Abstract
Pediatric corneal transplant is a highly demanding and technically challenging procedure for the cornea surgeon in today’s era. These cases pose unique challenges in clinical and surgical management. The indications of pediatric corneal transplant can be therapeutic, tectonic, optical, and cosmetic. Pediatric patients undergoing corneal transplants are at a high risk of graft infection, failure, rejection, dehiscence, and amblyopia due to young age, robust immune system, increased incidence of trauma, and compliance issues. The other factors contributing to graft failure can be allograft rejection, secondary glaucoma, corneal vascularization, multiple surgeries, vitreous prolapse, and lack of treatment compliance. A successful corneal transplant in children depends on meticulous preoperative evaluation, uneventful surgery, the expertise of a corneal surgeon, and regular and timely postoperative follow-up. Therapeutic and optical penetrating keratoplasty are the most commonly performed transplants in children. However, with the advancements in surgical technique and management protocol, the current focus has shifted toward lamellar keratoplasty. Lamellar keratoplasty offers early visual recovery and potentially fewer complications. Visual rehabilitation through corneal transplant in otherwise blind eyes can be a boon for the children. Recently, keratoprostheses have been promising in children with multiple graft failures. The current review gives insights into epidemiology, etiology, indications, clinical characteristics, investigations, management options, recent advances, and the future of pediatric corneal transplants. As surgical techniques continue to grow and comprehension of pediatric corneal transplants is improving, we can safeguard these eyes with the best possible anatomical and functional outcomes.
Keywords
Introduction
Corneal blindness is one of the major causes of visual impairment in the pediatric age group, which is more prevalent in developing countries. 1 Congenital corneal opacification can affect the visual axis and result in irreversible visual sequelae such as amblyopia and blindness. 2 Early corneal transplantation is recommended to safeguard these eyes to prevent visual deprivation, amblyopia, and permanent blindness. Severe corneal ulceration secondary to trauma and non-healing viral keratitis resulting in descemetocele, perforation, etc., requires timely therapeutic keratoplasty to have a good anatomical and functional outcome. 3 Pediatric keratoplasty was infrequently performed approximately five decades ago and was reserved only for patients with bilateral corneal opacification. Penetrating keratoplasty (PKP) remains the gold standard for corneal opacification due to varied etiologies. 3 The cumulative incidence of graft failure following PKP is approximately 35% at the 10-year mark. Immune rejection stands out as the primary factor contributing to the failure of the graft. Moreover, about 30% of corneal transplants undergo a minimum of one instance of immune rejection. 4 However, this usually varies with corneal etiology. The factors that make pediatric corneal transplantation a challenge are as follows: difficulty in examining the children, increased elasticity of sclera, positive vitreous pressure, increased incidence of secondary glaucoma, fibrinous uveitis, high risk of rejection, infection, amblyopia, and difficulty in communication with the children. 4 However, the pediatric corneal transplant has had better success rates with a better understanding of the existing techniques, intraoperative and postoperative complications, management, evolution of newer techniques, better access to eye care, and dedicated counseling. 5 Off late, with the advent of lamellar keratoplasties, pediatric corneal transplantation has undergone a massive revolution. There has been a shift toward deep anterior lamellar keratoplasty (DALK) for superficial corneal opacity and scarring due to keratoconus, although PKP can have equally good results in these cases. 6 Descemet stripping endothelial keratoplasty (DSEK), Descemet stripping automated endothelial keratoplasty (DSAEK), ultra-thin/nono-thin DSEK, Pre-Descemet endothelial keratoplasty (P-DEK), and Descemet membrane endothelial keratoplasty (DMEK) are increasingly being used over PKP for the management of congenital hereditary endothelial dystrophy (CHED), posterior polymorphous corneal dystrophy, Descemet membrane breaks due to forceps delivery, and Peter’s anomaly. 7 Pediatric keratoprosthesis is the replacement of the opaque host cornea with an artificial cornea. It has been kept as a last resort for managing recurrent graft failure in pediatric patients and corneal opacities with uncontrolled glaucoma. 8 However, pediatric keratoprosthesis is no longer used in the United States because the success rate in children is extremely poor. This review focused on the epidemiology, indications, classification, types of corneal transplant, preoperative preparation of patients, surgical technique, intraoperative and postoperative complications, postoperative management, prognosis, and recent advances in pediatric corneal transplantation.
Literature search
A literature search was done on PubMed, Google Scholar, EMBASE, Cochrane Library, Medline, PubMed Central, and Web of Science (Wos) from Clarivate Analytics till July 2022. The search strategy was composed of MeSH terminologies which included Title Search (TS) ‘ Pediatric keratoplasty’ or ‘Keratoplasty in children’ or ‘Pediatric corneal transplantation’ or ‘Congenital corneal opacity’ or ‘Adult keratoplasty’, with a variable combination of terms like ‘Indications’, ‘Types’, ‘Outcome’, with interposition of Boolean operators ‘AND’ and ‘OR’. The inclusion criteria were as follows: (1) All research articles focused on pediatric corneal transplantation; (2) Editorials, Review articles, Original articles, Case series, Case reports, Photo-essay, and Image and Letters; (3) Article search from (4) All articles in the English language. The exclusion criteria were all articles in a language other than English. In addition, the references of the articles were searched for the missed references.
