Abstract
Background:
Triamcinolone acetonide is a synthetic corticosteroid with multiple ocular uses. Like other corticosteroids, it too is associated with ocular side effects such as increased intraocular pressure (IOP), glaucoma and cataracts. Suprachoroidal administration of triamcinolone is hoped to reduce the ocular side effects of the drug, especially rises in IOP. Our systematic review and meta-analysis aims to study this phenomenon.
Objectives:
The purpose of this study was to evaluate the effect of suprachoroidal triamcinolone injections on IOP.
Design:
Systematic review and meta-analysis
Methods:
We utilized the Medline, Scopus and Cochrane databases for this review. Studies published till June 2023, which fulfilled the eligibility criteria, were included. Studies in the English language with adult participants who were administered suprachoroidal triamcinolone without any concurrent intervention were included, whereas studies that did not report the mean and standard deviation for IOP were excluded. The outcome of interest was IOP at 1, 3 and 6 months post-suprachoroidal triamcinolone injection. IOP values from included studies were extracted onto Review Manager version 5.4 for analysis.
Results:
Our search yielded 104 results, from which 22 papers were shortlisted for full-text screening. Finally, 12 studies were included in the analysis. Our analysis suggests a statistically significant increase in IOP in the first month after suprachoroidal triamcinolone injection but no significant differences at 3 and 6 months post-injection as compared to baseline.
Conclusion:
Elevated IOP at 1 month post-injection must be considered when using suprachoroidal triamcinolone and adequate safety measures must be taken. However, the absence of a significant rise at 3 and 6 months post-injection indicates that this modality is safer than other existing drug delivery methods.
Trial registration:
Registered with the Research Registry with the unique identifying number ‘reviewregistry1656.’ https://www.researchregistry.com/browse-the-registry#registryofsystematicreviewsmeta-analyses/registryofsystematicreviewsmeta-analysesdetails/648eefe65b6523002995eb21/
Introduction
Triamcinolone acetonide is a synthetic intermediate-acting corticosteroid, which has a role in the treatment of various ocular and non-ocular pathologies for its anti-inflammatory effects. It achieves this function via lipocortin production, immunosuppression and vascular endothelial growth factor (VEGF) downregulation. 1 Triamcinolone acetonide use is indicated for multiple ocular pathologies including thyroid eye disease, complicated chalazia, iridocyclitis, scleritis, macular oedema and allergic eye diseases. However, ocular and systemic use of corticosteroids has traditionally been associated with rises in intraocular pressure (IOP) as well as increased risk of glaucoma and cataracts. 2
The first reported use of corticosteroids for ophthalmic purposes was in the 1950s by Gordon and McLean. 3 Since then, there have been innovations in the variety of corticosteroids available, as well as their methods of administration. Topical application is generally preferred for anterior segment pathologies, but it has limited utility in posterior segment diseases due to ineffective penetrance and low bioavailability. The use of liposome preparations is a novel innovation that has been designed to counter this limitation but its effectiveness in humans for delivering triamcinolone acetonide is yet to be proven. 4 Periocular injections into the retro-bulbar, sub-tenon, sub-conjunctival and peri-bulbar spaces can also be considered. However, these techniques have been associated with unique complications such as orbital rim fat atrophy and granuloma formation. 5 Intravitreal use of triamcinolone rose to prominence in the 2000s due to its increased drug potency. However, it is also associated with many of the adverse effects linked with corticosteroids due to diffusion into the anterior segment. 6 Suprachoroidal injections of triamcinolone acetate are currently gaining traction after promising results in large clinical trials, due to less observed drug bioavailability in the anterior segment and significantly greater spread to the retina and choroid.7–9 This technique involves injecting into the suprachoroidal space, which is a potential space between the choroid and sclera. 10 Due to lower penetrance into the anterior chamber, it was hoped that this technique would reduce the adverse effects commonly associated with ocular corticosteroid use. Suprachoroidal triamcinolone acetonide received Food and Drug Administration (FDA) approval in 2021 after the PEACHTREE trial and is currently the only FDA-approved suprachoroidal-administered corticosteroid for use in uveitic macular oedema. 11 In all, 160 participants were involved in the PEACHTREE trial, which highlighted a statistically significant increase in best-corrected visual acuity and resolution of macular oedema. 7 Other prominent trials conducted to assess the efficacy and safety of suprachoroidal triamcinolone acetonide for the treatment of uveitic macular oedema include AZALEA, MAGNOLIA and DOGWOOD.9,12,13 Trials to observe the efficacy of suprachoroidal triamcinolone for treating diabetic macular oedema have also been conducted such as the HULK and TYBEE trials.14,15
