Abstract
Rituximab, an anti-CD-20 monoclonal antibody, has garnered increasing interest as a therapeutic option in orbital inflammatory disease (OID). We report our institutional experience with intralesional rituximab injections in two cases of orbital granulomatosis with polyangiitis (GPA), and additionally review the literature surrounding intralesional rituximab in OID. Two patients (a 74-year-old male (Case 1) and a 63-year-old female (Case 2)) with orbital GPA received intralesional rituximab injections at our tertiary institution. Case 1 had evidence of left optic neuropathy in extensive bilateral inferomedial orbital inflammation, while Case 2 had severe refractory and relapsing left orbital inflammation. Both patients received intralesional rituximab injections (Riximyo 10 mg/ml), administered weekly over 8–11 weeks. Rituximab injections were adjunctive to systemic induction therapy, including corticosteroids and rituximab infusion in Case 1, and a course of corticosteroids only in Case 2. Both patients demonstrated a clinico-radiological response to treatment. In conclusion, we present our experience with intralesional rituximab injections, used as an adjunctive treatment modality for orbital GPA, in a case of optic neuropathy and another of severe, refractory, and relapsing disease. Further prospective studies are required to define its utility and the long-term outcomes of intralesional rituximab injections in OID.
Introduction
Rituximab, an anti-CD-20 monoclonal antibody, has garnered increasing interest as a therapeutic option in orbital inflammatory disease (OID). Rituximab infusions have been utilized in the treatment of a range of OID, including nonspecific orbital inflammation (NSOI), orbital granulomatosis with polyangiitis (GPA), and IgG4-related orbital disease (IgG4-ROD).1,2 GPA is a rare autoimmune small vessel anti-neutrophilic cytoplasmic antibody (ANCA)-associated vasculitis (AAV). Orbital involvement occurs in approximately 5%–30.6% of GPA and may manifest as diffuse inflammation contiguous with sinonasal disease, a primary orbital granuloma, dacryoadenitis, or orbital myositis.3 –9 The 2021 American College of Rheumatology (ACR)/Vasculitis Foundation guidelines for the Management of AAV provide key recommendations and a framework for the management of GPA.10 –12 Currently, rituximab is the preferred agent for remission induction in patients with active, severe GPA, and is also the preferred agent for remission maintenance. 10 Medical management of orbital GPA includes systemic corticosteroid therapy in combination with various immunosuppressive agents, such as cyclophosphamide, methotrexate, or azathioprine.10,13 Intralesional rituximab injections have also been reported in the management of orbital disease, including NSOI and orbital lymphoma.14 –20
In this case series, we describe our experience with the use of intralesional rituximab injections in two patients at our tertiary center with orbital GPA. We will also summarize the relevant literature surrounding the role of intralesional rituximab in orbital inflammation.
Methods
Subjects
This was a retrospective case series, including patients diagnosed with GPA (as fulfilling the American College of Rheumatology/European Alliance of Associations for Rheumatology (ACR/EULAR) or International Chapel Hill Consensus Conference (ICHCC) criteria) with orbital inflammation treated with intralesional rituximab. Patients were treated at the Oculoplastics Unit at the Royal Adelaide Hospital (Adelaide, Australia). Data recorded included patient demographics (age at presentation, gender, relevant past medical history, and medications), clinical presentation, serum investigations (white-cell count (WCC), C-reactive protein, and autoimmune markers), magnetic resonance imaging (MRI) features, management course, and clinical outcomes. All research was conducted in accordance with the Declaration of Helsinki, and off-label use of intralesional rituximab was approved by our Institutional Drug and Therapeutic Board. All patients provided written informed consent for publication.
