inhibited hyperphosphorylation in tau protein-mediated to GSK-3β and β-catenin in Aβ-induced PC12 neurons, in keeping with this data for the in vitro model of AD. CBD also reduced Aβ-contents using SH-SY5Y transfected cells with amyloid progenitor (SH-SY5YAPP+), 145 as well and its treatment improved cognitive damage caused by AD in mice models. In a previous study, the research team investigated the role of TRPV2 in the biological mechanisms of CBD by comparing the behavior of human gingival mesenchymal stem cells treated with CBD (5 μM) and those that were not. CBD was found to suppress the expression of genes associated with AD, as revealed in a transcriptome analysis. 178
CBD and its derivatives in clinical trials
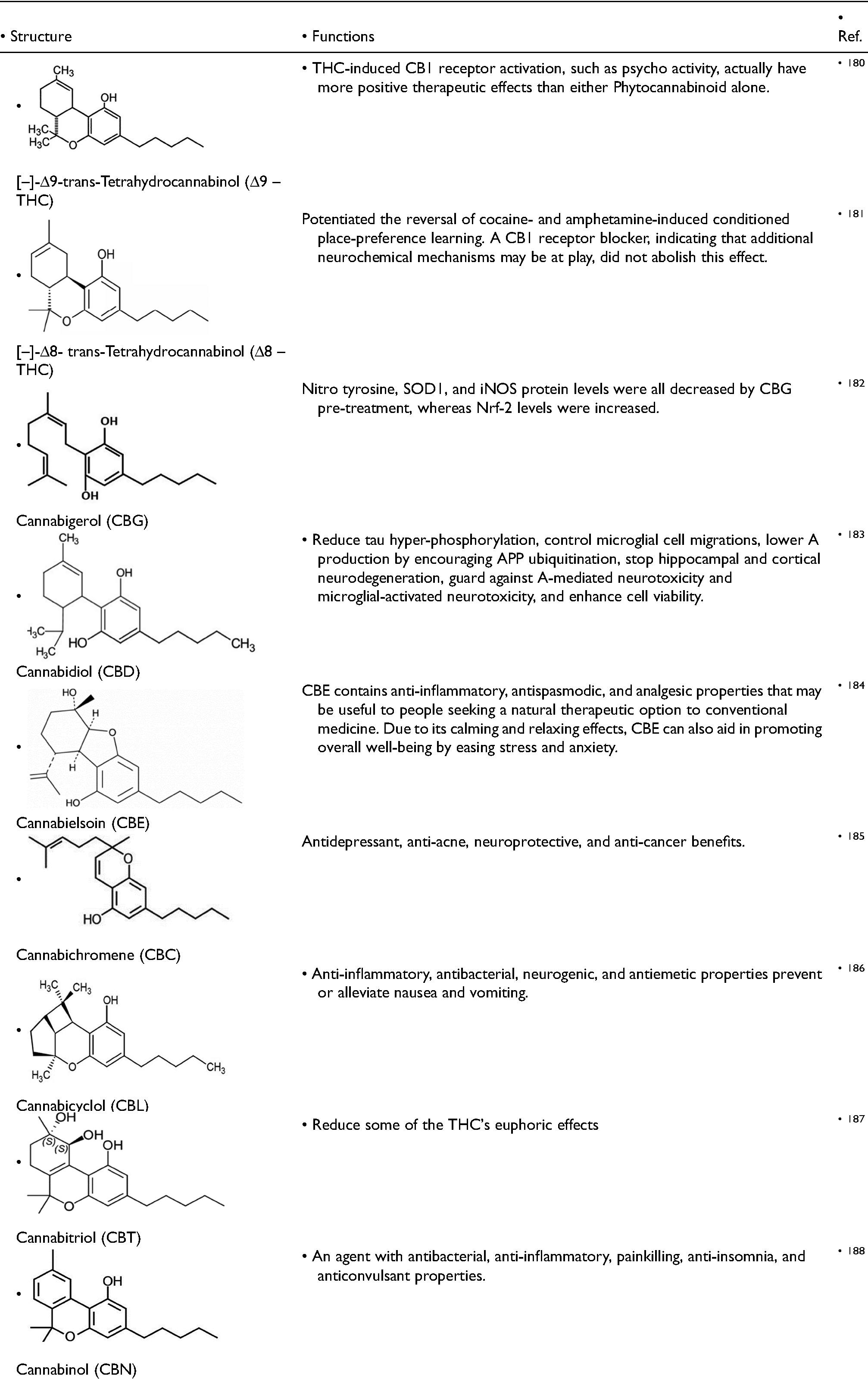
Mechoulam et al. initially identified the structure of CBD in 1963. 179 In 1977, researchers identified two optical isomers and determined the crystal structure of CBD. These isomers vary in the stereochemistry of the linkage between the resorcinol molecule and the terpene core, with the aromatic and terpene ring nearly perpendicular to each other. Natural cannabis plants contain [–]-CBD, but [–]-CBD can only be synthesized through chemical methods. According to the most recent research, all CBD-type cannabinoids that are present in nature have a consistent structure. Of [–]-Trans-[1R, 6R], which is thought to correspond to a negative optical rotation. There are some derivatives of CBD, which are used in clinical trials with their structures and functions (Table 2).
