Abstract
Demodex blepharitis is a chronic, progressive condition characterized by inflammation, ocular irritation, and erythema due to an overgrowth of mites in hair follicles and oil glands of the eyelids. AZR-MD-001(AZR) is a selenium sulfide-containing ophthalmic ointment that is a potential treatment option for Demodex blepharitis. We wished to complete a case study to identify the potential of this agent to treat Demodex blepharitis. A patient with chronic Demodex blepharitis applied AZR 0.5% to the right lower eyelid every other day at bedtime for 6 weeks, discontinued treatment for 6 weeks, then treated the left lower eyelid with vehicle only for 6 weeks, followed by use of AZR 0.5% for both lower eyelids twice a week for 6 weeks. Eyelashes were removed and pooled for evidence of mites and collarettes before and after the first treatment, and eyes were inspected for signs of blepharitis before and after each treatment period. After treatment with AZR 0.5%, resolution of blepharitis occurred in the treated eye, with no evidence of Demodex mites. The untreated eye showed no difference from baseline. After discontinuing for 6 weeks, the signs of blepharitis returned, similar to baseline. After 6 weeks of vehicle application to the left eye, no changes in signs were observed. After treatment with AZR 0.5% for 6 weeks to both eyes, complete resolution of blepharitis occurred in both eyes. No adverse events were reported. Treatment with AZR 0.5% was efficacious in eradicating Demodex blepharitis, which was not evidenced when using vehicle or a lack of treatment. This demonstrated a potential new use for AZR to treat Demodex blepharitis. Further studies are required to establish the safety and efficacy of this product.
Plain language summary
Blepharitis is a chronic condition with symptoms that include swelling and itching of the eyelids, and irritation of the eyes, and is commonly caused by an overgrowth of the Demodex mite on eyelids. Products with selenium sulfide have been shown to treat blepharitis, but these products have been difficult for patients to tolerate due to their other ingredients. AZR-MD-001 is an ointment that contains selenium sulfide that is applied to the margin of the eyelids and has been shown to relieve itching of the eyes in other studies. This case report describes the use of AZR-MD-001 in one eye of a patient who had an ongoing history of Demodex blepharitis over a period of several weeks, while the other eye was only treated with the carrier of the drug without the active ingredient (called the vehicle) to act as a control. The patient showed improvement in their blepharitis in the treated eye, with no change in the control eye. This suggests that Demodex blepharitis could be a potential use for this ointment. Further studies are needed to investigate the use of AZR-MD-001 in the treatment of patients with this condition.
Introduction
Blepharitis is a chronic, progressive condition characterized by inflammation, ocular irritation, and erythema, and can be caused by overgrowth of mites living on the skin and eyelashes. Sixty-eight percent of patients suffering from blepharitis have an abnormally high density of Demodex mites (ocular demodicosis) as a causal explanation. These patients present with symptoms of itching (28%), eyelid redness (21%), and watery eyes (15%), 1 along with collarettes, and signs of meibomian gland dysfunction (MGD). 2 Demodex mites contribute to blepharitis inflammation through mechanical damage as they burrow and lay eggs, along with chemical irritation from their digestive enzymes and the waste products released upon mite death. 3
Studies have also associated the density of Demodex mites with the presence of chronic blepharitis. 4 As the mites feed on eyelid skin and hair follicle cells and proceed through their life cycle, epithelial cells, waste, and eggs are deposited at the base of the lashes. Cylindrical dandruff (collarettes) forms around eyelashes, consisting mostly of keratins and lipids, and infested follicles show distension and epithelial hyperplasia with an increase in keratinization adjacent to the claws of the mite. 5 Thus, improving the lid environment with an agent that can remove keratinized material, treat ocular symptoms such as itch and/or redness, control the proliferation of Demodex mites, and only treat the affected area of the lid would represent characteristics of an ideal treatment for Demodex blepharitis.