Epidemiology
Worldwide, approximately 14 million children are suffering from blindness, 9 with childhood blindness prevailing at 8% in the South East Asian region. 10 Considering pediatric blindness, corneal pathologies are responsible for the majority of the cases.11,12 However, pediatric keratoplasty is a seldomly performed surgical procedure due to the technical complexity and high rejection rate. 13 In the United States, there are 4.16 infant corneal transplants for every 100,000 live births. 14 In Australia, 5% of the total corneal transplants were pediatric keratoplasties according to the Australian Corneal Graft Registry. 15 However, lately pediatric keratoplasty has gained popularity due to improvements in surgical techniques and postoperative care. A study done by Zhu and Prescott evaluated surgical trends in pediatric keratoplasty by obtaining data for the period between 2005 and 2017, from the members of the Eye Bank Association of America. They received data from 15 eye banks, which included that a total number of 2708 cases underwent pediatric keratoplasty. 16
Indications
There is a variation in the indications for pediatric keratoplasty from region to region. In the developed world, congenital corneal opacities are the most common indication for pediatric PKP, whereas in developing nations, acquired corneal scarring secondary to either infectious etiology or trauma represents the most common cause.3,16,17 A study done by Zhu et al.(8,10) showed that the most common indication for pediatric corneal transplant was corneal ectasia or thinning (33.7%), which was even more common in the adolescent age group of 13–17 years (56.3%). Congenital opacities accounted for 17% of cases in the age group of 5 years and below.8,10 A retrospective study done in the Indian population by Sharma et al. revealed the most common indications to be infectious keratitis (43%), congenital glaucoma (16.6%), and corneal trauma (11.2%). 17
Pediatric keratoplasty indications have been most commonly classified into four categories 18 :
Congenital
Acquired traumatic
Acquired non-traumatic
Regraft.
This classification was proposed by Stulting et al. and was revised by Al-Ghamdi et al. to categorize pediatric corneal opacities about visual prognosis post-keratoplasty. 19 Congenital, acquired non-traumatic, and acquired traumatic conditions account for 14–64%, 19–80%, and 6–29%, respectively. 20 (Table 1)
The summary of indications of pediatric keratoplasty.
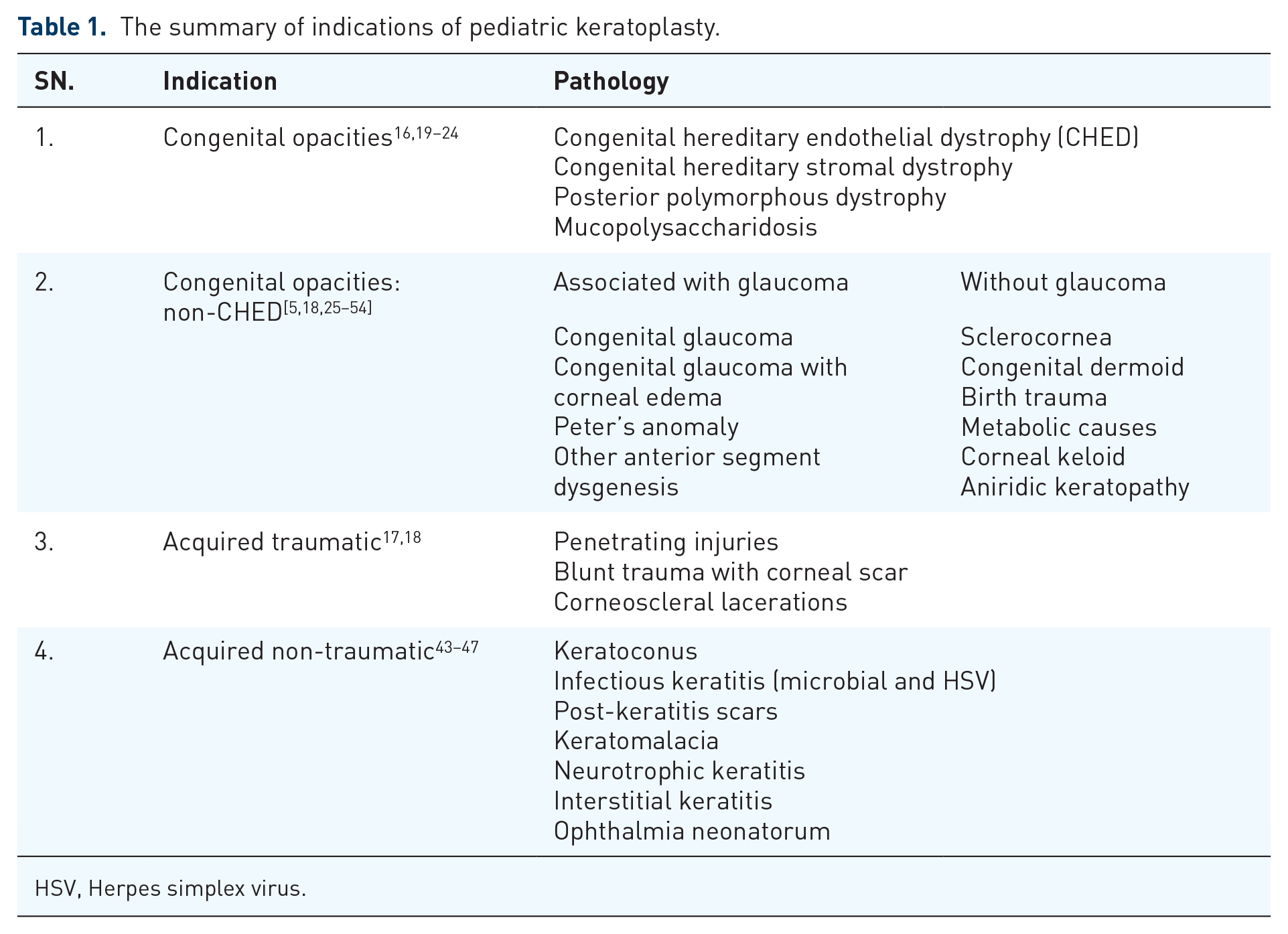
HSV, Herpes simplex virus.
Congenital opacities
Congenital hereditary endothelial dystrophy
CHED is characterized by non-progressive, bilateral, symmetrical corneal edema, and opacification present at birth or in early infancy, affecting the corneal endothelium. 21 The corneal edema is significant enough to cause stimulus deprivation amblyopia and enlargement of the globe resulting in axial myopia. This makes intervention imperative.
