Abstract
Background
Immunological responses to metal ions can occur in people with metal implantable devices. Nevertheless, patients are not routinely asked about metal hypersensitivity nor tested for it prior to surgical procedures. Moreover, we have not identified any published literature on patients’ experiences with this condition. We undertook this study to better understand the experiences of patients with metal hypersensitivity and to determine what information would help them make an informed decision about implantable medical devices with metal compositions.
Methods
This is a patient-oriented research using an interpretative phenomenological methodology. We enrolled 16 people from five countries (Canada, New Zealand, Spain, the UK, and the USA) who experienced metal hypersensitivity following implantable medical device. We collected data from in-depth, semi-structured interviews focused on pre-procedural patient education and postoperative experiences to elucidate the barriers and opportunities related to metal hypersensitivity. We analyzed the data using interpretative phenomenological analysis.
Results
The majority were white biological women between the ages of 34 and 65. None of the participants were informed about immunological responses to metals in implants during the consenting process. They encountered many challenges when symptoms of metal hypersensitivity occurred. The outcomes for the few who had the implants removed were mixed.
Conclusion
Hypersensitivity reactions to metal need to be discussed as part of the consenting process, and additional strategies are needed to mitigate this issue in the health system.
Keywords
Introduction
An implantable medical device is a device that is partly or completely introduced into the human body and remains in situ after the procedure.1,2 Implantable medical devices include stents, pacemakers, orthopedic devices, gynecological devices, dental implants, and surgical accessory devices like clips. It is estimated that between 5% and 10% of people in industrialized countries have implantable medical devices. 2 Globally, their use is on the rise because of the increasing demand for pain-reducing or life-saving procedures to maintain bodily function. In Canada, more than 130,000 hip and knee replacements were performed between 2018 and 2019, representing a 20–22% increase from 2017.3,4 The healthcare costs for hip and knee surgeries alone are more than C$1.4 billion.3,4 In the majority of cases, people with implantable medical devices have no adverse reactions—the device works as expected to treat the condition. 5
The human body maintains a tight homeostasis of metals; as such, even trace metals that are essential to normal bodily functions (e.g. copper, iron, and zinc) are known to have adverse health effects when the body is overloaded.6,7 Less known to clinicians is that metals in implantable medical devices (e.g. nickel, cobalt, and gold) can also have negative impacts on health. 8 These metals are considered inert due to their relative resistance to corrosion and presumed biocompatibility. 9 However, evidence suggests that in the body, metals corrode through slow and inevitable processes, such as mechanical wear and tear, physiochemical corrosion, and cellular-gated mechanisms. 9 Exposure to the released ions at sufficiently high enough concentrations for long periods of time can result in dysregulation of the normal metal homeostasis leading to toxic effects on the body. 10 There is a growing evidence that metals in these devices can trigger a delayed type IV hypersensitivity reaction when the ions bond with proteins in the blood. 11 Ultimately, the symptoms of this immune response can interfere with a person's ability to perform activities of daily living, thus negatively affecting their quality of life.
