Abstract
Purpose:
To evaluate the effect of preoperative nepafenac on pain following alcohol-assisted photorefractive keratectomy (PRK).
Settings:
Tel-Aviv Sourasky Medical Center—a tertiary medical center.
Design:
Observational case-series.
Methods:
Setting: Refractive center. Study Population: 205 PRK patients grouped randomly into five according to pain-management protocols: (1) paracetamol/ibuprofen (Parac-Ibupr group, n = 39), (2) high-dose oxycodone/naloxone only (Oxy-only group, n = 45), (3) oxycodone/naloxone and postoperative 0.1%-nepafenac (Oxy-Nep group, n = 36), (4) oxycodone/naloxone and preoperative and postoperative 0.1%-nepafenac (Nep-Oxy-Nep group, n = 42), and (5) preoperative and postoperative 0.1%-nepafenac only (Nep-only group, n = 43). Preoperative nepafenac was administered three times daily for 2 days. Main Outcome Measures: Mean and maximal pain levels (postop days 1–5), duration of tearing/photophobia, number of pain tablets taken, uncorrected visual acuity (UCVA), side effects and epithelial healing delay.
Results:
Mean pain scores differed significantly between groups (p < 0.001)—lowest in groups receiving preop nepafenac (Nep-only: 1.8 ± 1.6, Nep-Oxy-Nep: 2.3 ± 1.5) compared to the Oxy-Nep (3.2 ± 1.9), Oxy-only (3.8 ± 1.7), and Parac-Ibupr (4.8 ± 1.6) groups. Similar findings were observed with maximal pain scores. Total number of pain tablets taken was lowest in the Nep-only group. Duration of photophobia was shortest in groups receiving preoperative nepafenac (p < 0.001). Duration of tearing was longest in the Parac-Ibupr group (p < 0.001). Nausea/vomiting occurred in 20% of the Oxy-only group (p < 0.001). There were four cases of delayed epithelial healing—all in groups not treated with nepafenac. One-month UCVA did not differ between groups. No additional independent factors were found to be associated with pain except age.
Conclusion:
Adding preoperative nepafenac significantly reduced pain and photophobia with complete epithelial healing. Addition of oral opiates to nepafenac treatment had little analgetic benefit.
Keywords
Introduction
Since its introduction in the late 1980s, photorefractive keratectomy (PRK) has been one of the most common corneal refractive procedures performed worldwide. 1 Over the past decades, the procedure has been well-established to be safe and effective. 2 Nevertheless, the healing process following PRK is associated with significant postoperative pain and slow visual recovery. 2
Post-PRK pain appears shortly after surgery and persists over the first postoperative days as the corneal surface re-epithelializes. 3 Epithelial removal causes the release of inflammatory mediators, which affect nociceptors by increasing their excitability. In addition, laser ablation directly damages stromal nerve fibers and increases the inflammatory response via damage to stromal keratocytes. 4
Although quite variable, post-PRK pain can be significant and debilitating as the cornea is the most densely innervated human tissue. 5 Pain management following PRK has long been the subject of research but remains a challenge for refractive surgeons. Post-PRK pain management strategies include tissue cooling measures, application of a bandage contact lens, oral analgesics (including opioids, neuropathic agents, and nonsteroidal anti-inflammatory medications (NSAIDs)) and topical agents (including anesthetics, steroids, and NSAIDs).4,6 NSAIDs’ suggested mechanism of analgetic action involves the cyclooxygenase (COX) pathway by reduction of exposed nociceptor activity and reduction of inflammatory mediator levels. 4 Studies evaluating the role of topical NSAIDs in PRK pain management mostly evaluated their use either during the immediate perioperative period or postoperatively. 7 To our knowledge, the added value of preoperative nepafenac in this setting has not been evaluated previously.
Nepafenac is a topical NSAID differing from other topical NSAIDs by being a prodrug with low inherent COX-inhibiting activity. It is converted to an active form called amfenac after metabolic conversion through intraocular enzymatic hydrolysis. It is also characterized by excellent and rapid corneal penetration. 8 Nepafenac has a rapid onset of analgetic action, probably due to an inherent analgetic activity and a rapid saturation of the corneal epithelial layer. This is where corneal epithelial concentrations of nepafenac may be initially sufficient to attenuate corneal nociceptor prostaglandin formation without invoking an immediate conversion to amfenac. The conversion of nepafenac to amfenac occurs predominantly in intraocular vascular tissues, especially in posterior structures such as the retina and choroid, where higher hydrolase activity is present. 8 Therefore, earlier instillation of nepafenac in the preoperative period could increase its analgetic effect later on, once significant conversion to amfenac has occurred. Our group has modified the PRK pain management protocol in our institute to include preoperative topical nepafenac administration starting 2 days before surgery and topical nepafenac given 3 days after surgery.