Non-congenital hereditary endothelial dystrophy
Associated with glaucoma
Congenital glaucoma
Corneal decompensation secondary to congenital glaucoma is an uncommon indication for pediatric keratoplasty. 22 Entities resulting in congenital corneal opacification associated with glaucoma include congenital glaucoma, Peter’s anomaly with glaucoma, and CHED with glaucoma. 23 Intraocular pressure (IOP) should be controlled prior to PKP. Cyclodestructive procedures, trabeculectomy, and glaucoma drainage devices are the surgical options in pharmacologically resistant cases. This is important to reduce the size of buphthlamos before keratoplasty. However, penetrating glaucoma surgeries increase the risk of graft failure by breaching the blood–aqueous barriers. 20
Peter’s anomaly
Peter’s anomaly is one of the most common causes of congenital corneal opacification. 20 Currently, it is classified into two types. 24 Out of the two types, type I generally has better a prognosis due to fewer posterior segments and systemic associations with PKP being recommended within the first year of life. 55
A significant proportion of PKPs done in Peters anomaly usually undergo chronic graft failure and might even require repeat keratoplasties.25 –27,55 This is true for eyes that have severe diseases, require larger donor corneas, exhibit central nervous system (CNS) irregularities, or display anterior synechiae. These eyes tend to have less favorable outcomes compared to eyes without these characteristics. Given repeated graft failures, alternatives include posterior lamellar keratoplasty and optical iridectomy bypassing the corneal opacity.28,29
Other mesenchymal dysgenesis
The entities mesenchymal dysgenesis and anterior chamber cleavage syndrome include malformations of mesodermal tissues (corneal endothelium and stroma, anterior chamber angle, and iris stroma) along with an ectodermal contribution. The stepladder classification has divided them into peripheral, central, and a combination of both peripheral and central components. The spectrum includes disorders like Peter’s anomaly, Axenfeld anomaly, Reiger anomaly and syndrome, and posterior keratoconus. They have a high incidence of associated glaucoma. 30
Infrequently associated with glaucoma
Dermoid
Being classified as choristoma, they are whitish-yellow cones on the anterior ocular surface, consisting of ectodermal and mesodermal components. 31 They mostly cause peripheral opacification and require simple excision or lamellar keratoplasty. Occasionally, the entire corneal surface might be involved, associated with iridocorneal adhesions and cataracts. Tectonic PKP or lamellar patch graft and excision of dermoid is indicated in such cases. 32
Metabolic causes
Corneal opacification can be associated with many metabolic disorders. Characteristically, the cornea is transparent at birth and is followed by progressive opacification. 20 Mucopolysaccharidoses (MPS) are a group of lysosomal storage disorders affecting the glycosaminoglycan (GAG) catabolism which forms a major constituent of cornea. 33 Associated open-angle glaucoma, 34 pigmentary retinopathy, and optic atrophy can negatively affect the visual prognosis. 35 Although re-opacification of the donor tissue is common because of the GAGs present in it, surgery can be performed at an early age. Furthermore, the lifespan is also short in these cases if the disease is uncorrected. Thus, early surgery allows visual rehabilitation in the limited time available. 35
Sclerocornea
It is a rare congenital, non-progressive, and non-inflammatory condition characterized by partial or complete scleralization of the cornea. This scleralization consists of vascularization, opacification, and flattening of the normal curvature. 36 It can be isolated or associated with additional ocular and systemic anomalies. 37 It is generally associated with a poor prognosis requiring repeated regrafts. 38
Corneal keloid
Congenital corneal keloids are benign lesions that might involve the complete corneal stroma. Associations with other ocular abnormalities are common, including iridocorneal adhesions, aniridia, cataracts with subluxated lenses, and anterior segment mesenchymal dysgenesis. They can also be associated with glaucoma. Superficial keratectomy or lamellar keratoplasty or PKP can be performed for visually significant lesions. 39
Birth trauma
Forceps delivery can result in Descemet’s tears, which are usually central and unilateral. 20 Posterior lamellar keratoplasty has been done for associated corneal decompensation. 40 A Descemet detachment alone can be managed by air descemetopexy. 41 Even if the corneal edema resolves, residual high astigmatism may require PKP or DSEK if contact lens wear is not feasible. 20
Aniridia
Apart from the absence of iris tissue, other ocular structural defects are often associated. Corneal lesions, termed aniridic keratopathy, include peripheral pannus and epithelial abnormalities, often involving the center, raising the need for keratoplasty. 42 Other defects such as macular hypoplasia, cataracts, amblyopia, and glaucoma are often responsible for poor visual outcomes. In addition to PKP, limbal stem cell transplantation might be required to rectify the underlying epithelial pathology. 43
Posterior polymorphous corneal dystrophy
It is a bilateral, autosomal dominant congenital anomaly of the Descemet membrane. 44 The defects at the level of the Descemet membrane can result in regular or irregular astigmatism and keratoconus. Also, since the manifestations of PPCD can be asymmetric, this can result in anisometropic amblyopia. 45 In severe cases, endothelial decompensation can result in focal or diffuse corneal edema. As it progresses, opacification can occur. 46 Keratoplasty will be needed in 20–25%. If the corneal edema is not significant, lamellar keratoplasty can be opted as the surgical procedure of choice. 47
Acquired traumatic
Corneal scarring resulting from penetrating injuries is responsible for 8–26% of pediatric keratoplasties.5,19,48,49 The development of amblyopia, which can complicate the outcome, is of concern.
Acquired non-traumatic
In developing nations, acquired nontraumatic corneal opacities are a major indication for pediatric keratoplasty, with a study showing 71.32% of pediatric PKPs being done for it. 50 The most common indications include infectious keratitis and post-keratomalacia corneal melts.50 –52 Vitamin A deficiency, malnutrition, systemic diseases, and lack of nutrition predispose to keratomalacia.53,54 This contrasts with what is seen in the Western world.
