Abstract
Background:
New developments in artificial intelligence, particularly with promising results in early detection and management of keratoconus, have favorably altered the natural history of the disease over the last few decades. Features of artificial intelligence in different machine such as anterior segment optical coherence tomography, and femtosecond laser technique have improved safety, precision, effectiveness, and predictability of treatment modalities of keratoconus (from contact lenses to keratoplasty techniques). These options ingrained in artificial intelligence are already underway and allow ophthalmologist to approach disease in the most non-invasive way.
Objectives:
This study comprehensively describes all of the treatment modalities of keratoconus considering machine learning strategies.
Design:
A multidimensional comprehensive systematic narrative review.
Data sources and methods:
A comprehensive search was done in the five main electronic databases (PubMed, Scopus, Web of Science, Embase, and Cochrane), without language and time or type of study restrictions. Afterward, eligible articles were selected by screening the titles and abstracts based on main mesh keywords. For potentially eligible articles, the full text was also reviewed.
Results:
Artificial intelligence demonstrates promise in keratoconus diagnosis and clinical management, spanning early detection (especially in subclinical cases), preoperative screening, postoperative ectasia prediction after keratorefractive surgery, and guiding surgical decisions. The majority of studies employed a solitary machine learning algorithm, whereas minor studies assessed multiple algorithms that evaluated the association of various keratoconus staging and management strategies. Last but not least, AI has proven effective in guiding the implantation of intracorneal ring segments in keratoconus corneas and predicting surgical outcomes.
Conclusion:
The efficient and widespread clinical translation of machine learning models in keratoconus management is a crucial goal of potential future approaches to better visual performance in keratoconus patients.
Trial registration:
The article has been registered through PROSPERO, an international database of prospectively registered systematic reviews, with the ID: CRD42022319338
Plain language summary
Artificial intelligence has changed how we treat the eye disease keratoconus in recent years. This study examines the many keratoconus therapies available, including surgery and contact lens wear, and how artificial intelligence can improve the safety and accuracy of these procedures. We combed through numerous papers to locate this data. To achieve the best outcomes, several parameters and methods should be evaluated. According to the study, some elements from eye scans are more useful than others. The idea behind using artificial intelligence is to help patients see better and treat keratoconus more effectively.
Keywords
Introduction
The advent of artificial intelligence in ophthalmology 1 neural network algorithms and deep learning strategies in keratoconus (KC) diagnosis, 2 genetic, 3 and molecular biologic tests 4 alter the natural history of KC.5–7 This may allow us to think that KC is a different specialization of ophthalmology rather than just a clinical disorder.
First and foremost, a complete examination, including multimodal imaging8,9 public awareness, 10 and patient education 11 are critical components of the overall effort to battle this debilitating corneal disease.
Corneal cross-linking (CXL) known as the mainstay approach to impede progression of keratoconus 12 ; however, debates about ‘epi-on’, ‘epi-off’ procedures, ‘accelerated’, ‘pulsed’, and ‘customized’ CXL, and riboflavin solutions are continuing and clarification about ‘gold standard’ in CXL therapies need more investigation. 13 Patients with KC can present with high anisometropia 14 ; Therefore, it can be challenging to achieve best bilateral visual acuity with various contact lens designs (scleral, hybrid, and piggyback) without requiring invasive surgery15,16 or frames. In these cases, combined procedures may represent an option to provide these patients with the greatest visual potential bilaterally. The combination of cross-linking with a refractive operation (either two or three procedures such as photorefractive keratectomy17,18 with mitomycin-C applications, 19 phakic intraocular lenses (pIOLs),20,21 or intracorneal ring segment implantation) called ‘corneal cross-linking Plus’ halt keratoconus development while also correcting refractive error. 22
Noninvasive keratoplasty approaches such as Bowman’s layer transplantation, additive keratoplasty (i.e. corneal allogenic intrastromal ring23,24 or customized lenticule Implantation 25 ), and cellular therapies,26,27 have gradually unveiled increasingly accessible pathways in the field. However, there are conflicting reports regarding their effectiveness. Challenges to reaching a consensus about technique and materials and methods for cross-linking,28,29 laser surgeries,30–32 implanting rings,33,34 and keratoplasty’s approaches request a robust nomogram.
The subjective realm of AI contrasts with the linear statistical nature of traditional modalities and mathematical corneal models.35,36 Machine learning (ML), a subset of AI encompassing supervised, unsupervised, and reinforcement learning, has evolved as a critical tool in detecting KC using various types of neural networks (NNTs), automated decision trees, and support vector machines (SVM)2,37,38 (Figure 1).

Steps in artificial intelligence studies from data science to machine, and deep learning.
Prediction of unordered outputs (e.g. tomography) for discrimination of KC and normal eyes, becomes possible by supervised learning, whereas regression problems are better defined in suscept cases. 39
Using reinforcement programs (learning by trial and error) can assist in intrastromal ring implant surgery.2,36 The integration of clinical data, type of surgery, ablation depth, topographic, tomographic, and biomechanical data have showed dramatic effects in keratoconus management. 39 However, still the potential capability of most popular imaging modalities, such as anterior segment OCTs9,40 to use AI-applicable indexes has not been fully utilized.
The purpose of this study is to explain all of these features ingrained with artificial intelligence in treatment of keratoconus.
In the words of William Wordsworth ‘Let us learn from the past to profit by the present, to live better in the future’.
Methods
Study selection
A comprehensive search was done in the primary electronic databases, including PubMed, Scopus, Web of Science, Embase, and Cochrane, to identify all articles with the appropriate keywords (as mentioned below), till August 2023. It did not include language or type of study restrictions.
1- (‘keratoconus’)
2- AND ((algorithm) OR (machine learn*) OR (deep learn*) OR (artificial intelligence) OR (automatic) OR (neural network))
3- AND ((treat*) OR (manage*) OR (therap*) OR (surg*) OR (intervent*) OR (regim*) OR (medic*) OR (prescrip*) OR (cure*))
The search results in each database were included in Endnote software, and then duplicate articles were removed. After that, eligible articles were selected by screening the titles and abstracts. For potentially eligible articles, the full text was also reviewed (Figure 2) in three phases: In the initial phase, three authors (F.D, S.N, and Z.G) reviewed the articles based on agreed-upon inclusion and exclusion criteria (using the search keywords Included and Excluded in the listed databases). During the second stage, the same three primary authors examined accessible, relevant, and valid articles and ranked them according to the Critical Appraisal Skills Programme (CASP) checklist. During the third stage, the authors shared their results with scores and separated the study references. If the three authors disagree on one of the references, it will be reexamined by the fourth author (O.F) who was not involved in the collection process. Finally, the relevant data were extracted and handled from selected studies in a quantitative, narrative way.

