Abstract
Purpose
This narrative review synthesizes the latest evidence on TeleStroke implementation and proposes a structured protocol for developing an advanced TeleStroke network.
Methods
A structured search was conducted in PubMed, Scopus, Web of Science, and Institute of Electrical and Electronics Engineers (IEEE) Xplore (2000-2024) using “telemedicine” OR “TeleStroke” combined with “regulatory aspects,” “ethics,” “information architecture,” “artificial intelligence (AI),” and “training.” The review sections were consensually defined by four vascular neurologists and two digital health experts. An advanced network was consensually defined as one that enables synchronous communication, secure data storage, compliance with data protection laws, remote neuroimaging visualization, integration with prehospital emergency systems, and a validated AI tool. Studies were analyzed for best practices, regulatory considerations, and AI-driven innovations.
Results
We included 142 publications between 2000 and 2024 in the final review. Key components of a TeleStroke network include deploying neurologists trained in cerebrovascular care and telemedicine, adopting secure cloud-based technologies for imaging and data storage, and utilizing blockchain to enhance data security. Embedded AI tools, such as neuroimaging analysis and natural language processing (NLP), expedite diagnostics and decision-making. Georeferenced algorithms direct ambulances to appropriate stroke centers based on patient severity and available resources. Integrating telemedicine into ambulance systems enables real-time consultation, while automated documentation within electronic medical records (EMRs) ensures continuity of care. Adhering to data privacy laws and maintaining shared clinical responsibility are essential for complete implementation.
Conclusions
A multidimensional approach integrating technology, regulation, and clinical training is crucial for an advanced TeleStroke network. AI-driven solutions, mobile platforms, and cloud-based infrastructure enhance clinical outcomes and metrics.
Background
The implementation of telemedicine solutions is a significant advancement in overcoming challenges related to access to specialized healthcare, particularly in geographically isolated areas or those with economic development limitations.1–5 Restricted access to specialists, like neurologists, in these regions can result in delayed diagnoses, inadequate treatment, and consequently unfavorable clinical outcomes for critical health conditions. 6 This scenario is particularly critical for patients with suspected stroke, whether ischemic or hemorrhagic, as the time to diagnosis and initiation of treatment is crucial for patient recovery and the minimization of functional limitations.
Stroke represents one of the most severe medical conditions, with a high risk of sequelae, in addition to having a profound social and economic impact. It became the leading cause of death in Brazil in 2021 and 2022, highlighting the urgent need for effective strategies for its management and treatment.7, 8 Rapid diagnosis and the implementation of appropriate therapies are essential to reduce mortality and improve functional outcomes for patients.8–10
In this context, telemedicine emerges as a viable and efficient solution. Studies demonstrate that telemedicine in stroke cases is equivalent to an in-person evaluation by a neurologist, significantly increasing the indication and application of reperfusion therapies such as intravenous thrombolysis, which is extremely useful for the recovery of ischemic stroke patients.11–13 The implementation of telemedicine services for stroke has been shown to improve patient functional outcomes, and it is becoming a cost-effective strategy with significant social impact13–17 (Table 1). The adoption of this technology not only provides access to the knowledge and experience of neurology specialists regardless of the patient’s geographic location but also optimizes resources, reducing the pressure on overcrowded health centers and allowing a more equitable 18 distribution of high-quality healthcare.12, 19, 20
Summary of Telemedicine Service Objectives.

Methods
This narrative review aimed to synthesize the latest evidence on TeleStroke network implementation and propose a structured protocol for establishing an advanced TeleStroke network. The term advanced was consensually defined as a network that ensures: Synchronous communication between healthcare professionals; Secure storage and logging of sensitive data; Compliance with data protection regulations; Remote visualization and manipulation of neuroimaging; Integration with prehospital emergency services; and Deployment of validated artificial intelligence (AI) tools to improve functional outcomes and care metrics.
Literature Search Strategy
A structured search was conducted using PubMed, Scopus, Web of Science, and Institute of Electrical and Electronics Engineers (IEEE) Xplore databases to identify relevant publications from January 2000 to February 2025. The following search descriptors were used: Primary terms: “telemedicine” OR “TeleStroke.” Sub-descriptors (used alone or in combination): “regulatory aspects,” “ethics,” “information architecture,” “AI,” and “training.”
The selection of these terms was guided by the predefined review sections, ensuring alignment with key aspects of TeleStroke network implementation.
Inclusion criteria comprised peer-reviewed articles, guidelines, and official reports that addressed any aspect of TeleStroke implementation aligned with the predefined framework for advanced networks, which includes synchronous communication, data protection, remote imaging, AI tools, and integration with emergency services. Exclusion criteria included non-English articles and abstracts without full-text access. After removing duplicates, two authors independently screened titles and abstracts, followed by full-text reviews. Disagreements were resolved by consensus. Ultimately, 130 articles were included.