The effects of metals in the body can range from topical dermatitis, to aseptic loosening of implants, to less characterized and complex autoimmune/inflammatory syndrome induced by adjuvant and systemic nickel allergy syndromes.12,13 Metal hypersensitivity is also proposed to be associated with autoimmune diseases such as connective tissue disease (e.g. rheumatoid arthritis and Sjogren's syndrome) and fibromyalgia.14,15 This suggests that hypersensitivity reactions to metal occur on a continuum, with an asymptomatic sensitization phase characterized by subclinical immune activation to symptomatic clinical phase with an associated increase in healthcare utilization. 16
Healthcare providers are obligated to obtain informed consent before any intervention can take place.17,18 Informed consent is not merely the act of signing forms, but it is an ongoing process between the provider and the patient that is grounded in the ethical principle of autonomy.19,20 The precise definition of autonomy as it relates to ethics is debatable, 21 but it is generally accepted to encompass independence from controlling influences and the ability to take intentional actions. 18 Respect for autonomy can be shrouded by various factors, including the degree of independence an individual has to exercise their self-directedness. 22 As such, three preconditions of autonomy have been suggested: intentionality, with understanding, and without controlling influences. 17 Based on these precepts, respect for autonomy is not an absolute but rather a continuum that depicts the degree of understanding and controlling influences the person has. 17 For healthcare providers to respect the principle of autonomy, they must disclose information about the medical condition(s) and treatment option(s) so that the patient can exercise their self-determination. Doing so supports the common morality principles of informed consent process, truth telling and confidentiality. 21
Respect for autonomy is also important in shared decision-making process that is central to the provider–patient interaction and patient-centered care.23–26 Shared decision-making is grounded in the principles of self-determination in that it is an approach that supports patients in their decision-making process. 23 It involves the clinician sharing the best available evidence so the patients can consider their options and make an informed choice in light of their values and preferences.23,26 Thus, shared decision-making is one way of addressing the three preconditions of respect for autonomy by enhancing the patient's understanding. 17
Before surgery, the performing surgeon must obtain consent from the patient. The informed consent process must include the following: (a) assessment of the patient's competence to understand and make decisions; (b) full disclosure about the nature of the surgery verbally or with information pamphlets by the surgeon; (c) assessment of comprehension of the disclosure; (d) the ability of the patient to act voluntarily; and (e) give consent, authorizing the surgeon to carry out the surgery. 26 The practice of attaining informed consent is fundamental to assuring respect for autonomy, but it also assures the healthcare provider abides by the principles of nonmaleficence, beneficence, and justice that are the cornerstone bioethics principles of modern medicine.18,27
How much information should be disclosed during the consenting process is debatable. But generally, the adequacy of the disclosure can be determined by one of these standards: a professional practice standard, which is information that a reasonable physician would provide; a reasonable person standard, which argues for information that any reasonable person would expect; and the subjective standard, which is when the provider conveys information that particular person would want to know. 19 Informed consent is required before patients undergo surgery involving the implantation of medical devices with metal composition. It is unknown whether information about the metal composition of the implantable medical device and the risk of a hypersensitivity reaction to the device are discussed as part of the consenting process. Thus, we undertook this interpretative phenomenology study to determine what information would have helped patients with experiences of metal hypersensitivity make an informed decision about receiving an implantable medical device.
Methods
Study design and participant selection
Using a patient-oriented research approach, 28 the lead author engaged with a patient with lived experience of metal hypersensitivity to ascertain their perspective on the phenomenon. We discussed the findings of our previous studies.8,29 The patient partner noted that the enablers and barriers identified by the healthcare workers about screening for hypersensitivity reaction to metal 8 did not consider the issue of informed consent. This discussion led to the overarching research question: “Among patients with metal hypersensitivity, what information would have helped them make an informed decision about metal implants?” We used interpretative phenomenology because it allows us to use the informants’ experiences as well as integrating our own experiences as healthcare providers to patients requiring surgery and those with hypersensitivity reactions. 30
Eligible participants had to be 18 years or older, have implants with metal composition, and have received the diagnosis of metal hypersensitivity. Patients in the process of being investigated for metal hypersensitivity were excluded. The study was approved by the University of Victoria Ethics Board (#19-0473). Following ethics approval, we used the snowball sampling to locate the first two participants. We then posted a research notice on a province-wide platform (BCReach) and on social media (e.g. Facebook). A laboratory diagnostic company specializing in metal testing became aware of our study and circulated the research notice.
Following the posting, we received emails from one person interested in participating, but who voiced the following concern: Considering you’re reaching out for volunteers in a community populated by many who are being refused care, this may not be the optimum method to gain trust. Setting up a response receipt would have been one painless option to show forethought.
Other emails received during this timeframe inquired about who was conducting and funding the research. To address these issues, we set up an automatic reply, and the email account was monitored daily. The first author also sent comprehensive emails to interested individuals to introduce herself and explain her interest in this area. After completing interviews with participants recruited from this social media posting, participants referred others to the study. It appeared we had gained the trust of the community.
Data collection
All participants gave verbal consent, and the interviews were conducted and transcribed using commercial software. The interviews occurred between December 2020 and May 2021 and lasted between 1 and over 2 h. The first author took field notes during the interviews, and emerging areas of concerns (e.g. psychological and social burden) were explored in greater detail in subsequent interviews. The transcripts were verified by a member of the research team before analysis. Demographic data were gathered to ascertain the social positioning of the participants. Informed by interpretative phenomenology, the interview guide had open-ended, neutral questions that were used flexibly to allow issues to be raised that were not previously thought of by the research team.