Intralesional rituximab administration
Informed consent was obtained from each patient following discussion surrounding the indication, method, benefits, and risks of intraorbital injections.21,22 Intralesional rituximab was administered under sterile technique, without procedural imaging guidance, by an experienced Oculoplastic surgeon. The site of intralesional injection was determined by the location of orbital involvement from prior orbital MRI (e.g., in Case 1, intra-orbital injections directed to the inferolateral and inferomedial floor of the orbit, correlating to the location of significant inflammation). Visual acuity and intraocular pressure were assessed pre- and post-procedure. First, Tetracaine 1%W/V was instilled into the eye as a topical anesthetic, followed by Chlorhexidine preparation. Rituximab (Riximyo 10 mg/ml) was injected into the affected orbit using a 30-gauge needle and directed toward the involved orbital space. Noting prior literature on the utility of intralesional rituximab in NSOI, a regimen of weekly injections was implemented to guide ongoing therapy and response.14 –16 In each case, the initial duration of treatment was set to a total of 12 weeks; however, it was ultimately guided by the clinical response (or lack of) and disease activity, and an informed discussion with the patient regarding ongoing symptoms and course of management.
Results
Case 1
A 74-year-old man presented to our tertiary referral center with left eye proptosis and diplopia. GPA had been diagnosed 5 years prior to a positive ANCA and renal biopsy demonstrating pauci-immune glomerulonephritis with fibrous and fibro-cellular crescents. He had preexisting sinonasal involvement managed by a rheumatologist and an otolaryngologist. Maintenance therapy consisted of azathioprine 100 mg twice daily and mycophenolate 1 g twice daily. Other past medical history included ischemic heart disease, hypertension, and prostate cancer treated with radiotherapy 1 year prior.
At presentation, he described increasing left eye proptosis over the preceding weeks, becoming more notable in the 4 days prior. There was associated epiphora and diplopia on left inferior gaze. There were no new sinonasal or upper aerodigestive tract symptoms.
On examination, right and left visual acuity was 6/7.5 and 6/12, respectively; a left relative afferent pupillary defect and intraocular pressures were 19 mmHg bilaterally. There was a long-standing saddle nose defect. Orbital examination revealed proptosis of 2 mm, global limitation of left extraocular movements predominantly in elevation and abduction.
