Abstract
Background:
Scleral allograft-reinforced cyclodialysis intervention can achieve sustained intraocular pressure (IOP) reduction by enhancing uveoscleral outflow in hypertensive patients with primary open-angle glaucoma (POAG) failing medical therapy.
Objectives:
To evaluate clinical outcomes of bio-interventional uveoscleral outflow enhancement surgery through 12 months of follow-up in POAG subjects who are inadequate responders to IOP-lowering medical treatment.
Design:
Prospective interventional real-world evidence trial.
Methods:
Patients with POAG and medicated baseline IOP >21 mmHg failing medical therapy underwent bio-interventional uveoscleral outflow enhancement surgery with an ab-interno allograft-reinforced cyclodialysis. Acellular allogeneic scleral bio-tissue was micro-trephined, shaped, and loaded in a delivery cannula for internal scaffolding. Endoscleral reinforcement of the cyclodialysis was then performed to maintain the internal uveoscleral filtration conduit. Effectiveness outcomes such as IOP and IOP-lowering medication use, as well as ocular safety and tolerability, were analyzed through 12 months post-op.
Results:
Fifty-one eyes were enrolled with a baseline medicated IOP greater than 21 mmHg. The average age was 70.9 ± 8.5. The mean best corrected visual acuity (BCVA) at baseline was 0.40 ± 0.32, and the mean medicated IOP was 25.7 ± 4.4 mmHg on 1.2 ± 1.3 IOP-lowering medications. In 83% of cases, visually significant cataract comorbidity was present and treated with adjunct phacoemulsification. The bio-interventional cyclodialysis surgery and scleral reinforcement were successfully performed in all cases. The procedures were well tolerated, and there were no visually significant or serious, vision-threatening ocular adverse events. Durable and sustained reinforcement of the cyclodialysis was achieved through 12 months of follow-up without migration, displacement, or attrition of the allograft bio-scaffold. At 12 months post-op, there was a statistically significant (
Conclusion:
Uveoscleral outflow enhancement can be surgically enhanced in an ab-interno approach through bio-interventional cyclodialysis with adjunct scleral allograft reinforcement to lower IOP in open-angle glaucoma patients who are inadequate responders to medical therapy.
Trial registration:
The study was registered with clinicaltrials.gov NCT05506423.
Plain language summary
In a real-world evidence study of consecutive case series of 51 hypertensive eyes of OAG patients with preoperative medicated IOP > 21 mmHg who underwent cyclodialysis intervention with bio-tissue reinforcement with or without concurrent cataract surgery, subjects achieved significant IOP lowering through 12 months of postoperative follow-up with a 41% decrease in IOP and 42% decrease in IOP lowering medications compared to baseline. Bio-interventional uveoscleral outflow enhancement through allograft-reinforced cyclodialysis can lower IOP and associated medication use in patients with hypertensive OAG.
Keywords
Introduction
Surgical approaches in the treatment of glaucoma aim to lower intraocular pressure (IOP) by increasing aqueous drainage and outflow.1,2 There has been an increased use of interventional approaches in the treatment of OAG, and micro-incisional glaucoma surgery (MIGS) is now the major glaucoma surgical procedure in the United States, accounting for more than 70% of all glaucoma surgical treatments. While these are significant advances in the surgical paradigm for glaucoma, there is a continued need for more effective surgical treatments beyond the trabecular outflow pathway, which physiologic studies estimate to account for about half of the aqueous drainage of the eye, with the rest of the outflow through the uveoscleral pathway.3,4 Unlike pharmacologic treatments for IOP lowering, where the primary, most effective therapeutic modality (e.g., prostaglandin analogues) targets exclusively the uveoscleral outflow pathway, there is currently no available FDA-approved surgical treatment for the uveoscleral outflow.5–7 This is a major gap in the existing glaucoma surgical paradigm, which is limited primarily to the trabecular outflow pathway, has only a modest IOP-lowering effect, and is mainly used as an adjunct to phaco cataract surgery in milder glaucoma cases.5,8–14
Cyclodialysis was one of the earliest glaucoma procedures for uveoscleral outflow enhancement, first introduced in 1907 and used for decades in patients with moderate-to-advanced glaucoma. While effective in some patients, its durability is limited due to the premature closure of the cleft and restenosis of the supraciliary conduit. Attempts to sustain the patency of the cyclodialysis over time have included the injection of stabilization materials such as viscoelastic and air, but have been of limited consequence given the transient nature of the outflow maintainer.15–21
New techniques are emerging to fill the need for durable biocompatible reinforcement of the cyclodialysis conduit. 22 One such approach is the bio-reinforced cyclodialysis using an allograft bio-scaffold for the sustained maintenance and stability of the cyclodialysis conduit. Next-generation bio-interventional approaches using micro-trephined allogeneic bio-tissue, such as scleral allograft, along with advanced interventional surgical instrumentation, allow unprecedented precision and specificity for atraumatic access to the supraciliary space. This multi-interventional procedure of cyclodialysis followed by allograft bio-scaffold with a highly targeted sub-millimeter intraocular scleral reinforcement aims to sustain the patency of the cyclodialysis conduit to achieve durable uveoscleral outflow augmentation and IOP lowering.