Study selection and exclude strategy algorithms.
Risk of bias (quality) assessment
Risk of bias was evaluated by three independent researchers (F.D, S.N, and Z.G). Using the Cochrane quality assessment tool, the risk of bias in each study included in the current meta-analysis was evaluated. This instrument included seven domains, including random sequence generation, allocation concealment, reporting bias, performance bias, detection bias, attrition bias, and additional sources of bias. Each domain was assigned a ‘high risk’ score if the study contained methodological flaws that could have affected its findings, a ‘low risk’ score if there were no flaws for that domain, and a ‘unclear risk’ score if there was insufficient information to determine the impact. If the trial met the criteria for ‘low risk’ in all domains, it was deemed a high-quality study with an extremely low risk of bias.
Results
This study aimed to conduct a comprehensive review of the relevant studies so far. Considering the heterogeneity and great diversity of the findings, we decided to review the studies with not only a systematic but also a narrative approach. Below we briefly and usefully mention the most important points of management and treatment of keratoconus.
Preoperative assessments and evaluating the progression of KC
There is no universally accepted staging system for KC; however, significant advances in artificial intelligence in recent years can help identify the proper indications for surgery. 14 Pachymetric 41 and tomographic indices 42 integrate with biomechanical evaluation of cornea, 43 anterior corneal higher order aberrations (HOAs), 44 and ultra-high-resolution Optical Coherence Tomography (OCT) 45 not only use preoperatively, but also for postoperative (epithelial remodeling 46 and anterior stromal demarcation) management. 47 Furthermore, regarding genetic aspects3,7 and metabolic imbalance of the disease, 48 recent reviews considered that micronutrients such as metal ions, vitamins, hormones, antioxidants, oral riboflavin, and Essential Fatty Acids, can deeply influence KC initiation and progression, particularly in the early stages of KC. 49 Hence, as early as holistic approaches is necessary to get rid of unfavorable circumstances and may prevent the further spread of the disease. 10
Non-surgical options to impede progression
The effects of oral riboflavin and solar radiation on keratometric stability and visual acuity have been demonstrated in the literature. 50 Topical antihistamine/mast cell-stabilizing treatments, nonsteroidal anti-inflammatory drugs (NSAIDs), and, on rare occasions, steroids can help decrease the often-coexisting signs of ocular allergies (vernal and atopic keratoconjunctivitis) specifically, which can lead to eye rubbing 11 (Figure 3). These drugs can also help in large papillary conjunctivitis, a typical side effect of wearing contact lenses. Steroids should only be administered after considering the increased risks of cataracts, glaucoma, and a weakened immune system. 51
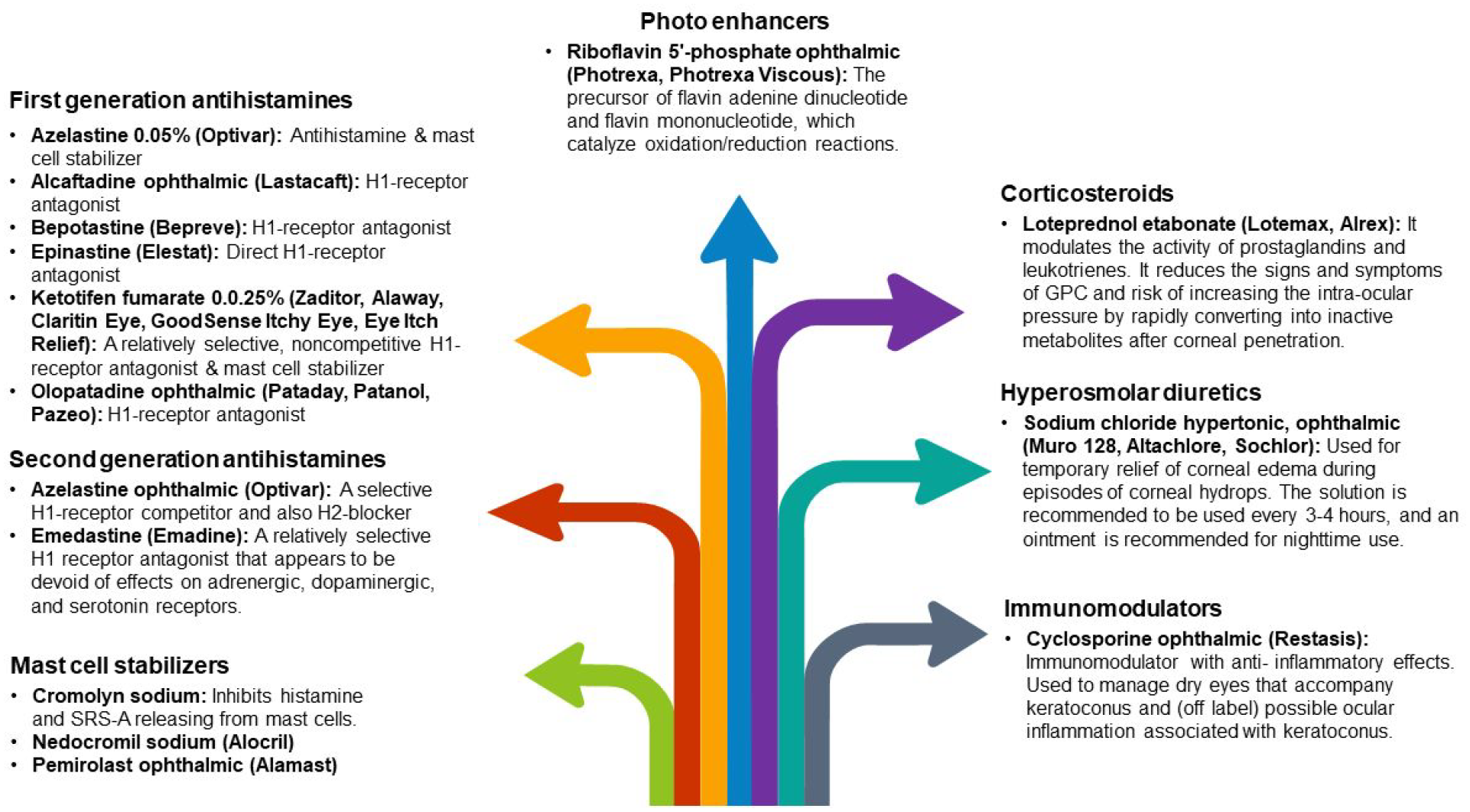
Hydrops episodes may necessitate treatment with hyperosmotic or topical steroid drops to minimize ocular edema and inflammation. Furthermore, an inflammatory component may play a role in ectasia development or initiation and can be managed with cyclosporine ophthalmic emulsion or components for a healthy ocular surface. 54
Eye rubbing can occur due to various factors including, but not limited to, fatigue, emotional stress, hormonal fluctuations, Computer Vision Syndrome, nutrients, irritation, dry eye, and allergy. As a result, KC awareness among allied health providers and thorough patient follow-up can significantly impact surgical results and planning. 11
Contact lenses
Different kinds of contact lenses, in addition to correcting visual distortion and astigmatism, can delay or prevent the need for keratoplasty in KC (Supplemental Figure 4). However, several factors are correlated with the visual improvement achieved with contact lenses in keratoconus eyes and a predicting model according to the pre-fitting data should be investigated.55,56
For a better contact lens fitting, tangential maps of Placido-based video-keratography with calculating the cone’s size and location help measurement of back optic zone radius (BOZR) and total lens diameter in rigid lenses and SynergEyes ClearKone hybrid lens fitting. 57 Furthermore, virtual sodium fluorescein fitting simulation software (Zernike polynomials and spherical harmonics) models the fitting characteristics of lens designs and represents the height data, such as sagittal corneal height. 58