MRI orbital scans demonstrated bilateral infiltrative contrast-enhancing soft-tissue masses in the inferomedial orbit, more extensive on the left side. The lesion encased the inferior and medial rectus, and in the left orbit, there was posterior extension toward the apex into the intraconal space with secondary enhancement of the optic nerve sheath (Figure 1). There was associated chronic mucosal thickening in the paranasal sinuses with evidence of prior sinus surgery.
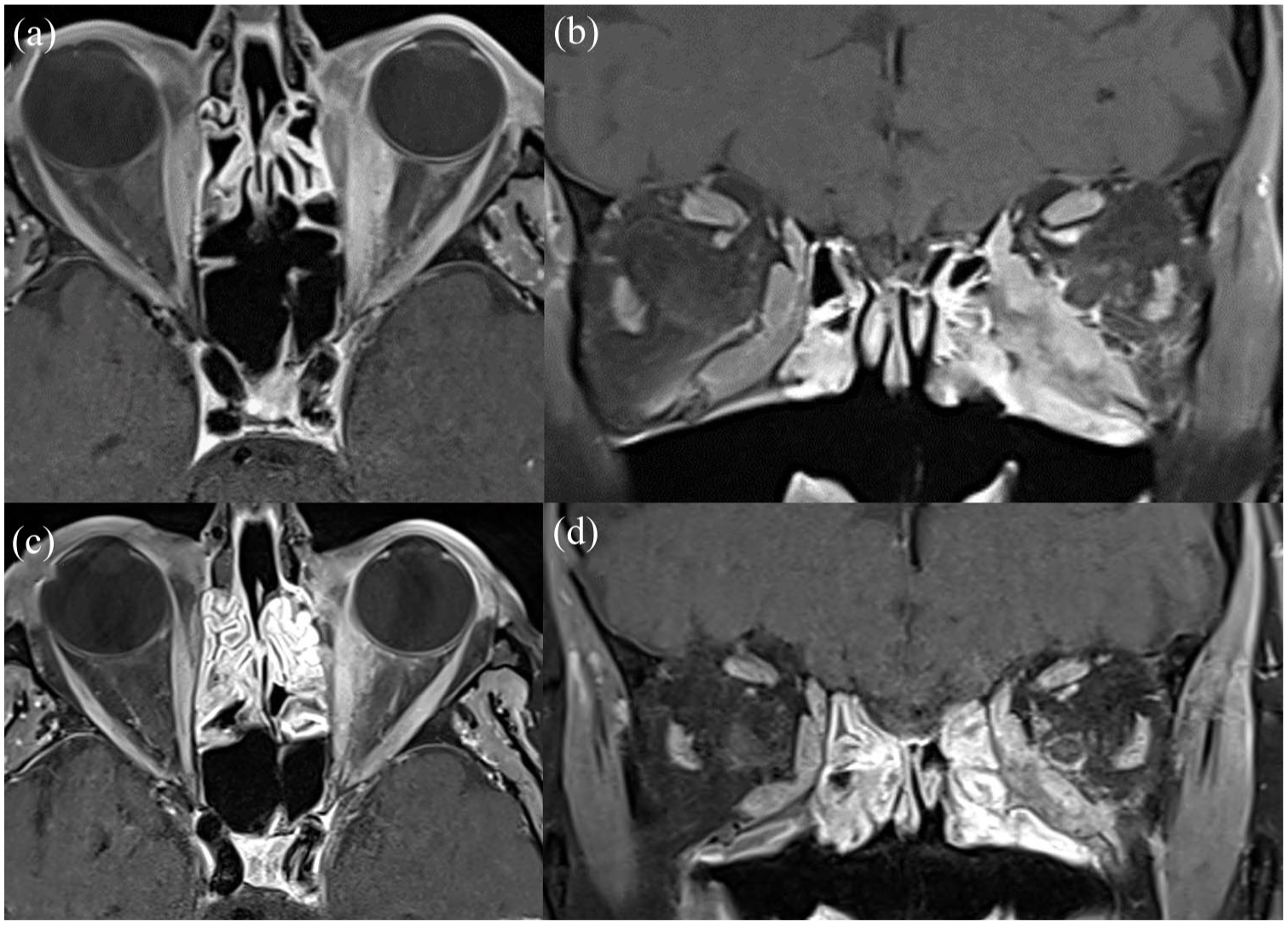
MRI orbital scans of bilateral orbital GPA (Case 1). (a and b) Pretreatment images demonstrate the bilaterally infiltrative contrast-enhancing soft-tissue mass in the inferomedial orbit with extension into the left intraconal space on axial and coronal FS CE T1, respectively. There is secondary enhancement of the left optic nerve sheath. The lesion encases the inferior and medial rectus, with posterior extension toward the left orbital apex. (c and d) Post-treatment images demonstrate reduced contrast enhancement in the aforementioned regions on axial and coronal Fat-suppressed contrat-enhanced T1-weighted imaging (FS CE T1), respectively.
Due to the extensive bilateral orbital disease and the presence of a left optic neuropathy, he was admitted under the Rheumatology service and commenced on 2 days of 500 mg IV methylprednisolone and IV rituximab as induction therapy. Additionally, an intra-orbital injection of rituximab (Riximyo) 10 mg/ml was administered to the left orbit, with 0.5 ml equally delivered to the inferomedial orbit and inferolateral orbit. He was tapered to oral prednisolone (1 mg/kg) starting at 70 mg daily. He was continued on azathioprine 100 mg twice daily and mycophenolate 1 g twice daily and underwent an additional two cycles of rituximab. The patient tolerated his first intralesional rituximab injection and proceeded to have weekly intralesional rituximab injections with a total of 11 injections over 3 months. There was resolution of proptosis, and steady improvement in ocular motility, though there was a residual RAPD. There was also radiological improvement in the size of the bilateral inferomedial orbital masses, with persistent enhancement of the left optic nerve sheath on MRI. There was an improvement in left VA to 6/6 and left visual fields. While the initial treatment plan was to treat for a total of 12 weekly injections, due to a plateau in clinical improvement in the final 3 weeks of treatment, a shared decision was made to cease treatment and monitor. Ophthalmic examination was stable with quiescent disease on prednisolone 5 mg daily and an additional treatment cycle of IV rituximab, at 3 months following his last intralesional rituximab injection.
