Abstract
Introduction:
Epiretinal membrane (ERM) formation following a successful rhegmatogenous retinal detachment (RRD) operation has been reported to occur in about 4%–15% of the cases. In this study, we collected vitreous samples from patients with RRD to identify whether the concentration of IL-6 is correlated with the presence of postoperative ERM. We aim is to identify whether the inflammatory cascade could be a potential key factor in the ERM pathogenesis.
Methods:
This was a prospective single-centre study where 42 eyes were enrolled. All patients underwent 25G PPV. The vitreous sample was collected before the infusion line was opened so that the sample would not be diluted. Then, the sample was centrifuged at 5000 g for 15 min at 15°C. Afterwards, the supernatant fluid was transferred to an Eppendorf tube and stored at −40°C. Electrochemiluminescence immunoassay (ECLIA) was employed for the measurement of IL-6 concentration (pg/ml). All patients had optical coherence tomography (OCT) scans at the macula at 4 weeks, 3 months and 1 year after primary RRD repair to identify the presence of the ERM.
Results:
All patients had a follow-up of at least 12 months. The mean BCVA of all 42 eyes at the end of follow-up period was 0.24 ± 0.31 LogMar. The presence of ERM was identified in 28.6% (n = 12) of the patients, and the mean IL-6 concentration was 573.96 ± 1179.58 pg/ml. It appears that the patients who developed a post op ERM had an IL-6 mean concentration value of 173.96 ± 191.25 pg/ml, and the patients with no post op ERM had 733.97 ± 1364.04 pg/ml with p-value = 0.04 < 0.05. Nevertheless, following a multivariate binary logistic regression model, the above results did not appear to be statistically significant.
Conclusion:
High concentration of IL-6 in the vitreous of eyes with RRD does not hold a significant role in the ERM pathogenesis. Our study identified the presence of a draining retinotomy as the most significant risk factor for ERM formation following a successful surgical RRD repair.
Keywords
Introduction
Rhegmatogenous retinal detachment (RRD) is a sight-threatening condition which constitutes a critical cause of irreversible visual impairment if left untreated. 1 The mechanism involves syneretic vitreous outflow into the subretinal space through a retinal tear or hole which separates the neurosensory retina from the retinal pigment epithelium (RPE). 2 This condition leads to photoreceptors homeostasis disruption with subsequent cellular death, occasionally prohibiting functional improvement even after successful surgical repair. 3
Pars plana vitrectomy (PPV) is an efficacious surgical procedure for the treatment of RRD with significant anatomical success rates. A relatively common sequelae following an uneventful RRD repair is the formation of epiretinal membrane (ERM). The incidence of ERM formation following PPV has been reported between 4% and 15%, while the incidence of PPV for removal of ERM ranges from 2.8% to 4.3%. 4 ERM is primarily composed of RPE cells, hyalocytes and retinal glia; interestingly, it has been speculated that its formation following RRD repair could constitute an early stage of proliferative vitreoretinopathy (PVR). Nevertheless, the exact pathogenesis of the disorder has not been fully understood yet. 5
Several studies have demonstrated an activation of both immune and inflammatory systems in the process of RRD. Indeed, elevated levels of cytokines, chemokines and growth factors into the vitreous cavity and subretinal space have been identified following RRD. Throughout literature, there is suspicion and evidence of a relationship between the aforementioned inflammatory mediators and the formation of PVR which constitutes the most common cause for RRD recurrence and postoperative visual impairment. 6 One of these factors is interleukin 6 (IL-6), while its expression and activity have been reported in eyes with RRD, either with or without PVR.