Consensus Process for Review Sections
The review sections were consensually defined by a multidisciplinary team comprising three vascular neurologists and two experts in digital health and computational sciences. The consensus aimed to address the most critical components for the successful deployment of an advanced TeleStroke system.
Data Extraction and Synthesis
The identified studies were screened for relevance and assessed for their contributions to the following core areas: Regulatory and ethical aspects: Legal frameworks governing TeleStroke implementation. Computational architecture: Network infrastructure, data storage, and interoperability solutions. AI integration: AI-driven decision-support systems and automated imaging analysis. Training requirements: Capacity building for neurologists and healthcare professionals using TeleStroke systems.
Given the narrative nature of this review, no formal meta-analysis was performed. Instead, findings were synthesized to provide best practices, technological strategies, and regulatory considerations. The proposed implementation framework was developed based on evidence from the literature and expert recommendations.
Initial Assessment, Objectives, and Planning
Definition of Objectives of an Advanced TeleStroke Service
First, an agreement with the stakeholders responsible for implementing the TeleStroke service must be established.21, 22 This agreement should clearly define the objectives of a care network6, 23–25 that ideally encompasses all stages of stroke care.
The objective for a TeleStroke service should be the implementation of a mobile telemedicine service in secondary and tertiary healthcare units, as well as a mobile prehospital service for the assessment, 26 transfer, 27 treatment, follow-up, and counter-referral of patients with suspected or confirmed cerebrovascular disease.20, 28 Additionally, the service should enhance access to early diagnosis and treatment of stroke, coordinating hospital and prehospital actors 29 (Table 1).
Needs Assessment
Staffing
Human resources for a stroke telemedicine service should include roles in coordination, management, information technology (IT), training, and regulation. These roles should be organized into tiers with quarterly goals for performance and engagement.19, 30 Each position is vital for efficiency and compliance, addressing activations, system issues, and technology implementation bottlenecks,31, 32 as detailed in Figure 1.
Roles of a TeleStroke Service Staff.
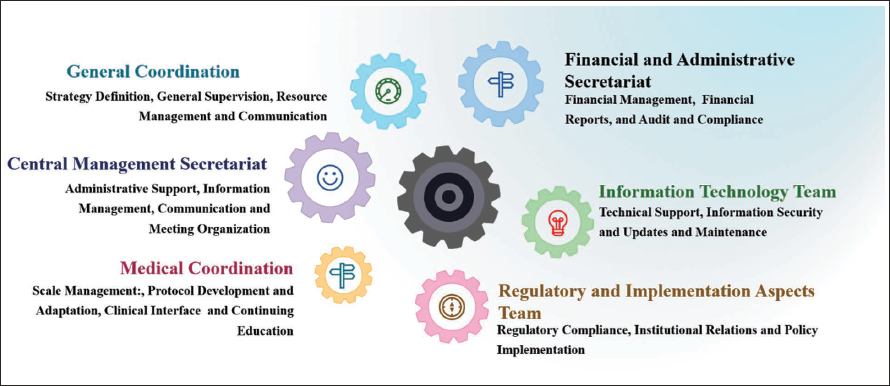
Neurologists. Neurologists must be registered medical specialists, hired under defined shifts, ensuring uninterrupted service coverage.22, 33 Preferably trained in vascular neurology (VN),34, 35 they should be certified in the National Institutes of Health Stroke Scale (NIHSS) and proficient in telemedicine. Responsibilities include patient documentation, adherence to ethical and data protection standards, and prompt teleconsultation responses.36, 37 Competitive benefits and performance incentives are recommended.21, 22, 37 Compliance with National Medical Council regulations governs practice scope, teleconsultation responsibilities, and electronic record requirements.38, 39 Neurologists must respond to all teleconsultation requests within 3-10 min of being called.22, 34, 35, 38, 39
Implementation Challenges
During the implementation of a TeleStroke network, several challenges may hinder service effectiveness. One critical barrier lies in the limitations of commercially available communication platforms, which often lack native integration with electronic health records (EHRs).40, 41 This impedes the documentation and sharing of clinical decisions among care team members and can fragment continuity of care. Additionally, many of these tools rely on generic or non-customizable alert systems, which may fail to adequately notify neurologists on call.22, 42 Ineffective alarm configurations or user interface issues can result in delayed responses or unavailability of specialists at critical moments, especially in high-demand scenarios.