Selenium disulfide and demodex
The sebaceous glands in the skin and the meibomian glands of the eyelid have similar anatomy and physiology, including embryonic development. 6 In the 1950s and early 1960s, the successful use of selenium medicated shampoos for the treatment of seborrheic dermatitis prompted several investigations of different formulations containing selenium sulfide as the active ingredient to be evaluated as a potential treatment for seborrheic blepharitis.7–11 However, these shampoos contained other ingredients, such as sodium laurel sulfate, that were irritating to the eye and could cause damage to the cornea. These publications then led to the testing of an ointment formulation containing selenium disulfide and a corticosteroid as a potential therapy for Demodex blepharitis. 1 In this study by Post and Juhlin, 14 patients with Demodex blepharitis were treated daily with an ointment containing selenium sulfide and corticosteroid over the eyelid margins for 30 min, followed by washing the ointment off the lids to avoid adverse events. After 14 days, the investigators reported complete eradication of the mites, with about 71% of participants showing “clinical cure” in their blepharitis. It is not entirely clear why this form of treatment was not pursued, but it is likely that concern over adverse events (AEs) and lack of commercial interest may have played a role.
Demodex mites have been recognized as an important cause of blepharitis and in recent years gained awareness by patients and the medical community following the introduction of XDEMVY® (lotilaner ophthalmic solution, 0.25%; Tarsus Pharmaceuticals, Irvine, CA, USA) which is used in the form of ophthalmic drops, two times a day for a period of 6 weeks to treat Demodex blepharitis. Lotilaner is an insecticide that kills Demodex mites by selectively inhibiting the GABA-Cl channels. 12 Lotilaner was shown to show collarette cure in Demodex blepharitis in 56% of participants and mite eradication in 51.8% of participants, with erythema cure in 31.1% of participants, leaving a large percentage of patients without full cure. 13
Azura Ophthalmics is developing AZR-MD-001 (AZR) 0.5% ophthalmic ointment as a novel treatment of MGD. The ointment contains selenium sulfide as the active ingredient. Selenium sulfide is a known keratostatic and keratolytic compound that has also demonstrated the ability to treat ocular itch, a known symptom of Demodex blepharitis, in phase II clinical trials in patients with MGD. 14 This previous study, in conjunction with the mechanisms of selenium sulfide, suggests that AZR would be a potential candidate for the treatment of Demodex blepharitis. The authors are employees of Azura Ophthalmics Ltd. and wished to identify if AZR had an effect on Demodex blepharitis to determine a potential path for future research. Here we report a case study of the use of AZR 0.5% in a patient with chronic Demodex blepharitis.
Methods
The patient was a 58-year-old male with a five-year history of blepharitis, who was not using any other ophthalmic medications to treat his condition, and gave consent to participate directly to the physician, as well as signing a consent form. Blepharitis due to Demodex was confirmed through a clinical evaluation and slit lamp biomicroscopy prior to treatment initiation via evidence of collarettes, confirmation of the presence of Demodex mites under a microscope in pulled eyelashes, eyelid edema, and erratic eyelash growth. The participant applied AZR 0.5% (Azura Ophthalmics, Ltd, Tel Aviv, Israel) to the right lower eyelid every other day at bedtime for 6 weeks, at which time three eyelashes were removed from the study eye, pooled, and inspected for evidence of Demodex mites. Inspection of lashes was completed by placing lashes on a glass slide, covering them with an artificial tear as an emulsifier, and placing a coverslip under the microscope so the mites could be counted. The patient was evaluated using slit lamp biomicroscopy for additional evidence of collarettes and lid condition. During this six-week treatment period, the left eye was used as a within-subject control and was left untreated. Pooling of lashes and a microscopic investigation were completed for both eyes after this period. The patient then discontinued treatment for 6 weeks, followed by treatment of the left eye lower eyelid only with vehicle every other day for 6 weeks at bedtime. Then treatment with AZR 0.5% was initiated to both lower eyelids twice a week for 6 weeks at bedtime. Treatment was then ceased, and the patient received a follow-up clinical exam 5 months after treatment was completed (see Figure 1). The investigator was not masked for these procedures.

Patient flow.
The patient received a clinical evaluation for signs of blepharitis in both eyes, and photos were taken before treatment initiation, as well as after each treatment or discontinuation period. Treatment (AZR 0.5% or vehicle) was applied by the patient at home using a washed finger to administer a thin layer over the lower eyelid of the indicated eye at the base of the eyelashes, followed by blinking to distribute the medication to both eyelids.
Results
Upon examination at baseline during the first assessment, significant collarettes, swollen lids at the base of the eyelash, and erratic eyelash growth in both eyes were observed prior to initiating treatment, confirming the diagnosis of Demodex blepharitis (Figure 2(a) right eye, and Figure 2(b) left eye). Three eyelashes were pooled from each eye and examined, which confirmed Demodex mite infestation.