IL-6 is a pleiotropic cytokine with a regulatory activity for several chemokines. It is known to be a trigger of the acute phase of inflammation and also acts as a warning signal for tissue damage, such as a retinal tear. 7 In eyes, IL-6 can be produced by both RPE cells and inflammatory cells in the subretinal space through the chemotactic signalling. As a result, IL-6 promotes the expression of fibroblasts and glial cells. 8 Additionally, IL-6 has been identified in the vitreous of patients with RRD, while elevated levels have been linked with the presence of PVR. Despite the lack of our understanding of the exact role of IL-6 in PVR formation, it has been reported that several cells found in PVR membranes may produce IL-6. 9
In view of the above, we sought to investigate whether patients with RRD and elevated levels of IL-6 in the vitreous cavity were at higher risk of developing ERM following PPV. This study aimed to characterize a potential correlation between the presence of IL-6 and the formation of ERM within 1 year of successful RRD repair.
Methods
The study was conducted at the ‘Attikon’ University Hospital of Athens, between 2018 and 2020.
This was a prospective single-centre study. Inclusion criteria were patients over the age of 18 years with primary RRD who underwent a single successful PPV. Patients were included regardless of the status of the macula to detachment. Exclusion criteria included history of uveitis, glaucoma, previous intraocular surgery within the past 6 months, diabetic retinopathy, genetic retinal dystrophies and vascular occlusion. In addition, patients were excluded if they had previous or concomitant treatment with oral steroids and previous anti-vascular endothelial growth factor or steroid injections.
At baseline, a comprehensive ocular examination was performed. Subsequently, patients were divided into three groups based on the duration of the retinal detachment. The latter was confirmed by evaluating the clinical findings with the duration of symptoms that patients disclosed. Our groups included patients with retinal detachment less than 11 days (Group A), between 11 and 30 days (Group B) and more than 30 days (Group C).
All patients underwent 25G PPV with cryotherapy or endolaser to the identified retinal breaks and gas tamponade with either sulphur hexafluoride (SF6) or perfluoropropane (C3F8). Silicone oil was not used as a tamponade agent in any of the cases. All operations were performed by one experienced vitreoretinal consultant surgeon. The vitreous sample collection was performed as follows: following trocar insertion, each sample was collected through the cutter in a 5-ml syringe before the infusion line was opened so that the sample would not be diluted. Then, the sample was transferred to an Eppendorf tube and centrifuged at 5000 g for 15 min at 15°C. Following this procedure, the supernatant fluid was transferred to a new Eppendorf tube and stored at −40°C. The electrochemiluminescence immunoassay (ECLIA) (ROCHE diagnostics, platform Cobas8000) was employed for the measurement of IL-6 concentration (pg/ml) according to the manufacturer’s instructions. Assay sensitivity for IL-6 was 1.5–5000 pg/ml.
All patients had regular follow-up appointments for 1 year. All patients had optical coherence tomography (OCT) scans at the macula at 4 weeks, 3 months and 1 year after the operation. The presence of ERM was defined as the presence of an epiretinal hyperreflective band with or without foveal sparring causing central foveal thickening. All patients who developed a recurrence of the retinal detachment were excluded from the study.
Except the duration of the RRD, other preoperational (pre-op) characteristics were analysed as well. Baseline characteristics are summarized in Table 1, and intraoperative characteristics are displayed in Table 2.
Participant baseline characteristics.

RRD, rhegmatogenous retinal detachment, SD, standard deviation.
Intraoperative characteristics.

Statistical analysis was performed using SPSS for Windows (version 24.0; SPSS, Chicago, IL, USA). T-test and logistic regression were performed to assess possible causes of post-PPV ERM formation.). P-values < 0.05 were considered statistically significant.
Results
In this study, we collected vitreous samples from 49 eyes with RRD during the process of PPV. Of the initial cohort, seven eyes were excluded; five developed re-detachment and two were lost from follow-up. Therefore, a total of 42 (42) eyes of 28 males and 14 females were analysed in this prospective study. The mean age was 63.8 years (range: 39–79 years). Concerning the macula status, 20 patients had an attached macula at the time of surgery, and 22 had a detached macula. Preoperative mean best corrected visual acuity (BCVA) of all participants was 1.26 ± 0.14LogMar. Twenty-one patients presented with RRD of duration less than 11 days, 16 patients between 11 and 30 days and 5 patients 30 days or more.