Scleral grafts have long been used for homologous scleral reinforcement in glaucoma surgery to cover and fixate the tubes of glaucoma drainage devices to the scleral wall.
23
The acellular, sterile donor tissue is readily available, highly biocompatible, and has long-term durability and structural stability—all essential and highly desirable biomaterial properties for an implantable material designed to function as a bio-scaffold for structural stabilization and reinforcement. Allogeneic scleral tissue is also hydrophilic, porous, and inert, and unlike conventional implantable hardware, it is biologically homologous to the native scleral tissue.24,25 This could impart significant advantages for healing, fibrosis, and foreign body reaction, which is predominantly driven by the mismatch of implant material stiffness and mechanical properties to those of the recipient’s native tissue.25,26 Unlike conventional implantable inorganic hardware, the natural conforming scleral allograft is bio-conforming and homologous to the underlying recipient endo-scleral wall, which makes it potentially less stimulating to fibrosis and cleft restenosis.
This investigation is the largest prospective series to characterize the clinical outcomes and performance of bio-interventional uveoscleral outflow surgery in the management of medically uncontrolled open-angle glaucoma (OAG).
Methods
We report the clinical experience from a prospective, interventional real-world clinical study of the efficacy and safety of a multi-interventional IOP-lowering procedure, namely a bio-reinforced cyclodialysis. The summary protocol is listed on Clinicaltrials.gov (NCT05506423). Fifty-one consecutive eyes with hypertensive primary OAG (baseline medicated IOP >21 mmHg) failing topical therapy underwent ab-interno cyclodialysis and adjunct bio-tissue reinforcement with allograft scaffolding following the completion of phacoemulsification cataract extraction. The study protocol adhered to the tenets of the Declaration of Helsinki and was reviewed and approved by the WCG central institutional review board (Princeton, NJ; https://www.wcgirb.com/). All patient data were anonymized and treated with confidentiality according to the tenets of the Declaration of Helsinki. During preoperative evaluation, all subjects met the inclusion criteria of diagnosis of primary open-angle glaucoma (POAG) with angles Shaffer grade 3+ in all four quadrants, with or without an operable cataract. Study exclusion criteria were baseline medicated IOP <21 mmHg, prior incisional glaucoma surgery (e.g., filtering procedure), significant myopia with axial length >24 mm, visual field loss within the central 10°, presence of a narrow angle or mixed angle glaucoma, secondary open-angle glaucoma, or clinically significant corneal pathology such as Fuch’s dystrophy.
The allograft used was minimally manipulated homologous donor scleral allogeneic tissue (AlloFlo™ by Iantrek, Inc., Providence, RI, USA) of elongated cuboidal form factor, 5 mm long and 500 µm wide. The tissue was prepared from larger scleral allogeneic patch graft material sourced from an eye bank. The allograft tissue underwent significant processing to create the final allogeneic bio-scaffold. It was processed in accordance with the highest standards of donor tissue screening, testing, and quality assurance as per the guidelines of the American Association of Tissue Banks. In addition, specialized micro-trephination and bio-tissue manipulation ensured high-precision engineered shaping and standardized sizing of the implants. The tissue was subsequently sterilized using gamma radiation and stored in sterile Tyvek packs to ensure a minimum shelf life of 12 months.