Treatment progression of the right eye before and after treatment of AZR 0.5% followed by discontinuation, and no treatment in the left eye. (a) Right eye before treatment. (b) Left eye at baseline. (c) Right eye after 6 weeks of treatment with AZR 0.5%. (d) Left eye after no treatment for 6 weeks. (e) Right eye after seven weeks post-discontinuation of AZR 0.5%.
After 6 weeks of treatment with AZR 0.5% on the right eyelid, there was a complete cure of the eye with disappearance of collarettes, eyelashes appeared shiny with no erratic growth, and the lids showed no evidence of edema (Figure 2(c)). Three eyelashes were pulled from the right eyelid and examined under a microscope, with no Demodex mites observed. The untreated (left) eye showed no change from baseline with continued evidence of Demodex blepharitis (Figure 2(d)). After the patient discontinued the therapy in the right eyelid, the Demodex infestation resumed (Figure 2(e)), but the collarettes observed appeared to be less numerous than at baseline. No difference was observed in the vehicle-treated (left) eye.
After treatment with the vehicle only (without selenium sulfide) to the left lower eyelid, no change in collarets or lid appearance was observed between baseline and 6 weeks (Figure 3(a)—before vehicle and Figure 3(b)—after vehicle).

No differences were observed in the left eye before (a) and after (b) 6 weeks of treatment with vehicle. (a) Left eye before treatment with vehicle. (b) Left eye after 6 weeks of treatment with vehicle.
After treatment of both eyes with AZR 0.5% twice a week at bedtime for 6 weeks, complete resolution of collarettes was observed with normal eyelash growth patterns and normal appearance of the lid margin (right eye—Figure 4(a) before treatment, and Figure 4(b) after treatment; Left eye—Figure 4(c) before treatment and 4(d) after treatment).
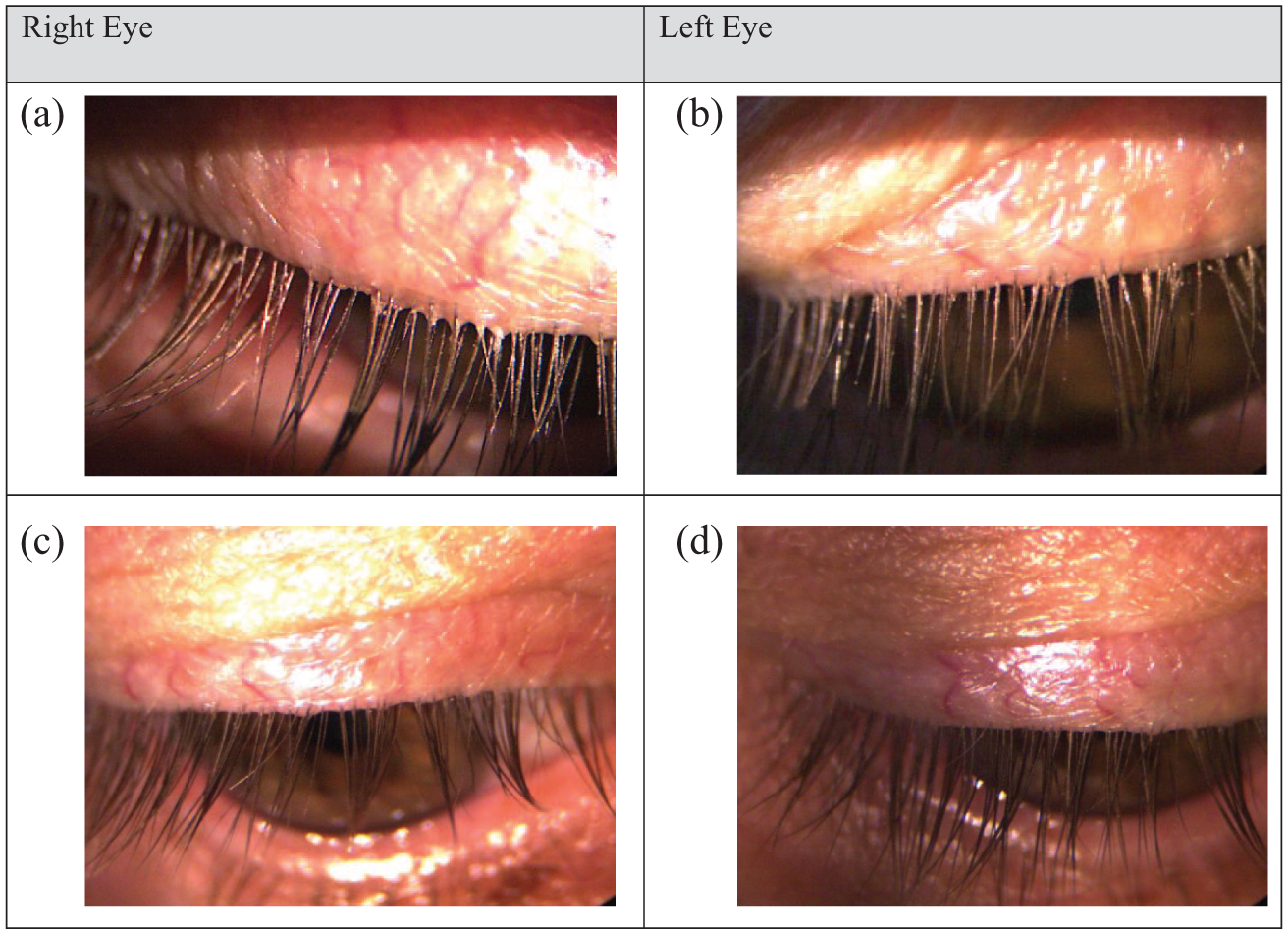
Signs of blepharitis in the right and left eye show complete cure in both eyes after 6 weeks of treatment with AZR 0.5%. (a) Right eye before treatment with AZR 0.5%. (b) Right eye after 6 weeks of treatment with AZR 0.5%. (c) Left eye before treatment with AZR 0.5%. (d) Left eye after 6 weeks of treatment with AZR 0.5%.
Five months following cessation of treatment, there was still a significant improvement compared to baseline (Figure 5(a) right eye and Figure 5(b) left eye) with only two and five collarettes counted on the right and left upper eyelids, respectively.