Regrafts
Regrafts in pediatric corneal transplants refer to the necessity of performing a second (or subsequent) corneal transplant surgery in children, following a previous graft. This situation may arise due to various reasons, including graft failure, rejection, infection, or other complications associated with the initial transplant. Pediatric corneal transplants are complex and carry unique challenges, often due to the size of the eye, the potential for rapid eye growth, and the difficulty in examining and managing younger patients. 38
Keratoplasty – Types
Penetrating keratoplasty
It is an open-sky procedure, where the host cornea is replaced with a full-thickness donor tissue. A study done by Zhu et al. has shown that among the various types of pediatric keratoplasty, PKP remains the most performed. 8 The outcome depends upon the initial indication. 56 Poor outcomes are generally associated with the presence of anterior segment dysgenesis, younger age, and congenital opacities, being limited by amblyopia and other ocular factors. Better outcomes are usually associated with CHED, 12 Peter’s anomaly type 1, 57 and keratoconus. 58 When compared to adult PKP, pediatric PKP offers a set of technical difficulties. As the cornea is less rigid in the pediatric age group, trephination of the host cornea is more difficult (Table 2). 59
A comparison between adult and pediatric keratoplasty.

Although the technical difficulty of the procedure is relatively less when compared to lamellar procedures, there are several inherent disadvantages of this procedure. The most common causes of failure include rejection, glaucoma, and infection, with rejection being seen in 50% of pediatric PKP. 60 However, with good follow-up and high-dose topical steroids initially, most PKPs will survive. The latter is expected, as a higher antigen load is transplanted and the immune response in the pediatric age group is more robust. Considering the complications associated and the advantages of the other surgical procedures, there has been a recent increase in lamellar surgeries [Figure 1(a)–(c)]. 8

Digital slit lamp images of the pediatric keratoplasty of (a) patient’s right eye post-penetrating keratoplasty depicting a large 8 mm well-opposed graft, graft edema, intact sutures, well-formed anterior chamber, a fibrinous membrane with few blood clots in the anterior chamber; (b) patient’s left eye post-penetrating keratoplasty depicting a 7.5 mm clear well-opposed graft with intact sutures; (c) patient’s left eye post-therapeutic keratoplasty depicting diffuse congestion, 7.5 mm clear well-opposed graft, intact sutures, an irregular pupil with aphakia; (d) patient’s left eye post-DALK depicting a relatively clear graft with intact sutures; (e) patient’s left eye post-DSEK depicting a well-opposed DSEK lenticule, clear cornea with stable IOL; and (f) patient’s left eye depicting a well-opposed DMEK graft with intact tunnel sutures and side port sutures.
Anterior lamellar keratoplasty
In anterior lamellar keratoplasty (ALK), there is a replacement of only the affected anterior stroma. This allows for better tissue strength and decreases the risk of rejection.61–64
Superficial anterior lamellar keratoplasty and automated lamellar therapeutic keratoplasty
Superficial anterior lamellar keratoplasty and automated lamellar therapeutic keratoplasty can be performed for opacities involving the anterior 250 µm and 300 µm, respectively.65,66 Common indications include Reis-Buckler corneal dystrophy, 67 healed keratitis, healed shield ulcer, congenital dermoid and Salzmann nodular degeneration, 68 corneal scarring, and choristoma. 66 Common complications include residual corneal pathology, mild interface haze, recurrence of underlying pathology, dry eye, and epithelial ingrowth. 69 The visual outcome directly correlates with the age at surgery. 70 A good visual outcome can be obtained if associated amblyopia is managed adequately. 66
Deep anterior lamellar keratoplasty
In DALK, dissection of the host tissue is done up till the level of Descemet membrane (DM) and a full-thickness graft without the Descemet’s membrane-endothelial complex is transplanted. DALK has been effectively done for several pediatric corneal pathologies including keratoconus, partial thickness corneal scar, exposure keratopathy, mucopolysaccharidoses (MPS), microbial keratitis, corneal dystrophies, superficial trauma, radiation keratopathy, and chemical injury.71–74 With DALK, as the host endothelium is preserved, the risk of endothelial rejection is eliminated and the higher endothelial cell count of a pediatric cornea is retained.74,75 In most developed countries, however, the main reasons for pediatric corneal opacities are congenital disorders such as Peters’ anomaly, sclerocornea, dermoid, or congenital glaucoma, The partial thickness technique does not work well in these children. Nor will it work well after corneal hydrops. In full-thickness or posterior lamellar surgeries, endothelial cell loss occurs during tissue handling and, if adult donor tissue is transplanted, the associated endothelial cell count will be relatively low. As children tend to have more years to live postoperatively when compared to adults, the significance of this endothelial loss is magnified, demanding a repeat graft. In case of failure, DALK grafts can be easily removed and followed by the placement of a secondary graft. The latter has a good visual and refractive outcome, without an accompanied increase in failure rate. 76 Lamellar keratoplasty is a good choice in children with partial (non-full-thickness) corneal opacities, such as anterior stromal infections, milder forms of keratoconus, and superficial trauma in which one can remove the diseased cornea and therefore preserve a normal Descemet’s membrane and endothelium. In most developed countries, however, the main reasons for pediatric corneal opacities are congenital disorders such as Peters’ anomaly, sclerocornea, dermoid, or congenital glaucoma. The partial thickness technique does not work well in these children [Figure 1(d)].
