Abstract
Dementia encompasses a broad spectrum of neuropsychiatric disease states marked by cognitive impairments that interfere with day-to-day functioning. Most dementias are complex phenotypes that result from a genome-environment interplay. Epigenetic regulation has emerged as a candidate mechanism for studying this interplay. In this narrative review, we discuss state-of-the-art evidence on environmental exposures relevant to dementia, including nutrition, physical exercise, psychosocial stress, and environmental toxins, and highlight epigenetic mechanisms that have been reported as a putative link between each exposure and dementia risk. We then discuss the clinical implications and future directions of this line of research. An improved understanding of the epigenetic mechanisms involved in dementia pathogenesis can promote the development of novel biomarkers for predicting outcomes but also targeted therapies to intervene early in the course of the disease.
Introduction
The term dementia refers to a broad spectrum of neuropsychiatric disease states marked by cognitive impairments that interfere with day-to-day functioning. The leading cause of dementia is Alzheimer's disease (AD), which leads to impaired visuospatial skills, aberrant conduct, and ultimately death. 1 AD is characterized pathologically by twisted beta-pleated sheet (amyloid) fibrils, including intraneuronal neurofibrillary tangles, extracellular amyloid-containing neuritic plaques, and cerebrovascular amyloid fibril deposits. 2 Alzheimer's disease neuropathologic changes are further defined by the accumulation of amyloid-β (Aβ) in the form of neuritic plaques and abnormally phosphorylated microtubule-associated protein tau in the form of neuronal neurofibrillary tangles. One of the most abundant proteins in the central nervous system, the amyloid-β protein precursor (AβPP), is crucial in the development of neuritic plaques, 3 The remaining cases of dementia include a variety of conditions with distinct neurodegenerative pathologies, such as vascular dementia, frontotemporal dementia, and dementia with Lewy bodies. In patients with dementia, a spectrum of neuropathologies may contribute to neurodegeneration, and some of the major neuropathology hallmarks may overlap. 4 Most dementias are complex disease entities thought to result from an intricate interplay between genetic and environmental factors. 5
AD and related dementias exhibit significant heterogeneity in clinical presentation, even among patients with similar genetic backgrounds. This variability is marked by genetic, demographic, neuropathological, cognitive, functional, neuropsychiatric, and biomarker factors. Genetic heterogeneity can arise from differences in mutations, such as those affecting AβPP processing, which influence Aβ deposition and disease course. Demographic factors like age, sex, education, and socioeconomic status also contribute to this heterogeneity, affecting disease progression and cognitive resilience. Neuropathological differences include the distribution and density of neurofibrillary tangles and Aβ plaques in various brain regions. Cognitive and functional heterogeneity is evident in the variability of cognitive profiles, daily living activities, and progression rates. Additionally, neuropsychiatric symptoms such as depression and apathy further diversify clinical presentations. These complexities necessitate tailored clinical and research approaches in AD to address the wide range of patient experiences and disease trajectories. 6
Among key mechanisms integrating environmental stimuli into the genome, epigenetic regulation has been studied as a possible hallmark of the aging process and a potential therapeutic target for age-related diseases. 7 López-Otín et al. 8 made this statement more specific by proposing the molecular hallmarks of aging. It has further been proposed that epigenetics may play a role in dementia pathogenesis. 9
The purpose of this review is to discuss state-of-the-art evidence on epigenetic mechanisms linking environmental exposure with dementia risk. To this end, the article presents an overview of environmental exposures associated with increased risk for developing dementia and highlights the epigenetic mechanisms reported as a link between environmental exposures and dementia. We then discuss the clinical implications and future directions of this line of research.