Moreover, the need for reliable, high-speed internet connectivity remains a barrier in remote or underserved areas. 20 In many areas, TeleStroke coverage had to be limited to daytime hours because of inadequate 4G/5G coverage in ambulances or emergency units.43–45 Staffing is another constraint: ensuring the availability of trained vascular neurologists 24/7 requires contractual adjustments, incentive structures, and shift optimization—factors often overlooked in the planning stages. Finally, resistance to workflow changes and digital tools from local teams can hinder adoption; in the early stages of the implementation framework, several spoke hospitals reported low compliance with teleconsult activation protocols due to insufficient training and unclear responsibilities.46, 47
Implementation in Resource-constrained Settings
Satellite internet, particularly low-Earth orbit (LEO) constellations such as Starlink and OneWeb, has emerged as a feasible option to provide real-time connectivity in underserved or geographically isolated regions. 48 Pilot studies in Alaska and rural parts of Australia and Chile demonstrated that these services could support the transmission of neuroimaging data and facilitate synchronous video consultations with acceptable latency and bandwidth for acute stroke care.19, 36, 49 Although these technologies come with initial setup costs, models based on government subsidies, public–private partnerships, or pooled financing among regional hospitals have proven sustainable in several low- and middle-income countries (LMIC) settings. 44
Additionally, bandwidth-adaptive platforms can sustain core TeleStroke functionalities under poor connectivity conditions. Systems such as store-and-forward telemedicine allow asynchronous transmission of computed tomography (CT) scans and clinical data, which are uploaded automatically once a connection becomes available. In Kenya and India, for instance, hybrid offline-first telemedicine platforms have enabled structured stroke triage using pre-installed NIHSS and modified Rankin scale (mRS) scoring modules embedded in mobile apps, allowing paramedics and non-specialist physicians to initiate evaluation even without internet access. 50 Data compression algorithms and AI-enhanced Digital Imaging and Communications in Medicine (DICOM) uploaders—already validated in LMIC settings—help reduce file sizes without compromising diagnostic accuracy. 51
Technical Resources
The proposed telemedicine mobile application 52 features are summarized in Table 2.
Telemedicine Mobile Application Features.
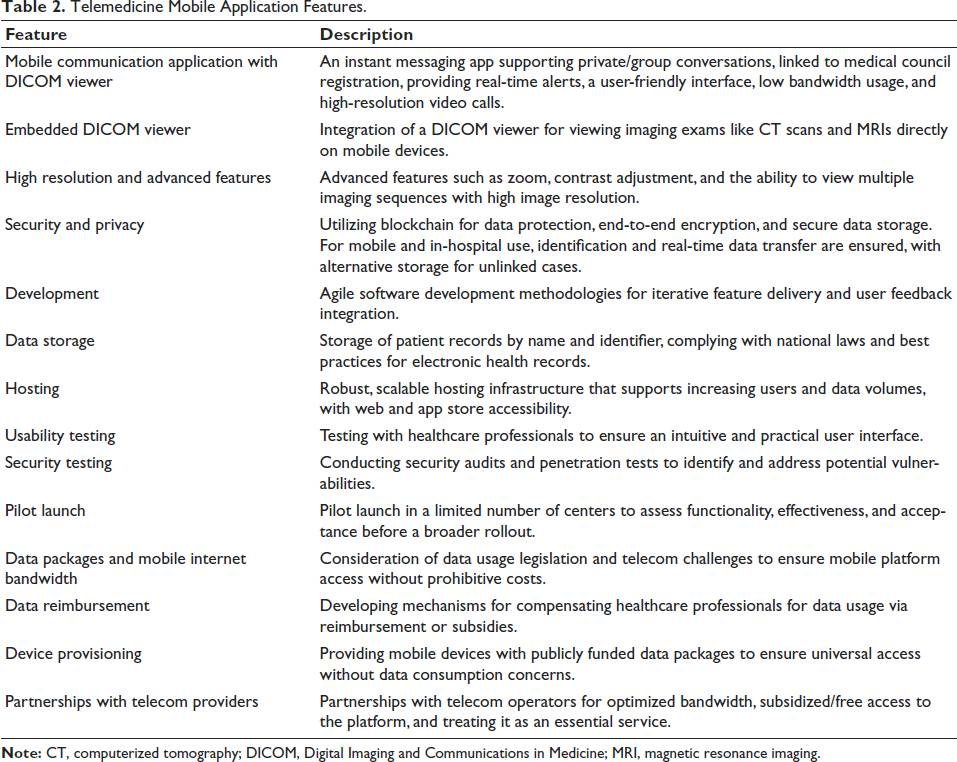
The adoption of strategies to mitigate the impact of data consumption on healthcare professionals is crucial for the program’s success and sustainability, enabling greater participation and effective implementation in various settings.15, 31, 37, 53
In addition, desktop-based platforms and mobile applications should be integrated. In particular, electronic medical records (EMRs) and imaging software should be integrated for collecting, registering, and viewing patients’ data, in a two-way traffic of information.31, 37, 54
Embedded Software in CT and Magnetic Resonance Imaging (MRI) Suites
Spoke hospital units must integrate secure software within imaging suites to automatically transmit DICOM files to a centralized cloud server. The software must support patient identification and employ data encryption, ensuring privacy and adherence to data protection standards.51, 52, 55
Cloud Server for Instant Access to Images and Text Interactions
