Abstract
Introduction
Technological advancements have increased the scope and range of home-use medical devices. These have allowed development of care at home. This requires appropriate understanding of patient's needs with regards to such devices for safe use of medical technology at home and minimise usability errors. This study sought to investigate patient experiences of usability and to identify future requirements for such devices
Methods
A qualitative mixed methods study was chosen. The participants were asked to complete a survey which included both multiple-choice and free-text entry questions. Participant recruitment was through purposive and snowball sampling approaches. All participants were required to have direct experience in using a medical device. Data were thematically analysed.
Results
There were 360 responses by patients, parents and carers. 503 devices were used by these participants. 99.4% used their device(s) on a daily basis. The largest group of responders were from type 1 diabetic patients. Multiple usability challenges were reported with respect to training, device interoperability, development of workarounds and the device-user interfaces. Device acceptance and their costs were also raised as issues for concern. A list of usability challenges was compiled.
Conclusions
Patients and other end users continue to experience multiple usability challenges in the use of medical devices at home despite guidance from regulatory bodies. Better post market studies are required to generate evidence to enable the improved design and safety of the device. This should be accompanied with an evaluation of improvements in patient satisfaction and their compliance with monitoring and treatment.
Keywords
Introduction
Medical technology for home use has seen a major growth in the recent years in.1,2 Changes in patient demographics, patient preferences, economic factors along with the aim of reducing hospital admissions and length of stay have greatly influenced the development and implementation of patient care at home.1–4 In UK (and EU), the increase in the ageing population has led to programmes aimed at empowering patients to take control of their health with support from medical technology.5–7 This is being achieved with the help of a number of programmes such as the NHS innovation Accelerator Programme, Clinical Entrepreneurs Programme and building of research and development centres throughout UK for medical devices through UK Clinical Research Collaboration.5,6 Furthermore, the Topol Review showed the importance of care at home and the principles of transfer of care from hospital to community setting. 8
Devices for use by patients at home bring additional challenges related to the diverse range of age, abilities, training and the constraints relating to the environment of use.1,9,10 Hence, study of device usability is essential to understand both their overall effectiveness, patient acceptance and any potential competitive advantages in this rapidly expanding market.11,12 Human factors (HF) principles ensure that the device interface is designed taking into account end-user capacity and needs and thereby minimise errors in use that might otherwise lead to harm. 1 Although HF principles have been successfully applied to other high risk industries for many years, they have had only limited application in healthcare technologies.9–11,13,14
Detailed documentation is provided by regulatory bodies such as MHRA (Medicines and Healthcare products Regulatory Agency) and FDA (Food and Drug Administration) on application of HF principles in the design of medical devices including the application of HF methods in the pre and post market stage.1,15 In EU, the IEC 62366 Medical Devices – Application of Usability Engineering to Medical Devices has been incorporated in the process of pre-market assessment of medical technology. 11
Despite this, patients and end-users of medical technology at home continue to be faced with multiple challenges in relation to their usability. Human factor studies involving patients in the home environment are rare. We conducted a systematic review on usability challenges faced by patients and found very few studies (n = 12) with direct patient involvement. Most were small studies involving a single type of device. Some of the reasons for this may be the difficulties in conducting these studies and manufacturer costs. 16
Most HF studies have taken part in an institutional setting 9 which may not adequately reflect the home environment.10,16 Delivery of safe and personalised care to patients relies on a good understanding of their individual needs. 17 Hence, HF studies in the home/community setting are essential, despite their challenges.
Patient-reported health outcomes have become more important in recent years. They provide information on treatment received, generate the evidence required to characterise the value of new treatments and monitor adverse events. They have been widely used across medical specialities. 18 Similarly, patient-reported device usability is related to their health outcomes. Data on factors affecting usability of home medical devices is scarce and studies are difficult to conduct.9,10,19 The Cumberledge report (2020) 20 recommended that patients should be engaged in their care and the importance of learning from their experiences.
This paper presents a qualitative study that collected data on direct patient and carer experiences with the use of medical devices in the home environment. It presents the usability challenges faced and their impact on safe use and specifically on i) patient/user usability challenges, ii) device usability requirements and iii) safe use of medical technology at home and potential usability errors.