Brief primer on epigenetics
The term “epigenetics” was originally coined by Conrad Waddington in the 1940s to refer to the study of the way through which genes and their products bring the phenotype into being. 10 However, nowadays the term usually encompasses the heritable chemical modifications that regulate gene expression without changing the underlying DNA sequence. Epigenetic mechanisms include DNA methylation, histone modifications, and microRNAs (miRNAs). 11 DNA methylation is a widely researched epigenetic alteration, most commonly occurring in the context of cytosine-guanine dinucleotides (CpG) that tend to cluster in regions called CpG islands. 12 DNA methylation regulates gene expression by changing the access that transcription factors have to the genomic region it occurs. 13 DNA methyltransferases (DNMTs), including DNMT1, DNMT3a and DNMT3b, catalyze the transfer of a methyl group from S-adenosyl-L-methionine (SAM) to the C5 position of cytosine. 14 5-hydroxymethylcytosine (5-hmC) should be distinguished from 5-methylcytosine (5-mC), as it is the first oxidative product in the active demethylation of 5-mC, an important molecule in switching genes on and off. 15 Ten-eleven translocation methylcytosine dioxygenases, such as TET1 and TET2,16,17 hydroxylate the methyl group of 5-mC, converting it to 5-hmC. Regarding histone modifications, several post-transcriptional modifications occur in histone tails. 18 Histone acetylation, catalyzed by histone acetyltransferases (HATs), is associated with transcriptional activation, whereas histone deacetylation, regulated by histone deacetylases (HDACs), is involved in transcriptional repression. 19 MicroRNAs (miRNAs) are small noncoding RNAs that act after gene transcription to influence the protein levels of target mRNAs. 20
Several epigenetic modifications have been implicated in dementia, including DNA methylation 21 ; histone acetylation, 22 methylation, 23 and phosphorylation 24 ; and miRNAs. 25 Epigenome-wide association studies have further attempted to identify exact genomic locations where relative epigenetic differences are found.26,27 Below we highlight epigenetic studies linking specific environmental risk factors with dementia phenotypes. Among candidate epigenetic mechanisms, emphasis is placed on chromatin modifications, namely DNA methylation and histone modifications, which have the most evidence to date for their involvement in dementia phenotypes.
Epigenetic mechanisms linking environment and dementia
Even though certain modifiable risk factors have been associated with dementia, a significant amount of risk remains unexplained. 28 At the same time, the field of epigenetics has significantly evolved since the early 1940s, when the British embryologist Conrad Waddington introduced this concept. 10 Since then, progress has been made in identifying the role that epigenetic mechanisms play in various diseases. Below are presented some of the most important risk factors that have been linked with dementia and the epigenetic modifications underlying this relationship.
Nutrition
One of the most established environmental manipulations influencing age-related diseases is caloric restriction (CR). It has been reported that a CR dietary regimen attenuates Aβ neuropathology, prevents the generation of Aβ peptides, and reduces neuritic plaque deposition in a mouse model of AD. 29 These effects are associated with the promotion of anti-amyloidogenic α-secretase activity, which plays a role in reducing the pathological accumulation of Aβ peptides. CR has also been found to ameliorate age-related behavioral deficits in mouse models of AD, while also demonstrating neuroprotective effects by enhancing neurogenesis, improving synaptic plasticity, and reducing oxidative stress. 30 Corroborating findings have been observed in other animal models, including squirrel monkeys 31 and C. elegans. 32
CR has been shown to exert neuroprotective effects against AD through mechanisms involving histone deacetylases (HDACs), particularly sirtuins like SIRT1 and histone acetyltransferases. SIRT1 induction via CR or pharmacological activators has been demonstrated to protect against neuronal loss and impairment in models of AD and Parkinson's disease by reducing oxidative stress and improving mitochondrial function. 33 CR's beneficial effects in AD models include the reduction of Aβ production and stabilization of tau protein, achieved through the modulation of chromatin structure and gene expression by HDACs and SIRT1. 34 Additionally, the ketone body β-hydroxybutyrate, a known HDAC inhibitor, extends lifespan and enhances stress response pathways, 35 which are protective against Aβ toxicity and neurodegeneration in AD models.
Histone acetyltransferases (HATs), such as CREB-binding protein (CBP), also mediate the neuroprotective effects of CR. CBP has been shown to mediate the beneficial effects of CR by enhancing histone acetylation, delaying age-related pathologies, and extending lifespan. 36 In transgenic models of AD, the reduction of CBP activity due to presenilin mutations leads to decreased acetylation at specific histone sites, contributing to cognitive deficits. This suggests that maintaining normal CBP levels and histone acetylation is critical for neuronal health. 37 Furthermore, drugs that facilitate histone acetylation, such as 4-phenylbutyrate, have been shown to improve cognitive function and reduce tau pathology in AD mouse models, 38 highlighting the potential therapeutic role of HAT activation.
