Abstract
Background:
The travoprost intracameral implant and cataract surgery both lower intraocular pressure (IOP).
Objectives:
We evaluated the safety and IOP-lowering effect at 3 months following administration of travoprost intracameral implant in combination with cataract surgery.
Design:
Prospective, 12-month, open-label, single-arm trial.
Methods:
We enrolled patients with age-related cataracts and open-angle glaucoma or ocular hypertension in the same eye. At baseline, patients were required to have an unmedicated mean diurnal IOP (average of 8:00 am, 10:00 am, and 4:00 pm IOPs) of 24 mmHg or greater, and an IOP of 36 mmHg or less at each of these three timepoints. On the day of the combined procedure (day 1), patients who had uncomplicated phacoemulsification cataract surgery received a travoprost intracameral implant. Follow-up evaluations occurred on day 2–3, week 2, week 6, and month 3 visits.
Results:
Sixty patients had uncomplicated cataract surgery and received a travoprost intracameral implant. There were no serious adverse events. Study eye adverse events were reported in 8.3% of patients. The most frequently reported adverse event was dry eye (6.7%). At month 3, the mean diurnal IOP change from baseline was −10.6 mmHg (95% confidence interval: −11.2, −9.9; p < 0.0001) from an unmedicated baseline mean diurnal IOP of 25.2 mmHg. In addition, at month 3, 97% of eyes had a 20% or greater mean diurnal IOP reduction from baseline, and 91.0% of eyes had a mean diurnal IOP of 18 mmHg or less.
Conclusion:
Administration of a travoprost intracameral implant combined with routine cataract surgery was safe. The sizable −10.6 mmHg IOP change from baseline at month 3 was both statistically significant and clinically relevant.
Trial registration:
NCT06061718, Travoprost Intraocular Implant in Conjunction with Cataract Surgery, https://clinicaltrials.gov/study/NCT06061718.
Plain language summary
Why was the study done? The travoprost intracameral implant is a drug delivery system that is placed directly in the eye to continuously release travoprost to lower intraocular pressure in patients with open-angle glaucoma or ocular hypertension. Cataract surgery also is known to lower intraocular pressure in normal eyes and to a greater extent in eyes with glaucoma or ocular hypertension. Placement of the implant in the anterior chamber angle of the eye and cataract surgery are both done through a small corneal incision. What did the researchers do? The researchers studied the safety and the reduction in intraocular pressure after administering a travoprost intracameral implant during the same procedure as removal of the cataractous lens and administration of an intraocular lens. Sixty patients with open-angle glaucoma had the combined procedure and were followed over a 3-month period to check their intraocular pressure and health of their eye.
What did the researchers find? There were no serious safety findings after the combined procedure, with less than 10% of patients having any ocular side effect. The most common side effect was dry eye. At month 3, mean intraocular pressure was reduced significantly from the untreated baseline pressure measured before the combined procedure. What do the findings mean? The study showed that it is safe to combine the placement of the travoprost intracameral implant in the eye during the same procedure as routine cataract surgery. The study also showed that there was a substantial lowering of intraocular pressure at 3 months in patients with open-angle glaucoma.
Keywords
Introduction
Cataracts and glaucoma are the leading causes of blindness worldwide.1,2 Both conditions are more common in individuals as they get older and require treatment to regain or maintain visual function. Removal of the opacified lens and replacement with an artificial intraocular lens (IOL) significantly improves vision or restores useful vision in patients with cataracts. Lowering intraocular pressure (IOP) using medical, laser, or incisional surgical techniques, or a combination thereof, reduces the risk of visual field deterioration in patients with glaucoma. 2
In patients with open-angle glaucoma (OAG) and cataracts, removal of the cataractous lens has been shown to lower IOP, with the magnitude of the reduction proportional to the preoperative IOP.3–5 A meta-analysis concluded phacoemulsification cataract surgery in patients with primary OAG lowers IOP by an average of 2.3 mmHg from a medicated IOP of 15.4 mmHg, 6 while two randomized well-controlled trials in patients with OAG demonstrated that at 24 months following cataract surgery, IOP was reduced by 5 mmHg from an unmedicated baseline of 25 mmHg.7,8 Patients with OAG undergoing cataract surgery may also be candidates for minimally invasive glaucoma surgery (MIGS). MIGS has shown a favorable risk-benefit profile resulting in greater IOP reduction than cataract surgery alone while also maintaining the same favorable safety profile for the combined MIGS-cataract surgery as for the cataract surgery alone.7,8 Several Schlemm’s canal-based MIGS (e.g., iStent inject, iStent infinite) use the same clear corneal incision and ab interno approach used for phacoemulsification cataract surgery.