The glaucoma surgery was performed following the phaco procedure when the eye was positioned for standard gonio-intervention by tilting the head and microscope for optimal intraoperative gonioscopy (Figures 1–5). After achieving adequate visualization and confirming an open-angle and accessible gonio-anatomy, a sectoral cyclodialysis cleft was surgically created using a cyclodialysis cannula. Subsequently, visco-cycloplasty was performed to augment the cleft and the supraciliary reservoir. The scleral reinforcement was then performed, and the bio-scaffold was deployed using the CycloPen micro-interventional system (Figure 1). Irrigation/aspiration was then performed to evacuate the viscoelastic and complete the procedure. Final postoperative gonio-confirmation was done to verify implant position with the proximal end “flush” at the iris root (Figure 6). A video of the surgical procedure is available at https://vimeo.com/1051502624/69502866eb?share=copy. Standard postoperative regimen of topical antibiotics and steroids is prescribed for 30 days (fourth-generation fluoroquinolone and prednisolone 4×/day). At follow-up postoperative visits, patients underwent full ophthalmic examination, including BCVA (Snellen), slit lamp evaluation with grading of inflammation, Goldman tonometry, gonioscopy, and assessment for ocular adverse events. Since this is a real-world evidence registry, clinical evaluation, IOP tonometry, and topical medication reintroduction followed the standard-of-care clinical practice paradigm of each physician. There were no pre-specified criteria for medication rescue or reintroduction. The registry study was monitored prospectively. An electronic data capture system was used for the collection of results.

The micro-interventional cyclodialysis system with a spatulated cannula tip containing the scleral allograft.

The homologous acellular allograft bio-scaffold deployed at the endoscleral surface to maintain the cyclodialysis filtration conduit for uveoscleral outflow enhancement.

First step of the multi-interventional procedure with the creation of an ab-interno, focal cyclodialysis.
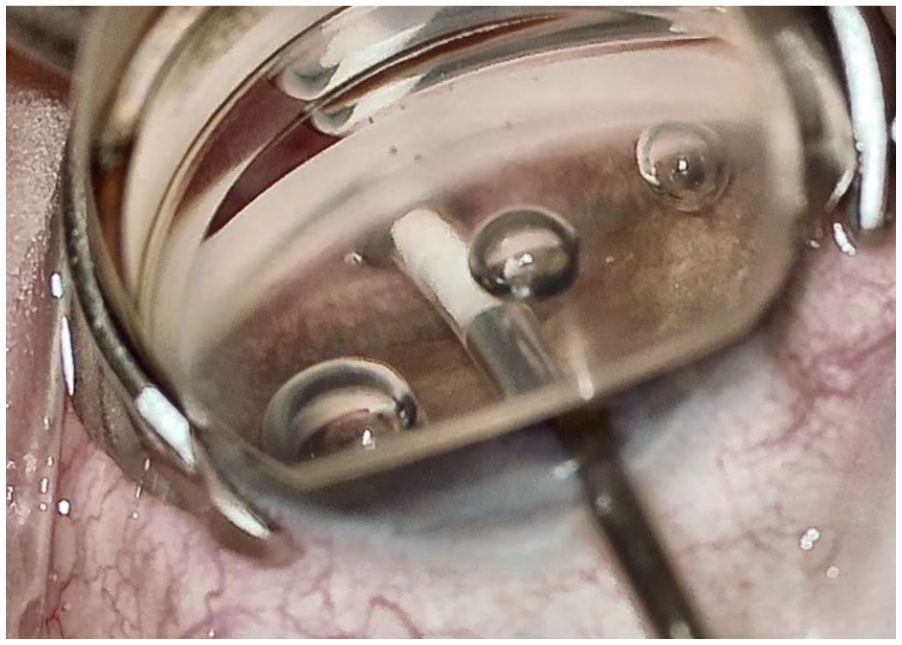
Step 2 of the multi-interventional procedure with the deployment of the allograft bio-scaffold for scleral reinforcement.