Providing a steeper BOZR, a softer touch to the corneal apex, and more significantly flattening the peripheral curves by the multi-curve design (which is made up of numerous spherical radii to create the required flattening shape) is the most common rigid lens design. Advanced technology introduced aspheric lenses, generally fitted steeper than multi-curve lenses with less sagittal height. Compared to slit-lamp biomicroscope dynamic fluo image simulation systems such as Medmont E300 (Precision Technology, Canada) and Pentacam AXL Wave, reported 74% diagnostic accuracy for contact lens fitting in KC patients. 59
Other technologies such as Eye Surface Profiler (Eaglet-Eye, Houten, the Netherlands), which measures up to 20 mm diameter of the anterior segment, and Cassini, i-Optics (The Hague, the Netherlands), which projects 700 spots, emitted from a multi-colored light-emitting diode on the corneal surface visualize the topographic deformations, caused by the corneal irregularity. 60
Keratometric measurements of the cornea’s spherical shape do not match the best soft lens fit; therefore, OCT’s new ML-based insights entered the field of contact lens fitting. The scleral sphericity shape, the corneal vault throughout the lens, and the highest attainable central clearance can all be measured. 61
Anterior segment OCT can also assess the impact of soft contact lens edge design and mid-peripheral shape on the epithelial thickness and central and peripheral tear film clearance in various hard lens models.61,62
Refraction and contact lens fitting in KC are complex issues due to irregular astigmatism and HOAs. 16 In addition to the imaging modalities, the models such as the Strehl (VSX) and the SyntEyes use corrections by custom ray tracing software (MATLAB R2020a) and represent the real eye. Other algorithms (free access at www.calculens.com) 15 and the hierarchical fuzzy system (from the expertise of experienced ophthalmologists during the lens evaluation) fine-tuned with the genetic algorithms, promote a final acceptable fit in keratoconus. 63
Nevertheless, the ability to manage contact lens-related problems is necessary.
Corneal erosion repeatedly occurs (even mesh-like scar in the cone’s stroma over time) because the tear layer declines: Reduce the optical zone diameter + The altered relationship between the base curve and the peripheral curves.
3 and 9 o’clock staining: Increase lens diameter/Decrease edge thickness/Make sure there is enough edge lift and lens centration.
Linear staining: Cleaning the lens/Removing rough edges from the joints of the peripheral curves / Experimenting with an aspheric design.
Apical staining: Reduce flattening of lens
Air bubbles on the corneal surface or ‘dimple veiling’ Over cone bubbles, around the cone, or at the lens periphery: Reduce the apical clearance level/Decrease BOZD/Minimize axial edge lift, respectively.
Proper lid cleanliness and treatment of dry eyes are also crucial to guarantee optimal wearing times.
Any lens has superiority or drawback (Table 1) and contact lens fitting need art and creativity, for instance despite decreasing the vault with time, the corneal thickness and physiology (Limbo kit test) did not change when the scleral contacts (Rose K2 XL) were tolerated for at least 8 h each day. Notwithstanding, longer-term research is required about variations of topographies, corneal thickness, and vault, as well as whether an objective test, like the Limbo kit, for limbal stem cell insufficiency, will stay negative over time. 64
RGP, rigid gas permeable.
Corneal cross-linking
The purpose of CXL is to strengthen the corneal stability and stiffness and thus arrest the progression of keratoconus. Several CXL methods have been developed that may differ in the procedure technique (i.e. Standard Dresden Protocol (Epi-Off), Accelerated cross-linking (Epi-Off or Epi-On), Pocket cross-linking, etc.), kinds of Riboflavin (such as Riboflavin with Dextran solution or Riboflavin with hydroxypropyl methylcellulose (HPMC) solution), U.V. irradiation, and oxygen usage 65 (Table 2).
Available riboflavin solutions for keratoconus cross-linking. It should be noted that the advised application procedure varies significantly between riboflavin solutions, for example, one drop every 30 s for 15 min for RIBOFAST and RIBOCROSS te, one drop every 2 min for 30 min for RICROLIN® TE and RICROLIN®+, with the recommended soak time for an iontophoresis-assisted delivery of RICROLIN®+ reduced to just 5 min. 65,66

BAC, Benzalkonium Chloride; EDTA, Edetate Disodium; HPMC, Hydroxyl Propyl Methylcellulose; Vitamin E-TPGS, D-alpha-Tocopheryl Polyethylene Glycol Succinate.
As Seiler et al. mentioned the most well-known challenges during the collagen CXL are: how rarely Riboflavin penetrates an epithelium that has not been broken, fast depletion of oxygen (especially at higher irradiances, Table 3), concerns of toxic effects on endothelium in thin corneas, and long regimen of collagen CXL (C-CXL).67,68
ACXL, accelerated corneal collagen cross-linking; min, minutes; nm, nanometer; mW/cm2, milliwatts per square centimeter.