More recently, intracameral sustained drug delivery systems, including procedural pharmaceuticals, have become commercially available in the United States. As with MIGS, procedural pharmaceuticals may offer favorable IOP-lowering effects when combined with cataract surgery. One such system, the travoprost intracameral implant, 75 µg (iDose® TR; Glaukos Corporation, Aliso Viejo, CA, USA), consists of a titanium implant containing a reservoir filled with travoprost which is capped with a membrane controlling the sustained release of travoprost. The implant is preloaded in a single-dose inserter to facilitate insertion directly through the trabecular meshwork of the anterior chamber angle where it is anchored into the sclera. 9 As with phacoemulsification cataract surgery, administration of the travoprost intracameral implant also involves an ab interno approach through a small clear corneal incision, thereby lending itself to administration in a combined procedure.
The travoprost intracameral implant 75 µg was approved in the United States in December 2023 for the reduction of IOP in patients with OAG or ocular hypertension (OHT). Travoprost was chosen as the FP-receptor agonist for the intracameral implant because of its history of safety and efficacy in reducing IOP in patients with OAG and OHT, as well as its high potency and selectivity.10–12 In addition, travoprost has a greater affinity for the FP receptor than latanoprost 10 or bimatoprost. 13 The physical state of travoprost as a viscous liquid allows it to completely fill the implant titanium reservoir. Twenty-four months following administration, approximately 16% of residual travoprost has been measured in explanted implants 14 and the aqueous humor concentration of travoprost-free acid 14 has been shown to be above that observed after dosing with travoprost ophthalmic solution, 0.004% 15 and above the established efficacious concentration. 16
Safety and efficacy studies conducted to support the U.S. approval of the travoprost intracameral implant were performed as standalone procedures.17–19 Results of the 36-month phase II trial demonstrated that iDose TR provided robust IOP-lowering and substantially reduced topical IOP-lowering medication burden for up to 36 months following a single administration. 19 A pooled analysis of the 12-month data from two phase III trials demonstrated that the IOP-lowering efficacy was statistically non-inferior to twice-daily administered timolol ophthalmic solution, 0.5%, and substantially reduced topical IOP-lowering medication burden. 20
The objective of the current 12-month study was to evaluate the safety and IOP-lowering effect of the travoprost intracameral implant administered in combination with phacoemulsification cataract surgery. Three-month results from this ongoing study are presented herein.
Materials and methods
Study design
This was a prospective, open-label, single-arm trial conducted at a single site in Armenia, using four experienced surgeons (three US surgeons and one Armenian surgeon). The trial was conducted in accordance with internationally accepted good clinical practices which embody the tenets of the Declaration of Helsinki, and was prospectively registered at Clinicaltrials.gov (NCT06061718). Approval from an independent ethics committee was obtained for the trial, and patients provided written informed consent prior to undertaking any study-related procedures.
Patient population
Patients who were 45 years of age or older, and who had a visually significant age-related cataract and OAG (primary, pseudoexfoliative, or pigmentary) or OHT, were screened for participation in the study.
At the screening visit, patients could be on 0–2 topical IOP-lowering medications in the study eye and were required to have an IOP (measured at 8:00 am or 10:00 am) that was no greater than 32 mmHg. Those on IOP-lowering medication(s) must have been able to safely undergo a washout period from their medication: 5 days for miotics and topical carbonic anhydrase inhibitors (CAIs), 2 weeks for alpha agonists, 4 weeks for beta-blockers, and 6 weeks for prostaglandin analogs (PGAs) and rho kinase inhibitors. In addition, prospective study eyes were required to have a Shaffer grade of 3 or 4 open angles on gonioscopy, normal anterior angle anatomy, and absence of peripheral anterior synechia, rubeosis, or other angle abnormalities that could hinder the proper placement of the travoprost intracameral implant; a minimum central corneal endothelial cell density based on age (2200 cells/mm2 for those who were 45 years, 2000 cells/mm2 for those 46–55 years, 1800 cells/mm2 for those 56–65 years, and 1600 cells/mm2 for those greater than 65 years of age); and central corneal thickness of ⩾440 and ⩽620 µm. Fellow eyes were required to have the best spectacle-corrected visual acuity (BSCVA) of at least 20/80 Snellen.