This is a first-of-its-kind systematic review and meta-analysis to evaluate the impact of suprachoroidal triamcinolone acetonide injections on the IOP. A meta-analysis has been conducted regarding the effect of intravitreal triamcinolone acetonide injections on IOP but no such literature exists for suprachoroidal delivery to better inform clinical practices. 16 Elevations in IOP can lead to multiple complications, so it is essential to evaluate the effect of suprachoroidal triamcinolone acetonide injections on the IOP to determine its safety. Elevated IOP is an established risk factor for glaucoma, which remains the most common cause of irreversible blindness worldwide. Raised IOP is postulated to induce mechanical degeneration of retinal ganglion axons passing through the lamina cribrosa. 17
Methodology
This systematic review and meta-analysis was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 18
Study selection
The literature search was conducted by two independent reviewers (ASG and AHK), who screened all published articles till June 2023 using the Medline, Scopus and Cochrane databases. The key terms utilized were ‘suprachoroidal’, ‘triamcinolone’, ‘IOP’ and ‘intraocular pressure’, and the Boolean operators ‘AND’ and ‘OR’ were utilized. The population of interest was adult patients who had been administered suprachoroidal triamcinolone. Both randomized and non-randomized interventional studies were included for comprehensiveness. Screening of articles was done by ASG and AHK based on the title and abstract using the software ‘Rayyan’. Both reviewers were initially blinded for screening, and this blind was removed once each reviewer had completed their screening. Any disagreements were resolved upon discussion. One reviewer (ASG) was responsible for full-text screening and data extraction onto an Excel spreadsheet. The data were rechecked by a second reviewer (AHK) to ensure accuracy and avoid errors. Furthermore, to assess the quality of the papers included in this study, two reviewers (ASG and AHK) utilized the NHLBI study quality assessment tools to evaluate the quality of the studies that were included. The Plot Digitizer software was utilized to extract IOP mean and standard deviation values when this information was not provided in the text but presented in graphs.
Eligibility criteria
The inclusion criteria were as follows:
(1) Studies containing patient data where suprachoroidal triamcinolone was utilized for any indication.
(2) Patients aged 18 years and above only.
(3) Articles in the English language only.
The exclusion criteria were as follows:
(1) Case reports, case series with a sample of ⩽10 eyes of patients, reviews, abstracts, conference presentations, editorials and expert opinions.
(2) Studies where interventions other than suprachoroidal triamcinolone were used concurrently, such as intravitreal anti-VEGF injections.
(3) Studies where the mean and standard deviation of IOP were not reported.
(4) Studies not conducted on humans.
Outcomes
The primary outcome assessed as part of this systematic review and meta-analysis was the IOP at 1, 3 and 6 months after administration of 4 mg/0.1 ml suprachoroidal triamcinolone. The unit used for measuring IOP was mmHg.
Analysis
Meta-analysis was performed by ASG using Review Manager version 5.4. IOP was considered continuous data. Mean difference was the effect measure used with a confidence interval (CI) of 95%, and a p-value <0.05 was considered significant. A random-effects model was used for this analysis. The IOP mean and standard deviation at study intervals of 1, 3 and 6 months post-suprachoroidal triamcinolone injection were compared to the IOP mean and standard deviation at baseline, to determine the impact of suprachoroidal triamcinolone injections on IOP. Heterogeneity was evaluated using the I2 value, with <30% representing low heterogeneity, 30–60% denoting moderate heterogeneity and >60% indicating high heterogeneity. Sensitivity analysis was performed with a leave-one-out method to determine if heterogeneity was disproportionately affected by any included study.
Results
The initial search strategy yielded 104 articles, across 3 databases. Removal of duplicates and screening based on title and abstract shortlisted 22 articles for full-text review. A total of 12 studies were included in the analysis, after a comprehensive evaluation of the shortlisted studies using the eligibility criteria. Details about the process are shown in Figure 1. The inter-reviewer agreement and reliability was assessed via Cohen’s Kappa coefficient, which was found to be 0.86. The 12 included studies all achieved high scores on the NHLBI quality assessment tool, signifying the acceptable quality of publication.

PRISMA flow diagram for the review process.
The characteristics of patients from the studies included in the analysis are given in Table 1. In all, 337 participants were included in the analysis, with 100% follow-up at 1 month. In total, 320 and 125 patients had follow-up data at 3 and 6 months, respectively.
