Abstract
Background:
Epigenetics studies the impact of environmental and behavioral factors on stable phenotypic changes; however, the state of the science examining epigenomic mechanisms of regulation related to secondary health conditions (SHCs) and neuroepigenetics in chronic spinal cord injury (SCI) remain markedly underdeveloped.
Objective:
This scoping review seeks to understand the state of the science in epigenetics and secondary complications following SCI.
Methods:
A literature search was conducted, yielding 277 articles. The inclusion criteria were articles (1) investigating SCI and (2) examining epigenetic regulation as part of the study methodology. A total of 23 articles were selected for final inclusion.
Results:
Of the 23 articles 52% focused on histone modification, while 26% focused on DNA methylation. One study had a human sample, while the majority sampled rats and mice. Primarily, studies examined regeneration, with only one study looking at clinically relevant SHC, such as neuropathic pain.
Discussion:
The findings of this scoping review offer exciting insights into epigenetic and neuroepigenetic application in SCI research. Several key genes, proteins, and pathways emerged across studies, suggesting the critical role of epigenetic regulation in biological processes. This review reinforced the dearth of studies that leverage epigenetic methods to identify prognostic biomarkers in SHCs. Preclinical models of SCI were genotypically and phenotypically similar, which is not reflective of the heterogeneity found in the clinical population of persons with SCI. There is a need to develop better preclinical models and more studies that examine the role of genomics and epigenomics in understanding the diverse health outcomes associated with traumatic SCI.
Introduction
Spinal cord injury (SCI) is a devastating condition that often yields permanent disability and challenges in medical care and symptom management. This once “ailment to not be treated” has over the years seen increased life expectancy resulting from advances in adaptive technology, pharmacologic intervention, and more robust understanding of SCI pathophysiology. 1 The SCI population is highly heterogenous in both American Injury Scale (AIS) level and the extent of permanent neurologic deficit. No two patients will have an identical pathophysiologic progression or clinical course post injury. 2 However, functional recovery continues to be hampered by a paucity of clinical treatment options. Regenerative approaches are ongoing with small progress; treatment strategies remain exploratory, and few have translated to patients (T2), practice(T3), or community (T4) on the translational research spectrum. 3 Post-injury management of secondary health conditions (SHCs) and complications is the key to longevity with good health and acceptable quality of life.
There are 3 generally accepted phases of recovery following traumatic SCI: (1) acute (hours to days), (2) subacute (weeks to months), and (3) chronic (>1 year) states. However, it is also accepted that more than 6 months post-injury is also considered chronic and may be more commonly reflected in preclinical studies. 4 The literature lacks consensus on how chronicity is defined likely due to the interindividual differences in functional recovery between human and preclinical pathophysiology. Thus, for the current review, we define chronicity as starting between 6 months to 1 year following injury.
It has been found that most of the research on SCI has focused on acute and chronic phases. 5 This is likely to be due to the interest in time-dependent inflammatory and immune responses post trauma. There is also increasing recognition that the long-term impact of traumatic SCI may be characterized as a sub-clinical inflammatory disease. A review by Noller and colleagues 6 discussed the contribution of persistent chronic inflammation in SCI resulting in common SHCs. Chronic inflammation is significant for individuals living with SCI because of the increased risk of cardiometabolic disorders that are the leading cause of morbidity and mortality in this population. Several researchers have shown SCI-specific inflammatory and structural markers found in the blood/cerebrospinal fluid (CSF) linked to injury and neurologic loss may be predictive of functional recovery in SCI.7 -10
Epigenetics is broadly defined as processes that may regulate gene expression and do not involve changes to the underlying DNA sequence: a change in phenotype without a change in genotype.11,12 Disruption of gene expression patterns governed by hyper, or hypo methylation may result in autoimmune diseases, cancers, and various other illnesses, in contrast to genetic changes, which are difficult to reverse. However, environmental (social and structural) influences on DNA methylation may be reversed if reduced or eliminated (discrimination, stress, smoking, etc.), and pharmaceutical interventions have also been used to reverse effects.13 -16 The field of epigenetic research seeks to discover how lived environment, social condition, psychosocial factors, and nutrition affect an individual’s expression of genetic information. 16 The 3 central mechanisms regulating epigenetics are (a) alteration of histone modification, (b) DNA methylation (DNAm), and (c) RNA regulation. 17
Neuroepigenetics is distinguished from classic epigenetics due to its specific application to neurons, which means modifications are not propagated to progeny cells as seen in non-neuronal cells. 18 However, the central regulating mechanisms remain the same. As more is being understood about environmental influences on disease development, multimorbidity, and health outcomes, epigenetic methodology opens the door to leverage biological and clinical data to inform SHC phenotypes in persons with SCI. Hence, to better understand the evolving science of epigenetics and its application in SCI research, we conducted a scoping review.