The purpose of the current study was to compare several PRK pain management protocols given at our institute and evaluate the role of preoperative nepafenac in post-PRK pain management.
Methods
The study was approved by the ethics committee and all data for the study were collected and analyzed in accordance with the policies and procedures of the Institutional Review Board of the TLV-0689-17 Medical Center and the tenets outlined in the declaration of Helsinki. An additional Informed consent for this study was not needed, which was also approved by the committee, as the treatment was part of the routine procedures and informed consent was obtained for the surgery itself.
Study participants
This retrospective study included myotext pic patients who underwent PRK by a single surgeon (A.H) between October 2022 and February 2023 at Enaim Medical Center, Tel-Aviv, Israel.
Inclusion criteria were age over 18 years; a stable refraction for at least 12 months; IOP less than 21 mm Hg.
Exclusion criteria were wearing contact lenses for a period of more than 2 weeks for rigid contact lenses and more than 5 days for soft contact lenses; history of autoimmune disease, diabetes, or previous ocular surgery.
Sample size calculation
Alpha level of 0.05 and beta level of 0.20, determined that each of the five treatment groups should have a minimum of 21 patients.
Treatment groups
As described in a previous paper, 9 different approaches for the postoperative pain management were used to improve postoperative patient care. Patients were not subjected to any additional selection criteria for the purpose of assigning them to the various treatment groups. We routinely evaluate pain and symptom levels using a daily phone questionnaire. In the current study, the patients were grouped into five groups according to the postoperative pain regimen:
No additional oral pain-relief medication was consumed.
Data collection and pain survey
The medical files of all eligible patients were reviewed, and the following demographic and preoperative data were extracted: age, gender, analgetic regimen, date of surgery, preoperative manifest sphere, and preoperative manifest cylinder. The following intraoperative data were extracted: optical zone diameter, ablation zone diameter, central ablation depth, and ablation volume. Data were obtained for both eyes of each patient. Recorded postoperative data included uncorrected visual acuity (UCVA) at 1 month after surgery, presence of any complications, and the status of epithelial healing.
In this study, data were obtained from a single eye per patient. The eye selected for analysis was the one with the higher value for each ocular parameter (such as ablation depth, treatment zone diameter, etc.) to ensure a more accurate reflection of the ocular data at the patient level. Since the primary outcome was pain and other patient-reported symptoms, which are assessed at the patient level rather than the eye level, this approach was deemed appropriate. The parameters used for this selection included Sphere, Cylinder, OZ, AZ Ablation Depth, and Ablation Volume.
Patients underwent a phone survey conducted daily over the first five postoperative days. All patients systematically contacted for the 5-day questionnaire by the same nurse who assigned specifically for that purpose and masked to treatments groups. The survey evaluated:
Visual Analog Scale (VAS) measures pain intensity where pain is graded on a scale of 0–10 where 0 indicates the absence of pain, and 10 indicates the worst possible pain. 10
The number of oral analgesic tablets taken in addition to the preset pain regimen.
The total number of analgesic tablets taken (including the preset regimen and any additional tablets taken.
Presence of additional symptoms: photophobia (as indicated by patients on a scale ranging from 0 to 10), tearing (as indicated by patients on a scale ranging from 0 to 10), nausea/vomiting (recorded as yes/no).
Surgical technique
Prior to surgery, each patient received three drops of a topical anesthetic (benoxinate hydrochloride 0.4%) in the conjunctival fornix, and an eyelid speculum was inserted.
Epithelial cells were removed with a dilute solution of 20% alcohol, using Balanced Salt Solution (BSS). The alcohol solution was instilled into an 8.5-mm alcohol well placed on the cornea. After 30 s, the alcohol was soaked with a sponge from within the well, and the well was removed. The cornea and ocular surface were irrigated with BSS to minimize toxicity to the limbal germinal epithelium. The epithelium was then easily removed using a hooky knife. Stromal excimer ablation was then performed using the Amaris 1050 rs (Schwind, Kleinostheim, Germany) excimer laser platform. A sponge soaked with 0.02% mitomycin C was placed on the stroma for exactly 20 s, immediately after ablation. Following thorough rinsing of mitomycin C, a PureVision (balafilcon A—a silicone hydrogel) soft contact lens (Bausch & Lomb) was placed over the cornea. Following surgery, moxifloxacin 0.5% (four times a day), dexamethasone 0.1% (four times a day), and artificial tears (four times a day) were prescribed. Patients were examined at 1 and 5 days, 1-, 3-, and 6-months post operatively and thereon as needed.
