Abstract
Keywords
Introduction
Macular holes (MHs) are a rare condition characterized by a full-thickness defect in the macula that results in loss of central vision. Gass was the first to propose that MHs form as a result of vitreoretinal traction, and optical coherence tomography (OCT) has since confirmed this mechanism.1,2 Since these early descriptions by Gass, it has become accepted that anterior-posterior vitreoretinal traction and tangential traction are responsible for MH formation. Kelly and Wendel proposed surgical treatment of full-thickness MH by removing the cortical vitreous, peeling membranes, and performing a fluid-gas exchange. This procedure, first described in 1989, further supported the role of tractional forces in the formation of MH.3,4 However, this accepted mechanism has come into question, as multiple reports of patients developing MH after both complete posterior vitreous detachment (PVD) and pars plana vitrectomy (PPV) have been reported.1,5–9
MH development after PPV is exceedingly rare, with an incidence of about 1%, 10 though limited evidence hinder accurate prevalence reports. The first cases of MH formation after PPV for retinal detachment (RD) were reported in 1988. 1 Since that time, several studies and small retrospective case series have described MH development after RD repair from pneumatic retinopexy, 11 PPV, 12 and/or scleral buckling.13,14 MH after RD repair is rare, with an estimated incidence of less than 1%. 15 Given its rarity, there has been little consensus on the mechanism and risk factors for MH development. One case series found that all eyes with MH had developed epiretinal membranes (ERMs) after primary RD repair (n = 25; of these, n = 14 post-PPV). 11 Similarly, another case series of 15 eyes with MH after PPV for rhegmatogenous RD (RRD) repair found possible associations with ERMs (73%); macula-off RRD (60%); high myopia, defined as spherical equivalence greater than −6.00 D (56%); and recurrent RRD (47%). 16 However, these studies lacked a control population and were limited by small sample sizes from a single institution, making it difficult to fully evaluate risk factors for MH development.
While these prior reports also focused on preoperative characteristics of the RRD, the present study aims instead to identify intraoperative and postoperative risk factors for MH formation after prior PPV for RRD repair. We hypothesize that certain intraoperative and postoperative factors may increase the risk for MH formation after PPV, such as the use of a 360-degree endolaser, drainage techniques, and the development of postoperative ERMs or cystoid macular edema (CME). By using a unique case-control design, this study compares these risk factors in eyes that developed MH after PPV for RRD vs eyes that did not develop MH after PPV for RRD but had similar preoperative RRD characteristics.
Methods
In this retrospective matched-pair case-control study, a chart review was performed to identify patients from 3 retina practices who underwent PPV for RRD from 2017 to 2024. Cases where MH developed after initial PPV were identified. Cases of concurrent MH at the time of RRD or of MH development prior to PPV were excluded. The demographic, preoperative, intraoperative, and postoperative data of the initial PPV were collected through manual chart review. Demographic information recorded included patient age and sex. Preoperative factors recorded included high myopia; lens status; presence of preoperative PVD, ERM, or CME; and RRD characteristics such as macular involvement, number of breaks, number of quadrants detached, and presence of a superior bullous detachment.
Intraoperative factors assessed included vitrectomy gauge size, use of a scleral buckle adjunct, type of tamponade, use of a 360-degree endolaser, use of triamcinolone, depressed shave, and drainage method (drainage through retinotomy, through the existing break, or using perfluorocarbon heavy liquid). Postoperative factors assessed were the presence of ERM or CME on postoperative OCT. Other outcomes assessed included time to MH formation, number of subsequent surgeries, and postoperative best-corrected visual acuity (BCVA) at 3-month follow-up after both initial PPV for RRD repair and subsequent PPV for MH repair.
Controls were then identified as patients who underwent PPV for RRD during the same time but did not subsequently develop MH by the end of follow-up. The cases were matched with controls from the same clinical site by the nearest neighbor propensity score matching method in a 1:1 ratio. The propensity scores were estimated with a logistic regression model adjusting demographic and preoperative characteristics at the time of RRD diagnosis, including high myopia, lens status, PVD at the time of RRD, macula involvement, number of breaks, and age. Exclusion criteria included undergoing PPV for indications besides RRD, having a MH at the time of initial PPV, and presence of lamellar holes.
Absolute standardized mean difference was reported for the matching variables between cases and controls 17 to show how balanced the distribution of variables was, with a value of 0.2 or less demonstrating well-matched samples. The Wilcoxon rank-sum test and Fisher exact test were used to evaluate intraoperative and postoperative risk factors after initial PPV for RRD repair. A Wilcoxon signed-rank test was used to analyze the vision acuity difference before and after the MH operation. The survival function for the time to MH development was estimated using the Kaplan-Meier method with a 95% pointwise CI. 18 All data analyses were performed using R version 4.3.1 with the survival package version 3.5.5 and MatchIt package, version 4.5.5 (R Foundation for Statistical Computing). 19 Statistical significance was set at a 2-sided P < .05.