Key exclusion criteria specific to patients’ study eyes included severe glaucomatous visual field loss (mean deviation worse than −12 dB using 24-2 SITA Standard or equivalent on a Humphrey Visual Field Analyzer); vertical cup-to-disk ratio greater than 0.8; prior glaucoma filtering surgery, stent, or MIG device implantation; prior argon laser trabeculoplasty or selective laser trabeculoplasty; prior administration of an intracameral implant or drug product; active corneal inflammation or edema; prior corneal or endothelial transplantation; corneal opacities or disorders that prevent adequate visualization of the intended implantation site; or significant choroidal, retinal, or optic nerve disorders. In addition, patients were excluded if they had active or suspected ocular or periocular infection in either eye. Women of childbearing potential were excluded from participation if they were breast-feeding, pregnant, or planning a pregnancy.
At the baseline visit, patients were required to have an unmedicated mean diurnal IOP (average of the 8:00 am, 10:00 am, and 4:00 pm IOP measurements) of ⩾24 mmHg, and ⩽36 mmHg at each of the three timepoints. In addition, those who were on a topical PGA at screening were required to have an unmedicated IOP at least 4 mmHg higher than the time-matched IOP value at the screening visit.
Patients were excluded from participation at this visit if they failed to complete the appropriate IOP-lowering medication washout period, if applicable, or if they had an active or suspected ocular or periocular infection in either eye.
On the day of the procedure (day 1), patients were excluded from receiving a travoprost intracameral implant if they had complicated cataract surgery including but not limited to anterior chamber IOL implantation, capsulorhexis tear or rupture of the posterior capsule bag, choroidal effusion or hemorrhage, detached Descemet’s membrane, zonular weakness, dehiscence or rupture, prolonged anterior chamber collapse, significant damage to the cornea, iris or anterior chamber, vitreous loss or vitrectomy, significant hyphema, and incomplete phacoemulsification or complications associated with posterior chamber IOL implantation.
A travoprost intracameral implant was then administered to all those with uncomplicated phacoemulsification and successful receipt of a posterior chamber IOL.
Study visits and procedures
Patients were assessed at seven visits: screening, baseline (a minimum of 1 day after screening if no washout was required, to 6 weeks later if the longest washout from prior IOP-lowering medication(s) was required), day 1 (day of the procedure, 3–30 days post-baseline), day 2–3, week 2, week 6, and month 3.
Study-related procedures included a pregnancy test for women of childbearing potential (at baseline), manifest refraction (at screening, week 6, month 3), Snellen BSCVA (at screening), pinhole Snellen visual acuity (VA) (day 2–3, week 2), Early Treatment Diabetic Retinopathy Study (ETDRS) BSCVA (baseline, week 6, month 3), slit-lamp biomicroscopy (at screening, baseline, day 2–3, week 2, week 6, month 3), conjunctival hyperemia assessment (at baseline, week 2, week 6, month 3), specular microscopy (at screening), gonioscopy (at screening, day 2–3, week 2, week 6, month 3), dilated ophthalmoscopy including assessment of cup-to-disk ratio (screening, month 3), pachymetry (at screening), and perimetry (at screening, if not conducted in the previous 3 months).
In addition, tonometry was performed at 8:00 am ± 30 minutes or 10:00 am ± 30 minutes at screening; at a single timepoint (only 8:00 am ± 30 minutes) on days 2–3, week 2, and week 6; and at 8:00 am ± 30 minutes, 10:00 am ± 30 minutes, and 4:00 pm ± 30 minutes at baseline and month 3. IOP was measured with a Goldmann tonometer using a two-person technique common to glaucoma clinical studies, in which one person looks through the slit lamp and turns the dial (with the dial readings masked), and a second person records the readings. 21
Grading scales were used to evaluate anterior chamber cells and flare, as well as conjunctival hyperemia. Anterior chamber cells were measured at the slit lamp and scored on a 6-point scale: grade 0 for <1 cell; grade 0.5 = 1–5 cells, grade 1 = 6–15 cells; grade 2 = 16–25 cells; grade 3 = 26–50 cells; and grade 4 = more than 50 cells. Anterior chamber flare was scored on a 5-point scale: grade 0 = none; grade 1 = faint; grade 2 = moderate; grade 3 = marked; and grade 4 = intense. Conjunctival hyperemia was assessed at the slit lamp comparing the redness of the patient’s conjunctiva to photographic images, and scored on a 5-point scale; grade 0 = normal; grade 0.5 = trace; grade 1 = mild; grade 2 = moderate, and grade 3 = severe.