All patients had a follow-up of at least 12 months. The mean BCVA of all 42 eyes at the end of the follow-up period was 0.24 ± 0.31 LogMar. The presence of ERM was identified in 28.6% (n = 12) of the patients, and the mean IL-6 concentration was 573.96 ± 1179.58 pg/ml (Table 3).
Postoperative outcomes.

ERM, epiretinal membrane; SD, standard deviation.
To evaluate whether there is a correlation between increased concentration of IL-6 in the vitreous and ERM formation, we divided the patients into two sub-groups: a group with post-op ERM formation and a group with no post-op ERM formation. Next, the mean IL-6 concentration in the two sub-groups was calculated, and it is illustrated in Table 4.
Vitreous interleukin 6 concentration – univariate analysis.
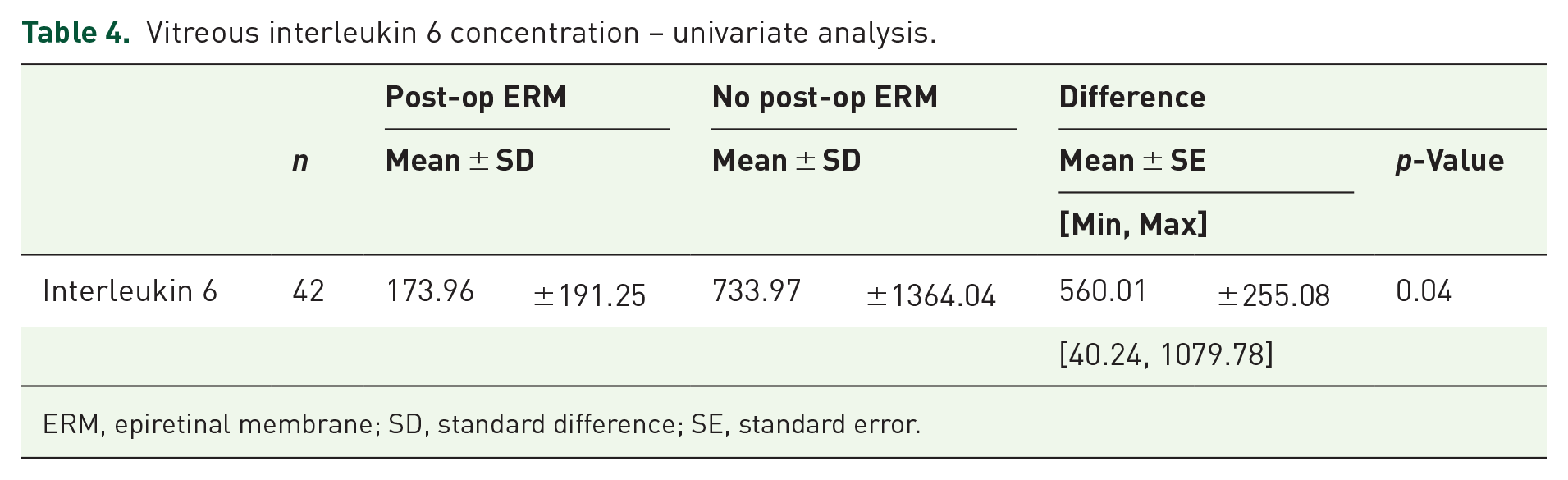
ERM, epiretinal membrane; SD, standard difference; SE, standard error.
It appears that the sub-group with the post-op ERM had an IL-6 mean concentration value of 173.96 ± 191.25 pg/ml, and the sub-group with no post-op ERM had 733.97 ± 1364.04 pg/ml with p-value = 0.04 < 0.05, which makes it statistically significant.