Scoping reviews differ from systematic reviews in that they do not aim to produce a critically appraised and synthesized result/answer to a particular question, but rather focus on (1) identifying certain characteristics/concepts in papers or studies and (2) mapping, reporting, or discussing these characteristics/concepts. 19 A scoping approach was chosen to guide this review to map and characterize studies investigating epigenetic regulation and their links to SHCs after acute and chronic SCI. We hypothesize that given the paucity of literature on this topic, the influence of epigenetics remains an understudied area for this population. The specific purpose of this scoping review was to examine the state of the science focusing on epigenetics that attempts to explain SHC and other conditions associated with chronic SCI.
Methods
A librarian-assisted literature search was conducted across 4 databases (PubMed; Embase; Web of Science; Scopus) using the search terms “spinal cord injury,” “spinal cord injuries,” “epigenetics,” “epigenome,” “histone modification,” and “chromatin modification.” An Endnote library was shared with 277 articles from the years 2010 to 2020.
Covidence review software (v2472 44496257; Melbourne, Australia) was used to facilitate abstract and full text review. For inclusion, articles (1) investigated SCI and (2) examined a mechanism of epigenetic regulation as part of the study methodology. Articles were further excluded if they were reviews, commentaries, book chapters, or unavailable in English. Articles were uploaded to the Covidence software, and 30 duplicates were removed, resulting in 247 articles remaining for title and abstract review. Two reviewers completed the abstract review (author

Prisma flow chart.
The software did not offer Cohen’s Kappa; however, a pilot practice of 7 articles was done among full text reviewers to ensure that inter-rater reliability was consistent. No conflicts were found, and reviewer responses to screening tools were consistent across sections and articles.
Results
Of the 23 articles included, 65% of studies examined mechanisms related to histone modification, with 26% of studies using DNA methylation (DNAm) and 9% using RNA regulation as epigenetic mechanisms. The studies occurred with mice (43%) and rats (44%) as the most commonly studied species. Only one human study was identified in this review. Most of the studies focused primarily on central nervous system (CNS) regeneration and plasticity, with only one article considered clinically relevant for SHC, such as neuropathic pain. We have divided and will discuss the included articles by epigenetic mechanism.
DNA methylation (DNAm)
DNAm is the most well characterized and most common epigenetic mechanism studied, with a role in the regulation of transcription, chromatin structure, genomic imprinting, and chromosome stability. 20 Methyltransferase enzymes (DNMT1, DNMT3A, and DNMT3B), are responsible for the transfer of the methyl group to DNA and are responsible for the mediation and maintenance of DNAm patterns (Figure 2). 20 Of the 23 articles, only 6 studies focused on demonstrating the contribution of DNAm in the process of nerve regeneration and remodeling after SCI. Two of those articles looked at the role of folic acid/folate pathway as a key methyl donor in the CNS, for CNS regeneration and repair, and as a target to reduce neuropathic pain.21,22

DNA methylation.
In an earlier study by Iskandar et al
21
an SCI regeneration model (dorsal column transection and conditioning sciatic nerve injury) was used to analyze changes in folate receptor (
Miranpuri et al
22
used a contusion SCI model in adult male Sprague-Dawley rates to analyze the effect of folic acid on matrix metalloproteinase 2 (
Shi et al 24 used Wistar rats to examine the role of abnormal DNAm in axonal regeneration and cell proliferation through histological observation, motor function assessment, and whole-genome bisulfate sequencing (WGBS) to detect gene methylation with Gene Ontology (GO) and KEGG pathway analyses. Histologically, they found several cavities, tissue necrosis, and glial scaring, with poor motor function in behavioral analysis. Bioinformatic analyses revealed 96 differentially methylated genes (DMG’s). Biological processes of the hypermethylated genes (50) were related to cell shape regulation and neurogenesis, while the biological processes enriched by the hypomethylated genes were related to brain development, protein phosphorylation, and ethanol response.