Surgical technique and perioperative medications
Prior to the cataract surgery and travoprost intracameral implant administration procedure, antibiotic eyedrops (levofloxacin 0.5%) were administered 4 times a day for at least 1 day.
Surgeries were performed under aseptic conditions in the hospital’s operating room. An additional drop of antibiotic was administered 30 minutes prior to surgery. The pupil was dilated with tropicamide, and topical anesthesia was administered. Patients underwent conventional (manual, small clear corneal incision) phacoemulsification surgery, with receipt of a hydrophobic foldable posterior chamber IOL preloaded in an insertion device.
Upon completion of the cataract surgery, the angle was inspected with a gonioprism and additional viscoelastic was injected into the anterior chamber. The travoprost intracameral implant, which is preloaded on a biocompatible single-use inserter, was advanced into the anterior chamber using the same temporal clear corneal incision used for phacoemulsification. The implant was inserted through the trabecular meshwork and anchored into the sclera on the nasal side. After withdrawing the inserter from the eye, the anterior chamber was irrigated and aspirated with a balanced salt solution to remove all viscoelastic, and the anterior chamber was inflated with saline solution as needed to achieve physiologic pressure.
For 1 week following the combined procedure, patients continued to administer their antibiotic eyedrops and also administered non-steroidal anti-inflammatory (ketorolac 0.5%) eyedrops.
Sample size and methods of analysis
Assuming a two-sided alpha = 0.05, a true mean change from baseline in mean diurnal IOP of −6.5 mmHg, and a common standard deviation (SD) of 3.5 mmHg at the month 3 visit, a sample size of at least 50 patients was determined to have greater than 90% power to demonstrate a statistically significant change from baseline in mean diurnal IOP.
The treatment effect of the combined procedure was assessed as the change from baseline in mean diurnal IOP (average of the 8:00 am, 10:00 am, and 4:00 pm IOP measurements) at the month 3 visit and was analyzed using a linear model with baseline mean diurnal IOP as a covariate for month 3. The mean change from baseline was tested against 0 using a least square (LS) means two-sided t test p value and 95% confidence interval (CI).
The primary analyses were performed on the intention-to-treat (ITT) population which included all treated patients. In this analysis, missing data as a result of withdrawal due to lack of efficacy, adverse events, or receipt of additional IOP-lowering medication(s) were multiply imputed using regression methodology on the worse half of patients, whereas missing data without withdrawal or withdrawal due to reasons other than lack of efficacy or adverse events were multiply imputed using randomized treatment-based Markov Chain Monte Carlo (MCMC) methodology to impute non-monotone missing data and using regression methodology to impute monotone missing data.
Sensitivity analyses to determine the robustness of the primary analysis of the primary efficacy endpoint were performed using various methodologies to impute missing data, as well as using observed cases. In the first sensitivity analysis, all missing data and data after intercurrent events were assumed to be missing at random with missing data multiply imputed using MCMC methodology to impute non-monotone missing data to produce a monotone missing data pattern and using regression methodology to impute monotone missing data. In the second sensitivity analysis, missing data without withdrawal were imputed using MCMC methodology, and data after intercurrent events were assumed to be missing not at random and were imputed using worse-half regression methodology. Finally, in the third sensitivity analysis, missing data or data after an intercurrent event for IOP were not imputed; rather, the analysis was based on the observed case.
Additional analyses, performed on the ITT population, included the percentage of patients with ⩾20%, 25%, 30%, and 35% reduction from baseline in mean diurnal IOP and the percentage of patients with mean diurnal IOP ⩽14, 16, 18, and 20 mmHg at month 3. For these analyses, multiple imputed values from the primary analysis of the primary endpoint were used to determine the percentage change from baseline.