Methods
A mixed methods approach was utilised to best enable the exploratory nature of the study given its recognised effectiveness in evaluating complex interventions.21–23
The patient survey was developed following a systematic literature review, PPI workshop with patients and members of the public and input from a HF specialist (MM). The survey was developed to assess challenges faced by patients whilst using a medical device at home. It included both multiple-choice and free-text entry questions. This was done to allow better coverage of patients’ different experiences.
Participant recruitment occurred through a purposive and snowball sampling approach. The study was advertised through a number of charities and research groups assisting patients who use devices at home. These included Anticoagulation UK, Barts Health Diabetes Group, Diabetes UK, Juvenile Diabetes Research Foundation (JDFR) and Kidney care UK. Additional study advertising was carried out through NIHR London IVD website and social media such as Dexcom G6 group on Facebook, LinkedIn and Twitter. Given the national distribution of these charities and research groups, patient and carers included were expected to live in different parts of the UK.
The main inclusion criteria were that participants used a medical device independently at home (as a patient, parent or carer) and were willing to participate in the study.
A participant information sheet was made available to participants prior to taking part in the study. The link to the questionnaire only allowed one response per participant. The responses were included if they were at least 70% completed.
All participants gave informed consent prior to taking part (incorporated at the start of the survey).
Data were collected on the following:
Patient-reported challenges Usability requirements for devices Safe use of medical technology at home and usability errors
The survey contained 32 questions exploring the above themes.
Thematic analysis was conducted as guided by the aims of the study. Other evolving themes were noted and presented. A section of the results was analysed by an independent assessor (MM) to reduce risk of bias in data analysis. The survey was designed in Qualtrics and data were collected through it.
A list of usability challenges was constructed based on the results of this study. The format used by the FDA on their usability recommendation document 1 was used to present the collected data. This enabled data presentation in an organised manner to aid designers and manufacturers of home use medical devices.
The Standards for Reporting Qualitative Research were utilised as a recognised and standardised method of reporting qualitative studies. 24
Results
The survey yielded a total of 540 responses from which 360 were at least 70% completed (94% of the 360 surveys were 100% completed). Hence, 360 responses were included in the study. The M:F ratio was 1:2. The mean age of participants was 39 years old. The youngest patient was 3 years old (parent completed the survey) whilst the oldest patient was 80 years old.
The largest group of participants responded to the advert placed via the JDFR and hence were type 1 diabetic patients of different ages (94%). This led to a young age group taking part in the study (31.4% of participants were over the age of 50 years).
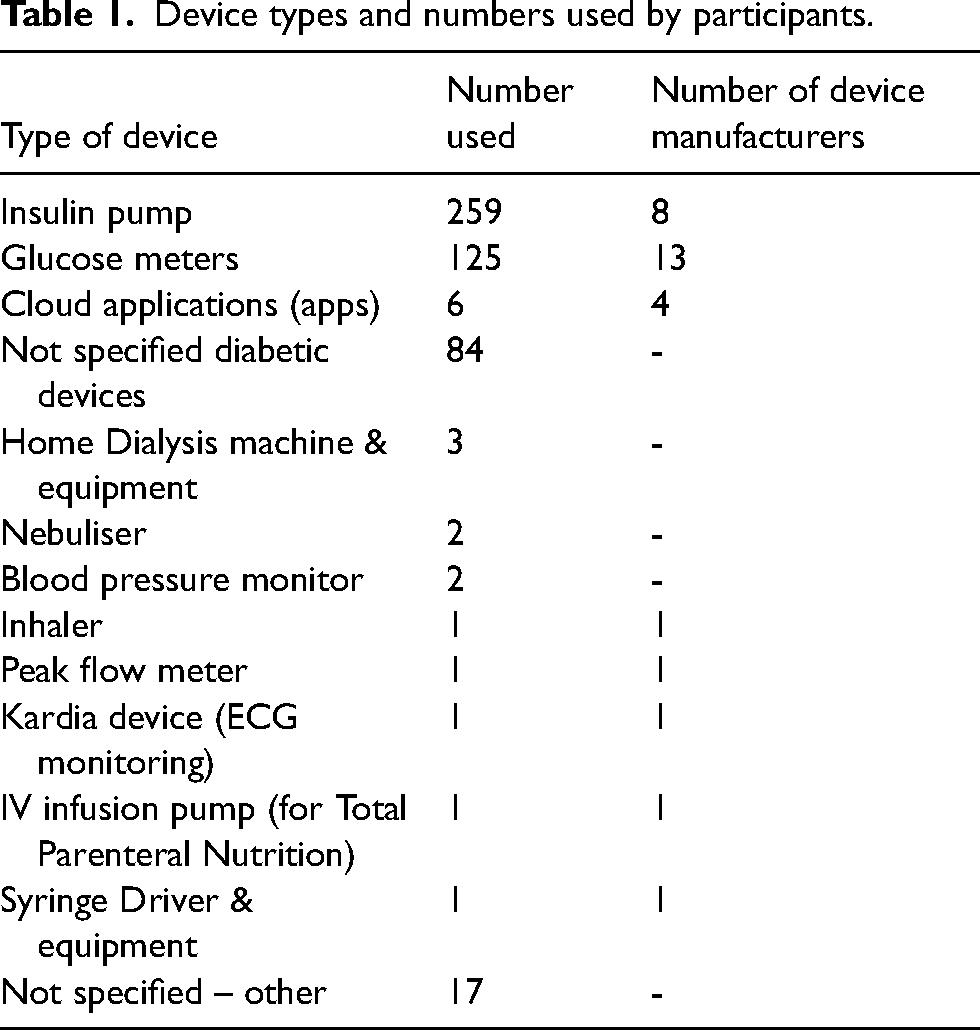
Table 1 shows the device groups used by patients in this study. Device manufacturers were reported for some of these devices. Table 2 shows the number of patients, parents and carers that took part in the study.