Allograft scleral reinforcement spacer implanted at the iris root for AC-clear deployment.
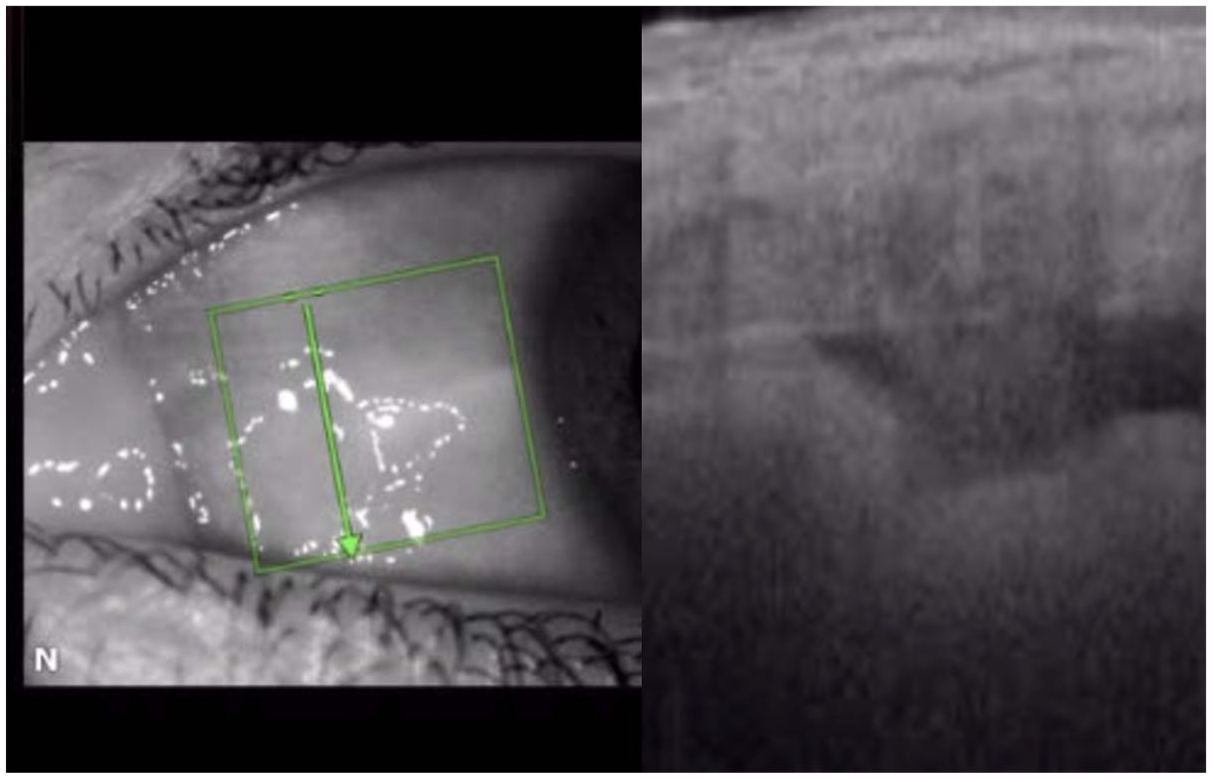
Optical Coherence Tomography (OCT) image of allograft bio-scaffold showing the cyclodialysis with the internal filtration conduit buttressed by the homologous allograft reinforcement.
Statistical analysis
Analytical methods for the results included descriptive statistics at the different time points expressed as means ± SD, with differences versus baseline through 12 months of follow-up. IOP and medication outcomes are analyzed using paired two-tailed
Results
Demographic and baseline characteristics of the 51 cases are presented in Table 1. At 1 year, only 3 out of 51 eyes (5.8%) were lost to follow-up due to patient relocation and study withdrawal. Three additional eyes underwent secondary intraocular surgery in the postoperative period (e.g., Xen and GDI) and were not part of the efficacy-evaluable cohort at 12 months.