Numerous studies have looked at the effect of nutrients and diets in the prevention of cognitive decline, which eventually results in dementia. Individual nutrients or food groups, however, might not accurately represent the inherently complicated human eating habits or a composite dietary pattern. Dietary patterns incorporate potential synergistic or interactive effects between multiple nutrients and foods, thus more accurately reflecting real-life conditions. 39 The dietary pattern most strongly associated with protection against dementia and cognitive decline is the Mediterranean diet (MeDi). Measured across various memory and executive function domains, MeDi, a diet based on fruits, vegetables, and fish, has been shown to delay cognitive decline and lower risk for AD.40,41 In a large prospective cohort study, MeDi was found to be inversely associated with dementia incidence after accounting for major cardiovascular risk factors. These results differed by dementia sub-type, sex, and education. 42 In a systematic review, however, the association between MeDi adherence and dementia risk has not been consistent, with lower risk of AD and all-cause dementia found, respectively, in four out of seven and zero out of five studies (with the other studies reporting null findings). 43 Notably, when the MeDi is combined with the Dietary Approach to Systolic Hypertension (DASH) diet, its positive effects on cognition may be increased. This hybrid diet—based on high intakes of fruits, vegetables, low-fat dairy products, whole grains, poultry, fish, and nuts and low intakes of fats, red meat, sweets, and sugar-containing beverages—has been shown to lower blood pressure that contributes to vascular pathologies, enhance cognitive functioning, and reduce the likelihood of developing AD.44,45 The MIND diet, also based on plant-based foods, is a combination of the Mediterranean diet and the DASH diet and has been associated with better cognitive function and reduced risk of cognitive decline.44–46
While cellular and molecular mechanisms underlying the interaction between nutrition and its effects on brain function have been widely investigated,47,48 we still know little about the epigenetic mechanisms underlying these phenomena. Early-life nutrition can induce long-lasting alterations in DNA methylation that influence a person's health and possible development of aging-related diseases throughout their lifetime. 49 Nutrients can exert their effects either by directly suppressing epigenetic enzymes like DNMTs, HDACs, or HATs, or by changing the availability of the substrate required for certain enzymatic activities. The expression of critical genes is in turn altered, affecting general health and longevity.
DNA methylation is mediated by the DNMTs and the methyl donor SAM. Vitamins B6, B12, choline, methionine, and folate regulate this process. One-carbon pathways donate and regenerate one-carbon units, including the methyl group that is essential for DNA methylation. When this process is altered, aberrant DNA methylation occurs, which is linked to several autoimmune diseases and neurological disorders. For example, folate, which is a water-soluble B vitamin, is a source of one carbon for the synthesis of SAM, which is required for DNA methylation, establishing a connection between folate metabolism and phenotypic alterations through DNA methylation. 50 Choline is another methyl donor nutrient that can influence the DNA methylation status and hence affect gene expression. 51
Histone modifications and miRNAs are also affected by diet. For example, dietary fibers derived from vegetable consumption and the polyphenol kaempferol, mainly found in vegetables but also in some fruits, can induce histone post-translational modifications by inhibiting HDAC activity. 52
Early-life nutrient exposures that can occur through pregnancy and breastfeeding have also been linked with cognition and dementia. Obesity is considered an important risk factor for cognitive decline. Parental obesity is associated with altered DNA methylation at multiple genes. Maternal obesity can influence the expression of multiple genes in the fetal brain, possibly altering the developmental program of key fetal brain regions involved in neurological disorders in later life. 53 Paternal obesity is associated with hypomethylation of the gene encoding insulin-like growth factor 2 (IGF2) in newborns. 54 IGF2 is a growth factor with effects on fetal skeletal muscle growth and development, while it also has a known insulin-like action that can contribute to obesity risk in adults. 55 Imbalanced maternal intake of specific nutrients, such as folate and vitamin B12, has been associated with global DNA hypomethylation in the offspring that is normalized by omega-3 fatty acid supplementation delivered 3 months postnatally. 56 This also highlights the importance of the interactions between different nutrients in modulating the expression of multiple genes linked with long-term neuroprotection and cognition.