Device types and numbers used by participants.
Who completed the survey.

Participants often used more than one device with the 360 patients using a total of 503 devices between them. 99.4% of participants used their device(s) on a daily basis. Only 2 participants used them 2–3 times a week.
43% of participants reported to have researched and chosen their own device (often self-sponsored). For diabetic patients, specialist nurses had recommended the device in 31% of cases whilst GPs (general practitioner) and hospital specialists had only recommended 4% of devices (2% each).
Patient-reported usability challenges
Patient-reported multiple challenges in relation to device use at home including the following:
Training received
Whilst 87% of participants had received training prior to device use, only half of them (51%) stated that it was useful to very useful and only 15% reported being confident to very confident in device use following the training session.
Confidence in detecting errors
Only 45% of patients stated they were confident in detecting an error or problem with the device. The remainder were either not confident (18%) or somewhere in between (38%).
Device interoperability
73% of devices were used with other devices such as monitors and treatment devices. Interoperability problems were reported to occur all the time by 7% of patients, sometimes by 33%, rarely by 38% and 22% reported to not have experienced such problems.
The device is ‘ok’, the interaction of the device with the user needs some work. I wear the pump on my belt, but that doesn't work when going to the toilet, or when wearing pyjamas etc. Having a solution that considers the ‘24/7’ of life with a device rather than just the acute medical use of it would be really helpful.
Development of workarounds
Many patients had developed workarounds to overcome the problems they faced. Most related to receiving abnormal results and trust on device readings.
Thematic analysis revealed 69 different workarounds developed by patients and carers to overcome problems with device use. The largest group (55%) used manual testing with an alternative device to ascertain whether the results were correct. Alarms and error messages were often the cause of the problems and were only used by 2.5% of patients to identify and manage a problem. A smaller group of experienced users (12.5%) were able to correlate the results with the clinical symptoms whilst 14 (23%) were able to correlate the results with data trends recorded by the device. Hence, their experience allowed them to recognise erroneous results that would otherwise be treated with the incorrect dose of medication (insulin).
Device acceptance
Device acceptance was variable. Whilst patients were happy to have available technology to help with the management of their condition, some had reservations about how this made them feel and the effect on their private life.
Pros
Made control of my diabetes easier and 24hour sensor reassuring to pre-empty Hypoglycaemia and take action. Wife calls kit 3rd item in our relationship
The problems with the pump are massively outweighed by the improvement to my lifestyle. It has small irritating features but my life has changed so much since using a pump, it is a tiny burden to bear.
The quality of life has increased since I use these devices.
Cons
Visually there feels alien to me it makes me aware I have a medical condition
It's quite bulky and can affect my sleep quality (rolling onto it)
There aren't really any practical cases suitable for women. I put mine in my bra which is not always ideal.