The corneal epithelium not only serves as an oxygen barrier but also uses significantly more oxygen than the stroma, thus complicating the issue of oxygen availability using transepithelial techniques. 65 Using supplementary oxygen may be helpful (especially during an epi-on surgery). After several months of follow-up, it was reported to have improved corneal curvature and visual acuity without significant side effects. 69
On the other hand, CXL in the thin cornea (less than 400 µm in advanced cases of keratoconus, pellucid marginal degeneration, post-LASIK ectasia, etc.) also has several challenges. 29 Methods like artificially thickening the cornea by using hypotonic Riboflavin to swell the cornea to a thickness of 400 μm before and during UV-A irradiation29,70 or placing a riboflavin-soaked contact lens on the eye (contact lens-assisted CXL: CACXL) 71 strength and protect the thin cornea. However, they have their own drawbacks, such as unpredictable effects and suboptimal corneal strengthening. 72 Sub400 (as a new procedure developed by Hafezi et al. to individualize total energy during CXL) according to intraoperative Pachymetry delivers a U.V. dose to avoid irradiating the safety zone without using artificial thickening techniques for thin corneas. 73 Focused cross-linking by imaging techniques and nonlinear optical cross-linking (NLO CXL) unlike UVA cross-linking uses not one photon but two, increasing the likelihood of generating subsequent radicals and photoactivation of Riboflavin.13,74 The also enables fine and deep x–y–z dimensional adjustments over the area of femtosecond laser-cross-linked tissue micromachine epithelial channels. 74
There is not yet definite protocol to confirm which outcomes or complications may occur, such as haze, scarring, sterile infiltrates, endothelial damage, excessive or no flattening, and failure post-CXL. Using AI in genetic 3 and biomarker 48 evaluation joined with longitudinal corneal data can predict future disease progression and enable the identification of eyes that may benefit from as early as intervention. 39
Since young age affects keratoconus progression, Kato et al. conducted a conventional neural network (CNN) to predict progression using patients’ age and corneal tomography data. 75 Similar to other studies, keratoconus progresses differently depending on age: middle-aged patients progress slowly, but young-onset patients progress faster.76,77
Previous research indicated that age and Rmin (Pentacam HR’s minimum sagittal curvature of the corneal frontal plane) were pivotal factors influencing keratoconus progression in patients who had been monitored at least twice after their initial visit. 28
Both an axial corneal frontal plane map and a pachymetry map, as well as a combination of the two, exhibited comparable area under the curve (AUC), sensitivity, and specificity values. Considering that each corneal topography/tomography device includes the axial map of the corneal frontal plane, it’s feasible for deep learning (DL) to clinically predict keratoconus progression. 75 The diagnosis rate may not be enough for keratoconus specialists, who empirically determine the indication of CXL based on clinical stage and age. However, it may help non-specialists like family practitioners, general ophthalmologists/optometrists, or other ophthalmologists decide if patients should see corneal specialists trained in CXL. 75
Different modalities of therapeutic refractive surgeries in keratoconus
Although KC poses a severe negative impact on quality of life and a significant financial burden on individuals and public health systems, an appropriate technique for refractive procedure has been challenging topic of studies.78–80 Cross-linking, as a ‘green light’ for keratoconus stabilization, opened a new door of therapeutic refractive surgery to ‘reshape’ the keratoconic cornea and postpone or even avoid corneal transplantation.18,81
Previous studies on patients with grade I–II keratoconus confirmed the safety and efficacy of ‘minimized-volume ablation’ with accelerated cross-linking on the Schwind AMARIS 750 excimer laser guidelines (Schwind eye-tech solutions GmbH, Kleinostheim, Germany). 82 The ‘Central Corneal regularization (CCR)’ protocol on the iVis Suite modified excimer laser ablation treatment framework (iVis Technologies S. r. l., Taranto, Italy) was proven to enhance corrected and even uncorrected visual acuity. 83
Further studies on using the Central Corneal Regularization/Photorefractive Keratectomy (CCR/PRK) technique in conjunction with C-CXL found it to decrease Kmax and HOAs more efficiently than C-CXL alone. 84 Although the amount of removed tissue was reduced by topographic C-CXL in the Athens protocol, 17 and uncorrected and corrected far visual acuity improved with 94.4% visual stability after 1 and 10 years. The main disadvantage of combined PRK and C-CXL is the limitation of 400-μm corneal thickness after ablation, making it impossible for patients with advanced keratoconus. Another protocol of combined PRK and CXL that reported improved astigmatism and visual acuity, and delay in the progression of keratoconus without producing considerable corneal thinning, is the Tel Aviv protocol.85,86 The excimer laser is used to achieve 50 μm laser ablation of the anterior stroma and epithelium, with astigmatism correction half of refractive astigmatism (along the same axis).85,86 The last alternative procedure is the Cretan procedure, characterized by transepithelial Phototherapeutic Keratectomy (PTK) in combination with CXL,13,87 generating better refractive outcomes and vision than mechanical epithelial removal (Figure 4 shows different combined modalities in KC).

In comparing simultaneous CXL versus sequential CXL because cross-linked tissue is ablated with the sequential CXL method, stability remains a significant concern, and data on this subject is sparse.93–95 In addition, with topography-guided excimer laser therapy, the majority of the tissue from the inferior ‘reddest’ steepest portion of the cornea (frequently associated with the thinnest region of the cornea near the cone’s tip) is destroyed. 95 Although non-topography-guided ablation for myopia and astigmatism would likely result in less tissue ablation in patients (especially in the thinnest portion of the keratoconus cornea) with excellent corrected distance visual acuity (CDVA) and subjective manifest refraction. 96 Nevertheless, the fundamental challenge of reducing the degree of stromal ablation (mean ablation depth) per spherical equivalent refraction and spherical equivalent remains unpredictable. 97
HOAs, mainly spherical aberration, coma, and corneal irregularities (simultaneous or sequential CXL31,98–100), cause more stromal ablation, more biomechanical disintegration, and reverse any benefit from a previous or concomitant cross-linking procedure. 30
By focusing primarily on a few HOAs, novel wavefront-guided methods have recently been devised to reduce tissue loss. PRK in suspect KC based on Placido NNTs [the Corneal Navigator software of the OPDScan II aberrometer/corneal topographer (Nidek Co. Ltd., Gamagori, Japan)101–103 was safe and effective. 104 Based on a recent study by Kanellopoulos, the customization platform of InnovEyes artificial intelligence software proceeds a prism-like ablation to correct the calculated HOAs optimally. 105 Collected data (including wavefront, Scheimpflug tomography, pupillometry, iris recognition, and axial interferometry length) of the Pentacam AXL Wave (Oculus, Germany) in a personalized treatment planning transfer to the EX500 excimer laser (Alcon, Wavelight, Erlangen, Germany). It also corrects the internal tilt between the real anterior corneal surface orientation and the ray-tracing orientation. It prevents patient tiredness and miosis by bilateral conduction of wavefront and unilateral performance of the tomography and interferometry before being repeated in the other. Compared to employing anterior corneal surface data or wavefront data alone, 106 the management of progressive keratoconus using CXL paired with innovative excimer laser customization and independent ray tracing enable to normalize distorted optics associated with corneal ectasia.