Safety analyses were performed on the safety population which included all treated patients. Adverse events were coded using the Medical Dictionary for Regulatory Activities. Any adverse event that occurred on/after the initial treatment was considered a treatment-emergent adverse event; only treatment-emergent adverse events are reported herein. Other ocular safety data included visual acuity, slit-lamp biomicroscopy, conjunctival hyperemia assessment, dilated ophthalmoscopy (including vertical cup-to-disk ratio), and gonioscopy.
Results
Disposition
A total of 74 patients were screened between September and December 2023 for participation in the trial, of whom 14 failed to meet eligibility criteria, primarily due to their inability to commit to attending follow-up visits or to failure to provide a reliable and interpretable visual field.
All 60 patients who met the inclusion criteria and none of the exclusion criteria, successfully underwent the combined procedure (i.e., phacoemulsification with the implantation of a posterior chamber IOL and administration of a travoprost intracameral implant). In addition, all 60 patients completed month 3 of the trial, and were included in the ITT and safety populations.
Patient demographics and baseline ocular characteristics
All patients were White, and 61.7% (37 of 60) were female. The mean (±SD) age of the patient population was 67.0 ± 6.60 years, with 58.3% (35 of 60) aged 65 years or older.
Baseline ocular characteristics are shown in Table 1. All 60 patients were diagnosed with OAG, including one with pseudoexfoliative glaucoma. Thirty-five patients (58.3%) were on 1 or 2 IOP-lowering medication classes at screening, of whom 20 (33.3%) were on a CAI, 19 (31.7%) were on a beta blocker, 10 (16.7%) were on a PGA (all of whom were on tafluprost), and 3 (5.0%) were on an alpha 2-agonist.
Ocular baseline characteristics.

One patient had pseudoexfoliative glaucoma.
ETDRS, Early Treatment Diabetic Retinopathy Study; IOP, intraocular pressure; PGA, prostaglandin analog; SD, standard deviation.
Mean IOP, measured at 8:00 am (±30 minutes) or 10:00 am (±30 minutes) at screening was 19.1 ± 5.13 mmHg. Unmedicated baseline mean diurnal IOP (average of the 8:00 am, 10:00 am, and 4:00 pm IOP measurements) was 25.2 ± 1.07 mmHg.
Efficacy
A statistically significant and clinically relevant change from baseline in mean diurnal IOP of −10.6 mmHg (95% CI: −11.2, −9.9; p < 0.0001) was observed at month 3 (Figure 1). Mean diurnal IOP was 25.2 ± 1.07 mmHg at baseline and decreased to a mean of 14.6 ± 2.53 mmHg at month 3. Since only small numbers of patients (1 at week 2, and 2 at each of week 6 and month 3) experienced an intercurrent event (i.e., used additional IOP-lowering medication), mean diurnal IOP reductions from baseline in the various sensitivity analyses were similar to the primary analysis of the primary endpoint.

Mean IOP at screening (while patients were on pre-study IOP-lowering medication, if applicable), baseline (following a washout period from pre-study medication), and month 3 (following cataract surgery and travoprost intracameral implant administration). At screening, IOP was measured at a single time of day (8:00 am or 10:00 am). At baseline and month 3, IOP represents a diurnal assessment (based on 8:00 am, 10:00 am, and 4:00 pm measurements). Error bars represent standard deviation.
The month 3 reduction from baseline in mean diurnal IOP was statistically significant and clinically relevant in categories based on sex (male, female), age (<65, ⩾65 years), baseline unmedicated mean diurnal IOP (<26, ⩾26 mmHg), number of IOP-lowering medications at screening (0 or 1, 2), and in the subgroup of patients on a PGA at screening (Table 2).
Mean change from baseline in mean diurnal IOP (mmHg) at month 3 for the overall study population and by subgroup.