Physical exercise
Both in human populations and animal models, exercise has been found to positively influence brain function, improving memory and learning and promoting healthy brain aging. 57 In a large meta-analysis that included several cohorts and 33,816 individuals, higher levels of physical activity were associated with a significant reduction in dementia risk. 58 Although the level of possible activity is more limited for older adults with osteoarthritis or other motor disabilities, even minimal amounts of exercise have been shown to help prevent dementia in this population, supporting the role of exercise in at-risk groups. 59 However, randomized controlled trials revealed conflicting results regarding the impact of exercise on cognition and the brain, raising some concerns about the effectiveness of exercise in preventing dementia. 60 It should also be noted that exercising in the setting of high air pollution in urban environments and cities might not be beneficial because air pollution intake increases considerably due to an increased ventilation rate and particle deposition fraction that can negatively influence brain function. 61
Physical exercise can induce DNA methylation changes by regulating DNMTs and demethylases. It has been shown that DNMT1 and DNMT3b levels in the hippocampus of young adult rats can be decreased by a single exercise session. This, however, does not apply to aged rats, even when a longer exercise protocol is followed. 62 In contrast to DNMTs, enzymes involved in DNA demethylation are regulated by physical exercise in aged animals. A 2-week voluntary wheel training in sedentary rats increased the level of hippocampal Tet1, 63 while a 4-week physical exercise increased the hippocampal 5-hmC content, counteracting the decreased Tet1 and Tet2 expression induced by aging and, in turn, promoting cognitive functions of aged mice. 17 Additionally, physical exercise has been reported to increase the level of activated MeCP2 (Methyl-CpG binding protein 2), a protein binding to methylated DNA (i.e., a DNA methylation reader). 64 Wheel-running exercise increased activated MeCP2 and the expression levels of the brain-derived neurotrophic factor (BDNF) in the rat hippocampus. 65
Increased BDNF expression induced by physical exercise was also a common mediator of enhanced cognitive ability in other rodent studies. 66 More specifically, studies have reported reduced HDAC2 and HDAC3 expression 67 as well as increased global H3 acetylation and reduced DNMT and HDAC levels, 68 resulting in increased BDNF expression. Finally, voluntary wheel running upregulated 20 miRNAs and downregulated 12 miRNAs in the hippocampus of adult mice, enhancing cognitive function. 69
Psychosocial stress
Studies on stress and health show that cumulative or severe adverse childhood experiences are significantly associated with increased risk for dementia.70–72 The association between stress exposure and cognitive impairment has been supported by preclinical studies. Maternal separation in mice has been reported to dysregulate microglia function, facilitate Aβ plaque formation, and worsen cognitive function. 73 In line with these preclinical observations, childhood maltreatment and loss of a parent or other relatives during early life have been linked to higher dementia risk.74,75 Exposure to chronic stress and lifetime trauma during adulthood has also been associated with a decline in cognitive functioning assessed in both midlife and later life. 76
Life adversity can have epigenetic effects relevant to human health that can last throughout life and could even be passed on to future generations. Psychosocial stress has been shown to induce changes in DNA methylation and gene expression that are relevant to aging-related and other disease states. 77 For example, a number of studies have found associations between psychosocial stressors and promoter methylation of the gene encoding the glucocorticoid receptor.78–80 Stress has also been found to synergize with aging to alter DNA methylation across the epigenome and at specific genomic sites, such as the gene encoding the stress-responsive molecule FK506-binding protein 5 (FKBP5/FKBP51).81,82 FKBP5 hypomethylation was associated with enhanced FKBP5 expression in immune cells, heightened peripheral inflammation, and cardiovascular risk. 81 Notably, the FKBP5 protein has also been shown to influence other aging-related processes and disease phenotypes relevant to dementia, including tau degradation and neurodegeneration.83,84 Life adversity has been further associated with epigenetic regulation of BDNF. 85 Maternal separation in rats resulted in biphasic changes in histone 3-lysine 9 dimethylation (H3K9me2) at the Bdnf IV promoter. H3K9me2 initially decreased into young adulthood before reversing into a significantly increased histone demethylation in midlife. These changes in H3K9me2 inversely correlated with BDNF exon IV transcription and hippocampal neurogenesis. 86 In conclusion, evidence supports epigenetic regulation as a highly plausible link between psychosocial stress and cognitive decline, but the underlying mechanisms have not been adequately examined.