A cloud server must securely store DICOM files,51, 55, 56 enabling instant access for neurologists during teleconsultations. Backup and redundancy policies 54 ensure data integrity, while a nationally located server adheres to local laws, facilitating regulatory compliance with standards like the General Data Protection Law (GDPL), and enhancing data privacy and security management.57–59
Storage Technology
Blockchain technology ensures secure, immutable, and encrypted data storage, enhancing transparency and auditability while preserving patient confidentiality.58, 60–63 Data access will comply with health privacy regulations, restricted to authorized personnel, and guided by necessity and minimal sufficiency principles to ensure controlled and monitored access. 62
Compliance with the GDPL (or Local Equivalent)
All data storage and processing operations will strictly adhere to the principles established by the GDPL, including transparency, purpose, adequacy, necessity, security, prevention, non-discrimination, and accountability. This compliance not only reinforces the commitment to protecting patients’ data but also establishes a foundation of trust with the platform’s users.22, 57–60
Application Selection Criteria
The proposed selection criteria for TeleStroke applications are represented12, 64–66 in Figure 2.
Application of Selection Criteria.

Prehospital Protocols and Assessment
Healthcare professionals must be trained in advance on the use of telemedicine technology and stroke care protocols, including the rapid assessment of stroke patients using the NIHSS, 67 the mRS, 68 and prehospital validated screening tools 69 (Table 3). The use of the FAST-ED (face, arms, speech, time, eyes, and denial/neglect) scale70, 71 is recommended, as discussed in the working groups that are implementing the stroke care pathway for screening patients potentially with proximal vessel occlusion.
Prehospital Stroke Scales.
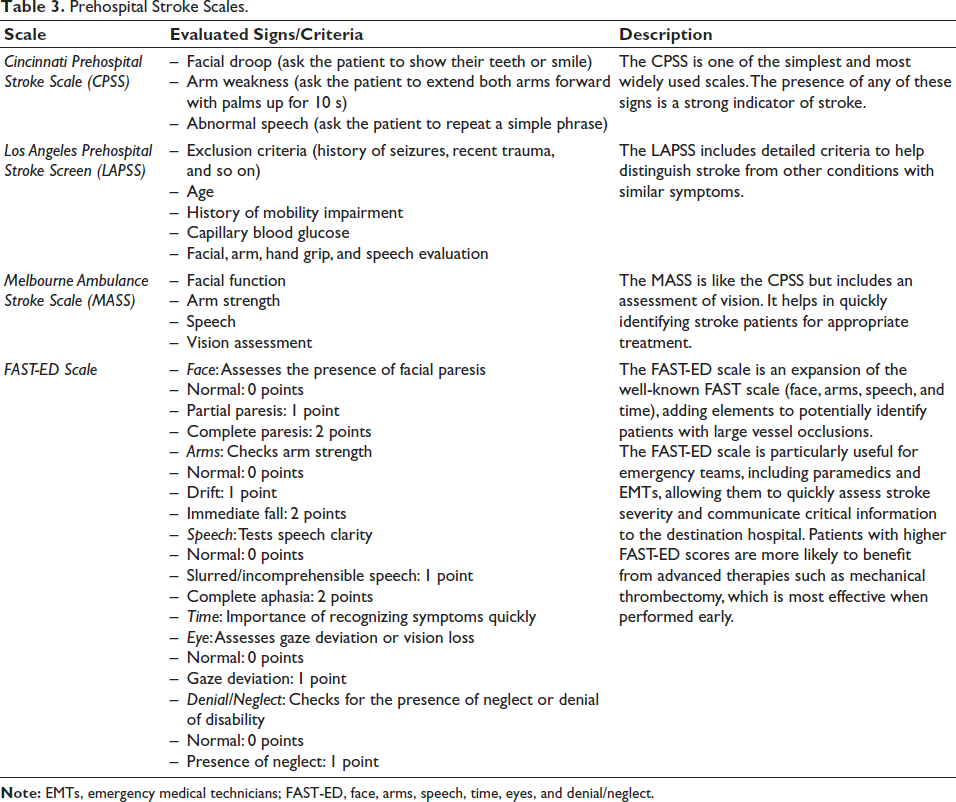
Recording teleconsultations in hospital and emergency contexts demands accuracy, security, and seamless integration with EMR.22, 58, 60 Ideally, telemedicine platforms should enable direct teleconsultation activation from the EMR via specific tools, supporting efficient communication.61, 72–75 Post-consultation, the EMR should be automatically updated with specialist notes, diagnoses, and recommendations, ensuring comprehensive documentation. Both requesting and responding physicians must electronically input and sign their contributions for accessibility and care continuity.54, 76
When Integration Is Not Possible
If direct integration with the EMR is not feasible,61, 77 the telemedicine application must maintain its records. The steps include an independent management of secure databases for teleconsultation details,46, 47 ensuring patient access and legal compliance.50, 61, 63, 73, 78 Records must include patient identification 78 and comply with privacy regulations, employing encryption and access controls to protect sensitive information.59, 62
Integration with a National Unified Health Record System
For countries with a national unified health record system,40, 79, 80 the telemedicine platform must consider integrating with the national system to enable automatic transfer of teleconsultation records,41, 81–83 ensuring consolidated access for healthcare professionals. 78 Additionally, it should grant patients full access to their teleconsultation data, fostering transparency and empowerment in healthcare management.