IOP values of individual studies included in this systematic review and meta-analysis.

IOP, intraocular pressure.
The forest plots for analyses of IOP levels (mm Hg) at 1, 3 and 6 months, against baseline, are given in Figures 2 to 4, respectively. The cumulative mean IOP pre-injection was 14.3, while the mean IOPs 1, 3 and 6 months post-injection were 15.3, 15.1 and 14.9, respectively. The mean increase in IOP 1, 3 and 6 months post-injection were 1.00, 0.87 and 0.68, respectively. Our results suggest a significant increase in IOP in the first month after suprachoroidal triamcinolone injection compared to pre-injection levels, (p < 0.00001 95% CI, 0.60–1.07). However, no significant difference from baseline IOP levels is noted at 3 months (p = 0.09 95% CI, −0.03 to 0.44) and 6 months (p = 0.53 95% CI, −0.46 to 0.89).

Analysis of mean IOP at 1 month post-CLS-TA injection, 12 studies reported IOP at baseline and 1 month post-injection.
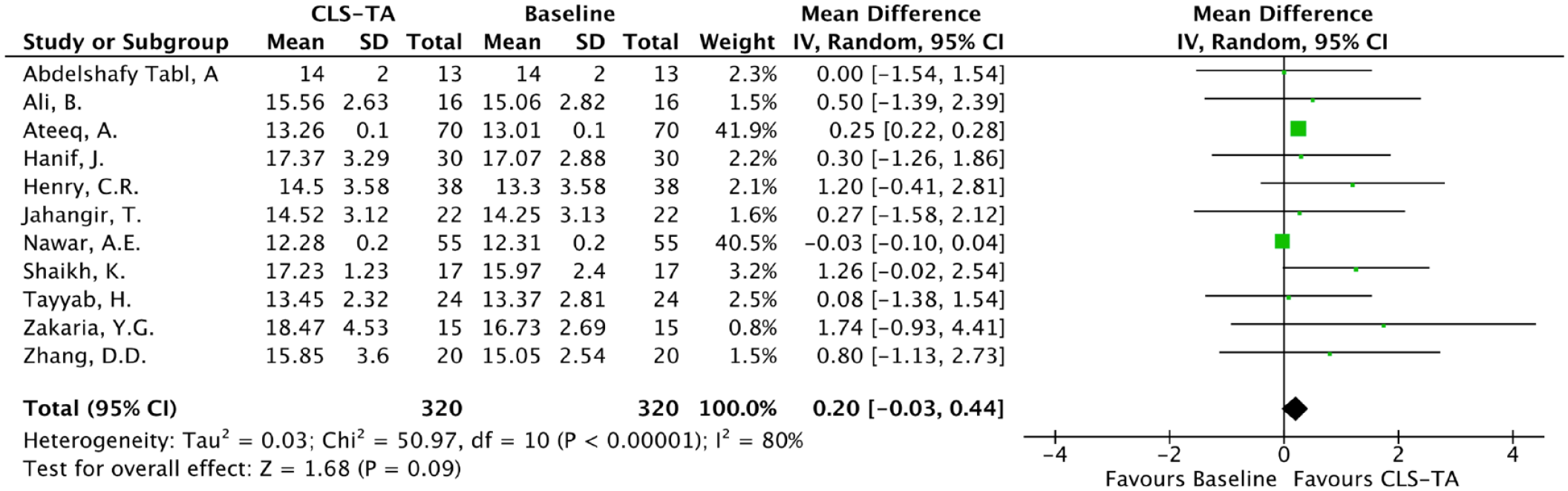
Analysis of mean IOP at 3 months post-CLS-TA injection, 11 studies reported IOP at baseline and 3 months post-injection.

Analysis of mean IOP at 6 months post-CLS-TA injection, four studies reported IOP at baseline and 6 months post-injection.
Heterogeneity was noted to be 53%, 80% and 42% for the analysis at 1, 3 and 6 months indicating moderate, high, and moderate heterogeneity, respectively. On sensitivity analysis at 1 month, removing the study by Abdelshafy Tabl et al. resulted in heterogeneity reducing to 6% (low), with a statistically significant increase in IOP still being noted (p < 0.00001 95% CI, 0.77–0.92; Figure 5). Sensitivity analysis at 3 months showed that removing the study by Ateeq et al. resulted in heterogeneity reducing to 0% (low) and no significant IOP increase was seen (p = 0.89 95% CI, −0.11 to 0.10; Figure 6). Moreover, sensitivity analysis at 6 months showed that removing the study by Henry et al. resulted in heterogeneity decreasing to 0% (low), and no significant increase in IOP was seen (p = 0.54 95% CI, −0.07 to 0.12; Figure 7).