Another study by Shi et al 25 reported results of a bioinformatic analysis of data to identify common hypermethylated and demethylated genes from DNAm sequencing of samples taken before and after sciatic nerve injury in rats. 25 Although the focus of this paper examined common DMG’s among SCI and non-CNS injury (sciatic nerve injury), one area of interest is identifying protein-protein interaction networks specific to SCI that may have shared pathways significant to SCI-related SHC. The findings of this study indicated 12 DMGs that are involved in biological regulation processes, such as cellular signaling and molecular functions of protein binding. Of particular interest was the Hippo signaling pathway given its activation by many demethylated genes important to neuroregeneration. 25
Sun et al,
26
examined the perturbations of hydropymethylcytosine (5hmC), an oxidation product of DNAm, on gene expression related to cell death after SCI. In other neurological conditions such as TBI, stroke, and sporadic amyotrophic lateral sclerosis (ALS), 5hmC has been found to play an important role in ischemia and perfusion to the brain and spinal cord. A contusion SCI model was used in rats at the T9-T11, followed by quantitative real-time polymerase chain reaction analysis (qRT-PCR) and hydroxymethylated DNA immunoprecipitation (hMeDIP) assays. Results revealed that global change in DNA 5hmC levels significantly increased at 6, 12, and 24 hours after SCI. Additionally, gene expression changes were found to increase in the presence of ten-eleven translocation family (
Lastly, DNAm was used to examine the developing spinal cord and its response to environmental input during locomotor development related to hindlimb activity.
27
Doherty et al
27
focused on brain derived neurotrophic factor (
Histone modification
The bulk of the studies examined histone modification, the covalent, post-translational modification of histones. This epigenetic regulatory process impacts gene expression by altering chromatin structure (phosphorylation, ubiquitination, acetylation, and methylation) or recruiting histone modifiers, such as histone acetyltransferase (HAT), histone methyl transferase (HMT), histone deacetylase (HDAC), histone demethylase (HDMT), kinases, and E3-ubiquitin (Figure 3).28,29 Six studies focused on histone acetylation, 6 examined histone deacetylation, and 3 examined histone demethylations. Histone acetylation is regulated by HAT and HDAC enzymes that add or remove acetyl groups to the N-terminal histone tails and together maintain acetylation homeostasis and play a role in plasticity transcription and regeneration-associated genes.
30
Evidence demonstrates that downregulation of HDACs has positive effects on learning, memory, and synaptic plasticity. Seira and colleagues
31
assessed the potential effect of trichostatin A (

Histone modification.
De Menezes et al
32
measured glial fibrillary acidic protein (