Financial costs
Some patients (43%) are choosing their own devices which leads to them privately funding the management of a chronic condition such as type 1 diabetes. This leads to lifelong financial constraints as seen in these participant comments.
I have used my pump and cgm* together to build a closed loop artificial pancreas this has been a huge move forward in my treatment but is not supported directly by the NHS
Help us self-funders to be NHS funded! We are out here doing our best, keeping ourselves healthy and reducing the future burden on the NHS but not being helped and bearing the financial burden too.
The fact for g6**. You have to pay £156 a month
*cgm = continuous glucose monitoring, **g6 – Dexcom G6 device,
Usability requirements for devices
A list of usability challenges was created which was converted into a list of usability requirements (Table 3). The device-user interface challenges were the most commonly reported problems. As can be seen from Table 3, the list is extensive and includes many known items from the usability checklist presented by MHRA and FDA. Many challenges were reported especially in relation to the size, alarms, interface, adjuncts and training received. Some of the participant comments on this issue are presented below.
Personally, a more discrete design would be nicer, the ‘Walkman’ name colleagues have given it is somewhat embarrassing, reducing the cable tug would also improve comfort for wearing through perhaps a supportive dressing or better designed excess cable control system (I.e. excess cables retracts so it can't be caught or it can be adjusted by wearer)
I am a designer, and I am struck by the way in which no matter how wonderful these devices are they have not been fully designed for a consumer market. They are still firmly clinical devices and could be greatly improved by much greater design input from users. Examples being you need separate inserters for sensors and infusion sets, why can’t they be designed to use 1 insertor? The CGMS sensor is remarkably fiddly and relies upon cannula plasters etc, that is a clinical solution for a domestic environment.
Consumables and software are not coordinated for eg why do cannula sets not have a colour code which then relates to a full amount pre-programmed also colour coded in the pump so that it cannot be wrong? Why to pump screens not rotate? They are worn devices and it is far easier to look at them if they can be read upside down? That said these devices are great and we do have greater control so I think we are getting to a point where we can begin to think about new measurements to inform us about control out of target range e.g., mmol/hours might be an idea.
If they can be designed for a non-clinical environment I feel that we can have greater compliance and therefore better control.
Bulky handset, difficult to press buttons, hard to see at night, information summary substandard compared to how this kind of info could be displayed on your phone
Patient-reported usability challenges.

Safe use of medical technology at home and factors leading to usability errors
Safe use of medical devices at home is dependent on multiple factors. The user experience is important in safe use of devices. Another factor is the ability to recognise device malfunction and take appropriate steps. Technology that involves a device – drug combination, has the added risk of incorrect dosage, and drug interactions in the absence of assistance from a healthcare professional. Table 4 shows some of the factors reported by patients. Patients’ comments can be found below:
Alarm did not sound even though canula had become detached ended up in hospital.
DKA* due to pump failure. Happened so quickly!!! Caught me off guard
For the developers to take on board that the device is not being reliable enough
Usually, technical issues with a faulty handset that connects with my pump. It is a repeated issue for which I've had replacements. I would like to change device to a more reliable one but this option is not available to me due to having to continue until warranty expires. *DKA – Diabetic keto acidosis
Factors affecting safe use of devices by patients.

Usability errors in using medical devices in the home environment were reported to be related to i) repeated erroneous results leading to mistrust in the machine reading, ii) time lag between system update and new readings given by machine and iii) a mismatch between the designer and manufacturer expectations and real-life experiences of patients and end users. One common complaint was that the device would not allow for identification of error source. Even when an error message was given, this was often in a code format which was difficult for patients to identify. Patients also reported a shorter device life than expected leading to the need for alternative device use. Below are some of the participant comments:
Not waterproof. The GUI (graphical user interface) is rather menu happy, looks like the interface was stolen off a 1990s Symbian phone.
The device comes with a clip to attach it to your waistband. It was nice and slim but kept breaking. They redesigned it and now it's so bulky the device stands off the waistband, is not discreet in the slightest and i’m more likely to knock it off or catch the tubing so I have given up on using it.