Data analysis
Using interpretative phenomenology analysis, each transcript was read by three members of the research team (DD, JC, and JAL). Detailed analysis was performed one transcript at a time to code the transcript, while rereading to allow for new ideas and insights to be generated. The second stage involved identifying and defining the emerging themes. These themes were discussed and then the steps were repeated with the next transcript. Any new themes were clarified in previous transcripts; thus, data analysis was iterative and consisted of returning to each stage, modifying themes as needed. The findings were discussed with the patient partner and several of the participants to ensure authenticity and trustworthiness.
Results
Of the 21 people who responded, 16 participants were enrolled. Nine (56%) were from the USA. An interpreter was used for one participant for whom English is a second language. Most of the participants were biological women (14/88%) and racially identified as white (12/75%). All were between the ages of 34 and 65 (Table 1). Ten indicated that they had a university education, and they included an artist/singer, lawyer, accountant, journalist, and surgeon. The experience of metal hypersensitivity was an economic burden on seven of the participants, who lost employment. One participant said, “I lost my career. We were losing our house.” Another noted, “I was very ill, couldn’t work and I’m self-employed, so it was a bad time.”
Demographic characteristics of participants.
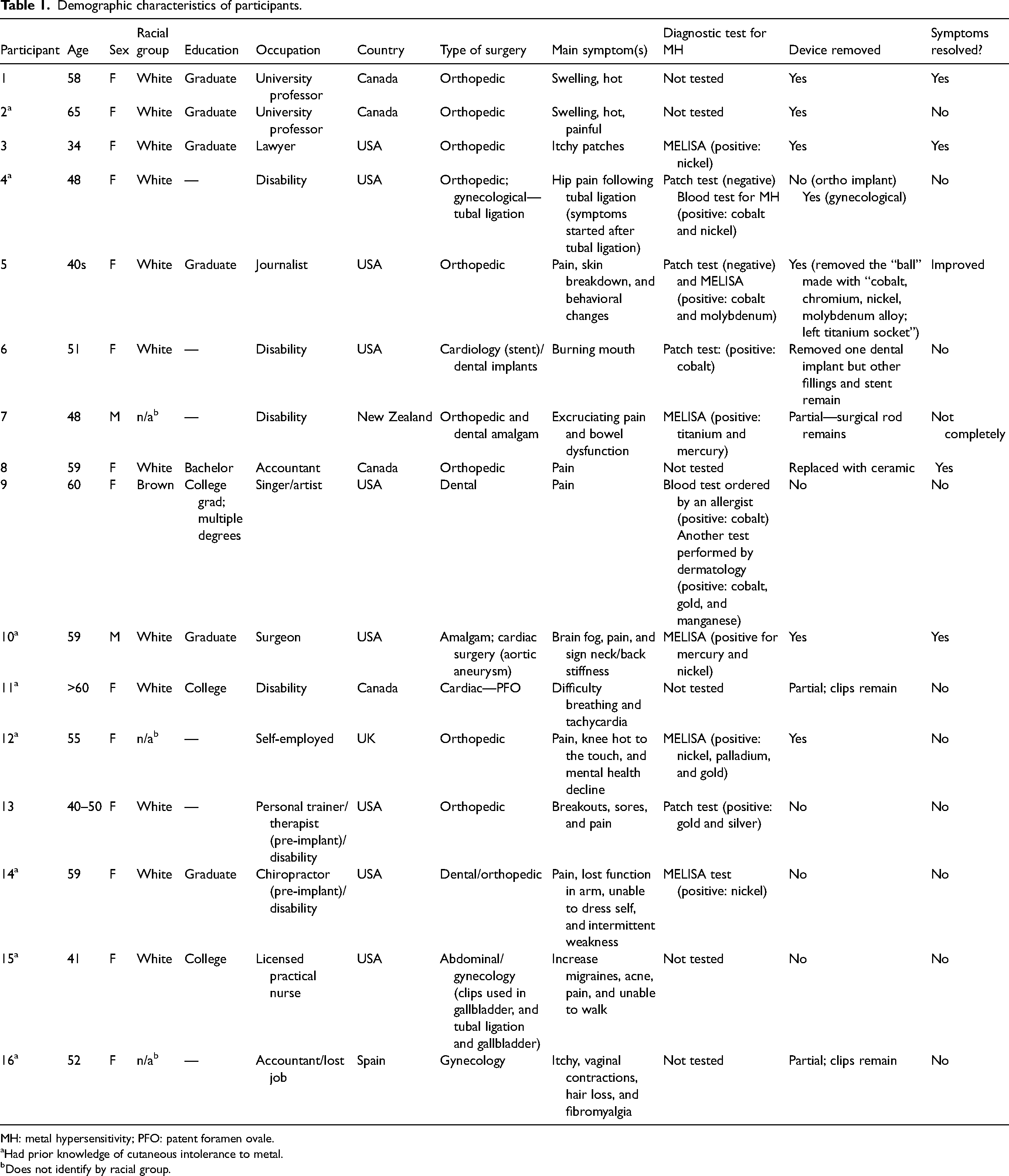
MH: metal hypersensitivity; PFO: patent foramen ovale.