This study was approved by the Institutional Review Board at both Vanderbilt University and Washington University in St. Louis, and the requirement for informed consent was waived. The study adhered to the Declaration of Helsinki tenets and followed Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Results
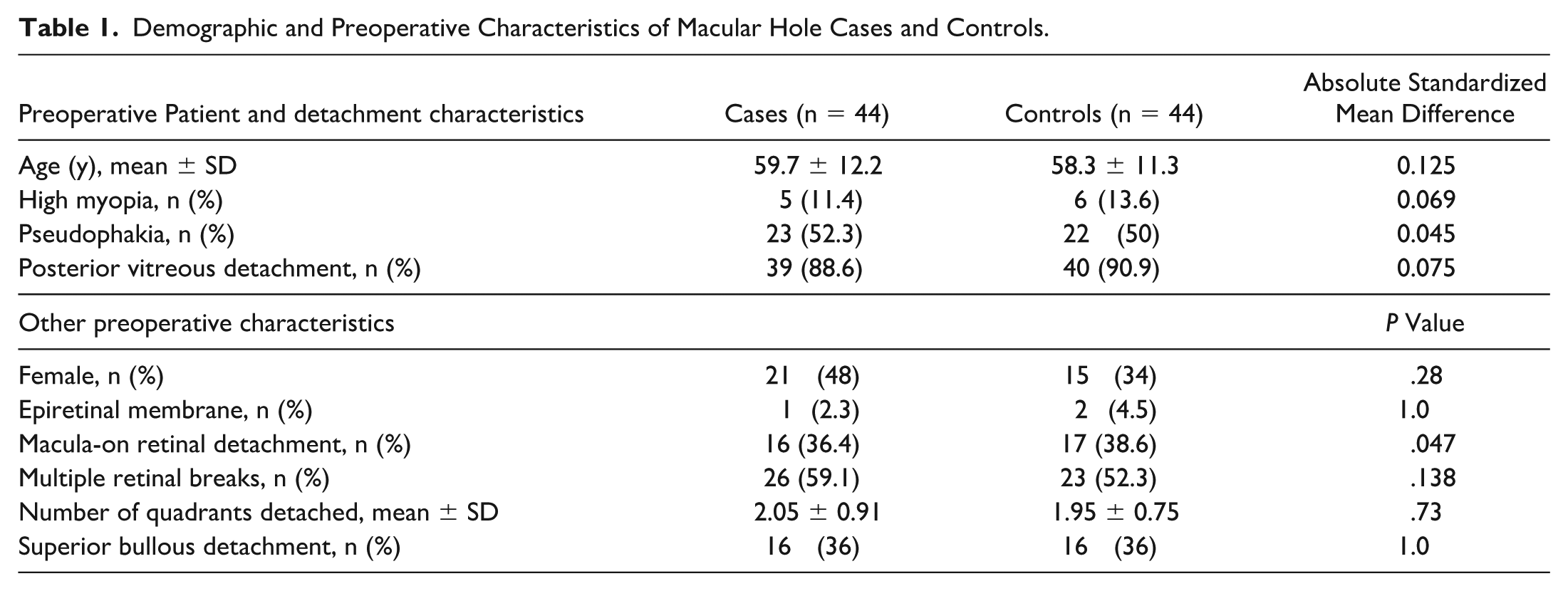
Forty-four cases of MH after PPV for RRD were successfully matched with 44 controls. All absolute standardized mean difference values between cases and controls were less than 0.2, indicating a good match 17 (Table 1). Among the patients who developed MH after PPV, the mean ± SD age at the time of initial PPV for RRD repair was 59.7 ± 12.2 years. Forty-eight percent of patients were female, 11.4% of patients had high myopia, and 52.3% of the patients were pseudophakic. The majority of eyes in both groups (88.6%) already had a PVD when they presented with RRD. Thirty-six percent of cases were macula-on RRDs and 59.1% had multiple retinal breaks. Cases and controls were well-matched without significant differences between cases and controls in demographic and preoperative characteristics (Table 1).
Demographic and Preoperative Characteristics of Macular Hole Cases and Controls.