Femtosecond-assisted deep anterior lamellar keratoplasty
In femtosecond-assisted deep anterior lamellar keratoplasty (FDALK), a laser program is used to place bladeless, precise lamellar cuts. 77 An improved biomechanical profile of the tissue is achieved with FDALK. As a result, early suture removal can be done, decreasing the risk of rejection. 78 Good results have been reported in pediatric eyes for indications including keratoconus and congenital glaucoma.64,79,80
Posterior lamellar keratoplasty/endothelial keratoplasty
In posterior lamellar keratoplasty, only the Descemet-endothelium complex is replaced. 81 It is associated with a better wound safety profile as it is a closed-system procedure and a relatively low risk of rejection. This allows for faster rehabilitation and early tapering of steroids. The latter minimizes the risk of ocular hypertension and steroid-induced cataracts.82 –85
Descemet stripping automated endothelial keratoplasty
In DSAEK, in addition to the Descemet-endothelial complex, a small amount of stromal component is also transplanted. Indications in the pediatric age group include CHED,74,75 buphthalmos, 86 posterior polymorphous corneal dystrophy, 87 and Descemet membrane breaks due to forceps delivery. 40 Among these, CHED is the most common indication. 26 Although DSAEK has several advantages, due to the irregularity of the interface, the optical clarity might be inferior to PKP.88,89 Ramappa et al. reported 10-year outcomes of DSAEK in 180 pediatric eyes and found improved and safe outcomes with this technique. 90
Descemet stripping endothelial keratoplasty
The terms DSEK and DSAEK are used interchangeably in the literature. 91 Both techniques of lamellar keratoplasty are similar but differ in the preparation of the donor graft. In DSEK, the graft is obtained using manual dissection, whereas in DSAEK, the process is automated using a microkeratome which creates a more regular interface [Figure 1(e)].92,93 Another surgical procedure that is very similar to DSEK and DSAEK is Pre-Descemet’s Endothelial Keratoplasty (PDEK). In this procedure, the pre-Descemet’s layer (Dua’s layer) is transplanted along with the Descemet-endothelial complex. 94 The advantage of having pre-Descemet’s layer in the donor tissue is its role to act as a splint. This allows for better tissue handling, decreasing endothelial cell loss. 95 However, PDEK is rarely performed these days.
Descemet’s membrane endothelial keratoplasty
In DMEK, only the Descemet-endothelial cell complex is transplanted without the stromal component. This allows for an interface without posterior surface aberrations. 96 The literature on DMEK in the pediatric age group is limited. It has been performed in a child with Kearns–Sayre syndrome 97 and posterior polymorphous corneal dystrophy 60 with favorable outcomes. Other studies have shown encouraging results for DMEK done for endothelial dysfunction in the pediatric age group [Figure 1(f)].25,98,99
Pediatric keratoprosthesis
A keratoprosthesis allows for the replacement of the opaque host cornea with an artificial cornea. Boston Keratoprosthesis (KPro) is most commonly employed, the backplate being available in both adult and pediatric sizes. 100 Indications include congenital corneal opacities, 101 multiple graft failures, 102 congenital glaucoma with the decompensated cornea, 103 keratitis-ichthyosiform-deafness syndromes, 104 and lacrimal grand choriostoma. 105
Advantages of KPro include minimal refractive error and attainment of a clear visual axis in the immediate postoperative period. This minimizes the risk of amblyopia. In addition, the risk of rejection is eliminated, faster posterior segment evaluation can be done, and greater postoperative comfort is achieved.105,106 Disadvantages include lifelong care and follow-up. Due to a more pronounced inflammatory response in the pediatric age group, the incidence of retroprosthetic membranes is higher than in adults. 100 Similar to the adult counterpart, glaucoma remains a concern postoperatively. 107 However, pediatric keratoprosthesis is no longer used in the United States because the success rate in children is extremely poor. Many of the children who have pediatric keratoprosthesis end up having extrusion, infections, and often lose all their vision and their eyes.
Patient selection
A detailed history, ophthalmic examination, and other investigations become imperative to decide between surgery and other treatment options. Onset and duration of the opacities, and antenatal and perinatal history become imperative as suboptimal visual recovery can result from amblyopia and other ocular comorbidities. An important prerequisite for the success of pediatric keratoplasty is the ability of the family to follow rigorous and prolonged postoperative care. Pediatric keratoplasty should be avoided if any factors preclude the same. As the child will usually require examination and surgery under general anesthesia, systemic fitness is of paramount importance.
A comprehensive ocular examination should be followed. Visual potential should be assessed. The presence of comorbidities might impact the visual outcome negatively. Alternative treatment options should always be considered. In cases of large corneal scars or opacities, sectoral iridectomy can be a viable option. Irregular astigmatism can be corrected using rigid gas-permeable contact lenses. A localized opacity can be managed using ipsilateral rotational autokeratoplasty.108,109 A partial thickness scar can often be intervened with lamellar keratoplasty where the risk of rejection is relatively less, and rehabilitation is faster.
Timing of surgery
The optimal timing for surgical intervention depends on the onset of opacities. For congenital opacities, to curtail the development of amblyopia, the transplant can be done between the second and third months of life. 110 In bilateral congenital opacities, the second eye should be intervened within 2 weeks of the first surgery. 111 This minimizes the risk of amblyopia and the number of examinations under general anesthesia. For patients with acquired opacities and who are still within the amblyogenic age range, it is advisable to perform the surgery within 3 months of onset. When the timing of presentation is outside the amblyogenic age range, in unilateral opacities surgery can be delayed until the patient’s maturity is achieved.
The relationship between age and rejection has been demonstrated by two studies. Lowe et al. concluded that graft survival becomes better if the transplant is done at an older recipient age. 7 However, more recently, Karadag et al. found that age had no bearing on the risk of graft failure in patients aged 12 years and younger. 112
Donor tissue
The average endothelial cell density of an adult donor graft is 2000 cells/mm2 or above, whereas in graft tissue obtained from younger donors, the endothelial cell density is higher. 113 In a study done by Huang et al., the average endothelial cell density in pediatric donor tissue was 1.5–2 times higher when compared with the minimal adult endothelial cell density (2000 cells/mm2). This is especially important when planning for DSAEK, where a significant endothelial loss is expected due to tissue handling. 114 However, in the United States, surgeons use a donor cornea with a minimum endothelial cell count of approximately 2600 cells/mm2 or more. However, using a pediatric donor cornea for PKP can be disadvantageous due to the physical and refractive properties of the tissue.115,116 The tissue is difficult to handle during PKP due to its flaccidity, elasticity, and thinness. Also, the extremely steep curvature of pediatric donor tissue can result in a high myopic anisometropia, which is difficult to correct. 116 This makes amblyopia management difficult. However, this can be advantageous in unilaterally aphakic patients as it decreases the hypermetropic error. 115 Pediatric donor tissue also has a higher risk of rejection in PKP due to a higher antigen expression. 117
For pediatric PKP, Lekhanont et al. have recommended using donor tissue from young donors aged between 4 and 30 years. 118 Zhu et al. showed that there is a predilection for requesting younger donor tissue among surgeons. 8 We recommend a donor age of more than 5 years for PKP. Pediatric donor tissue is more suitable for DSEK. Although the pediatric donor tissue is thin and flaccid, the host cornea tends to structurally support it, resulting in acceptable mechanical stability. 114 Also, with the DSEK antigen load transplanted is relatively less.