In regards of the correlation between the duration of the RRD and the presence of ERM, we found that ERM was present in 23.8% (n = 5) of patients in Group A, in 25% of patients in Group B (n = 4) and in 60% (n = 3) of patients in Group C (Figure 1). These results had a p value of 0.25 > 0.05, which makes them not statistically significant.
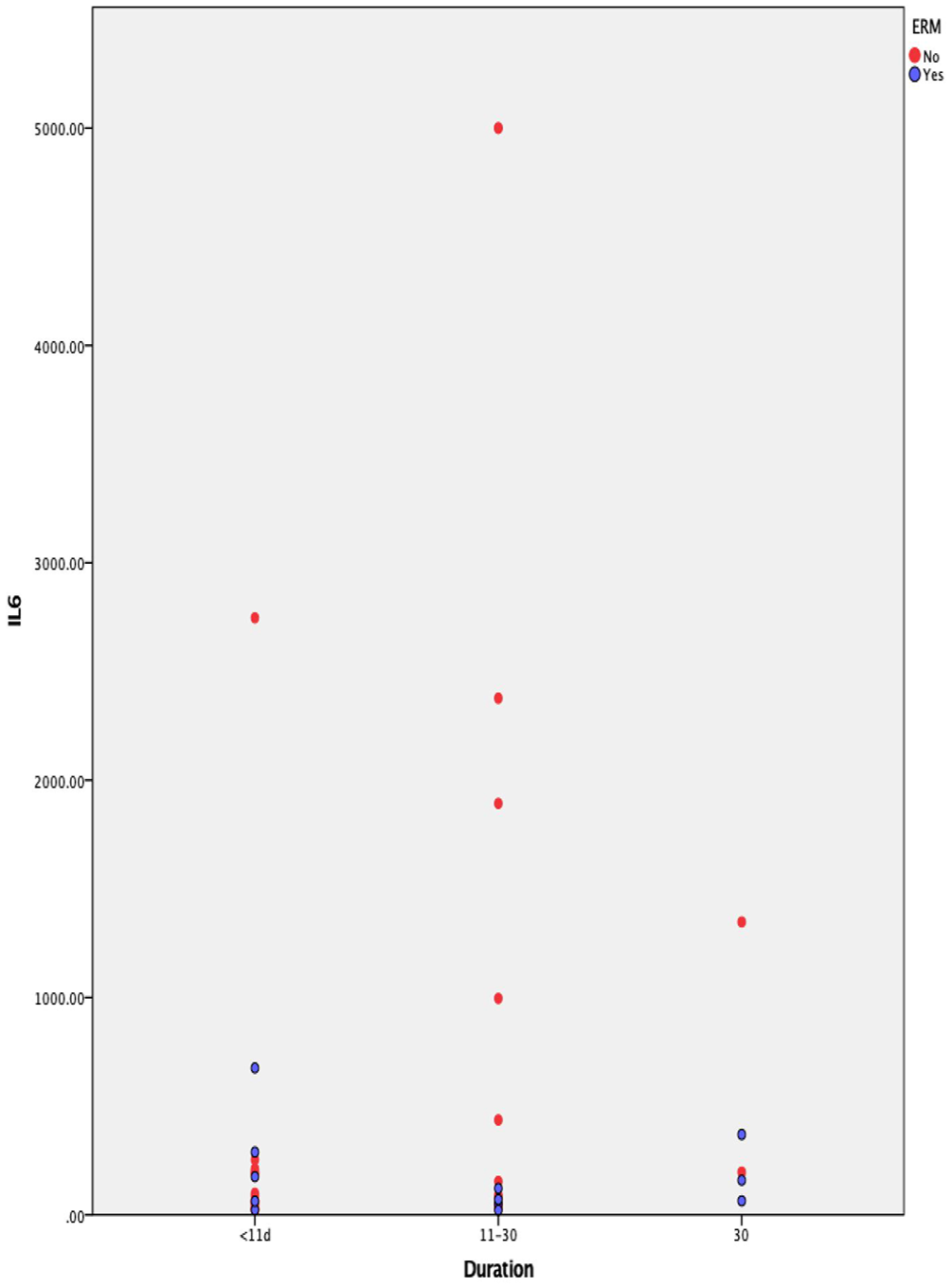
Distribution of vitreous IL-6 concentrations according to clinical sub-groups of retinal detachment.