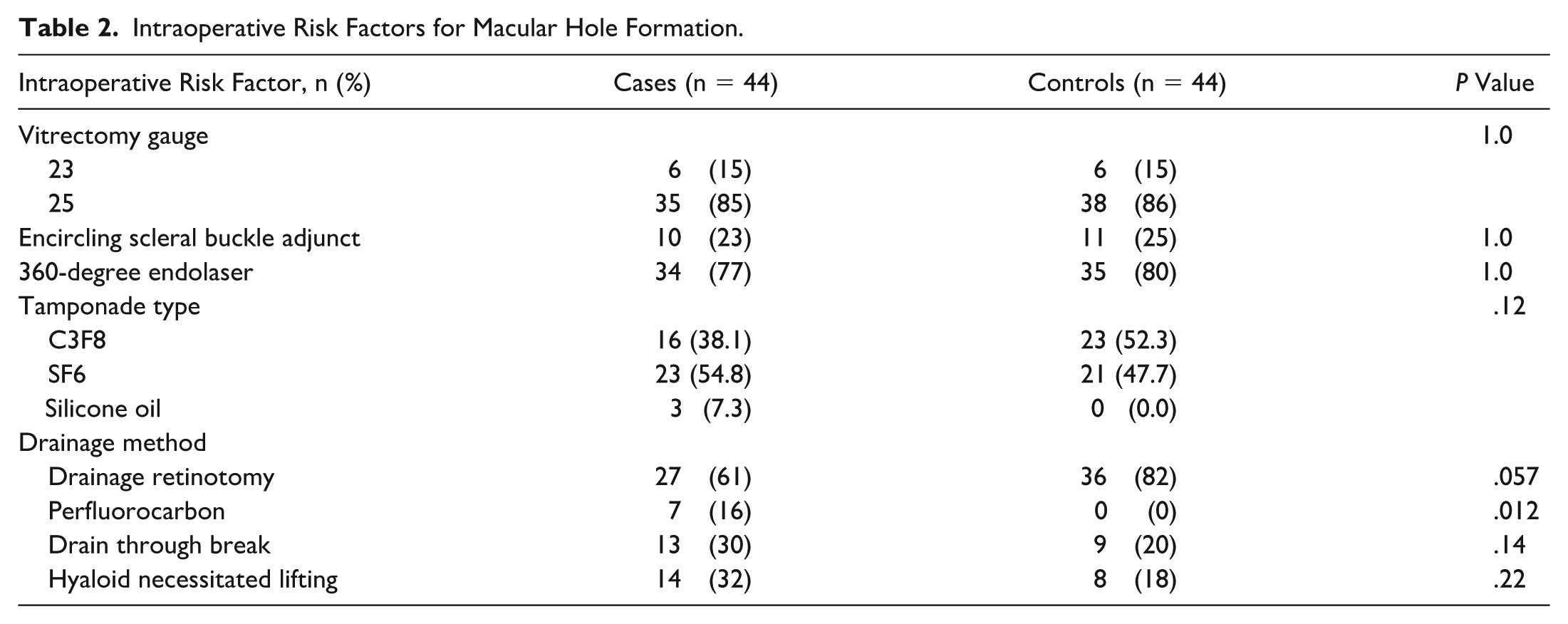
When analyzing the intraoperative risk factors for MH formation after PPV, there was a higher percentage of perfluorocarbon usage in patients who eventually developed MH than in patients who did not (16% in cases vs 0% in controls; P = .012). The frequency of drainage retinotomy was lower in patients who eventually developed MH (61% in cases vs 82% in controls), with this difference approaching statistical significance (P = .057). No differences were found for drainage through the break or other intraoperative factors, including use of a scleral buckle, use of a 360-degree endolaser, type of tamponade used, or lifting the hyaloid (Table 2).
Intraoperative Risk Factors for Macular Hole Formation.
Single surgery success rate for RRD repair did not differ between the MH case and control groups (P = .22). There was no difference in BCVA between cases and controls at the time of initial RRD presentation (P = .58), but BCVA was significantly worse in patients who eventually developed MH at the 3-month postoperative timepoint after initial PPV (0.89 ± 0.65 logMAR for cases vs 0.44 ± 0.37 logMAR for controls; P < .001). Eyes that developed MH had a significantly higher rate of postoperative CME (74% or 31 case eyes vs 10% or 2 control eyes; P < .001; Table 3). Postoperative ERM was also more frequent in eyes that formed MHs, though the difference was not statistically significant (61% or 27 case eyes vs 43% or 9 control eyes; P = .19; Table 3).
Postoperative Risk Factors for MH Formation.

Abbreviations: CME, cystoid macular edema; ERM, epiretinal membrane; MH, macular hole; RRD, rhegmatogenous retinal detachment.