It's bulky and large to be attached to a child 24/7. The buttons are hard to press so even when he could start to manage with supervision he will be unable to
Button size, types of alarms, volume of alarms, screen clarity, text size, metal case makes Bluetooth erratic, cheap-n-tacky construction, black cases (I'm a hippy chick not an undertaker!!!),
Discussion
Patients and end-users experience multiple usability challenges in the use of medical technology at home, despite recommendations by regulatory bodies. Often patients feel that devices are designed for the clinical environment and that the variability in end users or the home environment have not been taken into consideration. The training required for device use is essential for both safety and learning troubleshooting techniques. The level of training however is variable and in this study it did not correlate with the level of confidence in device use.
The majority of participants in this study had chosen their own device based on their personal research. This shows an independent decision-making process although some groups of patients would appreciate support from clinical teams. Additionally, many patients are self-financing their devices and treatments which may become a lifelong financial commitment leading to other constraints.
The device-user interface is the largest issue where improvements could be made that might enhance patient experience and therefore, compliance with treatment. Alarms aim to help the user identify a problem with the device. However, very often they are reported to be the source of the problem. This relates to them being in codes and difficult to understand, being too loud or too quiet and being triggered by other factors (e.g., environmental factors).
The interface between the device and user is vital for safe and effective interaction. An understanding of the interface is therefore essential for optimal device design and design of safe and user-friendly devices.2,25 This can only be achieved through interaction with the end users. 17 Patients often do not make a distinction between different types of technology (i.e., medical vs non-medical). A standard interface of commonly used features would allow for better usability. 26 Usability engineering allows for optimisation of medical technology and should be used accordingly in this area. 27
The interoperability with other devices and data transfer through Bluetooth for example are problematic and not always reliable. This requires further research and improvements in their interoperability with other complex devices. Difficulties using a device were found to be directly related to the compliance with medical treatment. Other concerns related to the increased connectivity are related to software security, device privacy, device security and risk of device hacking. 28
Usability challenges are expected to be greater for patients with physical or sensory impairment. Studies have found that medical equipment use difficult and potentially hazardous in this group of patients. 27 Errors in use can lead to patient harm and can be related to poor device design especially when the device interface is complex. 15
Recommendations by regulatory bodies are not part of compliance requirements although it is expected that most manufacturers will apply them to the design process.1,15 The difficulty stands in the fact that many usability challenges are not apparent until the post market stage. A HF approach is hence advised for both the pre and post market stages with a role in the risk vs benefit management throughout the device lifecycle. 15
An inappropriate design results in error-prone devices leading to increased stress for end users. 29 As a result, workarounds develop to overcome the mismatch between design and end user needs.29,30
Device design is directly related to patient safety and quality of care. An inappropriate design can lead to system vulnerabilities by diverting attention away from medical care. In the pre-market assessment stage, work as imagined is evaluated which makes it insufficient for assessment of device adequacy in real life.17,29 Designing for heath at home presents many challenges to designers of medical devices. This study has identified a long list of usability challenges reported directly by end users. Further work is required to design effective methods of gathering post market usability data from end users with the aim of improving device safety, patient experience and aiding designers and manufacturers.
Study limitations
Whilst this study engaged directly with patients, majority of the data was collected from diabetic patients belonging to younger age groups. This was not intentional on this study, it may reflect the extent of involvement of this patient group in their care and their willingness to take part in clinical studies. We are aware that this presents limitations to this study. Future work should include other patient groups including those with disabilities and older age groups which may experience further usability challenges.
Conclusions
Despite clear guidance from regulatory bodies multiple challenges still exist in the use of medical devices in the home environment. Further work is required to address the knowledge gaps and improve methods of carrying out post market studies with direct patient/end-user involvement and to consider wider patient groups. Emphasis should be given to the design of appropriate post market studies with direct patient involvement as the most reliable approach for improving design and safety of devices and increasing patient satisfaction and compliance with treatment.
Supplemental Material
sj-doc-1-cri-10.1177_25160435241230143 - Supplemental material for Usability of home use medical technology: The patient perspective
Supplemental material, sj-doc-1-cri-10.1177_25160435241230143 for Usability of home use medical technology: The patient perspective by Arkeliana Tase, Peter Buckle, Melody Z. Ni and George B. Hanna in Journal of Patient Safety and Risk Management
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was granted by the Joint Research Compliance Office (JRCO) at Imperial College London. SETREC reference: 20IC5714.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of responsibility
I, Arkeliana Tase as the primary author for this study, accept full responsibility for the conduct of the study, full data analysis, presentation of results and decision to publish.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