Case 2
A 63-year-old female with a background of GPA with recurrent orbital and laryngeal involvement received intraorbital rituximab injections at our tertiary institution.
For the past 20 years, she had experienced recurrent orbital inflammation, primarily manifesting as left dacryoadenitis and diffuse orbital inflammation. These recurrent episodes were treated with tapering courses of oral prednisolone and intraorbital steroid injections. She had also previously received treatment with methotrexate, four cycles of rituximab (MabThera), and tracheal steroid injections for management of laryngeal stenosis. Of note, she had also experienced flares of her left-sided orbital inflammation following her second and fourth COVID-19 vaccinations (Comirnaty, Pfizer-BioNTech). Despite a previously elevated pANCA level, a lacrimal gland biopsy and laryngeal tissue biopsy failed to demonstrate histopathological features suggestive of an underlying specific vasculitic etiology. However, a repeat orbital biopsy for recurrent orbital inflammation 3 months prior to her latest presentation demonstrated features consistent with GPA, including granulomatous inflammation composed of lympho-histiocytic inflammatory cells admixed with eosinophils, with small/medium size vasculitis (Supplemental Material).
On examination, right and left visual acuity was 6/4.5 and 6/6, respectively, with normal intraocular pressures. There was 5 mm of left proptosis with upper-lid fullness, and global limitation of extraocular motility, primarily of abduction. There was no optic neuropathy. The right eye was examined normally. MRI demonstrated diffuse contrast enhancement and high T2 signal of the extra- and intraconal orbital fat with contrast enhancement and enlargement of the extraocular muscles (EOM) (Figure 2).

MRI orbital scans of left-sided diffuse orbital inflammation in orbital GPA (Case 2). (a and b) Pretreatment images demonstrate diffuse left-sided contrast enhancement of the extra- and intraconal orbital fat, with enlargement of the EOMs on axial FS CE T1 and coronal FS T2, respectively. (c and d) Post-treatment images demonstrate reduced contrast enhancement in the medial extraconal fat on axial FS CE T1, and reduced T2 signal within the intraconal and lateral extraconal spaces on coronal FS T2.
Due to concerns of progressive motility restriction and proptosis, a course of immunosuppressive therapy was recommended. Both the patient and her treating rheumatologist were reluctant to commence systemic rituximab infusions. Hence, intra-orbital rituximab injections were administered, alongside a tapering course of oral prednisone therapy. The patient received a total of eight intralesional rituximab (Riximyo) 10 mg/ml injections, weekly over 8 weeks. The patient noticed improvement of her left periocular fullness and erythema, with concurrent improvements in ocular motility. Interval MRI demonstrated mild improvement in the extent of contrast enhancement within the medial orbit; however, there was still diffuse enhancement throughout the left orbit. Due to a plateau of improvement in orbital signs, at 8 weeks post commencement of intralesional rituximab, the patient was offered systemic treatment with IV rituximab therapy and cessation of intra-orbital injections.