Additionally, Group A had a mean IL-6 concentration value of 264.11 ± 128.30 pg/ml, Group B 1026.31 ± 426.37 pg/ml and Group C 427.82 ± 235.29 pg/ml with p = 0.14 > 0.05, which also makes it not statistically significant.
Finally, we constructed a multivariate binary logistic regression model including the following variables: Drainage retinotomy (yes/no), sex, macula status (on/off), vitreous haemorrhage at presentation, duration of the RRD, extension of the RRD, number of tears/holes, intraoperative use of triamcinolone, type of retinopexy (Cryopexy vs Endolaser vs Both), type of tamponade (SF6 vs C3F8), location of tears (superior equator vs inferior equator vs both) and IL-6 concentration.
After excluding the not statistically significant variables, we found that only the drainage retinotomy was a statistically significant risk factor for the postoperative ERM formation, and it was associated with a seven-fold risk, 95% confidence interval (1.08, 45.44), p = 0.04 < 0.05 (Figure 2).
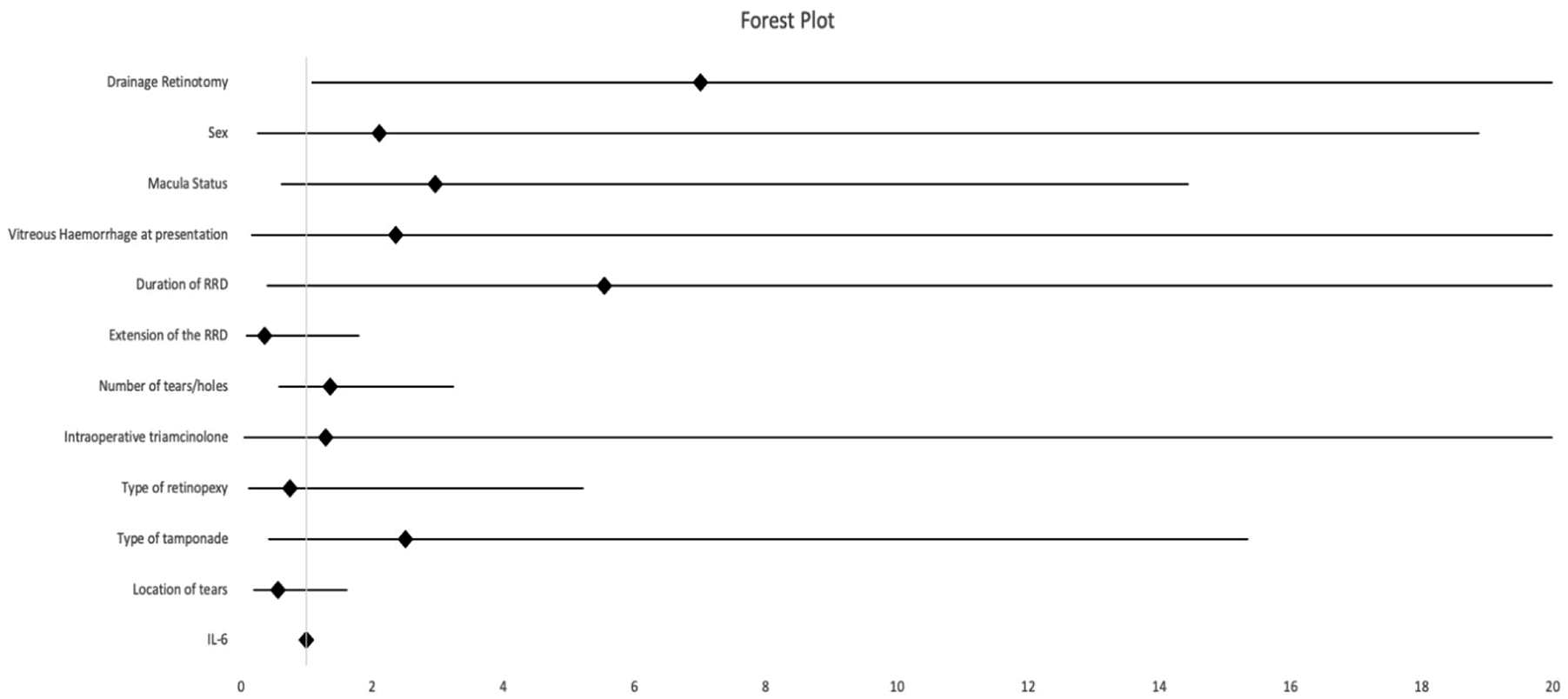
Forest plot showing odds ratios (OR) and 95% confidence intervals for variables included in the logistic regression model assessing risk factors for ERM formation.