Furthermore, since the anterior surface component that makes up for the posterior corneal irregularity will not be ablated, 107 the ray tracing-customized ablation should theoretically require less stromal tissue than the pure anterior surface topography-guided ablation while leading to less myopic shift simultaneously. Although a spherocylindrical shift still happens, when certain HOA is targeted (without compensating extra tissue removal), corneal tissue removal depths are decreased to the bare minimum necessary to alleviate irregular astigmatism. 106
In a study of Awwad et al. ML algorithm analysis of OCT evaluation for significant haze pointed that haze formation is more attributed to using of Mitomycin C (MMC) and CXL simultaneously, 108 and the interactions of MMC with immune system and change in level of cytokines and chemokines can induce haze. 30
On the whole, machine learning-based devices would simultaneously increase the predictability of therapeutic excimer laser surgery and cross-linking procedures.
Intrastromal corneal ring segments
As an alternative treatment for keratoconus, intrastromal corneal ring segments (ICRS) enhance visual acuity and reduce optical aberrations and refractive errors while correcting mean keratometric indexes.109,110 KC patients with contact lens intolerance who are 21 years old with a clear central cornea and a corneal thickness of 450 µm or more at the incision site are suitable candidates for an ICRS procedure. Patients with collagen vascular, autoimmune, or immunodeficiency disorders or those with severe atopy should not undergo an ICRS procedure.54,111
As mentioned by Ertan and Colin, the surgical procedures of implantation for all types of intracorneal ring segments, including Myoring (Dioptex, GmbH, Linz, Austria), INTACS (KeraVision, Inc., Fremont, CA, USA), AJL PRO (AJL Ophthalmic, Vitoria-Gasteiz, Spain), Keraring (Mediphacos, Belo Horizonte, Brazil), and Ferrara Intracorneal Ring (Ferrararing, Belo Horizonte, Brazil), are reversible and adjustable.112,113
By including high-order corneal aberrations, the predictability models could be enhanced. In other words, including the aberrometry factor could be a subtly indirect way to include the biomechanical corneal element in some measure. However, the indirect influence of aberrometry on corneal biomechanics is only a small part of the overall biomechanical effect. 77 As mentioned by Vega-Estrada et al., this is due to challenges in analyzing the cornea’s biomechanical characteristics in vivo in clinical settings. The precise contributions of the elastic and viscous components to the magnitude of these parameters are still not completely understood. 114
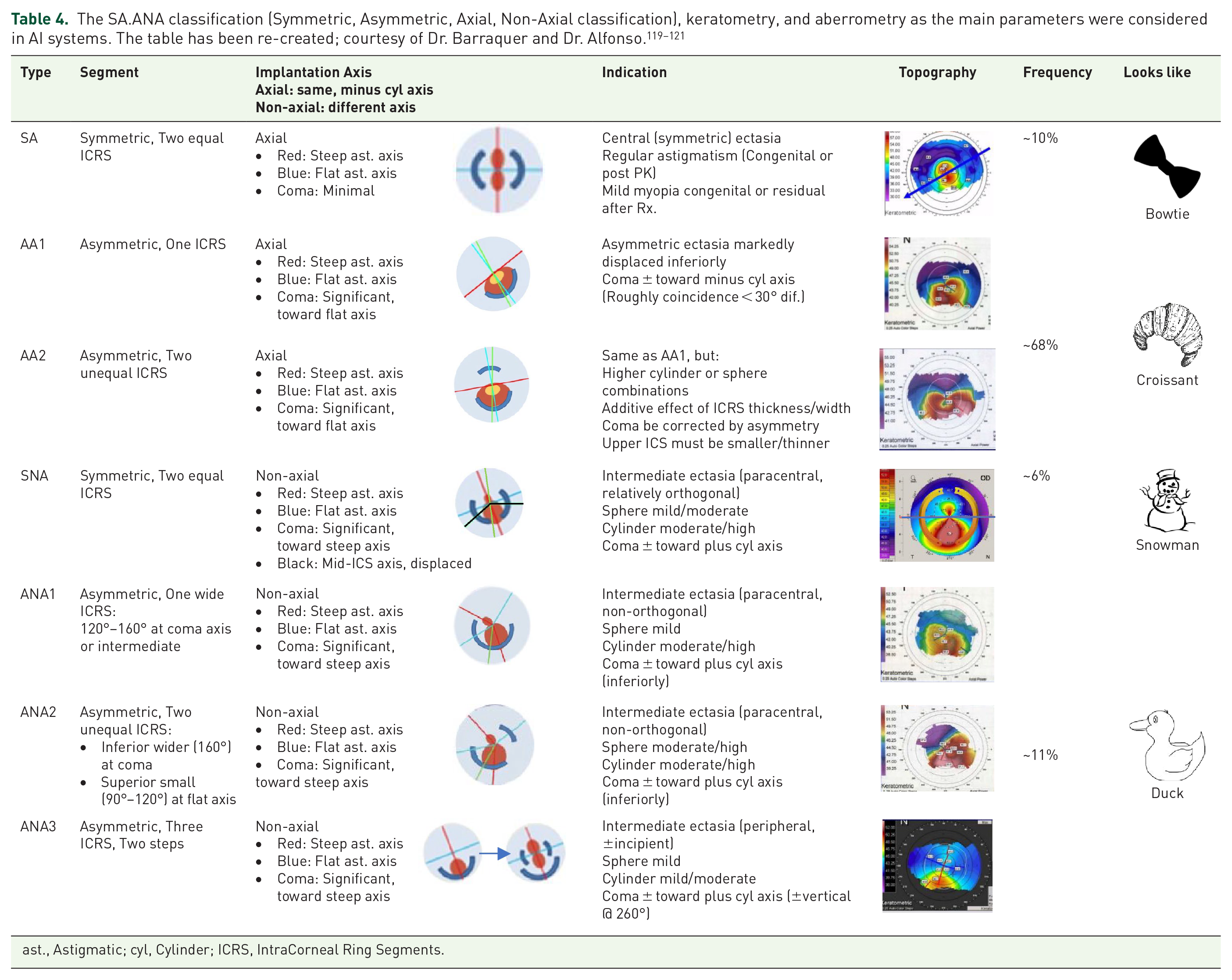
The intraoperative complications of intracorneal ring segments are incomplete channel development, suction loss, decentered channel creation, anterior or posterior perforation, shallow ring implantation, ring decentration and displacement into the anterior chamber, implant breakage, and epithelial defect or plug. 115 In spite of presenting many safety limit about segment thickness in relation to central thickness, and a plenty of nomograms about ICRS (Figure 5 and Table 4) by the main ICRS manufacturers (Figure 6 and Supplemental Figure 8), controversies about prediction of visual and refractive outcomes are still the most challenging.115,116

ast., Astigmatic; cyl, Cylinder; ICRS, IntraCorneal Ring Segments.