Baseline demographic characteristics.
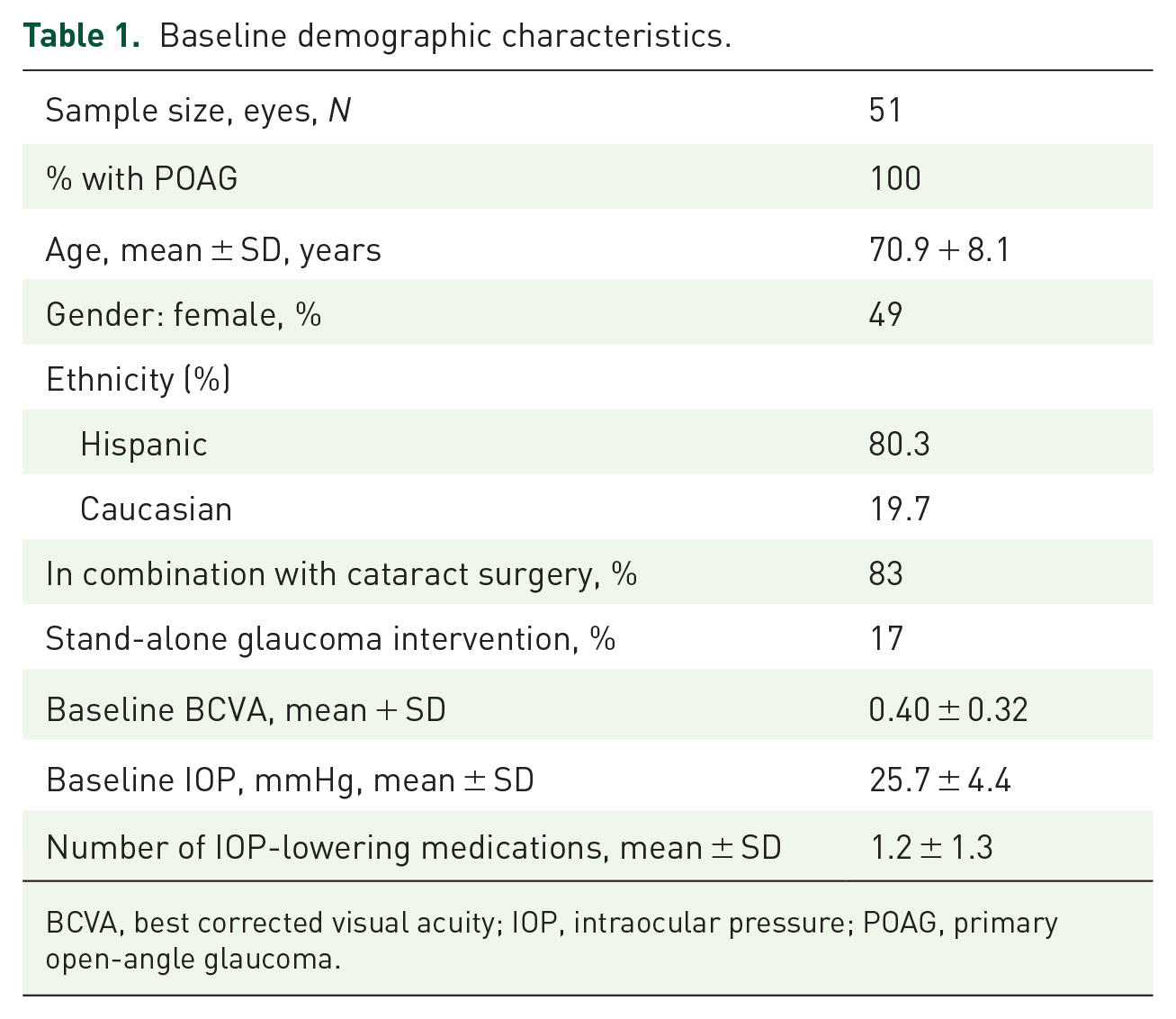
BCVA, best corrected visual acuity; IOP, intraocular pressure; POAG, primary open-angle glaucoma.