Derivatives of CBD and their various function.
Metabolism and toxic side effects of CBD in AD
Metabolism
CBD is primarily metabolized by the liver enzymes cytochrome P450 (CYP), particularly CYP2C19 and CYP3A4. 189 These enzymes break down CBD into various metabolites, some of which have therapeutic effects. The main metabolite of CBD is 7-hydroxycannabidiol (7-OH-CBD), which is known to be equally active as CBD. 190 AD is a neurodegenerative disorder characterized by the buildup of abnormal protein deposits in the brain. These deposits, called plaques and tangles, are thought to disrupt brain cell function and communication, leading to cognitive decline and memory loss. The potential interaction is the blood-brain barrier (BBB). The BBB is a highly selective membrane that protects the brain from harmful substances in the bloodstream. 191 CBD may have the ability to cross the BBB, 192 although the exact mechanisms are not fully understood. If impaired by AD, the BBB could allow for greater passage of CBD and its metabolites into the brain, potentially affecting its therapeutic effects. 193 AD may impair liver function, which could affect how CBD is metabolized. 194 A compromised liver might not be able to process CBD as efficiently, leading to higher levels of CBD and its metabolites in the body. This could potentially increase the risk of side effects.
Toxic side effects
CBD is generally well-tolerated, but it can cause side effects, especially at high doses.
195
While research on CBD for AD is ongoing, some potential side effects of CBD use to be aware of include:
Liver problems: CBD can interact with the liver enzymes responsible for metabolizing drugs. This can lead to elevated liver enzymes in some people, especially those taking other medications. Diarrhea: CBD can cause loose stools, especially at higher doses. Fatigue: CBD may cause drowsiness or fatigue, especially when taken in high doses. Drug interactions: CBD can interact with certain medications, including blood thinners, antidepressants, and anti-seizure medications. It is important to talk to your doctor about all medications you're taking before starting CBD.
Limitations of CBD in Alzheimer's disease
Cannabis and cannabis-derived compounds have not been approved by the Food and Drug Administration (FDA) to treat AD, and very few trials to evaluate the use of THC (dronabinol and nabilone) or CBD have been done or are ongoing. For example, Nabilone, a synthesized cannabinoid currently approved for the treatment of chemotherapy-related nausea and vomiting, was found effective in reducing symptoms of AD patients. 196 However, to patient safety, is become very important to closely monitor side effects such as sedation and may be cognitive decline. Considering cannabinoids as a therapeutic option, identifying an effective dosage and treatment time is challenging. It is known that molecular changes related to AD began several years before symptoms seen. As a result, neuroprotective and immunomodulatory effects of cannabinoids should be administered before AD is aggressive and prolonged in time. However, studies on the long-term effects of cannabinoids are not yet available. While studies on the long-term cognitive effects of heavy cannabis use suggest, cannabis negatively influences cognitive functions, such as episodic memory, attentional control, and motor inhibition. 197 for this reason, further studies to explore the short- and long-term effects of cannabinoids are urgently needed. Unfortunately, studies investigating cannabinoid drug-drug interactions are still limited. Several investigations would be fundamental to underpinning this critical point, considering patients with dementia take multiple medications, and cannabinoids could be included as an additional therapeutic strategy to tackling the symptoms of dementia.
Conclusion
In conclusion, the finding of this study indicates that cannabidiol/derivatives inhibit AD progression through various mechanisms and key hypotheses regarding AD pathology. Nave CBD can reduce Aβ, IL-6 expression in peripheral leukocytes, and retarded cognitive decline. Compare with other CBD derivatives, CBD carbamate derivatives notably reduced Aβ1–42 levels, restored cognitive function to a normal state, and exhibited superior behavioral performance when compared to donepezil. CBD decreases Caspase 9, Caspase 3, and cleaved PARP1 protein levels and shows Antiapoptotic effects during cognitive decline. It also shows anti-cholinergic activity by inhibiting AChE and BuChE. As a result, the expression of ChAT can be normalized. In terms of the neuroinflammatory process, the expression of proinflammatory miRNAs (miR-146a, miR-155, and miR-34a) associated with TLR and NF-κB signaling is reduce. Therefore, continued research efforts should focus on elucidating the precise mechanisms of action, exploring potential synergies with other AD medications, and optimizing CBD formulations and derivatization to maximize therapeutic benefits in AD patients. These observations underscore the significance of further research and exploration into the therapeutic applications of CBD in the context of AD.