Alfonso Morphological Classification of Keratoconus. The figure has been re-created; courtesy of Dr. Alfonso. 122
According to the study about optimizing outcomes of ICRS, alteration in asphericity and keratometry are the main parameters affected by ICRS implantation. 123 The mean absolute error values for asphericity and mean keratometry were 0.19 and 1.18, for the nomogram, compared to 0.11 and 0.09 for the algorithm of computational models based on ML. 36 Therefore, in agreement with Piñero and Alio readjusting the nomograms is required to provide more predictable results while taking into consideration biomechanical and aberrometric characteristics. 124
In 2017, Lyra et al. 125 analyzed and compared changes in the anterior and posterior corneal surfaces after ICRS implantation. K2, astigmatism, elevation at the thinnest point, and apex, and the maximum elevation in the central 4 mm reduced statistically significantly both anterior and posterior surface. However, asphericity in posterior surface did not show a statistically significant change.
Nearly all studies based on the third-generation nomogram126,127 found decrease of keratometry with improvement in visual acuity, but not asphericity.
The manufacturer’s nomograms and artificial neural network (ANN) groups were used to determine the number (1 or 2), arc length, and thickness of ICRS. Increased visual acuity, decreased spherical equivalent, and improved optical quality are all benefits of using ANN to guide ICRS for keratoconus patients.128,129
Phakic intraocular lenses
pIOLs are a beneficial treatment for reducing anisometropia when the patient has stable refraction or at least excellent enough best spectacle correction visual acuity without high irregular astigmatism, for example, after corneal surgeries like ICRS and collagen cross-linking or keratoplasty. Another crucial factor is considering the potential for post-pIOL laser (PRK-CXL and correct HOAs) to address remaining unfavorable refractive outcomes. 54 Optimal patient work-up prior to pIOL implantation (angle-supported anterior chamber, iris-claw anterior chamber, and posterior chamber) has been depicted in Figure 7.

The pre-operative works up in phakic IOL implantation.ML- and DL-based devices can become a tool for predicting the ICL vault and subsequently determining the correct ICL size (the main issue in phakic IOL work up). All part of the figure has been gathered under the CC-BY-NC (Creative Commons Non-Commercial License).130,131
AI-based studies showed the results of the vectorial analysis could be deemed satisfactory because compensating for astigmatism in keratoconus patients is far more challenging than in other cases.132,133 The main causes of this are keratoconus’s irregular astigmatism profile and the other, which is connected to the first, because it might be challenging to get refraction in keratoconus patients, particularly in those with severe ametropia. 134
To attain the targeted optimum vault and ideal size, different modalities have been proposed, and some of them take advantages of AI, such as the manual mode of WTW computation from the Eyemetrics toolbox, the Orbscan IIz slit scanning approach, 135 summation methods with Pentacam HR, 136 ultrasound biomicroscopy (UBM) for STS measurement,134,137 AS-OCT for ATA diameter, 138 crystalline lens rise (CLR), and lens vault. 137 Even though, ideal size has still the main significant repercussion during phakic IOL (intra-ocular lens) implantation.
Using ML and DL can utilize pIOL implantation. Especially in choosing the correct size of the implantable Collamer lens (ICL; STAAR Surgical Co., Monrovia, CA, USA) as a posterior phakic lens, ML-based techniques had been helpful. 139 Appropriate ICL sizing establishes a safe postoperative ICL vault, the space between the ICL and the crystalline lens. The optimal ICL vault, according to the widespread view, is 500 μm and should not be longer than 1000 μm. Angle-closure glaucoma abnormalities and abnormally large pupils are linked to a higher vault after ICL implantation without a center hole. In contrast, anterior subcapsular cataracts are more likely to develop in patients with lower vaults. 139 Maximum CLR in angle-based evaluation (Baikoff) by 300, and sulcus-based evaluation (Saketa) by 600, have been described.
In a study comparing the traditional nomogram, all ML techniques, including the random forest regressor, the gradient boost regressor, the linear regressor (LR), and the support vector regressor, offered lower mean absolute errors and higher percentages of eyes within 50–200 µm of the targeted ICL vault (p < 0.01). 21
Collagen cross-linking and intrastromal corneal ring segments help to stabilize topography before phakic IOL implantation. In the progressive KC eyes, combined CXL, ICRS, and PIOL implantation is a reliable, safe, and efficient treatment. Integration of ML with C-CXL + ICRS for cases with higher unusual astigmatism and even worse visual acuity, whereas C-CXL + TG-PRK for patients who require unusual astigmatism correction but have higher visual acuity, can optimize KC management strategy near to emmetropia.54,77,140
Keratoplasty in keratoconus
Penetrating keratoplasty (PK) as the main technique of graft gives a predictable visual recovery; however, immunologic rejection, graft failure, cataract development, and secondary glaucoma are still the main drawbacks. In spite of several advantages of Deep Anterior Lamellar Keratoplasty (DALK) such as post-DALK ectasia, and rejection due to preservation of the host endothelium and Descemet’s membrane (DM),141,142 however, there are controversies between rate of performance of DALK. 143 It is essential to consider indication for DALK and compare different surgical techniques. 144 The most commonly used methods are the Anwar (or big-bubble) and Melles procedures. 145
Melles pioneered using an air bubble in the anterior chamber as a stromal depth indicator to create a mirror image of the dissector blade. Dissection by method of Melles and femtosecond-assisted DALK (F-DALK) without big-bubble, both, may cause decreased visual acuity and contrast sensitivity due to remaining irregular stromal thickness in KC patients. 146 Thus, it would be better to complete F-DALK by injecting the air bubble into the residual stromal bed, thereby creating a big bubble to expose the DM. 147 Shehadeh-Mashor et al. compared F-DALK with manual trephination in patients with KC, iatrogenic ectasia, and scar. The visual outcomes were similar but femtosecond laser (FSL)-assisted mushroom showed earlier visual recovery compare to manual DALK with straight-edge configuration. 148 Anwar proposed baring DM with the ‘big-bubble’ (BB) technique, which pumps air into the deep stroma and produces a big bubble between the stroma and DM. 145 Three types of bubbles can form when air is injected into the cornea’s stroma, including type 1 BB [the most prevalent type of BB occurs between the stroma and the pre-Descemet’s layer (PDL)]; Type 2 BB (between the DM and the PDL) has less resistance (than type 1) to physical shock, and the third form is the mixed BB in which both type 1 and type 2 exist, either fully or partially. 141
Managing the development of BB and predicting the ratio of type 1 created over type 2 has an essential effect on the success rate. In comparing a type-1 bubble with 2, a type-2 bubble presents a risk of perforation, which will be forcefully converted to mushroom-shaped or full-thickness PK, also causes an increase in the intraoperative problems of DALK and, finally, a higher risk of forming a double anterior chamber postoperatively. 149 However, there is no difference in the best-corrected visual acuity, depending on whether type-1 or type-2 bubbles are present during pneumatic dissection.