Mean medicated IOP was 25.7 + 4.4 mmHg, and subjects were persistently hypertensive and uncontrolled despite being on an average of 1.2 IOP-lowering medications.
All cases had successful multi-interventional procedures of focal cyclodialysis with adjunct scleral bio-reinforcement (Figure 1). There were no vision-threatening intraoperative or postoperative complications (Table 2). In most cases, there was minimal blood reflux from the supraciliary cleft, which is expected and was not associated with any significant visual sequelae. There were no severe or persistent inflammation, and postoperative hyphema was mild and transient without any cases of clinically and visually significant bleeding. Also, no bio-tissue migration or corneal touch was observed through the 12-month follow-up period. Operated eyes had good visual acuity outcomes, increasing from an average baseline LogMAR BCVA of 0.4 ± 0.32 to 0.73 ± 0.23 at 12 months (
Key safety outcomes.
Intraoperative and postoperative complications observed after allograft-reinforced cyclodialysis.

Fully resolved by month 6; Not visually significant.
With phaco cataract surgery and diabetic retinopathy. No hypotony.
One case of Ahmed tube, one case of Xen implant, one case of Cyclophotocoagulation (CPC)/Goniotomy, and one paracentesis for IOP reduction. No cases requiring anterior chamber wash-out. No wound-related events such as wound leak, dehiscence, or need for suturing.
BCVA, best corrected visual acuity; IOP, intraocular pressure.
The two cases of CME postoperatively were in the setting of cataract surgery and pre-existing diabetic retinopathy, and fully resolved in the postoperative period. At no time during their postoperative follow-up was the IOP in either case below 14 mmHg nor was there any concern of maculopathy or prolonged inflammation.
Overall, the IOP-lowering effect was robust, immediate, and sustained through 12 months postoperatively (from 25.7 + 4.4 mmHg at baseline to 15.7 + 4.4 and 15.2 + 4.5 mmHg, 1 and 12 months, respectively) with a concurrent significant reduction in the number of IOP-lowering medications (1.7 + 1.3 at baseline to 0.9 + 1.2 and 0.7 + 0.9, 1 and 12 months respectively). 77.8% of eyes achieved a 12-month IOP of <18 mmHg with the same or fewer number of IOP-lowering medications (Tables 3 and 4; Figure 7). The statistical significance of the IOP-lowering effect was maintained in a sensitivity analysis with further imputation for the cases undergoing secondary surgical intervention.