Had prior knowledge of cutaneous intolerance to metal.
Does not identify by racial group.
Theme 1: not informed
All the participants indicated they did not receive any information about metal hypersensitivity during the consenting process. One expressed his concern about metal hypersensitivity to the mid-level provider (physician assistant), who was unaware of it. Another participant said they initiated the discussion during the consenting process based on their own research, which led to pre-operative testing that showed sensitivity to nickel. Overall, participants believe they were not properly informed of the risks associated with the metal composition of the implant noting the single most important thing would be to: “just [tell] people this can happen…[even] in rare occasions. This is what it looks like or could look like this. If you have these reactions, here's what you can do.” In addition, participants with dental implants noted it is important to include in the consenting process an explanation of galvanism and how it can potentially affect long-term health.
Theme 2: suspecting but not knowing
Participants described a broad spectrum of physical and psychological symptoms at the onset of metal hypersensitivity. They described “swelling, redness and pain,” “very itchy spots [that] seemed like they would get worse every year,” “my fingers … were blistering and I was losing all the skin,” (pompholyx eczema) and cessation of body hair growth. The locations of the cutaneous symptoms were diffuse, including the face, back, neck, hands, mouth, and arms. Overall, “horrendous” pain was the chief complaint, which appeared to be progressive and “nothing [prescribed analgesic] touched the pain.” Participants described “the pain [as] horrific … far worse than anything I’ve ever experienced before in my entire life, and it never improved.” In some cases, the pain was severe enough to affect activities of daily living: “I was barely able to walk,” “carry bags of grocery,” “I had to change my car from a manual transmission to an automatic transmission,” “I literally lost the ability to dress myself. I had to move in with my sister.” The participants kept a symptom log to determine patterns and took this information with them when they sought assistance from healthcare providers.
Participants also described experiencing psychological issues during their ordeal. Three participants experienced “severe brain fog” that impaired their ability to concentrate. One participant stated, “I was left like this for two years, my mental health suffered enormously. I was very badly clinically depressed, but the depression did not respond to any medication.” Another said, “After feeling like I couldn’t cope anymore with the crazy, seemingly unrelated health issues that started to majorly and negatively impact my passion, I considered suicide. This is not living. This is a death sentence not only for me, but for my family [who] have to watch me deteriorate day after day, month after month.”
When the symptoms initially surfaced, providers informed some participants, “well, it's part of the healing,” and were dismissive (n = 16), which led to patient mistrust because “I feel there's no one I could be honest with or talk to about it because I feel like they’re all just going to prescribe me an antidepressant and shove me out the door.” Another said, “It's maddening and it's disheartening to go to doctors that don’t believe this is a problem and they don’t believe their patients. My concerns are dismissed.” Reflecting on the experience, one person said, “I would never have [had] the surgery…I would have stayed sobbing in pain… because at least my brain worked.” Ultimately, participants felt their “surgeon[s] had essentially washed [their] hands of [them],” especially because in most cases, they had standard follow-up care before symptom onset. One participant presented to the 6-week follow-up with excruciating pain, and another had ongoing pain immediately after surgical clips were inserted during tubal ligation.