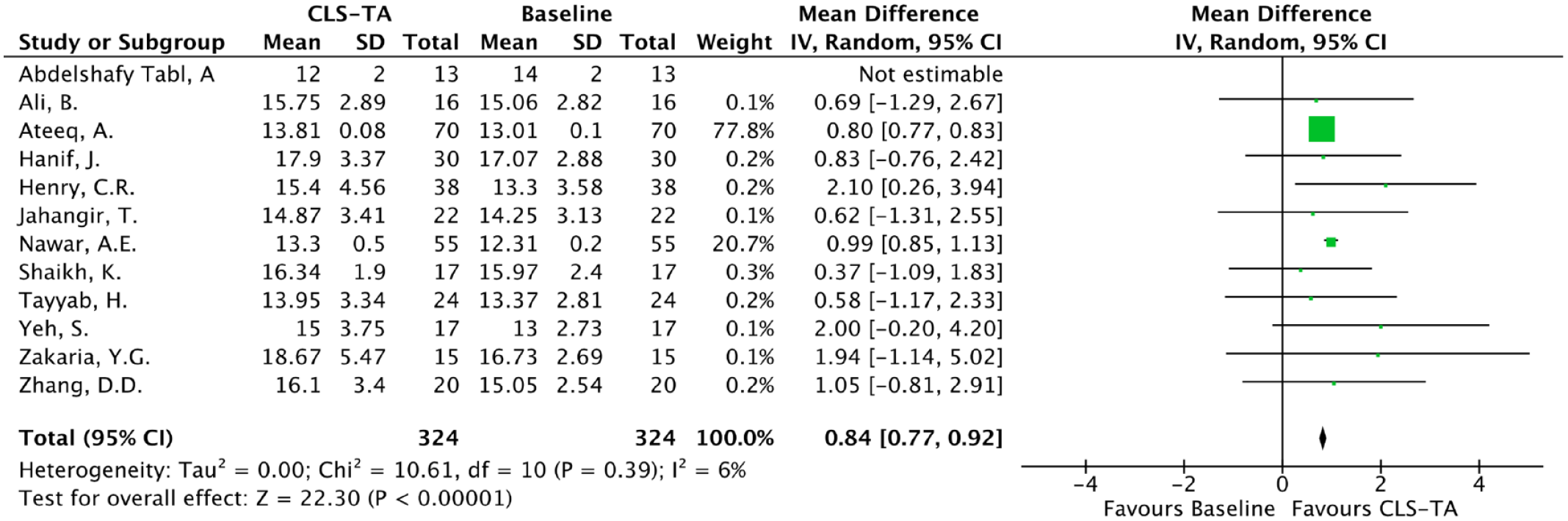
Sensitivity analysis of mean IOP at 1 month post-CLS-TA injection.

Sensitivity analysis of mean IOP at 3 months post-CLS-TA injection.
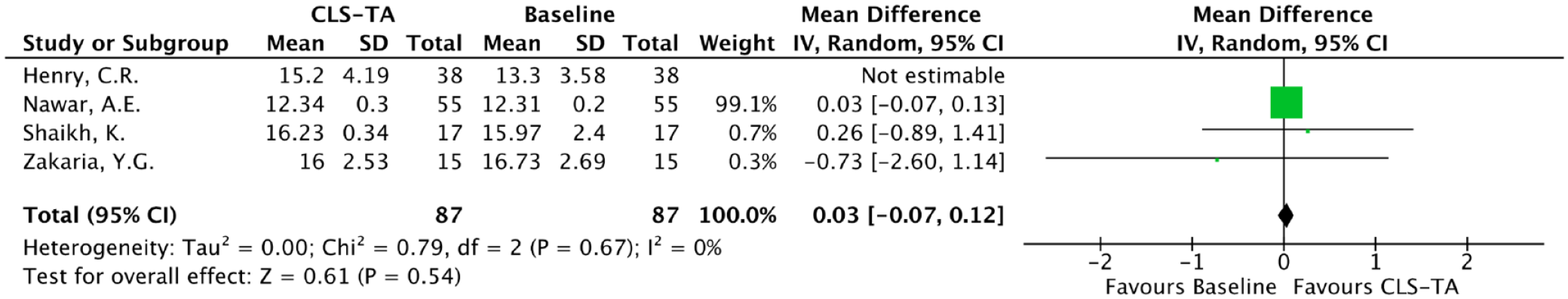
Sensitivity analysis of mean IOP at 6 months post-CLS-TA injection.