Ethical and Regulatory Aspects
A detailed record of the opinions provided by the teleconsultant, as well as the information received that supported those opinions, should be maintained.84, 85 The physician requesting the opinion or consultation from another physician may or may not be solely responsible for the treatment, as well as for the decisions and recommendations made to the patient.46, 86
The teleconsultant is responsible for providing a quality opinion, based on the available information and in accordance with medical practice standards. They must ensure that their recommendations are grounded in clinical evidence and best practices.63, 87
Although the requesting physician remains primarily responsible for the patient’s treatment, the teleconsultant shares responsibility, particularly regarding the accuracy and appropriateness of the opinion given. 86 In some jurisdictions, responsibility may be shared, where both physicians can be held accountable in case of an error or omission. 88
At the conclusion of the teleconsultation, the involved healthcare professional will write a report. This report must be inserted into the patient’s medical record and also into the shared/unified medical record if the patient has one. 60 Additionally, the report will be sent to the responsible physician and the requesting physician (if they are not the same) through a secure messaging system or equivalent platform in asynchronous cases.
Care Protocol
Healthcare professionals must first be trained in the use of telemedicine technology and stroke care protocols, including rapid assessment using prehospital screening scales and/or the NIHSS 67 via video or not, the mRS, 68 and prehospital screening scales. It is suggested to use the FAST-ED scale for screening patients potentially with proximal vessel occlusion and, therefore, potentially eligible for mechanical thrombectomy.25, 89, 90
Screening scales can be embedded in a mobile application within the emergency mobile system, serving as a prerequisite to opening the case discussion.69, 91, 92 Only after reaching the minimum score on at least two scales is the communication screen with the Neurology on-call team activated.92, 93
A diagram of the assistance and transfer of suspected stroke cases is illustrated in Figure 3.
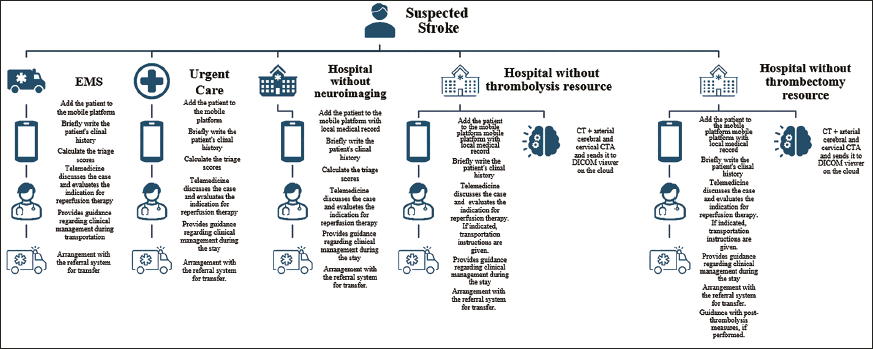
Activation Model
The initial stroke screening should adopt patient identification and documentation via secure platforms and determine the urgency of referral for treatment.71, 91 Ideally, identify the patient by civil registration or medical record from the unit initiating the request. For patients on public roads or in mobile services, national identification documents and the date of birth can be useful for patient identification. The person initiating the request should also include essential clinical data, such as triage scale score, NIHSS, and mRS, along with a summary of the main complaint and other relevant clinical information. In cases of direct audio and video activation, patient identification data must be completed.94, 95 After the call, both the requesting physician and the specialist must individually document the discussion and the proposed treatment plan. Voice-to-text platforms can be used for transcribing teleconsultations via audio or video, respecting privacy and data security standards. 96 Teleconsultation supports continuity of care, enabling alignment with local teams and optimizing dehospitalization42, 97 within the Stroke Care Line framework.14, 98, 99
Georeferencing
Integrating georeferencing into a mobile teleconsultation platform optimizes stroke patient transport by aligning real-time patient data with hospital capacity,28, 100, 101 ensuring timely access to the most appropriate care. 52
The implementation model of this algorithm begins with the collection and integration of critical data about the hospital’s installed capacity, including the availability of essential resources such as CT scans, intensive care units (ICUs), and mechanical thrombectomy equipment. These data are provided by local managers and are kept up to date to reflect the real capacity of healthcare services. Thus, the mobile platform can access this information to assess which units are prepared to treat the type of stroke that the patient is presenting.44, 102
The georeferencing algorithm processes this clinical data in combination with the ambulance’s location and the available hospital resources. The choice of hospital is based on proximity and the ability to provide the necessary care, considering the urgency and specificity of the required treatment.103, 104 The platform embedded in ambulances can also be integrated with the bed regulation systems of registered hospital units.