Despite the experience, some participants express deep admiration for the primary surgeon. One said, “I love the surgeon who put it in. He's great. And he thought he was doing absolutely the right thing. I mean, I had to have it done because I had such bad scoliosis that it can start to impact your organs. And I mean, other than the metal allergy, the surgery was a great success.” Others were unhappy with their healthcare providers (e.g. surgeons and general practitioners) and their lack of academic curiosity to investigate and diagnose their problem. One said, “getting [the surgeon] to agree to the surgery [to remove the implant] was another whole incredibly bad experience. And then to think about I had to go [through] the trauma of going under surgery [again] with him after we had had this kind of altercation was very, very disturbing.” Though some had poor interactions with their general practitioner, others commented “my GP … was pretty good in listening, he didn’t write me off.”
Theme 3: self-advocacy
All participants were asked to talk about how they came to know they had metal hypersensitivity. Eight noted they were not aware of any sensitivity to metal until after the device was implanted. Despite some participants having prior history of metal intolerance, they did not disclose this information, mainly because they were not prompted by their healthcare provider. Interestingly, one participant who provided their healthcare team with a list of their allergies, including nickel, said this was disregarded, and a device containing nickel was implanted. Because of this, the participant believes having a postoperative list of devices at discharge including closing material would be helpful. Other participants wished that “somebody believed [them] sooner” or “explained to [them] why [they] couldn’t have the device taken out sooner.” When prompted to discuss this feeling of “not being believed,” one participant noted, “I was obviously taking photos of my arm … so I was obviously feeling like I want to show you what I’m talking about, because sometimes by the time I got to see someone or it would be like I wanted to say, look this is bad, this is not right.”
Arriving at the actual diagnosis of metal hypersensitivity was a long process for all the participants, with one person noting, “I had been trying to figure out for a very long time why this was happening. No doctor ever was like hey this is what's wrong with you.” Another stated, “I decided I would go and prove it unequivocally, that they would not believe that I was allergic to Nickel systemically. And I wanted to prove that I was.” Ultimately, all participants used word of mouth from people with similar symptoms or online resources to “guess” their way to the diagnosis. “I started asking questions and really pushing buttons and asking for my medical records, and every time I’d have a doctor's visit, I would ask for those to be printed out and have them include the doctor nurse notes. And I just started Googling.”
Once they had the information about metal hypersensitivity, some did not consult the surgeon who implanted the device but received the diagnosis of metal hypersensitivity and treatment from a secondary healthcare provider. When asked why they did not approach the surgeon who implanted the device, they commented that they were cleared from a surgical perspective. Some thought that if they had been able to consult with the primary surgeon, they may have been diagnosed sooner.
Another factor that appeared to complicate the diagnosis period is that participants attributed their symptoms to the events occurring immediately before the symptoms. One participant said, “dismissing [the symptoms] as [brought on by] physiotherapy probably wasn’t good, I probably should have contacted the surgeon at that stage.” Regardless of their assumptions, the healthcare providers from whom they sought care (e.g. physiotherapist, emergency room provider, walk-in clinics, rheumatologist, and general practitioners) were unable to “connect the dots” between the presenting symptoms and the implant. Participants believe there is a “huge gap” in the health system where “there is just no curiosity [about the patient] just looking down and writing and not even … scheduling follow ups.” Their general impression was that they were ignored because “the medical profession is supposed to be the expert.” One participant noted that “the medical profession seemed to dismiss [the] fact that I know my body better [now] than I ever will. I’m the one living in it – they’re not.” Even with the information about metal hypersensitivity, many of the participants described having problems trying to determine the metal composition of the device in their body.
Theme 4: validation
Only one participant underwent testing preoperatively, and nine underwent testing at the time of explantation. Those who were tested at the time of diagnosis and device retrieval underwent in vitro testing with MELISA laboratories (Germany). They described difficulty in obtaining in vitro testing, mainly because health insurances do not cover the cost. Also, the logistics of drawing blood and sending it to Germany are challenging. All participants also encountered difficulty finding surgeons who “believe” hypersensitivity can occur and are willing to perform the surgery to retrieve the device. “Some doctors believe it and some doctors don’t. The ones you think would, don’t. And the ones you think don’t, do.” One participant noted, “I was very thankful to find him because from being in those metal groups … a lot of people say they have a hard time finding a surgeon who will believe them and take it out.” Interestingly, it appears the surgeons who were willing to remove the device had prior tacit knowledge of metal hypersensitivity: one participant noted, “He said he had seen this before with stainless steel, he said it happens a lot less often with titanium.”