Hutson et al 33 examined the influence of environmental enrichment versus standard housing (control) on Cbp-dependent histone acetylation-mediated axon regeneration. Enriched environment (EE) offers opportunities for increased motor, social, and sensory activity and has been found to enhance neural connections and improve functional recovery on behavioral testing.34,35 They found that by exposing mice to EE, cAMP-response element binding protein had a mediation effect on histone acetylation; thus, changes in chromatin that increase regenerative capacity of neurons result in increased sensory and motor functions. 33
The only human study in this scoping review was conducted by Goldhardt et al
36
They examined the impact of a single body weight support gait training session on oxidative stress parameters,
Rudman et al
37
examined the role of bromodomain and extraterminal (
Abdanipour et al
39
tested valproic acid (VPA), a known HDAC inhibitor, in rats after SCI. HDAC inhibition results in transcriptional reactivation by inducing histone acetylation through HDAC enzyme suppression. Their findings support earlier studies which also report neuroprotective effects of VPA and increased
Qi and Wang
42
examined class IIa HDACs, which differ from class I and class IIb HDACs by their sequence and structural organization, in that they rarely interact with histone tails or the removal of acetylated lysine groups. To better understand the epigenetic mechanism of macrophages (used to transition from phenotype M1 associated with pro-inflammation to phenotype M2 found to suppress excess inflammation, prevent axonal damage, and improve locomotor function), a male mice contusion model was tested with
Hervera et al
44
also studied HDAC3 to determine its role as a novel mechanism to discriminate between axonal regeneration and regenerative failure in dorsal root ganglia (DRG) sensory neurons. Conducting both in vivo and bioinformatic analysis, they found that reduced HDAC3 activity allowed for increased histone acetylation and regenerative gene expression in DRG. However, HDAC3 phosphorylation is calcium dependent and spinal injury does not induce the calcium-dependent protein phosphate 4 (
Lee et al45,46 and Ni et al
47
investigated the epigenetic regulation of angiogenesis and vascular regeneration in SCI. The study of post-SCI vascular regeneration by Ni et al
47
examined mRNA levels of
In both Lee et al studies,45,46 histone H3K27 demethylation
Lee et al
46
contribute findings to the critical role of histone H3K27 demethylase
RNA regulation
miRNA is a class of non-coding small RNA that functions to downregulate gene expression through mRNA cleavage, translational inactivation, and deadenylation (Figure 4). 16 Using small interfering RNAs (siRNAs) to inhibit the interphase phase of the cell cycle, Wang et al 48 studied epigenetic silencing of cyclinD1 in bone-marrow-derived mesenchymal stem cells (BMSCs) for protective effects in rats after SCI. Cyclins are proteins that function to progress the cell cycle by binding a group of enzymes called cyclin-dependent kinases (CDKs) which phosphorylate target proteins. 49 CyclinD1 specifically is responsible for progressing the cell cycle from G1 to S phase. 50 Inhibition of CyclinD1 expression is associated with downregulation of pro-inflammatory cytokines, 50 which may be a potential therapeutic target for pro-inflammatory conditions such as SCI.
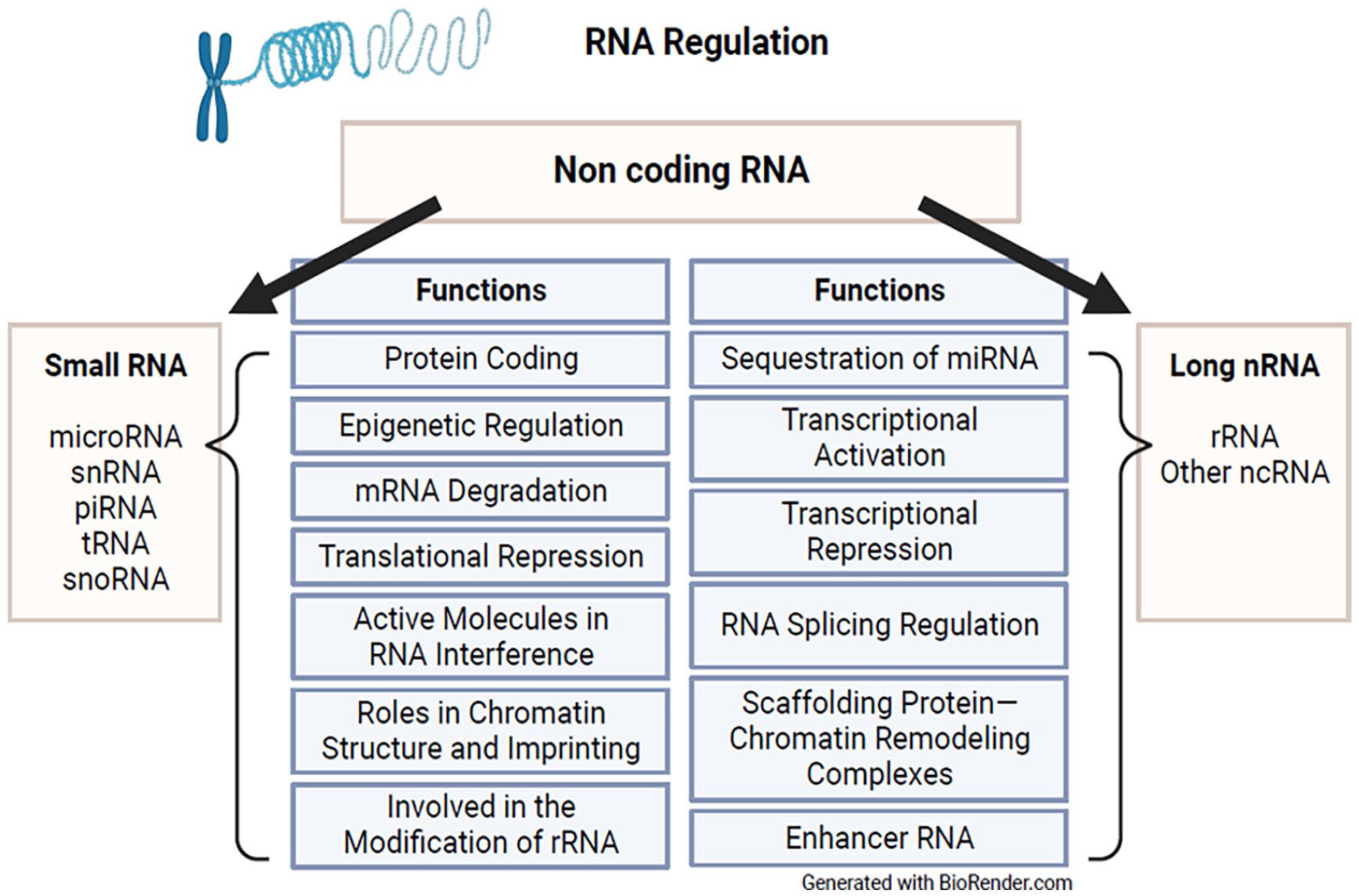
RNA regulation.