Strengths and limitations should be kept in mind when translating the findings on psychosocial stress from rodents and other model organisms. Human psychosocial environments are far more intricate and multifaceted, making phenotypes such as stress and dementia also more complex to understand. By enabling controlled experimental manipulation, studies in model organisms can yield mechanistic insights about certain biological processes conserved in humans, but their findings and potential implications should be interpreted with caution.
Environmental toxins
Environmental toxins can come in various forms, such as air pollutants, persistent organic pollutants, toxic metals like arsenic, mercury, lead, cadmium, nickel and manganese, polycyclic aromatic hydrocarbons, tobacco smoke, and aflatoxin B1. 87 Killin et al. provided a systematic review of environmental toxins linked with dementia risk. 28 Air quality measured by levels of nitrogen dioxide and carbon monoxide was shown to have a dose-response relationship with dementia risk. 88 Second-hand tobacco exposure was also found to be associated with severe dementia syndromes. 89 Accordingly, long-term improvements in ambient air quality has been associated with lower dementia risk in older women. 90 Toxic metals have also been associated with dementia risk. 28 Furthermore, exposure to fine particulate matter and ozone levels above standards in megacities like Metropolitan Mexico City is linked to increased AD risk, likely as a result of increased neuroinflammation and neurodegeneration. 91 Notably, air pollution, particularly exposure to ultrafine particulate matter and industrial nanoparticles, poses significant early risks for neurodegeneration.
Little is known about the epigenetic and other molecular mechanisms through which environmental toxins may contribute to dementia risk. Studies in both cultured cells and human cohorts suggest that metals such as mercury and manganese are associated with increased AβPP expression, circulating Aβ peptides, and AD risk.92,93 Convergent findings across rodents and humans also suggest that prenatal particulate matter exposure is linked to dysregulation of biological pathways relevant to neuronal survival and neurodegeneration, infant neurodevelopmental delays, and changes in miRNA levels found in placental extracellular vesicles.94,95 While these early findings suggest that environmental toxins can epigenetically regulate the expression of molecules involved in dementia pathology, 96 further research is needed to understand the underlying mechanisms.
Clinical implications and interventions
Acetylcholinesterase inhibitors such as donepezil, rivastigmine, galantamine, and tacrine as well as the N-methyl-D-aspartate receptor antagonist memantine are medications currently approved by the FDA for the treatment of AD, but they can only improve symptoms rather than modifying disease progression. An improved understanding of the epigenetic underpinnings of dementias may open opportunities for developing new treatment strategies for these devastating diseases. Experiments in model organisms have provided early support for the potential role of targeting DNA methylation and histone modifications in dementia. 97 Increases in the methyl donor S-adenosyl-L-methionine have been demonstrated to reduce the production of AβPP and Aβ through hypermethylation of genes involved in relevant biological pathways.98,99 Small molecules acting as HDAC inhibitors can pass the blood-brain barrier, delaying the onset and progression of symptoms and enhancing cognitive performance in animal models of AD. 100 Several drugs known to act as HDAC inhibitors, including trichostatin A, sodium 4-phenylbutyrate, and valproic acid, have shown benefits across both cell and rodent models of AD, collectively contributing to decreased Aβ production and tau pathology, enhanced neuroprotection and synaptic plasticity, and improved cognitive functioning.38,101–104 Targeting these epigenetic modifications could pave the way for new, personalized therapies for neurodegenerative disorders. However, there is much research needed to be done before such therapeutic modalities become clinically applicable.
Conclusions/ future directions
As schematically summarized in Figure 1, epigenetic regulation is a key candidate for understanding how environmental factors contribute to dementia risk, but much evidence is still needed before reaching conclusive evidence on specific mechanisms. An improved understanding of the epigenetic mechanisms involved in dementia pathogenesis has the potential to promote the development of novel biomarkers for predicting outcomes but also targeted therapies to intervene early in the course of the disease. We thus hope that this narrative review will spur future studies on this important line of research.