A survival analysis of the MH cases showed median time from the initial PPV for RRD to diagnosis of MH was 190 days (95% CI, 104-418 days; Figure 1). Furthermore, 65.9% of cases (95% CI, 53.3%-81.5%) developed MH more than 90 days from the time of initial PPV for RRD. Of those who underwent additional PPV with membrane peeling for MH repair (n = 42), 95.1% of eyes achieved anatomical closure of the MH at the end of the follow-up period. The median BCVA was 0.70 logMAR (interquartile range, 0.40-1.20) at 3-month follow-up after MH repair, a significant improvement from the initial 1.00 logMAR (interquartile range, 0.53-1.68) BCVA at time of MH diagnosis (P = .001).
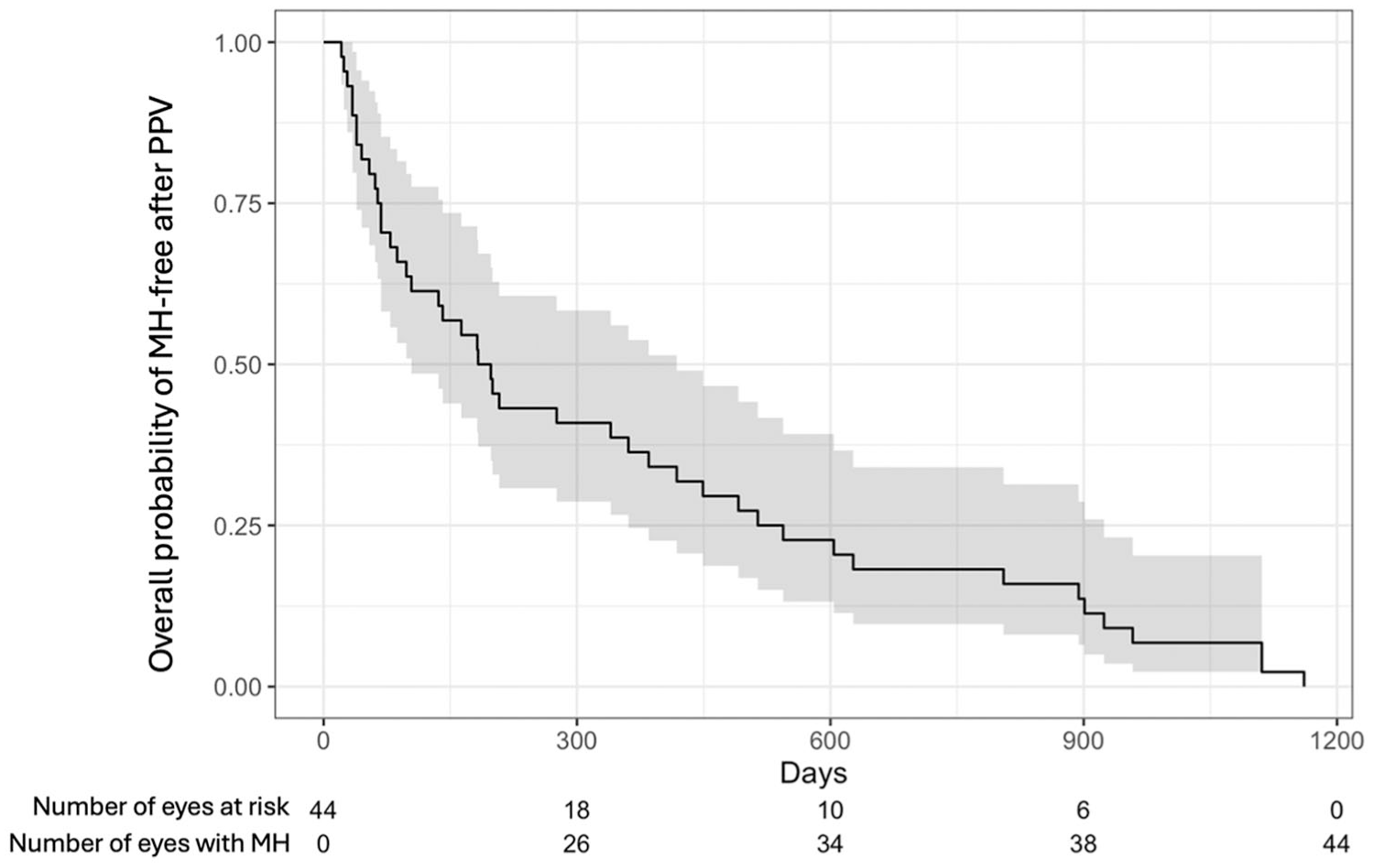
Survival plot of MH development over time. Abbreviations: MH, macular hole; PPV, pars plana vitrectomy.