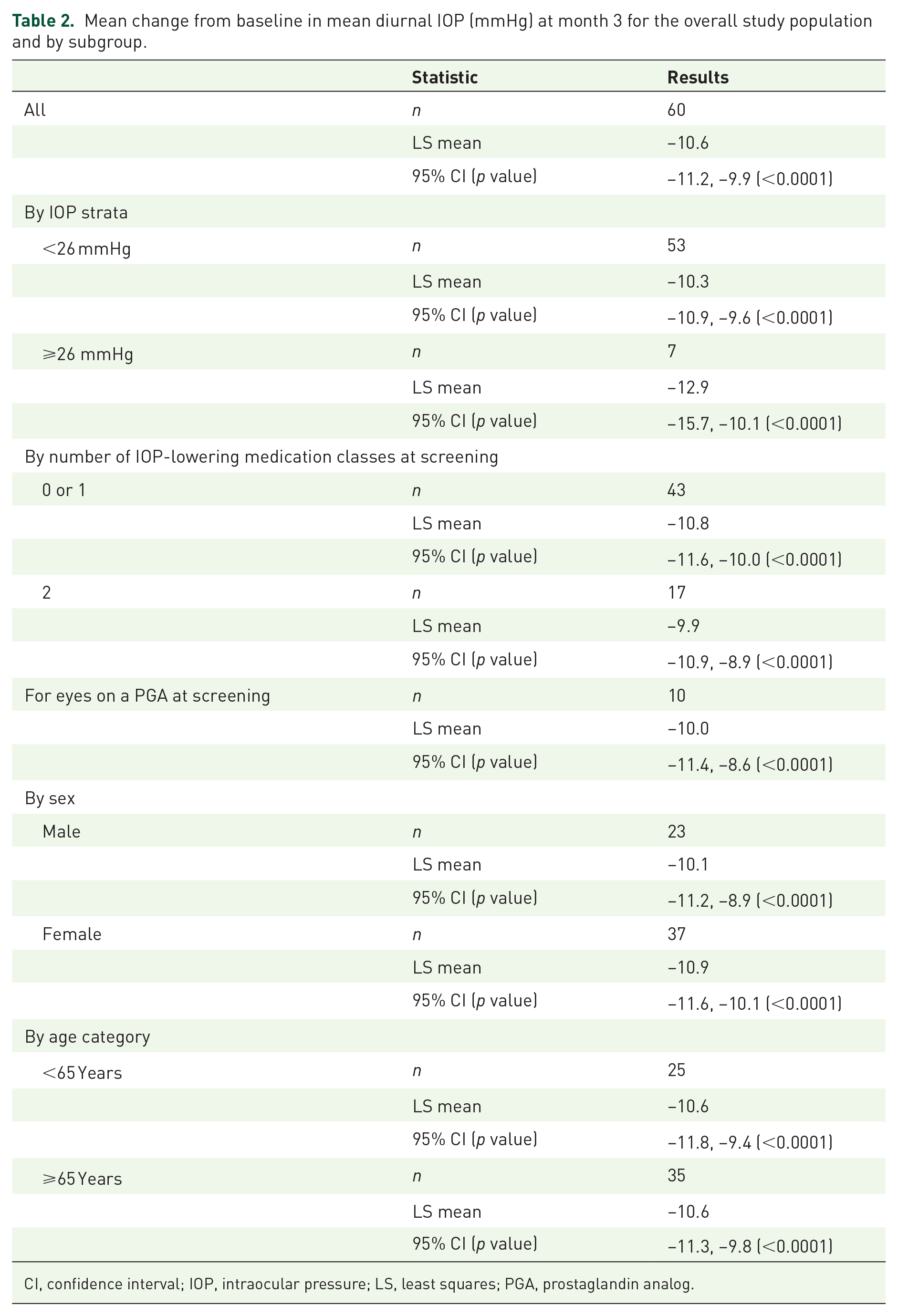
CI, confidence interval; IOP, intraocular pressure; LS, least squares; PGA, prostaglandin analog.
In addition, the month 3 reduction from baseline in mean diurnal IOP was numerically similar in males and females, as well as in those <65 and ⩾65 years, whereas numerically greater reductions from baseline were observed in subgroup of patients in the ⩾26 mmHg stratum versus <26 mmHg stratum, and in the subgroup of patients on 0–1 IOP-lowering medication classes at screening versus those on two medication classes at screening (Table 2).
At month 3, 96.7% of patients achieved a ⩾20% reduction from baseline in mean diurnal IOP (Figure 2) and 91.0% of patients achieved a mean diurnal IOP of ⩽18 mmHg (Figure 3).

Percentages of eyes achieving mean diurnal IOP reductions from baseline of ⩾20%, ⩾25%, ⩾30%, or ⩾35% at month 3. Error bars represent upper 95% confidence interval.