Discussion
The purpose of this meta-analysis was to determine the effect of suprachoroidal triamcinolone injections on the IOP at 1, 3 and 6 months to evaluate its safety. We discovered a significant rise in IOP at 1 month but no significant rise in IOP at 3 and 6 months as compared to baseline. This indicates that suprachoroidal triamcinolone does not have long-term detrimental effects on IOP but there is a short-term IOP rise. There was low heterogeneity at 1, 3 and 6 months after sensitivity analysis, which is indicative of minimal variability in the data included in our study and the reliability of our results.
Our findings were consistent with the study conducted by Nawar, who also observed a significant rise in IOP with the use of suprachoroidal triamcinolone after 1 month but no significant rise at 3, 6, 9 and 12 months after injection. 8 Most other studies included in this meta-analysis all noted a rise in IOP 1 and 3 months after injecting suprachoroidal triamcinolone but interestingly, this rise was not statistically significant and was associated with a p-value >0.05.9–10,22–26 The exceptions to this were the studies by Abdelshafy Tabl et al. and Ateeq et al. Abdelshafy Tabl et al.’s study was the only study conducted solely in patients with refractory diabetic macular oedema due to epiretinal membrane, and they found a decrease in IOP 1 month after injection. 19 Ateeq et al. on the other hand noted a statistically significant rise in IOP at 1 month as well as 3 months post-injection. 20 However, there was consensus regarding there being no statistically significant rise in IOP at 6 months post-injection. A 2016 meta-analysis conducted to determine the association between intravitreal triamcinolone and IOP rises noted that there was a significant rise in IOP 1 month and 3 months post-injection. 12 The incidence of IOP rises greater than 10 mmHg and cataracts have been reported to be as high as 22% and 81%, respectively, after the administration of intravitreal triamcinolone.27,28 Steroid-induced glaucoma has also been reported after the use of intravitreal triamcinolone. 29 This would indicate that the suprachoroidal route is safer than the intravitreal route for triamcinolone administration with regard to the long-term effects on IOP.
An IOP range from 10 to 21 mmHg has long been accepted as a standard due to a low likelihood of this pressure range causing glaucomatous damage in a majority of patients. 30 Therefore, it is important to evaluate the probability of suprachoroidal triamcinolone elevating the IOP above 20 or causing large rises in IOP. The studies included in this meta-analysis have reported 13 patients with notable IOP rises, of which 12 resolved upon the use of topical IOP-lowering medications and 1 resolved spontaneously. Henry et al. reported that 6 patients had an IOP rise >10 mmHg and 2 out of 38 patients (5%) had an IOP >30 mmHg. Four of these patients were treated with one IOP-lowering medication and three were treated with two IOP-lowering medications. Zakaria et al. reported that two patients had an IOP rise >10 mmHg, which resolved with the use of topical anti-glaucoma drops. Tayyab et al. reported one occasion where IOP rose from 19 to 24 mmHg at 1 month but this was controlled on medications and returned to 16 mmHg at 3 months. Ali et al. reported three patients using IOP lowering medications and 1 out of 16 (6%) participants experienced increased IOP to 20 mmHg that reduced to 18 mmHg in the second month. Yeh et al. reported that one patient experienced an IOP elevation to 36 mmHg within 30 min after the injection but this returned to normal levels with no treatment. No surgical intervention was required to control the IOP elevation in any of these patients.