The cannula or needle insertion into the stroma is another essential point for successful BB development. 150 An approach that is too superficial often results in extensive emphysema of the cornea without creating a bubble and can potentially generate a type-2 bubble, either entirely or partially. On the other hand, an insertion that is too deep can perforate the DM.
As an alternative to manual dissection when pneumatic dissection fails, Ophthalmic Viscosurgical Devices (OVD) provided profound plane separation; for parameters like corneal densitometry improvement and visual restoration, OVD showed a statistically significant higher results versus eyes dissected with air in the 1 and 3 months postoperative though they became comparable in the latest follow-ups. 151
In many cases, there is a risk of DALK conversion to PK and perforation risk, even in expert hands. 152 A lamellar approach can still be used in such cases, especially if the scars do not disrupt the visual axis. However, post hydrops-cases are not good candidates for DALK, even manual, and the procedure will need to be modified to a PK intraoperatively if a large tear is caused (larger than 2 clock hours). Decentered grafts and surgical sutures are two main issues in PK of keratoconus patients.
Decentered grafts can create significant irregular astigmatism in the visual axis; Keratoconus patients may benefit from employing same-diameter trephines for both donor and host tissues, which shrinks the donor button and lowers postoperative myopia. However, when the anterior lens-to-retina length is less than 20.19 mm, a reduction of donor size could result in significant postoperative hyperopia.153–157
The surgeon can choose interrupted sutures, combined continuous and interrupted sutures, single continuous sutures, or double continuous sutures following placing the four cardinal 10-0 nylon sutures. 158 In several conditions, interrupted sutures should always be the closure method of choice, including:
Where a partial or complete suture removal in one region of the graft is likely to be needed during the postoperative period: a. Pediatric keratoplasty (sutures becoming loose too quickly) b. Vascularization in the host cornea (occasionally seen after a hydrops episode or contact lens-related keratitis) c. Multiple previous rejections d. Other inflammatory concomitant conditions
2. To close big, decentered grafts near the limbal area due to their higher rejection risk. 158
DALK has several advantages over the oldest technique, PK, in terms of duration of visual recovery, endothelial cell density loss, and immunological graft rejection rates. However, oversizing the graft for better integration is the primary source of the visible myopic shift and corneal steepening, particularly during big bubble DALK. 159 Still, more research on long-term results is needed to assess the influence of DALK on keratoconus. 160 According to a meta-analysis, BCVA, refractive, and topographical cylinder were comparable between patients who underwent PK and those undergoing DALK. 161
DALK is the first surgical option for patients with keratoconus and possibly other corneal stromal pathologies with normal endothelium. Because of its benefits, such as better preservation of globe integrity, reduced intraoperative complications, and elimination of endothelial graft rejection, DALK can be considered an alternative to PK for this condition.
Postoperative corneal ectasia following PK for KC is predicted to occur in 6–11% of individuals 20–25 years after surgery. 162 The term ‘recurrent keratoconus’ has no established cause and even clear meaning and criteria. A range of mechanisms have been put forth, including the insufficient excision of keratoconic host tissue during PK, particularly for grafts of 7 mm recipient diameter; grafting donor corneas with subclinical keratoconus; the development of the ectatic disorder; the discharge of degradative enzymes from an anomalous host epithelium; and the presence of variations in the Bowman membrane after epithelium-stroma. 162 Failure to thoroughly remove the damaged tissue may cause the host to develop keratoconus, possibly involving donor tissue. Despite several management options for post-PK ectasias, such as contact lens fitting, wedge resection, corneal sutures, IOL implantation, overlay DALK, peripheral reconstructive and annular lamellar keratoplasty, tuck in lamellar keratoplasty, and repeating PK, their limitations are more than primary graft. 163
Artificial intelligence and robotics in keratoplasty, either PK or DALK (i.e. keratoplasty using a femtosecond laser with OCT guidance), can involve less of a steep learning curve and ultimately cause high satisfaction for both physician and patient (Figure 8).