Discussion
Literature surrounding the utility of rituximab in the management of OID is growing. There are numerous case reports, case series, and clinical trials describing the outcomes of rituximab infusions in the management of OID and as escalation therapy for refractory or relapsing disease.1,2,23 Rituximab infusions in orbital GPA have focused on providing an effective induction treatment to induce remission and an alternative treatment modality for refractory or relapsing disease.23 –25 Rituximab infusions have been demonstrated to be equivalent and potentially more successful in inducing remission compared to traditional agents, such as cyclophosphamide.11,12,25 Although the use of intralesional rituximab injections has been reported in NSOI, its application to orbital GPA remains less defined. In situations of systemic organ-threatening or vision-threatening GPA, independent of orbital involvement, systemic treatment remains the first-line treatment.10 –12
The use of intralesional rituximab in orbital disease was first proposed in an interventional pilot study in 2011 for the management of primary ocular adnexal lymphoma, and since this study, there have been additional case reports and series describing its use.17 –20 Savino et al. described an induction regimen of weekly 5 mg intralesional rituximab (MabThera 5 mg/ml; Roche, Basel, Switzerland) injections for 1-month for the treatment of four patients with orbital and lacrimal gland B-cell lymphoma. In cases of incomplete response after induction therapy, patients received a higher dose of rituximab (10 mg/ml) for an additional three injections, spaced 4 weeks apart. The total dose administered ranged from 40 mg to 140 mg over a period of 8–16 weeks. 17 Other regimens described include weekly 15 mg injections (10 mg/ml) for 4-weeks followed by 6-monthly injections for treatment of two patients with conjunctival Mucosa-associated lymphoid tissue lymphoma (MALT) lymphoma; 18 a single 50 mg rituximab injection (50 mg/1 ml) for extra-nodal MALT lymphoma of the lacrimal gland; 19 and 50 mg intralesional rituximab monthly (for up to a total of three injections) for low-grade conjunctival lymphoma. 20
Intralesional rituximab has been utilized as both a first-line induction therapy and in steroid-refractory or relapsing orbital inflammation. Published studies detailing its use in orbital inflammation have been summarized in Table 1.14–16 While cases in the literature describe the use of rituximab as a primary isolated treatment, our reports are more exploratory in nature, and thus, the role of intralesional rituximab was more adjunctive, with the observed clinical response (or lack of) guiding its ongoing administration and alternative management. In our series, injections were directed toward the relevant orbital mass (e.g. inferotemporal and inferonasal in inferior orbital inflammation in Case 1), or if diffuse, then a peribulbar injection was administered in the inferotemporal quadrant. There have been no prior reports of injections in orbital myositis.14 –16 The first reported use of intralesional rituximab in NSOI was described in 2015 by Savino et al. 14 This series included three patients with NSOI, one with an extraconal inferior mass, one with a unilateral lacrimal gland mass, and one with bilateral lacrimal gland masses. In all three patients, systemic corticosteroids were initially trialed with only mild response and improvement, and/or relapse of disease during tapering. Patients subsequently received weekly 10 mg rituximab (MabThera®; Roche, Basel, Switzerland, 100 mg/10 ml) injections over a month, directed to the orbital mass and/or involved lacrimal glands. Two patients received two cycles of intralesional rituximab, and all patients demonstrated a response to treatment. 14 Furthermore, Miller et al described a retrospective study including nine patients with idiopathic dacryoadenitis over a 2-year period treated with 50 mg intralesional rituximab (50 mg/1 ml) to the lacrimal gland, with a statistically significant decrease in the orbital inflammation score. All patients achieved complete resolution within 4 weeks of commencing treatment; however, one patient experienced relapse of disease at 8 months, which was responsive to a further cycle of intralesional rituximab. There were no adverse effects recorded over a mean follow-up period of 28.9 months. 15 More recently, Demirici et al. described the use of intra-orbital rituximab injections in 18 patients with NSOI with lacrimal gland involvement. In this cohort, 89% of patients demonstrated a clinical response, of which 78% were complete and 11% partial (⩽30% decrease in lesion diameter). The remaining 11% of patients did not respond to three injections of intra-orbital rituximab and subsequently required systemic corticosteroid and methotrexate. 16 Within our two cases, repeated weekly injections of rituximab (10 mg/ml) were administered, while within the literature, a single higher dose (i.e., 50 mg/ml) has been administered to the lacrimal gland with response. In our reported cases, an empirical duration of treatment was set at 12 weeks with weekly monitoring of clinical response; however, there remains limited data available to guide the optimal duration of treatment. Although clinical improvement was observed following 11 and 8 weeks of treatment, respectively, it remains difficult to comment on the efficacy and durability of response given the concurrent use of systemic treatment, variable severity of disease, and orbital inflammation, and consequently, the plateau in clinical improvement observed. Further observational studies are necessary to guide the dose, frequency of injections, and duration of therapy.14 –16
Summary of published literature detailing the use of intralesional rituximab in orbital inflammation.