Statistical analysis
Data were recorded in Microsoft Excel and analyzed using XLSTAT (version 2023.1.2, Microsoft). For parameters recorded from both eyes, the maximal value from either the right or left eye was chosen for analysis since the primary outcome of the study is pain level. The normality of distribution was assessed using the Shapiro–Wilk test. Continuous variables were compared between the groups using the Kruskal–Wallis test. Analysis was performed for both mean and maximal pain levels recorded on each of the first five postoperative days. Repeated measures ANOVA was used to analyze and compare pain levels throughout the 5-day study period with pairwise comparisons of the treatment groups done using the Benjamini–Hochberg correction for multiple comparisons. Univariate evaluation of factors associated with pain levels was performed using Spearman correlation. A multivariable model using analysis of covariance (ANCOVA) was then constructed, including the significant and nearly significant univariate factors found. A two-sided p value <0.05 was considered statistically significant in all analyses. All presented means are accompanied by their respective standard deviations.
Results
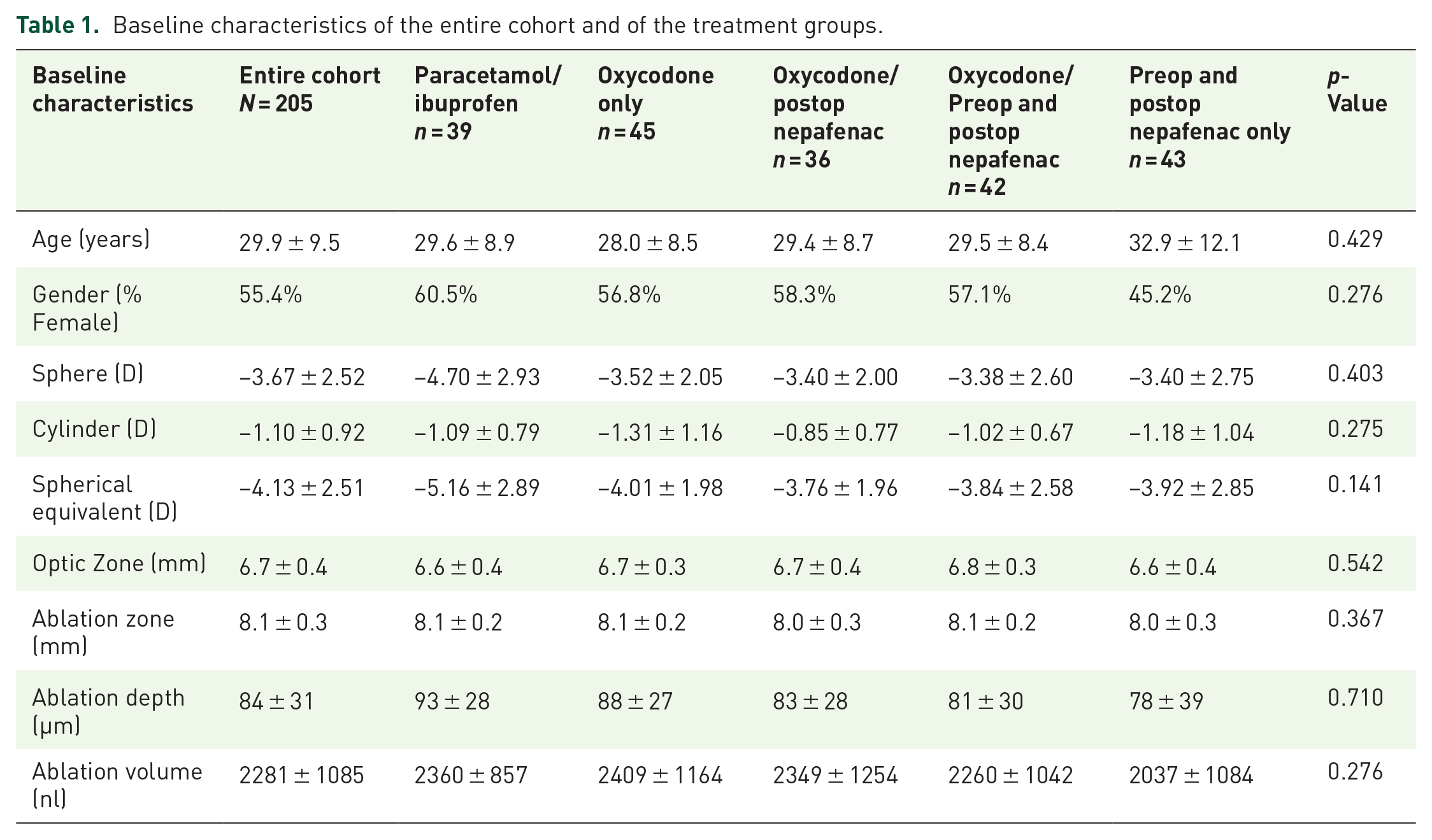
A total of 205 patients were included of which 112 were female (55.4%). Mean age was 29.9 ± 9.5 years. There were 39 patients in the Parac-Ibupr group, 45 patients in the Oxy-only group, 36 patients in the Oxy-Nep group, 42 patients in the Nep-Oxy-Nep group, and 43 patients in the Nep-only group.