Discussion
It is well established that rhegmatogenous retinal detachment ignites a pathological upregulation of interleukins which regulate a localized low-grade inflammatory reaction in addition to wound healing. 11 A number of studies have supported that levels of IL-6 in RRD eyes have been significantly higher than in healthy eyes, with even more increased levels in those complicated by PVR. To the best of our knowledge, the association of IL-6 values with the presence of ERM in patients who underwent RRD repair has not been evaluated yet.
In this study, the incidence of ERM following successful PPV repair was 28.6%, which is higher than what has been previously reported. This difference could be potentially explained by the fact that, in addition to clinically significant ERM cases, patients with asymptomatic ERM were also included in this study, while previous studies mainly focused on the assessment of patients with symptomatic ERM. 16
Concerning the association between the duration of the RD and the presence of an ERM, we performed a univariate analysis which showed that patients with more than 30 days duration of RRD (Group C) had 80% presence of ERM comparing with 31% in Group A (<10 days) and 33% in group B (11–30 days). Despite these results were not statistically significant, they were in line with previous findings, which suggested that the longer duration of detached macula revealed a higher possibility of postoperative ERM formation. 17 Therefore, we speculate that a potentially larger patient sample could enable significant outcomes and shed more light on this association.
According to our univariate analysis, the sub-group of patients who developed ERM during the 12-month follow-up period displayed a lower mean concentration value of IL-6 as compared to the sub-group that did not develop ERM. At first glance, this result appears counterintuitive, especially considering IL-6’s known pro-inflammatory properties. However, this association did not persist after adjusting for other variables in a multivariate regression model. This suggests that the observed difference in IL-6 levels may have been confounded by other factors – most notably the presence of a drainage retinotomy, which emerged as the only independent predictor of postoperative ERM formation. Therefore, despite our study enabling this outcome, we could not support that IL-6 might have a protective role against ERM formation, given the properties of IL-6 and its role in inflammation.
Interestingly, our findings might stir controversy since a potential association between cytokines and ERM pathogenesis has been previously reported. 18 More specifically, it is implicated that Müller glia and macrophages trigger a fibrotic activity by releasing specific soluble factors like tissue growth factor beta (TGF-β), IL-4, IL-6, IL-13, and platelet-derived growth factor (PDGF)18,19. In addition, increased levels of IL-6 have been found in eyes with idiopathic ERM compared to eyes with macular holes. The authors suggest that an inflammatory and profibrotic cascade is implicated in the ERM pathogenesis and imply that patients with idiopathic ERM should be cautiously considered as a healthy control group. 20 Our results highlight the importance of accounting for potential confounders when evaluating the role of inflammatory markers in ERM development. While elevated IL-6 has been implicated in proliferative vitreoretinopathy (PVR) and idiopathic ERM, our study suggests that preoperative IL-6 concentration in eyes with rhegmatogenous retinal detachment (RRD) alone may not independently influence postoperative ERM formation.