IOP over 12 months of follow-up.

IOP, intraocular pressure.
IOP-lowering medication use over 12 months of follow-up.

IOP, intraocular pressure.
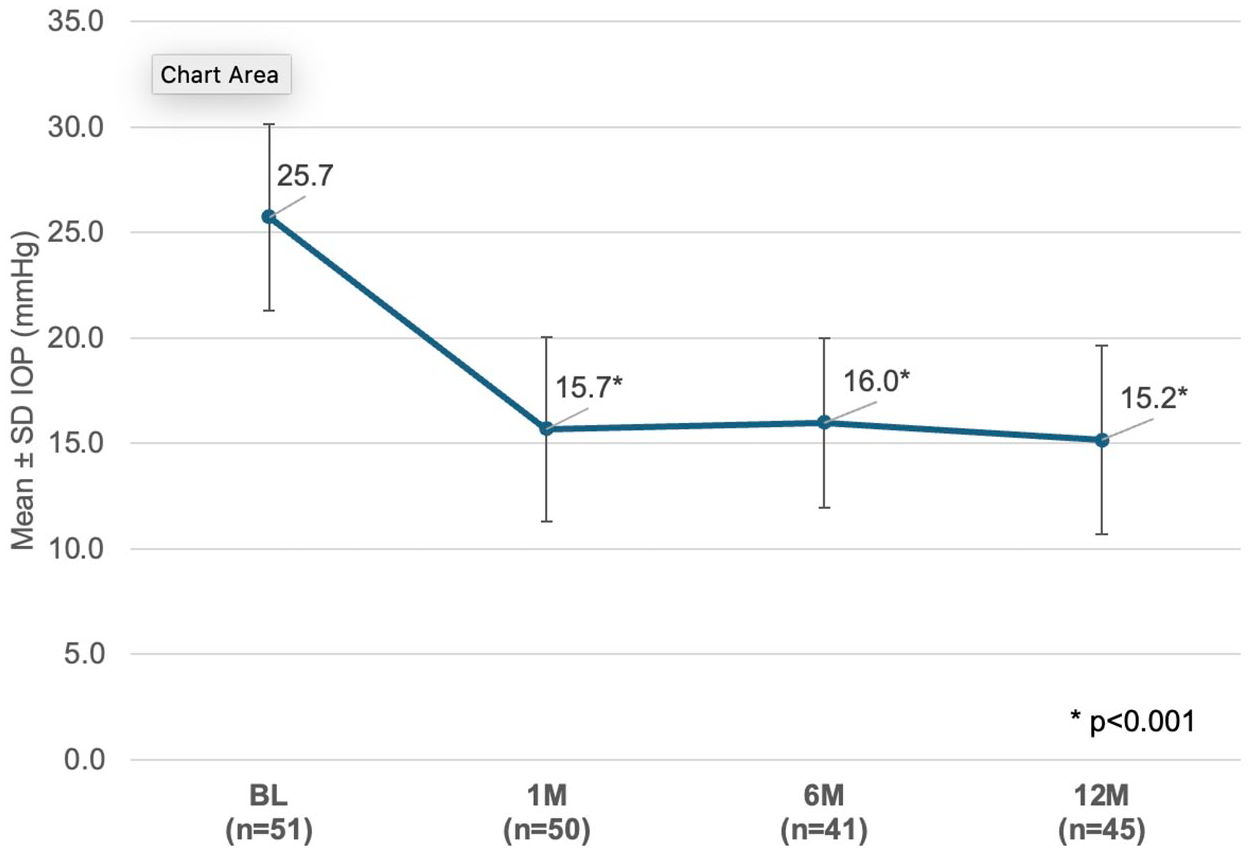
IOP through 12 months of follow-up.
In the smaller stand-alone cohort (N9), where no adjunct phacoemulsification surgery was performed, there was also a statistically significant 30% reduction in mean medicated IOP from 29.3 ± 6.4 mmHg at baseline to 20.6 ± 6.7 mmHg at 12 months with a concurrent reduction in IOP-lowering medications (
Discussion
We report on the clinical outcomes of a bio-interventional surgery for uveoscleral outflow enhancement using next-generation cyclodialysis with adjunct scleral allograft bio-reinforcement. Our results demonstrate a robust and sustained IOP-lowering effect through 12 months of follow-up, with more than 10 mmHg mean medicated IOP reduction along with a significant reduction in IOP-lowering medication use. The safety profile is characterized by few adverse events, both in the operative and long-term postoperative period.
Our results further validate the modified surgical approach of bio-scaffolded cyclodialysis originally described by Ianchulev et al., 27 which is designed to improve on the conventional surgical technique. The incidence of ocular adverse events and complications was low and consistent with that of an interventional procedure. This is related to the advanced surgical cyclodialysis instrumentation, which allows for the construction of a controlled, high-precision supraciliary cleft in an atraumatic, gonioscopic, ab-interno approach. The durability of the treatment effect was also distinctly enhanced with the lack of early cleft restenosis and no rebound IOP within the first 90 days post-op—the characteristic hallmark of conventional cyclodialysis. The sustained IOP-lowering effect through 12 months of follow-up is likely the result of the durable uveoscleral outflow channel reinforcement, whereby the permanent allograft biomatrix provides the structural scaffolded support to maintain the stability of the internal filtration cleft. There were no reports of an extended inflammatory response, no incidence of prolonged uveitis, nor peripheral anterior synechiae, which indicates a highly inert, biocompatible, and homologous allograft material.