The effect of steroids on IOP has been well documented and many ideas have been put forward to explain this phenomenon. 5 The rise in IOP can be severe enough to cause a form of open-angle glaucoma due to a reduction in aqueous outflow. It has been postulated that corticosteroids may cause changes to the trabecular meshwork by elevating extracellular matrix protein expression, increasing the resistance to flow. It has also been theorized that steroid use results in the stabilization of lysosomal membranes and inhibition of phagocytosis of aqueous debris by endothelial cells of the trabecular meshwork, resulting in debris build-up. 31 Suprachoroidal injections of triamcinolone, however, have been associated with decreased penetrance into the anterior chamber.5,24 Studies have shown that after suprachoroidal injections, the posterior retina is exposed to 523,910 times more steroids than the aqueous. 32 A statistically significant association has also been noted between higher volume of steroid and greater IOP elevation. 32 Therefore, less volume of triamcinolone in the anterior chamber would result in a smaller rise in IOP, which is the case in suprachoroidal injections due to the non-even and majorly posterior distribution.33,34 This could explain why the IOP returns to baseline faster than other methods of corticosteroid administration. The reduced bioavailability in the anterior chamber may result in faster drug clearance, which could cause quicker normalization of IOP as compared to other delivery mechanisms that have greater anterior chamber bioavailability. There is a need for more research on this topic to explain the exact mechanism for the rise in IOP that has been observed up to 1 month post-suprachoroidal triamcinolone.
There are multiple clinical implications of this systematic review and meta-analysis. The statistically significant rise in IOP 1 month after the administration of suprachoroidal triamcinolone indicates that stringent IOP monitoring is required during the first month post-operatively to avoid any IOP-related complications. Additionally, IOP-lowering medications could be considered prophylactically to prevent glaucomatous damage or other complications associated with increased IOP for the first month after the triamcinolone injection. The absence of a significant rise in IOP at 3 and 6 months indicates a decreased need for regular follow-up appointments after 3 months to monitor IOP and highlights that suprachoroidal triamcinolone does not have a long-term detrimental impact on IOP. There is a need for larger studies as well as randomized control trials on suprachoroidal triamcinolone use to strengthen our conclusions, as well as better reporting of IOP means and standard deviations to facilitate reviews, such as this one, that aim to statistically evaluate the safety of using suprachoroidal triamcinolone. There is a need for more literature analysing the prevalence of other corticosteroid-related adverse effects after suprachoroidal triamcinolone administration.
There are a few limitations of this study which must be acknowledged. Unfortunately, some landmark trials such as PEACHTREE, MAGNOLIA, HULK and TANZANITE could not be included in this systematic review and meta-analysis due to the unavailability of mean and/or standard deviation values for IOP.7,26,27,35 However, despite this drawback, our analysis still contains 12 studies with data on 337 patients and low heterogeneity after sensitivity analysis. Eight out of 12 studies included in this meta-analysis were non-randomized, depicting a lack of randomized studies on suprachoroidal triamcinolone with available IOP mean and standard deviation values in the available literature. Additionally, due to the unavailability of raw data for each included study, we were unable to calculate the cumulative percentage of patients with IOP elevated over 25 and 30 mmHg at 1 month post-injection. This could have helped understand the incidence of significant IOP elevations and provided more knowledge regarding the safety profile of suprachoroidal triamcinolone administration. Publication bias may also be present as only published literature was considered for this study. Restricting ourselves to Medline, Scopus and Cochrane databases to ensure high-quality standards for the included studies may have excluded other potential studies in unpublished and grey literature, the inclusion of which could potentially have altered our findings.
Conclusion
Suprachoroidal triamcinolone has proven efficacy in treating macular oedema but its association with elevated IOP at 1 month must be considered and adequate safety measures must be taken. The absence of a significant rise in IOP at 3 and 6 months post-injection indicates that suprachoroidal triamcinolone is safer than other modes of ocular corticosteroid delivery, such as intravitreal injections, with respect to long-term IOP elevation. Further studies are required to determine the prevalence of other corticosteroid-associated ocular adverse effects in patients who have been administered suprachoroidal triamcinolone.
Supplemental Material
sj-docx-2-oed-10.1177_25158414241228671 – Supplemental material for Effect of suprachoroidal triamcinolone on intraocular pressure: a systematic review and meta-analysis
Supplemental material, sj-docx-2-oed-10.1177_25158414241228671 for Effect of suprachoroidal triamcinolone on intraocular pressure: a systematic review and meta-analysis by Altamash Shahriyar Ghazanfar, Aly Hamza Khowaja and Haroon Tayyab in Therapeutic Advances in Ophthalmology
Supplemental Material
sj-pdf-1-oed-10.1177_25158414241228671 – Supplemental material for Effect of suprachoroidal triamcinolone on intraocular pressure: a systematic review and meta-analysis
Supplemental material, sj-pdf-1-oed-10.1177_25158414241228671 for Effect of suprachoroidal triamcinolone on intraocular pressure: a systematic review and meta-analysis by Altamash Shahriyar Ghazanfar, Aly Hamza Khowaja and Haroon Tayyab in Therapeutic Advances in Ophthalmology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