Once the decision is made, the mobile telemedicine platform automatically issues a notification to the selected hospital.104–106 This notification includes detailed information about the patient, such as identification data and current clinical condition, as well as any triage scale results indicating the need for specialized treatment.
Additionally, the patient’s identification data and clinical condition are sent to the hospital’s regulatory center.63, 87, 107
Benchmarks
A robust example of large-scale TeleStroke implementation is provided by the German Stroke Society, which developed detailed national recommendations for organizing teleconsultation services within a TeleStroke network. 108 These networks operate through structured partnerships between tertiary stroke centers and peripheral hospitals, ensuring 24/7 access to neurovascular expertise. The model includes well-defined processes for initial patient registration, bidirectional video consultation, neuroimaging co-assessment, and documented therapeutic decision-making. Each teleconsultation adheres to strict data protection standards and includes quality assurance mechanisms such as time-stamped process tracking and standardized reporting templates. Importantly, the system allows even semi-elective cerebrovascular consultations and the structured coordination of inter-hospital transfers, enhancing access to thrombectomy and specialized care.
In parallel, the TeleSpecialists® network in the United States demonstrates the scalability of a private TeleStroke provider model. Operating across more than 200 hospitals, the system offers real-time neurologist access, integrates cloud-based imaging and documentation, and incorporates performance dashboards to monitor quality metrics. 42 Evidence from this network shows significant reductions in door-to-needle times and increased rates of intravenous thrombolysis in previously underserved hospitals. 1
An additional instructive case comes from Spain, where a comprehensive national survey revealed that 12 of the 17 autonomous communities have implemented TeleStroke networks, covering over 22% of the country’s population. 109 These programs range from regional networks—such as in Andalusia and Catalonia, which integrate emergency medical services and vascular neurologists—to smaller provincial or hospital-based systems. Andalusia’s centralized model is particularly noteworthy: it allows 93% of the population to reach a vascular neurologist within 30 min via telemedicine. 109 Despite challenges such as suboptimal videoconferencing quality and limited registry access, these programs have increased the accessibility and equity of stroke care, especially in rural areas.
Another notable case is the South Australian TeleStroke Network, 36 which successfully implemented a regionally coordinated service covering rural and underserved areas. Through a hub-and-spoke model supported by trained stroke physicians and standardized workflows, the network demonstrated reduced stroke mortality and improved adherence to reperfusion therapy protocols. 36 The initiative emphasized digital infrastructure investment, clinical training, and mobile imaging integration, making acute stroke treatment feasible in geographically dispersed locations. Similarly, the Neurovascular Network for Stroke Patients (NEVAS) network in Bavaria (Germany), 16 maintained robust clinical outcomes and time metrics throughout the COVID-19 pandemic by relying on telemedicine-supported stroke triage and management, even in emergency-declared periods.
In the United States, the Remote Evaluation of Acute Ischemic Stroke (REACH) TeleStroke program, 12 which connects rural hospitals with academic stroke centers, has shown sustained benefits in expanding access to reperfusion therapies and decreasing the rate of unnecessary patient transfers.