In female Sprague Dawley rats, cyclinD1 was transplanted with BMSCs into the lesion area of the SCI using siRNA plasmids. CyclinD1 was found nominally expressed by Western blotting and qRT-PCR, confirming that siRNA transfection was successful. Observations of histological changes confirmed decreased tissue edema, inflammatory cell infiltration at the site of the SCI lesion, and smaller syrinx in groups with si-cyclinD1 + BMSCs and BMSC groups compared with the control and sham groups. Additionally,
In another study investigating the subacute phase of SCI, Wang et al
48
sought to use bioinformatic analysis to construct and integrate ceRNA network of critical genes, miRNAs, long non-coding RNAs (lncRNA), transcription factors, and signaling pathways. Gene expression data sets were used from the gene expression omnibus (GEO) database to define the “SCI group” as mice with SCI acquired by contusion. DEG analysis was performed using GO and KEGG enrichment. They found 4 genes (
Discussion
The results of this scoping review suggest that there is an increasing enthusiasm for epigenetics and neuroepigenetics in SCI research, especially given the increased complexity when considering the different cell types that exist in the brain and spine. Further, the findings from this review offer exciting insights into neural circuits and synaptic plasticity involved in learning new information. The emerging sub-discipline of neuroepigenetics describes the “discoveries of a wide variety of roles for epigenetic molecular mechanisms in the central nervous system (CNS) regarding acquired behaviors, CNS disorders, neural plasticity, neurotoxicity, and drug addiction.” 51 Current evidence suggests that in the presence of lost neuronal acetylation homeostasis, short- and long-term chromatin architecture changes, resulting in the development of neurodegenerative disorders, such as Parkinson’s disease (PD), multiple sclerosis (MS), stroke (CVA), and Alzheimer’s disease (AD), which has significance post-SCI neurodegenerative changes. 52 Notably, the histone acetylation-deacetylation balance modulates different cell types, such as inflammatory and immunological cells, which influence the severity of blood-brain barrier dysfunction, axonal demyelination, oxidative stress, and injury impact.53,54 Applying neuroepigenetics to SCI research could lead to new approaches in rehabilitation where re-learning motor activities could improve functional outcomes.
This scoping review also reinforces the dearth of studies that examine the potential of clinically applied precision health within the rehabilitation setting, particularly for individuals living with SCI and managing SCI-associated SHCs. Many of the studies included in this review examined changes in the acute or subacute post-SCI phase. None of the studies looked at these gene expression changes in chronic SCI, thus leaving a larger gap in the significance of chronicity on these mechanisms. Understanding gene expression and its regulation translates to more accurate diagnosis and treatment for clinically significant changes in phenotype. Wagner 55 presented the Rehabilomics research framework, offering a translational path for rehabilitation research that combines omics technology with personal biology, physiological responses to environmental exposures, and biopsychosocial outcomes based on the World Health Organization International Classification of Functioning, Disability, and Health domains. However, this adapted model primarily focuses its application on traumatic brain injury (TBI) research. While TBI and SCI share similarities in primary and secondary injury cascades following mechanical injury, the study of SCI and the chronic SHCs that follow remain understudied. Individuals living with SCI are living longer and have increased and/or unpredictable levels of risk for developing a wide range of SCI-driven SHCs. The identification and use of prognostic biomarkers are major steps in developing decision trees, predictive tools, personalized interventions, and treatments.