The significant long-term IOP-lowering effect demonstrated in this cohort of hypertensive eyes failing medical therapy is likely the result of several factors. The multi-interventional bio-reinforced cyclodialysis procedure with adjuvant allogeneic bio-tissue is distinctly different from prior suprachoroidal devices, supraciliary stents, and uveoscleral outflow procedures by way of the unique mechanistic approach to aqueous filtration and the high degree of homologous biocompatibility of the implant. The primary mechanism of aqueous outflow augmentation is through the native micro-interventional cyclodialysis rather than an exogenous implantable hardware. The cyclodialysis cleft is small, controlled, and made of native resident tissue, which inherently minimizes any hardware-related foreign body reaction—a significant concern with supraciliary devices. The cyclodialysis filtration conduit is further augmented using visco-cycloplasty with injection of viscoelastic to create a larger supraciliary reservoir and further enhance filtration. Furthermore, the allogeneic scleral scaffolding and reinforcement not only provides structural stability and a durable framework for the internal cleft, but does so in a highly biologically advantageous way—using not only a highly inert biocompatible material, but a homologous allograft. It has been demonstrated in the implant literature that homologous minimally modified allograft materials are less likely to induce macrophage activation, fibroblastic response, and local tissue damage. In addition, the non-resorbable scleral acellular matrix is highly collagenous, porous, and hydrophilic, which allows for aqueous conductivity and access to the internal cleft reservoir for trans-scleral and suprachoroidal outflow.
With almost 40% reduction in mean medicated IOP from baseline through 12 months, our results provide further support to the growing evidence of the compelling efficacy of uveoscleral outflow augmentation as a potentially superior approach to trabecular pathway interventions. Several studies and a recent meta-analysis28–30 demonstrate that surgical enhancement of the uveoscleral outflow seems tooffer one of the highest IOP-lowering effects of all internal non-penetrating angle-based surgeries—exceeding the effect seen with trabecular interventions. This is not unlike the clinical experience from pharmacotherapy for glaucoma, where the primary and most efficacious treatment is with the therapeutic class of prostaglandin analogues, which act exclusively through the uveoscleral outflow agents. This is further supported by the emerging clinical results of another suprachoroidal stent, the MiniJect device, where uveoscleral outflow enhancement provides a sustained effect over 2 years of follow-up of close to 40% reduction in mean IOP. 31 While the bio-interventional cyclodialysis procedure demonstrates comparable efficacy to what is seen with non-trabecular suprachoroidal devices such as MiniJect and CyPass, 32 the absence of rigid intraocular hardware, along with the anterior chamber–clear non-protruding deployment within the cyclodialysis cleft, significantly reduces the impact on the corneal endothelium.
There are several limitations of our study, many of which are related to its real-world evidence design. While prospective in nature, this is a non-randomized trial without a phaco-only control group. The stand-alone cohort is small, as the patient population reflects the community practice paradigm where most angle-based glaucoma surgery is performed in combination with cataract procedures. Phaco-emulsification cataract surgery has a modest additive effect for IOP lowering, which likely accounts for part of the cumulative efficacy captured in this study. In addition, the standard real-world practice paradigm does not allow for baseline or terminal wash-out of IOP-lowering medications so all of the IOP outcomes are medicated. There are also no prescribed medication reinstitution criteria, and the treating physician used their own judgment and discretion on how to titrate postoperative pharmacotherapy. Gonio-imaging of the implant postoperatively with Ultrasound Biometry (UBM) or Optical Coherence Tomography (OCT) was not collected as a part of this real-world study and will be a subject of future investigation to demonstrate the postoperative dynamic evolution of the cleft and the supraciliary space. Future clinical studies can address some of these outstanding questions and provide comparative clinical evidence to other trabecular and non-trabecular outflow enhancement interventions, particularly for moderate-to-advanced and stand-alone OAG treatment, where the clinical utility of uveoscleral intervention is most applicable.
Conclusion
Sustained, safe, and clinically meaningful IOP reduction was achieved through 12 months in medically uncontrolled POAG eyes following a novel scleral allograft bio-reinforcement of a cyclodialysis.