Evidence of the minimal return of blepharitis signs 5 months after discontinuation of treatment of AZR 0.5%. (a) Right eye 5 months after discontinuation of treatment with AZR 0.5%. (b) Left eye 5 months after discontinuation of treatment with AZR 0.5%.
Throughout all treatment durations, no AEs were observed or reported by the participant for the vehicle or AZR 0.5%.
Discussion
Selenium sulfide is a keratolytic agent that has demonstrated the ability to eradicate Demodex mite infestation 1 and to treat seborrheic blepharitis, some of which is likely associated with Demodex infestation. In laboratory testing, selenium sulfide has been shown to repel Demodex mites at a low concentration of 0.5% and to kill them at higher concentrations of 4%. 15 In the botanic literature, selenium, a trace element found in soil, is known to be an insect repellent. 16 Collarettes, a sign of Demodex blepharitis, are also made of keratin, and therefore, it is expected that their removal should be enhanced by the breakdown of disulfide bonds by selenium sulfide, in addition to removing the mites’ food source. 17 A past study demonstrated selenium sulfide eradicates mites in patients with Demodex blepharitis at a 0.5% concentration; however, it was used in combination with steroids, and was used every day, and was washed off after 30 min. 1 In addition, some of the formulations described in the literature from the 1950s and 1960s contained surfactant agents, which may have caused unwarranted AEs.7–11
The patient in this case report showed consistent eradication of signs of Demodex blepharitis after treatment with AZR 0.5%, and this result was not seen in the untreated eye, during a period of drug discontinuation, or in the vehicle-treated eye. After initial treatment in one eye followed by discontinuation of the treatment, the reappearance of the condition may have occurred due to a reinfestation, possibly originating from the fellow eye that was left untreated, or potentially through migration from other parts of the face or eyebrows.
AZR 0.5% has been developed for the specific use over the lid margin of the eyelids, and the formulation and manner of application have been developed to ensure ocular safety, which had been a concern with previous formulations using selenium sulfide.14,18 This case study corroborates previous data of selenium sulfide products in blepharitis and supports the hypothesis of the use of AZR 0.5% for the treatment of Demodex blepharitis. Future studies are needed to fully understand AZR 0.5% potential use for the treatment of this condition in a larger population.