Embeddable AI Tools
In addition to neuroimaging visualization and manipulation tools, algorithms for processing radiology images can be embedded in TeleStroke platforms. 110 These algorithms should be hosted on cloud servers to maintain the speed and efficiency of the TeleStroke platform.110–112
These tools use deep learning algorithms trained to identify signs of large vessel occlusions (LVOs) in computed tomography angiography (CTA) images.113, 114 Additional neuroimaging processing capabilities include the detection of intracranial hemorrhage, subarachnoid hemorrhage, and automated scoring of collateral assessments and the Alberta Stroke Program Early CT Score (ASPECTS).37, 115, 116
Natural language processing (NLP) tools can analyze large volumes of unstructured clinical data, such as medical notes, radiology reports, and EHRs. In cases of ischemic stroke, NLP can help identify patterns in symptoms and clinical signs within medical records that suggest an ischemic stroke.117, 118 This enables automated alerts that expedite response times for healthcare professionals. Additionally, these tools can be integrated into clinical decision-support systems to aid in interpreting complex data, such as imaging results or medical histories.117, 118
Speech-to-text technologies offer the ability to transcribe verbal interactions between healthcare professionals and patients or among medical team members. These tools enable physicians to document cases and medical orders quickly and accurately, without manual typing.111, 119–121 This can streamline the care process and ensure that all essential information is recorded immediately. Furthermore, healthcare professionals can use voice commands to interact with EMRs or to activate specific stroke treatment protocols. 121
The benefits of these tools include reducing diagnostic and treatment times.49, 122, 123 However, significant challenges remain, such as the need for AI model training on data specific to the local population, integration of these technologies with existing systems, and ensuring data privacy and security.119, 120, 124
Moreover, AI has shown increasing relevance in addressing several implementation barriers in TeleStroke services. In the Catalonia Stroke Program (Spain), AI-based tools were integrated into regional imaging systems to automatically identify LVOs and prioritize patients for thrombectomy referral. This not only improved decision-making accuracy but also reduced treatment delays in high-volume centers. 17 In Switzerland, the use of AI-assisted real-time video analysis in prehospital settings enabled paramedics to perform remote NIHSS scoring, facilitating early stroke identification and streamlining communication with stroke units. 24 Beyond acute triage, AI has also enhanced TeleStroke documentation and coordination. In a US pilot study,117, 118 NLP tools were employed to extract structured data—such as NIHSS, mRS, and thrombolysis decisions—from free-text emergency department notes, improving continuity of care and enabling real-time audits of stroke care metrics. In South Korea, a rural TeleStroke initiative 106 implemented AI-driven decision-support protocols to guide non-specialist physicians through acute stroke management steps, based on patient symptoms and uploaded imaging, resulting in faster therapeutic interventions and greater protocol adherence. 106
Training Proposal
The integration of TeleStroke training into VN fellowship programs is a strategic response to the increasing demand for TeleStroke. This can be a strategy for the sustainability of services; however, training initiatives are still scarce in many programs, especially among those in LMIC. 125
Pioneered by the McGovern Medical School, this model involves a formal TeleStroke rotation, allowing fellows to conduct remote consultations under structured supervision. Training encompasses orientation to telehealth platforms, shadowing experienced stroke attendings, and progressively independent patient evaluations. Didactics, such as case conferences, further support skill development. 126
Training in TeleStroke involves developing a comprehensive set of skills to ensure effective, efficient, and professional remote stroke care. The evaluation of fellows should focus on the following core competencies
127
: Technical skills: Proficiency in using telemedicine tools, such as Zoom, adjusting the field of view, and maneuvering robots (if applicable). Fellows should recognize and address connection issues, including knowing whom to contact for technical assistance. History-taking: The ability to gather an efficient yet detailed patient history relevant to acute therapy decisions, including contraindications and recommendations to treatments like intravenous tissue plasminogen activator (IV tPA), treatment windows, vital signs, and laboratory results. Imaging interpretation: Competence in remotely reviewing imaging to identify acute stroke findings, such as early ischemic changes, hyperdense signs, or hemorrhages, and to guide therapy decisions. Handling complex cases: Knowledge of managing challenging clinical scenarios, such as recent myocardial infarction or previous strokes (ischemic or hemorrhagic). Neurological examination: Skill in conducting quick, accurate stroke and neurologic assessments, and in communicating complex NIHSS components to staff. Team communication with spoke staff: Ability to emphasize urgency during stroke codes, communicate plans clearly, and expedite treatment, such as calculating tPA dosing. Team communication with spoke physicians: Proficiency in discussing findings and treatment plans. Team communication with patients and families: Clear and concise communication of clinical assessments and plans, including obtaining consent. Identifying endovascular candidates: Recognizing potential candidates for endovascular therapy and navigating transfer logistics, including Emergency Medical Treatment and Labor Act (EMTALA) rules and coordination with hubs. Professionalism: Developing strong rapport with spoke physicians, nursing staff, patients, and families, ensuring a collaborative and respectful environment.