There were no significant differences between the groups in baseline and demographic characteristics (Table 1).
Baseline characteristics of the entire cohort and of the treatment groups.
Pain
Mean VAS pain scores for all 5 days were 1.8 ± 1.6 for Nep-only, 2.3 ± 1.5 for Nep-Oxy-Nep, 3.2 ± 1.9 for Oxy-Nep, 3.8 ± 1.7 for Oxy-only and 4.8 ± 1.6 for Parac-Ibupr.
Maximal VAS pain scores were 4.0 ± 3.1 for Nep-only, 5.0 ± 3.0 for Nep-Oxy-Nep, 5.9 ± 2.8 for Oxy-Nep, 8.0 ± 1.8 for Oxy-only and 8.5 ± 2.0 for Parac-Ibupr (Table 2 describes the mean pain score for each day in each group).
Mean VAS scores of each treatment group throughout postoperative days 1–5.

VAS, Visual Analog Scale.
A repeated-measures ANOVA determined that mean VAS pain scores differed significantly between the treatment groups throughout postoperative days 1–5 (p < 0.001). A pairwise comparison found the Nep-only group to have the lowest VAS pain scores (significantly lower than those of all other groups except for the Nep-Oxy-Nep group, Table 3). The Parac-Ibupr group had the highest VAS pain scores (significantly higher than those of all other groups except for the Oxy-only group, Table 3; Figure 1).
Pairwise comparison of the treatment groups using the Benjamini–Hochberg correction for multiple comparisons.

p < 0.05

VAS scores of each treatment group throughout postoperative days 1–5.
The number of pain tablets taken in addition to the pre-prescribed pain regimen was 3.0 ± 2.8 tablets in the Parac-Ibupr group, 2.5 ± 2.1 tablets in the Nep-only group, 2.4 ± 2.1 tablets in the Nep-Oxy-Nep group, 1.8 ± 2.2 tablets in the Oxy-only group, and 0.2 ± 0.9 tablets in the Oxy-Nep group. In a pairwise comparison, the Oxy-Nep group was the only one significantly different than any of the other groups, showing the lowest number of pain tablets taken in addition to the pre-prescribed regimen (p < 0.001 for all comparisons of this group).
The total number of pain tablets taken (including both tablets comprising the planned pain regimen and tablets taken in addition to the pre-prescribed pain regimen) was 5.8 ± 3.2 tablets in the Parac-Ibupr group, 4.0 ± 3.2 in the Nep-Oxy-Nep group, 3.9 ± 2.4 tablets in the Oxy-only group, 2.6 ± 1.6 tablets in the Oxy-Nep group, and 2.5 ± 2.4 tablets in the Nep-only group. In a pairwise comparison, the Nep-only group had the lowest total number of tablets taken, significantly lower than the Oxy-only (p = 0.042) and the Parac-Ibupr (p < 0.001) groups.