Minor AE in one case of mild temperature elevation with response to 500 mg oral paracetamol.
AE, adverse effect; F, female; M, male; NSOI, nonspecific orbital inflammation.
In this series, intralesional rituximab was initiated for patients with severe refractory/relapsing disease or optic neuropathy. In Case 1, the injections were commenced as an adjunct to systemic infusions, while in Case 2, it was used as an alternative due to physician and patient preferences. Return to baseline VA of 6/6 with only residual optic neuropathy was observed in Case 1, while Case 2 demonstrated clinical and radiological improvement. Relapses and refractory disease were not observed within the follow-up periods reported. Adverse effects (AE) of systemic rituximab infusions include the risk of infusion reactions (fever, chills, rash, anaphylaxis, acute respiratory distress syndrome), immunocompromise leading to reactivation of or opportunistic infections, lymphopenia, dermatological/mucocutaneous reactions, and cardiac arrhythmias. 26 Localized intralesional administration of rituximab may help to ameliorate these systemic AE; however, the long-term and extensive AE profile for intralesional administration remains to be determined. There were no reports of AE experienced by our patients during or after intralesional rituximab administration.
The limitations of this study include the small sample and limited long-term follow-up available for these patients. Rituximab does not yet have approval from the Australian Therapeutic Goods Administration for use in intralesional administration to orbital lesions in patients with GPA. In both cases, the investigators received individual patient use approval for intralesional administration by our institutional (Central Adelaide Local Health and Network) Drug and Therapeutics Committee. Within the literature, the description of intralesional rituximab has been primarily focused on the management of refractory OID, including NSOI and orbital GPA. Within these cases, there have been failed attempts at controlling orbital inflammation with systemic corticosteroid therapies requiring escalation of an immunosuppressive drug regimen. The concurrent use of intra-orbital and systemic infusions of rituximab has been similarly depicted within our cases.14,15 It remains difficult to ascertain the exact clinical utility of intralesional rituximab within these reported cases due to the concurrent administration of systemic rituximab. There is existing literature surrounding the experimental utility of intralesional rituximab in other OID; however, further controlled studies are necessary to determine its application as a primary therapy in orbital GPA. There are yet to be any reports of its application as a first-line modality, and its utility and long-term outcomes remain to be defined.
In conclusion, we present our initial experience with intralesional rituximab injections, used as an adjunctive treatment modality, alongside systemic treatment, for orbital GPA in a case of optic neuropathy and another of severe, refractory, and relapsing disease. The clinical response from intralesional rituximab injections remains to be defined, and further prospective studies are required to investigate the utility and long-term outcomes of this treatment modality.
Supplemental Material
sj-docx-1-oed-10.1177_25158414251368027 – Supplemental material for Intralesional rituximab injections for orbital granulomatosis with polyangiitis: a case series and literature review
Supplemental material, sj-docx-1-oed-10.1177_25158414251368027 for Intralesional rituximab injections for orbital granulomatosis with polyangiitis: a case series and literature review by Terence Ang, Jessica Y. Tong and Dinesh Selva in Therapeutic Advances in Ophthalmology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