Several studies on F-DALK as an alternative robot-assisted surgery for KC have been published. Still, only two have compared its outcomes to manual trephination DALK (M-DALK).167,168 M-DALK and F-DALK were compared in a larger study, and identical visual acuity values were found; however, the M-DALK group had higher myopia and mean keratometry values. 167 In long follow-ups, neither evident benefit of F-DALK over manual trephination, nor of one cut configuration over the others, has been found. 168 The femtosecond laser could standardize the big-bubble technique in DALK, reducing the risk of intraoperative complications and allowing good refractive outcomes. 168 Both with and without OCT guidance, FSL can help establish a stromal channel for air injection into the cannula. The FSL is used to incise and remove the anterior stroma. Then, the air is supplied by a cannula or needle with the guidance of OCT to create a big bubble, which is subsequently unroofed by a second FSL laser cut. 169
As an AI-analyzable machine, AS-OCT can detect corneal layer rearrangement, providing surgeons with crucial information for surgical planning, both in terms of timing and method.13,164 For instance, AS-OCT is an important test for post-PK ectasia planning, which helps the surgeon identify and gauge the donor graft’s diameter, the extent of the graft-host thinning, and the precise location of the undamaged corneal tissue surrounding the graft. 170 AS-OCT parameters showed statistically significant changes in the post-PK KC group versus groups undergoing PK for other diseases as a control. 170
Higher keratometric parameters, including Ks, AvgK, Sph, and Kmax, heightened the corneal ectatic changes after PK. However, astigmatism changes in post-PK eyes should not be used for measuring the progression of KC due to the similarity of astigmatism (regular or irregular) and HOA between groups. 170 The logic for using ACD as an indicator of corneal protrusion is supported by positive correlation of ACD with corneal protrusion parameter, even though ACD can also depend to the lens opacity. In short, KC eyes long after PK show inferior graft and host corneal thinning, and corneal protrusion. Corneal power parameters such as Kmax or Ks can be analyzed to monitor KC progression after PK via AI. 170
Artificial intelligence and OCT image data cannot only detect, classify, and optimize keratoplasty but also can take advantage to predict need to keratoplasty.171–173 Recent reports found that intelligent algorithms can accurately forecast the effectiveness of BB creation in DALK. 174 KC eyes formed more BBs than corneal-opacified eyes. That study merely tested if an accurate algorithm could anticipate the creation of a huge bubble with limited data without using known related factors. The determination success rate was 78.3% for successful and 69.6% for failed big bubble, suggesting the feasibility of an automatic judgment system. Due to the short sample size, this pilot study could not determine the accuracy of individual and special indications for DALK, such as herpetic scars, traumatic scars, solely KC, or corneal dystrophies. 174
Finally, ML strategy can promote new developments, both in diagnose and treatment modalities. As the same way and according to the global studies, the incidence of keratoplasty has decreased dramatically. Individualized course of treatment based on disease stage determine the most optimized action to achieve emmetropic eyes.
Discussion
New developments in artificial intelligence, particularly with promising results in the early detection and management of KC, have favorably altered the natural history of the disease over the last few decades. However, usually, the sample size is not only sufficiently large in medical studies, keratoconus severity and also incidence varies widely due to different classifications; 38 hence, direct comparison between studies is difficult. 37
In spite of the limited clinical use of existing AI models, however, incorporation of them could enable surgeons in patient selection and decision-making (Figure 9).

In a retrospective cohort of KC patients, Kato et al. 75 created a CNN model to predict progression using the axial map, corneal thickness, and patient age. The use of a CNN (VGG-16) DL model showed an accuracy rate of 81.4%. KC patients will first be categorized using a semi-supervised and unsupervised DL model in order to develop a disease progression detection strategy. The study used 29 factors retrospectively acquired from topography, tomography, clinical, and demographic data to categorize KC patients using a Bayesian deep neural network and to cluster patients into four groups using a Gaussian mixture model. The capacity to predict treatment results for KC patients is still impacted by variable corneal characteristics. For procedures like ICRS, using AI-based models may enable professionals to take a more advantageous approach.36,116 The first neural network to predict changes in keratometry and corneal astigmatism after ISCR implantation was introduced by Valdeé-Mas et al. The best models had an error of less than1D (0.97D corneal curvature and 0.93D astigmatism). 116 Later, in a comparison with clinical data for ICRS (Ferrara segments), Lyra et al found the optimum mean absolute error value to be 0.19 for corneal asphericity and 1.18D for mean keratometry values. 36
These results are encouraging, but they need to be revised further before being used in the surgical decision-making process. Researchers may ultimately discover more benefits in developing models to maximize this more recent breakthrough given the parallel development of topographically directed collagen cross-linking. Researchers discovered that OCT factors (in addition to a great roles in anterior segment surgeries) had a greater impact on the model than topographical imaging variables, indicating that minor differences between groups would be easier to spot through corneal morphology. 177 However, the combined strategies was still able to deliver the best results. 177
As Shetty et al. mentioned the best management of keratoconus is multistep and multifaceted diagnosis and treatment 175 ; therefore, several features, in conjunction with innate artificial intelligence, are already underway. The necessity for this comprehensive systematic overview is depiction of key points about management strategies in keratoconus.
Overall, a large portion of AI-based keratoconus studies is about ANNs. ANNs fall within the artificial intelligence branch of ML, which, in the words of Arthur Samuel, ‘gives computers the ability to learn without being explicitly programmed’. 178 The clinical outcomes of an ANN maximize the predictability of surgical treatments in keratoconus. The initial phase of any algorithm in ML is data processing based on the weights of connections between neurons. This is necessary because the computer must be designed to maximize performance using data from prior experience. Regarding each step of keratoconus management, the AI system can help the physician identify the best individual plan. 128 However, to validate this system, more study with distinct datasets is required. As the program improves its learning, ANN’s predictability will improve due to the ongoing incorporation of fresh examples.
Conclusion
The latest advancements in ophthalmology have made it possible for patients with keratoconus to receive an early diagnosis and effective visual rehabilitation under much safer conditions. Each patient can receive a customized course of treatment based on disease stage and treatment goals. The efficient and widespread clinical translation of ML models in keratoconus management is a crucial goal of potential future approaches to have best possible visual performance in KC patients.
Supplemental Material
sj-docx-3-oed-10.1177_25158414241232258 – Supplemental material for Keratoconus: exploring fundamentals and future perspectives – a comprehensive systematic review
Supplemental material, sj-docx-3-oed-10.1177_25158414241232258 for Keratoconus: exploring fundamentals and future perspectives – a comprehensive systematic review by Sana Niazi, Zisis Gatzioufas, Farideh Doroodgar, Oliver Findl, Alireza Baradaran-Rafii, Jacob Liechty and Majid Moshirfar in Therapeutic Advances in Ophthalmology
Supplemental Material
sj-png-1-oed-10.1177_25158414241232258 – Supplemental material for Keratoconus: exploring fundamentals and future perspectives – a comprehensive systematic review
Supplemental material, sj-png-1-oed-10.1177_25158414241232258 for Keratoconus: exploring fundamentals and future perspectives – a comprehensive systematic review by Sana Niazi, Zisis Gatzioufas, Farideh Doroodgar, Oliver Findl, Alireza Baradaran-Rafii, Jacob Liechty and Majid Moshirfar in Therapeutic Advances in Ophthalmology
Supplemental Material
sj-png-2-oed-10.1177_25158414241232258 – Supplemental material for Keratoconus: exploring fundamentals and future perspectives – a comprehensive systematic review
Supplemental material, sj-png-2-oed-10.1177_25158414241232258 for Keratoconus: exploring fundamentals and future perspectives – a comprehensive systematic review by Sana Niazi, Zisis Gatzioufas, Farideh Doroodgar, Oliver Findl, Alireza Baradaran-Rafii, Jacob Liechty and Majid Moshirfar in Therapeutic Advances in Ophthalmology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.