Percentages of eyes achieving mean diurnal IOP values of ⩽20, ⩽18, ⩽16, or ⩽14 mmHg at month 3. Error bars represent upper 95% confidence interval.
The 8:00 am LS mean IOP change from baseline at the week 2, week 6, and month 3 visits is shown in Figure 4. The reduction from baseline was sustained over the 3-month evaluation period.

8:00 am IOP change from baseline across visits. Error bars represent 95% confidence intervals.
Prior to surgery, 58.3% of patients (35 of 60) were on one or more topical IOP-lowering medications. At the month 3 visit, only 3.3% of patients (2 of 60) were using a topical IOP-lowering medication (a topical CAI); these 2 patients had a mean diurnal IOP of 12.5 and 11.5 mmHg.
Safety
Adverse events
A summary of adverse events and display of adverse events in the study eye is presented in Table 3.
Summary of adverse events and adverse events in the study eye.

All adverse events were mild, with a single patient experiencing an adverse event (hyphaema) that was related to study treatment. Hyphaema was observed the day following the procedure, was mild, and had resolved by the subsequent scheduled visit (week 6).
An adverse event of conjunctival hyperemia was reported in the study eye of a single patient. At the baseline visit, the conjunctiva had a normal (grade 0) appearance. At the week 2 visit, mild hyperemia (grade 1) was observed, and by the week 6 visit, the conjunctiva returned to normal (grade 0). Adverse events of dry eye were reported in the study eye of four patients, of whom three also had dry eye in the fellow eye. These patients were treated with ocular lubricants.
Other safety assessments
On day 2–3 following the combined procedure, most patients had grade 0 (<1 cell) anterior chamber cells, with the remaining 30% (18 of 60) having grade 0.5+ (1–5 cells) anterior chamber cells. Moderate corneal edema was observed in a single patient (1 of 60, 1.7%) at day 2–3; 13.3% (8 of 60) had mild corneal edema, and the remaining 85% (51 of 60) had no corneal edema. Anterior chamber cells and corneal edema were resolved in all patients by the week 2 visit. There were no observations of epithelial abnormalities, endothelial folds, corneal opacification, anterior chamber flare, or posterior capsule opacification at any follow-up visit.
Mean BSCVA was 81.1 ± 3.65 ETDRS letters at the month 3 visit, with four patients (6.7%) having a worsening of five ETDRS letters from baseline for which no discernable cause was observed via slit-lamp biomicroscopy or dilated ophthalmoscopy, and no patient had a worsening of more than five letters.
There were no abnormal findings of the macula, retinal periphery, retinal vessels, or vitreous on dilated ophthalmoscopy in any patient at the month 3 visit. In addition, gonioscopy confirmed the implant to be anchored and in its original position in 100% of patients at each follow-up visit. Optical coherence tomography (OCT) of the macula was performed only if there was clinical concern about vision impairment or ophthalmoscopic macular findings.
At the baseline visit, conjunctival hyperemia was graded as normal in all patients. No patient had moderate or severe hyperemia at any follow-up visit. Trace hyperemia was observed in one patient (1.7%) and mild hyperemia in five patients (8.3%) at the week 2 visit, whereas a single patient was observed to have trace hyperemia at the month 3 visit.
An IOP elevation resulting in the use of adjunctive IOP-lowering medication occurred in two patients. One patient experienced a time-matched reduction of only 1 mmHg from baseline at study day 2 (i.e., from 25 to 24 mmHg at 8:00 am) and was prescribed topical dorzolamide TID; subsequent IOP reductions week 2 through month 3 visits ranged from 9 to 12 mmHg. The second patient experienced a time-matched increase from baseline of 2.5 mmHg at the week 2 visit (i.e., from 24 to 26.5 mmHg at 8:00 am) and was prescribed dorzolamide BID, resulting in an IOP reduction from baseline of 6 mmHg at the week 6 visit and 12 mmHg at the month 3 visit.
Discussion
This is the first study evaluating the safety and IOP-lowering effect of travoprost intracameral implant administration in combination with cataract surgery.