Theme 5: moving forward
All participants expressed a strong sense of duty to educate healthcare workers and the public about this issue. In terms of systems improvement and suggestions, they highlight a need for information and disclosure about the risk of metal hypersensitivity and comprehensive discussion about alternatives to metal prior to the surgery. They suggested having an option for testing, although there was no consensus among participants about whether testing should be done before or after surgery. The majority advocated for pre-operative testing; they recognized that simply asking about a previous history of metal sensitivity was inadequate because many did not know if they had metal sensitivity. For post-implant care, participants suggested that the signs and symptoms of metal hypersensitivity be included in discharge material, and that there is follow-up care with a healthcare provider who is knowledgeable about metal hypersensitivity and can provide in-depth assessment about symptoms. They also recommended having an implant card that details the metal composition of the device and closing devices so the information is readily available when issues arise. They advocated that more resources are allocated to research to improve our understanding of the immune responses to implantable medical devices. Ultimately, the participants asked that healthcare providers be more understanding, accountable, and relational. They also highlight a need to address the power imbalance between physicians and patients so that patients can feel they are being heard.
Discussion
Based on our scoping review, 29 this is the first qualitative study to explore the experiences of patients with hypersensitivity reactions following implantable medical devices. The findings highlight a major theme of “unknowing” on the parts of the patients and the healthcare system. Consistent with the literature, participants described nonspecific symptoms that can be associated with many issues that are considered normal findings in the acute postoperative period. The psychological symptoms described are not commonly reported, but Green et al. 31 demonstrated that cobalt and chromium metallosis is associated with neuropsychiatric and depressive symptoms. Despite their challenges, participants demonstrated self-advocacy by tracking and researching their symptoms to arrive at a diagnosis of metal hypersensitivity. In vitro testing was a source of validation for some participants, and device removal led to a beginning of a new norm: some participants experienced complete resolution of symptoms and others had ongoing issues. These findings highlight issues with how people experience the healthcare system and its management of device-related adverse effects.
Our main research question pertains to consenting and what information needs to be disclosed to assure informed consent. The findings demonstrate that information about hypersensitivity reactions to metal is not routinely discussed pre-operatively during the consenting process. Informed consent is grounded in the principle of respect for autonomy and codified into law. It also reflects common morality because bioethics guidelines of “particular morality” for healthcare providers and alike disregard the ethical concerns of importance to society. 32 Moreover, studies show factors associated with medical malpractice litigations include poor doctor–patient relationships resulting in devaluing patient's views, delivering information poorly, and failing to be attentive to the patient's perspective.33–36 Our findings suggest participants prefer the reasonable person's standard for information disclosure during the consenting process. This allows for discussion about the risk of metal hypersensitivity and for the patient to clarify their values and preferences.
It is unclear why the risk of metal hypersensitivity was not disclosed during the consenting process to any of the 16 participants, but it might be due to the controversy in the literature about whether clinicians are convinced about the evidence in this area. 8 Hallock et al. 37 demonstrated that, despite knowing about metal hypersensitivity, few orthopedic surgeons screened for it. 38 In the literature, metal hypersensitivity is noted as a “rare” effect, 39 although the epidemiological data in this area is incomplete because the condition is underreported and difficult to diagnose. 40 Nonetheless, prevalence rates as high as 78% have been reported in people with failed or poorly functioning joints. 41 A recent prospective study of 207 patients undergoing titanium plate cranioplasty found 82 (40%) were sensitive to material in the device pre-operatively. 42 Post-operatively, 11 (5.3%) patients experience delayed scalp erosion and had reoperation. Findings show that patients with sensitivity to one or more metals in the device pre-operatively had 17 times higher implant failure rates compared to those without sensitivity, and those with sensitivity to four or five of the device components had the highest risk of implant failure rates—odds ratio of 18 and 43, respectively. Overall, higher rates of metal sensitization have been observed post-operatively than pre-operatively—among people with complications compared to those without complications.43–46 Albeit fewer in number, there are reports of people with a known metal allergy and well-functioning implants. 47 These findings suggest the notation of this condition as “rare” is misleading.