Size of graft
Graft size is tailor made for every case concurring to the diameter of the host cornea. A 7.5 mm diameter graft is optimal for a normal-sized cornea of 10.5 mm diameter.18,19 A smaller graft size may be required in case of micro-ophthalmia/microcornea. Ideally for infants, a 6 mm host trephine with a graft trephine that is 0.5 mm larger is appropriate; while for older children, a 7 mm host trephine with a graft trephine 0.5 mm larger is preferable. Few studies have shown that when the graft size is smaller than 8.0 mm, the long-term graft survival is higher. 27 Oversizing the graft by 0.5–1 mm has its advantages and disadvantages. It allows for an easier closure and provides a deeper anterior chamber with a better morphological result, decreasing the incidence of postoperative glaucoma.38,52 However, with a larger button size, the rejection rate might increase as more antigenic material is in the proximity of the limbus. Also, an unwanted myopic shift might be induced which might induce amblyopia if not corrected. 119
Surgical procedure
Challenges in pediatric keratoplasty
Smaller anatomical configuration, reduced rigidity and augmented elasticity of cornea and sclera, thin and pliable cornea, severe posterior pressure with forward movement of lens-iris diaphragm, and impending lens expulsion are some factors that make pediatric PKP more challenging as compared to adults. The shortest possible time to complete the surgical procedure is advisable. Because of the thin pediatric sclera, utmost care should be taken to prevent global perforation.
Globe preparation
As pediatric sclera is less rigid than adult sclera, Flieringa ring of diameter 2–3 mm greater than corneal diameter should be applied in every case to stabilize the iris-lens diaphragm. 120 In addition, it provides scleral support, thus preventing scleral prolapse post-trephination of host tissue.
Corneal trephination
Trephination of donor tissue is analogous to that of adults. However, the elastic nature of pediatric corneal tissue makes corneal trephination a challenging procedure. 121
Graft suture
The donor cornea is then sutured using 16 interrupted 10-0 nylon sutures, with all the knots buried. A running suture is contraindicated in pediatric patients since such sutures loosen more quickly. Moreover, interrupted single sutures aid in earlier suture removal, thus avoiding suture-related issues. 122
Managing positive vitreous pressure
One of the major concerns in pediatric keratoplasty is extremely high positive pressure encountered intraoperatively. Lateral canthotomy can be considered in cases with smaller palpebral apertures. 118 Minimum external pressure should be applied due to speculum. One or two preplaced mattress sutures help secure the graft. The use of cohesive viscoelastic like healon GV or healon 5 is preferred to push the lens-iris diaphragm posteriorly. In extreme positive pressure, 8-0 silk or monofilament may be used to place cardinal suture.
Surgical modifications
A modified technique has been advocated for host cornea removal that reduces the risk of lens extrusion or expulsive hemorrhage.123,124 In this technique, host tissue is cut with corneal scissors in the same manner as with an adult transplant. However, the cutting of each quadrant is followed by suture placement in the host cornea approximately 45° from the cardinal positions. After the complete separation of the host cornea from the host bed, it is held in place with four 10-0 nylon sutures, covered with a cohesive viscoelastic, and the donor tissue is placed on top of the viscoelastic. The donor tissue is secured to the host bed using three cardinal sutures. After cutting the host corneal sutures, gentle removal of the host cornea is done from under the donor tissue through the area where the last cardinal suture will be placed. A layer of viscoelastic is maintained between the host and donor corneas throughout the procedure.
In another modified technique called the ‘sandwich technique’, the recipient corneal button is not completely excised and remains attached at a 3.0’ clock position. 125 Suturing is started after putting the donor button in the recipient opening. The host corneal button is excised after securing the donor corneal button with four cardinal sutures and suturing is continued. In cases of extremely high positive pressure, the intact recipient cornea is put back on the recipient opening. A few (4–8) cardinal sutures are applied and posterior pressure is reduced with intravenous mannitol. Once intra-operative pressure normalizes, the recipient corneal button is removed, and the donor button is sutured. Sutures can be anchored into the sclera when dealing with a thin host cornea.
Combined procedures
Combined surgery is performed in pediatric keratoplasty patients with concomitant ocular disorders. When deciding on combined surgery, the concurrent procedure is done before suturing the graft, thus minimizing trauma to the graft. 126 Pupilloplasty, lensectomy, and anterior vitrectomy should be done whenever necessary. An exception to the rule is the implantation of a glaucoma-filtering device, which is done after completion of keratoplasty. However, the concurrent procedure has poor primary graft survival. Pars plana vitrectomy before trephination has been advocated to prevent posterior pressure for patients who are at higher risk for developing extreme positive posterior pressure during PKP. In cases of refractory glaucoma requiring PKP, sclerotherapy is performed in two or three quadrants with two–four spots in each quadrant.
Postoperative care
Corneal transplantation is only the first step and has a long road ahead that requires diligent effort in the form of regular follow-up evaluations under anesthesia during the initial years. The young child is unable to explain any associated symptoms which adds to the complexity, congruously. The postoperative examination is started within 24–48 h after surgery and continued based on the age and cooperation of the patient. The cooperative and older patients are examined weekly in the clinic for all suture removals, whereas younger children are examined weekly during the first month and monthly for a year under anesthesia.