Key genes, proteins, pathways
The subacute phase after SCI is categorized by neuronal apoptosis, axonal demyelination, Wallerian degeneration, axonal remodeling, and glial scar formation, while the chronic phase is characterized by cystic cavity, axonal dieback, and maturation of glial scar.
3
These clinical features are reinforced by the findings of studies included in this scoping review, particularly as key genes, proteins, and pathways that were present in multiple studies, including brain derived neurotrophic factor (
Preliminary work by Bogie and colleagues,59,60 has shown the genetic influence of candidate DNA variants and single nucleotide polymorphism (SNPs) on intramuscular adipose tissue (IMAT) accumulation and recurrent pressure injury (PrI) risk. They have previously shown that varying levels of circulatory biomarkers may be indicative of recurrent PrI risk.59,60 Furthermore, genetic biomarkers, specifically those related to fatty metabolism, are indicative of persons with SCI at highest risk for recurrent PrI. 61 Histone H3K27me3 demethylase Jmjd3 has been found to be involved in wound repair and the inflammation process,62,63 highlighting the value of incorporating both genetic and epigenetic regulation of gene expression, as it may hold significance for the study of pressure injury and other SHCs after SCI. Thus, defining the epigenetic mechanisms linked to remodeling neural tissue would help researchers design therapeutic strategies to promote wound healing and tissue functionality after SCI among other potential targets.64,65
Chronic multimorbidity in SCI research
There is a key discussion absent in SCI research around epigenetics, chronic multimorbidity, and SHCs. Symptom science is “focused on quantifying subjective symptom experiences and measuring the biologic, physiologic, and omics underpinnings of symptoms and sequela common to health conditions and their treatments” 66 and recognizes the “complex relationships within and between symptoms both in a single chronic condition and among chronic conditions (ie, shared symptom cluster).” 67 Symptoms serve as critical clues to changes in health status (physical and biopsychological) and are the primary reason urgent or emergent care is sought, which may (or may not) lead to hospitalization and/or diagnosis of a newly developed SHC. This is of particular importance for SCI research, where individuals are likely to suffer higher rates of multimorbid SHCs and the management of accompanying symptoms.
Diseases can often be due to multiple factors including routine environmental exposures. 68 While we present the effects of epigenetics leading to the development of multimorbid SHC, that is, the gene-environment, it is very probable that multimorbid SHC directly lead to remodeling in the epigenome. Studies looking at the influence of changes due SHC on the epigenome need to address the dynamic variability inherent in epigenetic studies. Prospective studies to measure and compare pre-multimorbidity states are also challenging. Multimorbidity as previously described carries a significant burden for individuals living with SCI and is influenced by age and injury characteristics (ie, paraplegia; tetraplegia; etc.). These influences also carry significant complexity in the presence of the injury. Although the relationship between multimorbid SHC and epigenetic change is even less understood given the dearth of research in this field, we wanted to acknowledge it as an important consideration as more work is being done to develop this field.
Theoretical models have been developed to improve our understanding of the mechanisms that contribute to symptom behavior (ie, initiation, severity, temporality). However, symptom behavior and associated mechanisms has primarily been evaluated in other conditions, such as cancer and cardiovascular disease; only one study addresses symptom behavior in SCI research.69 -72 Development of a Rehabilomics theoretical model specific to SCI symptomology would provide guidance that would support the translation of bench findings to clinical management of chronic multimorbidity resulting from SCI.
Conclusion/Implications for Research and Practice
This scoping review has revealed some key epigenetic regulators of gene expression and adjacent pathways that appear to overlap many critical biological, molecular, and cellular processes following SCI. However, it also elucidates the need for more translational/applied clinical research as a next step from the bench to begin impacting the patient. Given the paucity of research that studies the concepts of epigenetics, SHCs, and/or their associated symptoms in SCI research (individual or integrated), more work in this area is critical to continue advancing our understanding of SCI-specific biobehavioral outcomes.
Footnotes
Acknowledgements
Authors acknowledge to the LeAnne S. Garcia for support with graphic development.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
LYG conceived the study overseeing overall direction, planning and led in the writing of the manuscript. LYG, KFK, T-FW, carried out the review of articles for final inclusion. All authors provided contribution to the acquisition, analysis or interpretation. All authors provided critical revisions to the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