The utility of pre-operative screening of patients undergoing device implantation remains highly debated in the literature, because there is mixed evidence related to a clinician's ability to accurately identify people who might be at risk.48,49 Of the available recommendations about screening patients,38,50–54 the majority noted that it is reasonable to do the following before surgery to predict and prevent dangerous complications of metal hypersensitivity: (a) perform a comprehensive history with diagnostic in vivo (cutaneous testing) or in vitro testing (lymphocyte transformation test where available) for those with a positive metal allergy history; (b) discuss with the patient the potential benefits and risks associated with hypoallergenic components; (c) discuss the potential outcomes of using hypoallergenic components; and (d) use an alternate device not containing the known allergen in patients adamant that hypoallergenic components are used or in patients with a positive metal allergy test. Post-operative recommendations include the following: (a) monitor inflammatory mediators in symptomatic patients and (b) consider using lymphocyte transformation studies in patients deemed to have metal allergy once the large differentials are ruled out.38,55–57 These recommendations align with those offered by the participants in this study, demonstrating an agreement between people with lived experiences and clinical experts.
Most of the participants in our study (88%) are biological women. This may be related to a higher willingness to or interest in participating in research on the part of women (e.g. gender base). Also, prior studies suggest that females have a higher risk of hypersensitivity because of exposure to jewelry.58–60 However, biological sex might also be a risk factor. Physiological evidence suggests women experience pro-inflammatory Th1 and anti-inflammatory Th2 cytokine states most noticeably during pregnancy.61–63 There is also evidence suggesting that a male fetus shifts the maternal immune system more into pro-inflammatory Th1 cytokine status than a female fetus. 64 Metal hypersensitivity involves a pro-inflammatory status. It is unclear what role pro- and anti-inflammatory states play in the susceptibility of women to metal hypersensitivity.
Lastly, all participants in our study racially identified as white and had university degrees, suggesting there might be a hidden disparity with regard to hypersensitivity reaction to metals in implants. Many of the participants self-diagnosed their condition through words of mouth while using their own financial and social resources to “prove” their diagnosis. Previous studies have not routinely reported racial grouping; thus, it is difficult to ascertain whether this is a new finding. However, our study does suggest racial minorities and those with lower education and socioeconomic status may be experiencing this issue but might be unable to obtain diagnosis and treatment due to their social location or because of general mistrust of the health system.
Limitations
Study limitations include selection bias and small sample size. We used the snowballing sampling and social media to recruit the sample. It is possible that studies using different sampling methods might yield different findings. For a qualitative study, the sample size was adequate, and data analysis revealed data saturation about the main research question about informed consent. However, saturation was not achieved in the lived experiences. Three groups emerged. The first comprised participants who had the device removed and had complete resolution of symptoms. Their quality of life was restored to pre-implant status. These individuals have higher overall satisfaction with both the surgery and management of metal hypersensitivity. The second group had the device removed with resolution of their initial symptoms but continues to experience nonspecific “autoimmune” concerns and is investigating reasons for it. Many studies indicate improved or complete resolution of clinical symptoms after removal of the device,65–68 but there is dearth of evidence on what proportion of patients report long-term immunological conditions after device explant. Further studies are needed to quantify the incidence of this and possible underlying mechanisms. The last group includes participants who still have the device in situ with no immediate plans for removal.
Conclusions
This qualitative study found that, during the consenting process, participants did not receive any information about the adverse effects of metals on the immune system. To address metal hypersensitivity, patients with implantable medical devices who have symptoms should be assessed post-operatively to rule out the large differential diagnosis associated with failed implants. Communication with patients is key to successful patient-centered care; thus, in patients with a history of metal hypersensitivity, the healthcare provider should have a long and detailed discussion pre-operatively to inform them about the risks and potential benefits of proceeding with surgery using hypoallergenic or regular components. Overall, the healthcare system needs to implement better patient selection processes that are analogous to what is currently done for organ transplantation and blood transfusion to assure patient safety. Future studies are needed to inform better patient selection for device implantation.
Footnotes
Acknowledgments
We wish to express our deepest gratitude to all the participants who entrusted us with their stories.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by the BC SUPPORT Unit and University of Victoria.
Ethics approval
This study was approved by the University of Victoria research ethics board.