Regulatory frameworks, such as the Accreditation Council for Graduate Medical Education (ACGME) milestones, highlight the need for nuanced supervision protocols, particularly distinguishing between direct and indirect supervision in time-sensitive scenarios. The post-pandemic regulatory environment underscores the importance of institutional collaboration to adapt to shifting supervision and billing requirements.126, 127
Emerging modalities like mobile TeleStroke, enabling prehospital consultations in ambulances, add a layer of complexity to training, requiring tailored approaches. Simulation-based training, virtual rotations, and inter-program collaborations are proposed to ensure equitable access to TeleStroke education across institutions, including those without established networks.126, 127
A formal evaluation framework categorizes trainees’ development into five levels, focusing on their ability to manage technical systems, conduct efficient assessments, communicate effectively, and coordinate complex care.
126
1. Level 1 a. Conducts a complete and relevant history and examination. b. Manages acute stroke patients remotely using telemedicine tools, addressing acute risk factors effectively. 2. Level 2 a. Performs efficient and complete assessments, identifying surgical or endovascular candidates and high-risk patients (e.g., intracranial hemorrhage). b. Resolves technical issues or seeks appropriate assistance when needed. 3. Level 3 a. Integrates history and examination findings into treatment decisions. b. Manages acute stroke cases without unnecessary delays, efficiently coordinating with spoke hospital teams for transfers or advanced care. 4. Level 4 a. Demonstrates proficiency in remote assessments and treatments with minimal delays. b. Facilitates patient transfers for higher levels of care, ensuring compliance with regulations (e.g., EMTALA). c. Communicates effectively with spoke physicians, staff, patients, and families, maintaining professionalism throughout. 5. Level 5 a. Serves as a role model for other trainees by demonstrating excellence in telemedicine consultations. b. Efficiently manages acute stroke cases, triages post-tPA patients, and coordinates advanced care and transfers seamlessly.
Financing Mechanisms and Sustainability of TeleStroke Networks
Public funding remains central, especially in resource-limited settings and rural regions where the benefits of TeleStroke are potentially highest but adoption is lowest. 43 Government programs may subsidize infrastructure costs—including telecommunication equipment and connectivity—while academic centers often serve as hubs, absorbing initial technology investments. 43 Partnerships with telecommunications providers have also emerged as viable models to expand broadband access and minimize fixed connectivity costs. In some models, costs are defrayed by pooling smaller hospitals into centralized service contracts, coordinated through regional networks or resource centers that aggregate demand and streamline credentialing and technical support.128, 129
Long-term sustainability requires ongoing strategies beyond the initial implementation phase. One critical pillar is integrating TeleStroke into national reimbursement frameworks, including procedure-based payments and bundled-care models. Despite growing evidence of cost-effectiveness, current reimbursement systems often fail to capture the operational expenses of TeleStroke at the originating site.129, 130 To offset this, hospital systems may explore direct billing for remote neurologist consultations and reallocation of avoided transfer costs. Sustaining clinician engagement is equally essential; structured training programs, digital literacy development, and the incorporation of TeleStroke workflows into clinical routines increase utilization and acceptance over time. Embedding TeleStroke practices within broader telemedicine strategies also promotes scalability, enabling health systems to leverage shared platforms, reduce redundancy, and achieve financial and operational resilience.43, 128
Conclusion
The use of appropriate information and communication technologies allows specialized medical treatment for people living in distant locations or with few professionals. TeleStroke allows to expand the actions of professionals and community health agents, integrating them into the services offered by hospitals and reference centers.
Our proposed protocol differs from existing TeleStroke models by offering a comprehensive and integrated framework that explicitly addresses the architecture and security of digital infrastructures, a topic often underrepresented in previous medical literature. In addition to highlighting traditional elements such as specialist availability and teleconsultation workflows, our review emphasizes critical innovations in software structure, including embedded DICOM viewers, blockchain-based secure storage, and compliance with national data protection regulations and EHR assessments. Furthermore, we reviewed advanced technologies such as AI for neuroimaging triage and NLP, .369solutions that ensure interoperability with EHR and national health data repositories. This multidimensional approach ensures that the proposed protocol is technically robust and suitable for broad implementation in diverse health systems.
Authors Contribution
João Brainer Clares de Andrade: Design, supervision, and data collection; Daniela Laranja G Rodrigues, Claudia Galindo Novoa, and Gisele Sampaio Silva: Supervision, data collection, and final review. Ivan Pisa, Sophia Oliveira Querobin, Marialdo Augusto Cordeiro Souza, Milena Roberta Guimarães Brianti, and Octavio Marques Pontes-Neto: Data collection and final review.
Footnotes
Declaration of Conflicting Interests
The authors declared no conflict of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was obtained from the relevant ethics committee or Institutional Review Board (IRB).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors Andrade, Laranja, Pontes-Neto, Martins, Novoa, and Silva received research grant from Ministry of Health (Brazil), and Pisa received research grant from National Council for Scientific and Technological Development (Brazil).
Informed Consent
The participant has provided informed consent for the submission of the article to the journal.