However, in contrast with the previously mentioned studies where the samples were collected from patients with an already present ERM, our patients did not have an ERM at the time of presentation. Also, the levels of IL-6 in the vitreous cavity following the surgical intervention could not be directly assessed, as this would require another invasive procedure. Consequently, since our regression analysis results do not show a statistically significant correlation between the pre-op mean concentration of IL-6 in eyes with RRD and the development of an ERM at the post-op period, we assume that the inflammatory state of an eye with RRD before the operation is unlikely to contribute to the formation of an ERM.
Additionally, according to our multivariate analysis, the only factor that showed to be linked with a significant risk for the formation of ERM was the presence of a drainage retinotomy. This finding is in line with previous reports from other studies. Additionally, it has been documented that the larger size of retinotomy has been associated with a higher risk of ERM development. It is a hypothesis that the pathogenesis that links ERM formation and drainage retinotomy may be associated with retinal-choroid-vasculature damage, followed by glial cell activation and migration of RPE and inflammatory cells on the retinal surface, subsequently resulting in fibrous membrane formation.12,13
In another study by Qian et al., they calculated that 36.2% of patients with complicated primary RRD that needed tamponade with silicone oil formed an ERM during the follow-up. They concluded that the risk factors that were associated with the formation of the ERM were preoperative PVR, preoperative choroidal detachment and photocoagulation energy. 14 In addition, a retrospective study by Hirakata et al. evaluated the risk of ERM formation in 226 patients that had RRD surgical repair using a multivariate regression analysis protocol. They concluded that pre-op vitreous haemorrhage (VH), multiple retinal breaks, re-detachment and RRD extension were significantly associated with post-op ERM. 21 In our study, no preoperational choroidal detachment and PVR was reported and there was no statistically important difference in patients who had cryopexy versus laser retinopexy versus both to whether they will form an ERM. Furthermore, the patients who failed to adhere to the primary repair were excluded from the study. Lastly, our multivariate regression analysis failed to show statistically significant relationship between ERM formation and the number of breaks or the presence of pre-op VH or the extension of the RRD. In our opinion, the smaller sample size of our study, together with the limitations of retrospective studies, could possibly excuse the non-compliance of our findings with the aforementioned studies.
Finally, a meta-analysis from Xu et al. investigated the role of steroids after RRD surgical repair. Throughout the articles they reviewed, they observed that there was no significant difference in the incidence of epiretinal membrane between patients receiving steroids and patients who received placebo. 15 This finding supports our theory that even though PVR and ERM might be associated, inflammation does not play a pivotal role in the pathological cascade of the ERM formation.
Our study has several limitations. First, IL-6 levels were measured only at the beginning of surgery and not during postoperative follow-up. As such, we were unable to capture dynamic changes in cytokine levels that may occur in response to surgical trauma or during retinal healing. Therefore, we cannot exclude the possibility that post-surgical elevations in IL-6 – independent of the preoperative baseline – may contribute to the development of postoperative epiretinal membrane. Even though further studies assessing longitudinal IL-6 levels at multiple postoperative timepoints would be valuable in clarifying its temporal relationship with ERM formation, we understand that such a task would face many challenges. Additionally, while we recorded the presence of a drainage retinotomy, we did not document its size or location. This limits our ability to investigate whether more extensive retinotomies could be associated with increased IL-6 levels and, consequently, with a heightened risk of postoperative membrane formation.
Conclusion
According to our findings, the level of IL-6 in eyes with RRD does not play a significant role in the ERM pathogenesis during the post-op period. Nevertheless, as previously mentioned, few studies correlate inflammatory cytokines with the presence of an ERM. Therefore, given that the literature on this topic is not extensive, we believe that further investigation with probably larger number of participants could produce more information in regards of the role of the inflammatory cascade in the formation of an ERM following a successful RRD repair.