Elements
substantial to record during care by the physician and to educate the family about
Early IOP measurement is vital as there is a high incidence of post-op glaucoma, especially in patients with Peter’s anomaly. 45
Optic disc examination and charting of axial length over time. 118
The biphasic care approach focuses on maintaining a clear graft and reversing amblyopia through long-term therapy. 20
Teaching family about penlight examination to detect any early postoperative complications. 118
Frequent correction of refractive errors. 20
Postoperative medication
Preventing graft rejection
The pediatric population, as compared to their adult counterpart, has a higher rate of graft rejection due to the robust postoperative inflammatory reaction,49,127–129 particularly during the first postoperative year.57,88 Accordingly, topical corticosteroids are used aggressively in the form of 1% prednisolone acetate eye drops every 1–2 h and then gradually tapered. Ointment tends to interfere with vision but is nonetheless provided for use at night as drops are often cried out. 118 Majority of the surgeons (56.3%) start tapering within the first month, 122 and others shift to less potent forms, such as fluorometholone, in 3–6 months. 20 Some surgeons (4.2%) 122 prefer 2% topical cyclosporin A (CsA) 121 and systemic CsA for repeat rejections or high-risk patients. 38
Preventing graft infection
Surgeons use topical antibiotics aggressively until corneal epithelization is complete.100,118 Topical eye drops of quinolones and polymyxin B–trimethoprim have been utilized safely in children.130,131
Preventing secondary glaucoma
Apart from its use in high-risk patients, topical CsA can be used as a steroid-sparing agent in cases with steroid-induced ocular hypertension. 132
Cycloplegics
As per previous studies, approximately 4.2% of surgeons use cycloplegic drugs to control inflammation and decrease ciliary spasms. 22 Atropine 0.25–0.5% is prescribed every other day for 1 or 2 weeks. Atropine should be discontinued once the inflammation settles considering the possible worsening of amblyopia. 133 Patients with Down syndrome and CNS disease are prescribed atropine with vigilance and should not be used in premature and sick infants. 134
Wound integrity and suture removal
Wound healing is considerably faster in infants and children than in adults; accordingly, removal of sutures can be commenced safely in 2–6 weeks. 126 The robust healing response comes with its set of problems as healing leads to contraction of the wound area and loosening of sutures, which may result in suture erosions. 118 Loose sutures irritate the eye, which can provoke rubbing, leading to an increased chance of infectious keratitis, microabscesses, and stimulation of vascularization, posing a danger of graft rejection and failure. 118 The complications occur insidiously as young children often cannot communicate their discomfort and any visual changes they notice. 20 Therefore, periodic examinations are recommended to inspect the same. 118
The timeline for suture removal differs with different centers. 20 Some surgeons remove all sutures in patients under 5 years of age within 1–3 months of surgery due to rapid healing response, whereas in children above the age of 5, some sutures are removed selectively after 6 months to lessen astigmatism as in adults. 118 On the other hand, some surgeons remove sutures in children under the age of 8 within 3 months and children older than 8 within 6 months. 20
Amblyopia therapy
Amblyopia therapy is the only independent prognostic factor for visual improvement after pediatric keratoplasty. 49 Dana et al. reported that visual improvement was noticed in a subset of children with ocular trauma after optical correction or amblyopia therapy though they had preoperative amblyopia. 134 There is unanimity on providing amblyopia therapy for visual rehabilitation, but the timing of care varies among surgeons. 109 Most surgeons (67.2%) commence after the postoperative period, some (26.6%) may commence before surgery, and very few (6.3%) defer to their comanaging pediatric ophthalmologist, and no one delays more than 6 months. 118
Postoperative complications
Graft rejection
Allograft rejection is considerably more common in children than adults due to a more robust immune system. 57 There is often a delay in diagnosing and treating because the pediatric population cannot communicate the early symptoms of graft rejection, such as reduced visual acuity and ocular discomfort. Well-established graft rejection is usually irreversible in children. 5 If graft failure happens during the amblyogenic age range, then regrafting is vital to advance the patient’s visual development. 133
Comer et al., in their case series, observed that 53% of rejection episodes were irreversible and resulted in graft failure, 135 and Vajpayee et al. also observed that in children who reported late, 22.5% of them had graft rejection with 55% of them being irreversible. 51 At the same time, heightened inflammatory response in an infant can push rejection to ensue rapidly and be less responsive to additional treatment. 20
In pediatric keratoplasty, graft rejection rates fluctuate between 22% and 43.4%.51,136 Outcomes of many studies indicated that graft rejection accounts for most cases of graft failures. However, it is essential to note that rejection is not the sole indicator of graft failure, as most rejection episodes are treated successfully. 137 McClellan et al. were triumphant in maintaining clear grafts with five out of six rejections. 58
CsA 2%, a potent immunomodulator drug, can be used four times a day with systemic steroids and then tapered to once a day over the next 3 months. 20 It specifically affects the early stages of antigen sensitization and subsequent proliferation of T cells and does not affect the antimicrobial arena. By contrast, steroids suppress ocular immunity overall and predispose to graft rejection. 20 Other complications seen with extended use of steroids such as glaucoma, delayed wound healing, and cataract formation are also avoided. 20 CsA is extremely valuable in avoiding suture-related complications in the pediatric population, as sutures can be removed early without delaying wound healing. However, in many parts of the world, it is not used due to unavailability.