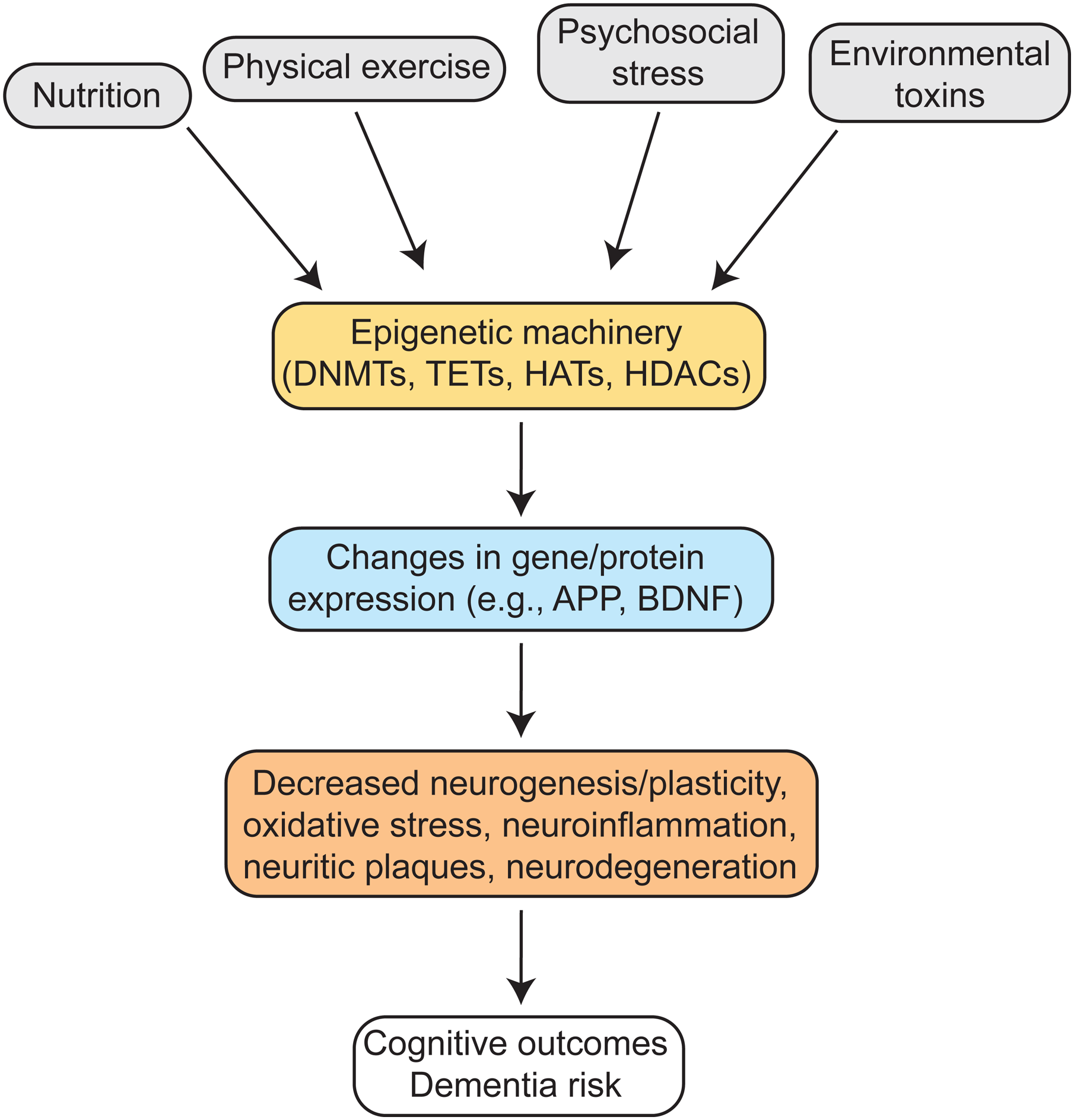
Simplified scheme summarizing evidence to date and model of epigenetic regulation as a link between environmental factors and dementia risk. APP: amyloid precursor protein; BDNF: brain-derived neurotrophic factor; DNMTs: DNA methyltransferases; HATs: histone acetyltranferases; HDACs: histone deacetylases; TETs: Ten-eleven translocation methylcytosine dioxygenases.
Footnotes
Acknowledgments
The authors have no acknowledgments to report.
Author contributions
Adamantia Koulouri (Writing – original draft; Writing – review & editing); Anthony S Zannas (Conceptualization; Supervision; Writing – original draft; Writing – review & editing).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