Additional factors associated with pain
Additional factors were analyzed for their association with pain, including age, gender, preoperative spherical equivalent, optic zone diameter, ablation zone diameter, and ablation depth. Spearman correlation analysis found age (r = −0.241, p = 0.001) to have a significant negative correlation with VAS pain scores (older age associated with lower scores). Ablation depth (r = 0.169, p = 0.016) was found to have a significant positive correlation with VAS pain scores (deeper ablation associated with higher scores). Ablation zone diameter (r = 0.127, p = 0.071) was found to have a nearly significant positive correlation with VAS pain scores (larger ablation zone associated with higher scores). Gender (p = 0.736) and spherical equivalent (p = 0.135) were not significantly correlated with VAS pain scores.
A multivariable model was constructed, including the significant and nearly significant univariate factors found (age, ablation depth, and ablation zone diameter) as well as the pain-treatment group type. The model found the pain-treatment group (p < 0.001) and age (p = 0.006) to be independent factors associated with VAS pain levels. Due to age being an independent factor associated with pain, the repeated-measures ANOVA comparing mean VAS pain scores between the groups was redone with adjustment for age, showing no substantial change in the results.
Additional symptoms and medication side effects
Mean duration of postoperative tearing was 1.8 ± 1.0 days for Nep-Oxy-Nep, 1.9 ± 0.9 days for Nep-only, 2.3 ± 1.4 days for Oxy-only, 2.5 ± 1.2 days for Oxy-Nep, and 3.0 ± 1.0 days for Parac-Ibupr. Duration of postoperative photophobia was longest in the Parac-Ibupr group compared to all other groups with no additional significant differences between any of the groups (Figure 2).

Proportion of patients with tearing of each treatment group throughout postoperative days 1–5.
Mean duration of postoperative photophobia was 2.0 ± 1.9 days for Nep-Oxy-Nep, 2.4 ± 1.4 days for Nep-only, 3.2 ± 1.7 days for Oxy-only, 3.9 ± 1.6 days for Oxy-Nep, and 4.2 ± 1.0 for Parac-Ibupr. Duration of postoperative photophobia differed significantly between the treatment groups (p < 0.001), significantly shortest in the Nep-Oxy-Nep group compared to all other groups except for the Nep-only group (Figure 3).

Proportion of patients with photophobia of each treatment group throughout postoperative days 1–5.
Nausea and vomiting occurred in 9 of 45 patients (20%) in the Oxy-only group and in 1 of 36 patients (3%) in the Oxy-Nep group. There were no reports of nausea and vomiting in any of the other treatment groups. The difference in nausea and vomiting rates between the Oxy-only and other groups was statistically significant (p < 0.001).
Epithelial recovery
There were four cases of delayed epithelial healing of which three cases were in the Oxy-only group and one case in the Parac-Ibupr group. All cases resolved following prolongation of bandage contact lens wear. There were no occurrences of postoperative haze.
Visual recovery
No differences in UCVA were found between any of the groups at 1 month after PRK (overall p-Value 0.480). Mean logMAR UCVA was 0.19 ± 0.14 in the Nep-only group, 0.20 ± 0.15 in the Nep-Oxy-Nep group, 0.21 ± 0.15 in the Parac-Ibupr group, 0.21 ± 0.17 in the Oxy-Nep group, and 0.25 ± 0.15 in the Oxy-only group.
Discussion
This study compared several analgetic regimens for the management of post-PRK pain and evaluated the added benefit of preoperative nepafenac given 2 days before surgery. Treatment groups who received preop nepafenac reported the lowest pain scores and shortest duration of tearing and photophobia, with no evidence of delayed epithelial healing or prolonged visual recovery. The treatment group who received preop and postop Nepafenac alone not only had the lowest pain scores but also the smallest number of pain tablets taken in total.