Graft infection
A child undergoing primary PK is at risk of developing bacterial keratitis, a severe complication resulting in graft failure and poor visual outcomes. In a developing country, up to 50% of cases have infectious keratitis (bacterial, fungal, acanthamoeba) that results in graft failure. The reported incidence varies from 10% to 50% in pediatric grafts. 138 Lower socioeconomic status and remote access to health care resulting in non-compliance to follow-up and compromise recognition of early symptoms. 60 Therefore, regular postoperative examinations should be targeted. Eyes with congenital corneal opacity or congenital glaucoma have a higher prevalence when compared to acquired causes. 20
Endophthalmitis
The reported rate of endophthalmitis after pediatric keratoplasty is 2%, with a higher risk in glaucomatous eyes undergoing multiple procedures. 139
Glaucoma
Glaucoma is a frequent complication either due to anterior segment dysgenesis or postoperative steroid application (5–9%). 118 Around half of the eyes of patients with Peter’s anomaly either have glaucoma preoperatively or develop postoperatively. 57 Raised IOP damages the optic nerve and endangers the survival of the graft, which can impact the visual prognosis of the child. 117
Persistent epithelial defects 125 and cataracts (2–7%)25,139 are the other postoperative complications that can compromise the visual prognosis. Amblyopia is one of the major factors limiting the visual prognosis in children (Table 3).
Enlist the complications of pediatric keratoplasty.

IOL, intraocular lens; IOP, intraocular pressure.
Cataract
The reasons for cataract formation after pediatric keratoplasty can be multifactorial. It can be secondary to the surgical trauma itself, the use of intraocular or topical corticosteroids, which are often required postoperatively to prevent graft rejection but can accelerate cataract formation, any associated anterior segment inflammation, which might stimulate lens changes or preexisting conditions or the primary disease that led to the need for keratoplasty in the first place (Figure 2). 140

Digital slit lamp images of the patient’s (a) right eye depicting a failed white opaque graft post-penetrating keratoplasty; (b) left eye depicting graft infiltrate with anterior chamber exudates; (c) left eye depicting anterior chamber hypopyon and suture infiltrate; (d) left eye depicting a patch graft post-corneal perforation in case with dermoid excision; (e) right eye depicting a clear penetrating keratoplasty graft with cataractous lens; and (f) right eye depicting extra-temporal sutures at 9’o clock post-wound leak.
Prognosis
The graft survival rates are less promising in children when compared with adults, and usually, graft failures occur within the first year of transplantation. 60 The success rate of grafts varies among studies and correlates with transplantation indications. Phakic eyes, acquired corneal scar, and late corneal decompensation in older children are associated with relatively good prognoses. On the contrary, patients with active infection or inflammation, multiple ocular anomalies, or those undergoing combined procedures have fewer promising results.2,53 Recently, improved survival rates are due to advances in cornea microsurgery and postoperative care. The mean graft survival time was 45.2 ± 5.8 months in a series of 35 children aged 2 months to 12 years. 111 Despite its technical challenges, pediatric keratoplasty has less endothelial cell loss over time than adults. 139
Limitations
In this comprehensive review of pediatric keratoplasty, certain limitations must be acknowledged. First, the review predominantly synthesizes evidence from studies with variable methodological rigor, which may lead to a selection bias and affect the generalizability of the findings. The majority of data derive from tertiary care centers, which might not reflect the broader surgical outcomes seen in different healthcare settings, particularly in developing countries. Furthermore, there is a lack of studies addressing long-term follow-up, which is crucial in the pediatric population due to the developmental nature of their visual system and the potential for late-onset complications. There is also a lack of uniformity in the reported outcome measures across studies, making it challenging to synthesize a conclusive assessment of the efficacy and safety of the procedure. In addition, given the rapid evolution of surgical techniques and postoperative care, some of the included studies may not represent the most current practices. Lastly, the review is limited by language bias, including only articles published in English, which may exclude relevant findings published in other languages and thus limit the comprehensiveness of the analysis.
Recent advances in pediatric keratoplasty
With the advancement in knowledge, availability of newer instruments, continued research, and innovation, the trend in pediatric corneal transplantation has shifted toward lamellar keratoplasties. Recently, intraoperative OCT has been used to guide lamellar dissection in DALK which has reduced the perforation and improved success rates. The new technique of femtosecond-assisted ALK (FALK) in conjunction with OCT-guided dissection has helped in excising the pathological tissue at a correct depth. Femtosecond-assisted dissection of donor lenticule has helped in achieving a smoother interface and better surgical outcomes. The major advantage of FALK is rapid visual recovery and it is a sutureless technique. DSEK and DSAEK are technically challenging pediatric keratoplasty techniques due to difficulty in scoring the Descemet membrane in children but are increasingly being employed due to smaller size wounds, reduced risk of suture-related problems, close globe surgery with early visual rehabilitation than PKP. Ashar et al. described non-Descemet stripping endothelial keratoplasty and compared it with DSEK and found almost similar outcomes with both techniques. 84 Asif et al. described the OCT-guided DSEK in CHED patients and found improved outcomes with this technique. 141 Soh and Mehta described selective endotheliectomy in Peter’s anomaly in a 21-month-old child and found improved outcomes following the transplant. 142 Table 4 depicts the review of the literature of all the major studies of pediatric keratoplasty.
A review of the literature of all the major studies of pediatric keratoplasty.

Conclusion
Pediatric corneal transplantation is a critical procedure that addresses various corneal pathologies in children, offering visual rehabilitation and ocular surface stabilization. Unlike in adults, pediatric grafts pose unique challenges, including amblyopia, higher graft rejection rates, and the intricacies of managing an immature immune system. Early intervention and meticulous postoperative management are paramount for graft survival. Successful outcomes often hinge on a multidisciplinary approach, integrating pediatric ophthalmology, corneal specialty care, and often pediatric rheumatology for systemic associated conditions. Innovative techniques, enhanced surgical instrumentation, and advanced understanding of immunosuppression have improved graft success rates over time. Still, postoperative challenges such as graft clarity, visual acuity, and refractive outcomes necessitate rigorous follow-up. Clinicians need to weigh the potential benefits against risks, tailoring the decision-making process to individual cases. Pediatric corneal transplantation remains an evolving field, necessitating continuous research and collaboration to refine techniques and optimize patient outcomes.