The study demonstrated statistically significant and clinically relevant IOP treatment effect at month 3, with a −10.6 mmHg IOP change from baseline in mean diurnal IOP. Few studies have reported on the early (month 3) IOP reductions in patients with OAG following phacoemulsification cataract surgery. Devience et al. reported a 2.5 ± 3.8 mmHg (corresponding to a 13.6% ± 25.0%) reduction 3 months following phacoemulsification cataract surgery in 90 patients with a mean preoperative IOP of 16.1 ± 3.0 mmHg. 5 Fakhraie et al. reported a 4.0 ± 3.2 mmHg IOP reduction (from a baseline of 19 ± 4 mmHg) 3 months following cataract surgery in OAG patients treated with latanoprost. 22 In contrast to our study, patients in the Devience and Fakhraie studies were maintained on their pre-study IOP-lowering medications.
Significant IOP reductions were observed in the overall patient population, as well as in subgroups based on sex, age category, baseline IOP strata, categories based on number of IOP-lowering medication classes at screening, and in the subgroup of patients on a PGA at screening. The clinical relevance of the IOP reductions also was supported by the high percentage of patients who achieved an IOP reduction of 20% or greater and patients with an IOP of 18 mmHg or less.
The −10.6 mmHg mean diurnal IOP change from baseline at month 3 observed in our study is greater than the approximately −6.6 mmHg change from baseline at month 3 reported by Sarkisian et al. in patients who received a travoprost intracameral implant. 18 These differences may be explained by the additional IOP-lowering effect of cataract surgery and the slightly higher baseline unmedicated diurnal IOP in the current study (25.2 mmHg vs 24.0 mmHg).
The combined procedure of travoprost intracameral implant administration and cataract surgery did not result in any serious adverse events; there was a single treatment-related adverse event (hyphaema), which was mild and resolved. There were no reports of pseudophakic cystoid macular edema (CME) during the 3-month follow-up in our study. Clinically significant CME affects approximately 1%–2% of patients following cataract surgery,23,24 with risk factors, including diabetes mellitus and complicated cataract surgery. 25 Preoperative use of PGA eye drops has not been found to increase the risk for pseudophakic CME 24 ; however, the development of CME with postoperative use of a PGA is controversial. Some studies report that postoperative use of PGA eye drops results in a greater incidence of pseudophakic CME whereas others have found no association between CME and PGA use following uncomplicated cataract surgery.22,24–26 In our study, patients with complicated cataract surgery were not eligible to receive a travoprost intracameral implant, and a single patient (1.7%) had a history of diabetes mellitus. There were no reports of persistent corneal edema or iritis, and only two patients (3.3%) had elevated IOP in the early postoperative period which was treated successfully with a topical CAI.
The 6.7% occurrence of dry eye observed in our study is similar to that reported in the trials conducted for the U.S. approval of the travoprost intracameral implant,17–19 and substantially less than the 37.4% of patients without preexisting dry eye who were reported in a meta-analysis to develop the condition following cataract surgery. 27
We acknowledge that there were several limitations to the study. The study was designed as a single-arm, open-label trial given the inherent difficulty in masking the surgeon and the observer conducting safety assessments (e.g., slit-lamp biomicroscopy, gonioscopy, and ophthalmoscopy). A second limitation is that the study was conducted at a single site. However, surgeries were performed by four different experienced surgeons. A third limitation was that all patients were White which is not fully representative of the demographic of patients with OAG and may have decreased the likelihood of observing postoperative safety findings since studies have shown that Black race is associated with a higher risk of postoperative IOP spike following cataract surgery, 28 and the study included only a single patient with diabetes, a comorbidity associated with increased risk of persistent postoperative inflammation. 29 A fourth limitation may be that although a clinical examination was performed at month 3, a more detailed evaluation of the macula via OCT or fluorescein angiography was not performed. However, no patient presented with decreased or blurred vision suggestive of pseudophakic CME. Yet another limitation may be the lack of endothelial cell density measurement at the month 3 visit. Cataract surgery has been reported to result in a loss of endothelial cell density, with the most substantial decline (11.6%) occurring within the first 3 months postoperatively. 7 A substantially smaller decline (1.9%) has been observed 3 months following administration of the travoprost intracameral implant. 18 A final limitation may be that the current analysis is only 3 months in duration. However, the study remains ongoing through 12 months to determine the longer-term safety and efficacy of administering the travoprost intracameral implant in conjunction with cataract surgery.
Conclusion
Travoprost intracameral implant administration in combination with phacoemulsification cataract surgery is safe, and results in a robust −10.6 mmHg treatment effect at 3 months which was both statistically significant and clinically relevant.