The role of nepafenac in pain management following PRK has been evaluated in previous literature, showing good efficacy.11 –16 All studies evaluated nepafenac given either postoperatively or perioperatively (up to 1 h preop). To our knowledge, none of these studies evaluated the added value of preoperative nepafenac. Three groups in the current study received nepafenac (Nep-only, Nep-Oxy-Nep, and Oxy-Nep). Of those, two groups received nepafenac 2 days before surgery in addition to postoperative administration (Nep-only and Nep-Oxy-Nep groups). Both these groups had significantly lower pain scores compared with the Oxy-Nep group that received nepafenac over the postoperative period only (p < 0.001, Figure 1). Preoperative nepafenac administration has been evaluated in cataract surgery patients. Oğurel et al. conducted a prospective randomized double-masked, placebo-controlled trial evaluating the effect of nepafenac given 3 days before cataract surgery on pain levels, showing significantly reduced pain in the group receiving preop nepafenac. 17
The rationale behind preoperative nepafenac administration is based on the fact that it is a prodrug whose activity is increased following intraocular conversion to amfenac. This conversion occurs predominantly in posterior intraocular vascular tissues, with higher hydrolase activity. 8 Therefore, more time is required to achieve higher potency. Nepafenac probably acts in several mechanisms to reduce pain. It does have rapid onset of analgetic action, which is probably due to an inherent analgetic activity. However, once converted to amfenac, the analgetic effect is amplified, possibly via a stronger reduction in inflammatory mediator levels such as prostaglandin. In prospective trials, preoperative administration of nepafenac has been shown to reduce intracameral prostaglandin levels and aqueous flare in cataract surgery patients.18,19 Future studies of similar design in PRK patients could provide more insight into the mechanisms by which the addition of preoperative nepafenac reduces postoperative pain.
Although pain is the main outcome measure of the current study, it is not the only component of patient discomfort following PRK. Patient well-being over the first postoperative days is also affected by photophobia, which can be considered part of the same process but is perceived by the patient as a separate component of postoperative discomfort. In the current study, the duration of both photophobia and tearing was shortest in groups receiving preoperative and postoperative nepafenac (Nep-only and Nep-Oxy-Nep).
Oral opiates are considered strong analgesics and are often prescribed following PRK. One downside of opioid use is common side effects such as nausea and vomiting. In the current study, the group receiving 10-mg oxycodone (Oxy-only group) had a significant portion of reported nausea and vomiting (20%), which was significantly higher than any of the other study groups. Also, pain scores in the Oxy-only group were relatively high compared to the other study groups, second only to pain levels of the group receiving a paracetamol/ibuprofen combo (Parac-Ibupr group). These findings suggest that use of high-dose opiates alone is associated with relatively low safety and analgetic efficacy in PRK patients. Moreover, opioid usage is unnecessary in patients who would receive topical nepafenac pre-and postoperatively. Differences between the group receiving preop and postop nepafenac only (Nep-only group) and the group receiving preop and postop nepafenac together with oxycodone (Nep-Oxy-Nep group) were small in all parameters evaluated in the study.
Delayed re-epithelialization is a known side effect of topical NSAID use, which, in addition to increasing patient discomfort and prolonging visual recovery, could potentially increase the risk of infectious keratitis and sterile corneal melt. 20 Caldwell et al. reported significantly larger epithelial defects in nepafenac-treated eyes than in placebo eyes at 2 days after PRK. 11 Trattler et al. discontinued a trial comparing nepafenac and ketorolac after enrolling seven patients only (out of 60 planned) due to slower re-epithelialization and greater haze scores in the nepafenac group. 14 In the current study, no occurrences of an epithelial healing delay were observed in any of the nepafenac-treated groups. Topical NSAIDS dangerous side effects include corneal melting or perforation after ablation which was not witnessed in this study. Both these aspects should be further validated in larger and longer prospective trials. In this study, patients were examined on the first day and on the fifth day; thus, epithelial closure time was not recorded.
Limitations of this study include its retrospective, non-randomized nature and the absence of daily measurements of epithelial defect size. The variation in the number of pain tablets consumed beyond the prescribed pain regimen among the groups could introduce additional confounding effects. Moreover, the study was conducted at a solitary facility, a factor that could potentially limit the generalizability of the findings to broader contexts.
Nevertheless, to our knowledge, this is the first study evaluating the added benefit of preoperative nepafenac on post-PRK pain levels.
In conclusion, the addition of preoperative nepafenac administered 2 days prior to PRK significantly reduced pain and photophobia with no evidence of delayed epithelial healing. The addition of oral opiates to nepafenac treatment had little analgetic benefit.
Value statement
What was known
PRK is a common and established refractive procedure known for its safety and effectiveness.
Postoperative pain and slow visual recovery are significant issues following PRK due to epithelial removal and inflammatory responses.
The gap in knowledge lies in evaluating the impact of preoperative nepafenac on post-PRK pain management in comparison to other pain management strategies.
What this paper adds
Evaluates the novel use of preoperative nepafenac in PRK pain management, which had not been previously studied.
Demonstrates that preoperative nepafenac significantly reduces post-PRK pain and photophobia, aiding in complete epithelial healing without additional opiates.
Provides insights into optimizing pain management protocols for PRK procedures by incorporating preoperative nepafenac administration