Conclusions
Given that existing case series of MH after PPV have small sample sizes, lack a control population, and focus on preoperative RRD characteristics, this study used a case-control approach to study intraoperative and postoperative risk factors for secondary MH formation after initial PPV for RRD repair. After matching the preoperative RRD characteristics, this study showed that the type of drainage method used and the development of postoperative CME were significant risk factors for secondary MH formation after PPV. Conversely, the use of adjunctive 360-degree endolaser, use of an encircling scleral buckle, or needing to lift the hyaloid (when there was not a complete PVD) did not significantly contribute to the risk of developing a secondary MH. Despite the vision-threatening nature of MHs, the study also demonstrates most eyes experience significant visual improvement after successful secondary MH closure, even after prior RRD.
As a prior PPV or development of PVD releases anterior-posterior traction on the macula, the pathologic mechanism for secondary MH formation after PPV has been performed may be related to environmental stress- and cellular senescence-induced inflammatory response and the formation of increasing tangential traction. 20 CME could be the result of a proinflammatory and fibrinolytic process that leads to glial-retinal attachment, which was seen in 76% of cases in this study. 21 Moreover, topical treatment of CME alone has led to the successful closure of secondary holes. 22 Without treatment, the spontaneous closure of MH happens rarely, ranging from 3% to 15% in previous studies. 23 The findings in this case-control study further validate those of other small case reports of cohorts, pointing to the need for aggressive treatment or prevention of postoperative CME in eyes undergoing PPV for RRD that may be at a higher risk for MH formation. Additionally, 61% eyes with MH formation developed ERM. These findings, while not reaching statistical significance, are consistent with earlier studies demonstrating that ERMs can cause tangential foveal traction and allow an MH to reopen after prior closure.11,24 Additionally, the late reopening of MH after successful closure with PPV has also been attributed to ERM formation. 15 While tangential force provides a plausible mechanism for MH formation, it alone may not fully explain its development, as ERM was not universally observed with MH. The combination of senescence-related processes and tangential forces likely contributes to the pathophysiology of MH formation.
When looking at intraoperative risk factors, this study found that 360-degree endolaser, tamponade type, encircling buckle adjuncts, and hyaloid lifting did not significantly increase the risk of a secondary MH formation. The slightly higher risk associated with perfluorocarbon use may reflect the more complicated nature of the retinal surgery or the presence of preexisting proliferative vitreoretinopathy as a confounding variable. Conclusions about the effects of intraoperative technique on MH development are not widely generalizable, and further investigation with a prospective study is needed.
Given the rarity of the disease, any study of secondary MH is limited by the small sample size and retrospective nature. Although a thorough review of operative notes and preoperative imaging was conducted, it is possible that coexisting MHs were overlooked at the time of RRD diagnosis, which is a potential confounder in this study. Another potential limitation is the variability in patient populations and differences between surgeons across the 3 sites. This was mitigated by matching controls and cases from the same clinical site. The specific characteristics of the MH, such as size and OCT findings, were not included in this study, which potentially limits our understanding of the mechanism of MH formation.
The study underscores the need follow-up after RD repair, as MH formation may occur several months following the initial repair. Surgeons should have heightened concern for potential secondary MH formation in eyes developing CME and ERM after initial RRD repair as this case-control study shows. Intraoperative technique, conversely, may not significantly contribute to the formation of secondary MH after PPV for RRD. Aggressive postoperative management of CME could theoretically prevent secondary MH formation and maintain excellent long-term visual outcomes after RRD repair. Further studies should focus on the mechanism of how CME and ERM contribute the MH formation and mitigating the risk of postoperative CME and ERM after RD repair.
Footnotes
Authors’ Note
Drs. Xie and Lin contributed equally to this work.
Ethical Approval
This study was approved by the Institutional Review Boards of Vanderbilt University and Washington University in St. Louis.
Statement of Informed Consent
Given the retrospective nature of the study and the de-identification of patient information, no ethical approval was needed, and the requirement for informed consent was waived.
Data Availability
The de-identified data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
All authors have completed and submitted the ICMJE disclosure form. Dr. Thomas is on the advisory boards of AbbVie, Alimera, Bausch + Lomb, and Eyepoint; is a consultant to Avesis and Bausch + Lomb; and is on the Genentech speaker’s bureau. Dr. Finn is on the advisory boards of AbbVie, Apellis, Eyepoint, Genentech, and Iveric Bio, and is a consultant to Genentech. None of the other authors declared potential conflicts of interest with respect to the research, authorship, and/or publication of the article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
