Abstract
SARS-CoV-2 affects the nervous system directly by neurotoxic action, by binding to angiotensin-converting enzyme-2 (ACE2) receptors or indirectly by inducing cytokine storm leading to disruption of the blood-brain barrier, immunological mediation, increasing blood coagulation and as a trigger for autoimmune-mediated demyelinating injuries in the central nervous system. In COVID-19 neuro-ophthalmological manifestations are not so common. Optic neuritis is the result of optic nerve inflammation and has varied causes. In many patients, signs of inflammation are not visible on the fundus, and it usually manifests as papillitis-anterior neuritis, retrobulbar neuritis or visible optic nerve oedema. We are reporting a case of a middle-aged adult diagnosed with myelin oligodendrocyte glycoprotein (MOG) antibody-positive optic neuritis of the right eye post-COVID-19 disease. Routine biochemical and haematological investigations, including electrolytes and hepatic and renal functions, were normal. In cerebrospinal fluid (CSF) – glucose 63.8 mg/dL, protein 39.1 mg/dL and ADA – 1 µ/L. No oligoclonal bands of immunoglobulin G (IgG) were seen on high-resolution electrophoresis. Serum Anti-MOG-antibodies were positive. A gadolinium-contrast magnetic resonance imaging (MRI) of the brain and orbits shows post-contrast enhancement in the superior aspect of the right intraconal soft tissue. The right optic nerve appears bulky and heterogeneous with peripheral post-contrast enhancement along its entire length suggestive of neuritis. A diagnosis of MOG antibody-positive optic neuritis was made, and the patient was treated with an injection of Methylprednisolone with intravenous immunoglobulin. Each day, the evaluation of the right eye showed remarkable improvement from finger counting to 6/6 vision. The patient was discharged on the 9th day of admission. We can conclude that early diagnosis was essential for improving the long-term outcome of the patient.
Introduction
With increased learning about the mechanisms of SARS-CoV-2 transmission, pathophysiology and interactions, it became clear that COVID-19 severely affects patients, and aside from respiratory symptoms, COVID-19 can lead to neurological symptoms and complications directly or indirectly. SARS-CoV-2 affects the nervous system directly by neurotoxic action, by binding to angiotensin-converting enzyme-2 (ACE2) receptors or indirectly by inducing cytokine storm leading to disruption of the blood-brain barrier, immunological mediation, increasing blood coagulation and as a trigger for autoimmune mediated demyelinating injuries in the central nervous system (CNS). 1 Optic neuritis is the result of optic nerve inflammation and has varied causes. In many patients, signs of inflammation are not visible on the fundus, and it usually manifests as papillitis-anterior neuritis, retrobulbar neuritis or visible optic nerve oedema. 2 In COVID-19 neuro-ophthalmological manifestations are not so common.
Myelin oligodendrocytes glycoproteins (MOG) are located on the outermost surface of the myelin surrounding oligodendrocytes. They are found exclusively in the CNS. When there is dysregulation and disruption of the blood-brain barrier, these antibodies against MOG enter the CNS and cause MOG antibody disease (MOG-AD), a distinct form of demyelinating disease. The most common presentation of this disease is optic neuritis. 3
Here we are reporting a case of a middle-aged adult diagnosed with MOG antibody-positive optic neuritis of the right eye post-COVID-19 disease.
Case report
A 44-year-old female presented to the emergency department with a gradual loss of vision in the right eye for the last 5–6 days, starting with grittiness and heaviness in the right eye. Two weeks before the onset of right eye pain, the patient was diagnosed positive for COVID-19 by polymerase chain reaction after developing a fever, generalized body ache and cough for which he was home quarantined. Symptomatic treatment was given to the patient and no hospitalization was required. The loss of vision was unilateral, not associated with pain, no similar complaints in the past, no family history of any neurological complaints, no history of flickering of light, loss of colour vision and no history of hypertension, diabetes or other cardiovascular diseases, or any history of trauma. Routine biochemical and haematological investigations, including electrolytes and hepatic and renal functions, were normal. In cerebrospinal fluid (CSF) – glucose 63.8 mg/dL, protein 39.1 mg/dL and ADA-1
Patient pertinent laboratory values.

C-ANCA, antineutrophil cytoplasmic antibodies; GPL, unit 1microgram of IgG; MOG, myelin oligodendrocyte glycoprotein; MPO, myeloperoxidase; MPL, unit 1microgram of IgM.
Higher mental functions: conscious, cooperative, coherent and oriented to time, place and person GCS: E4V5M6. Cranial nerves: All are normal except the second nerve (optic nerve); in the right eye there is no perception of light and in the left eye, there is 6/6 vision. Fundus evaluation was normal (no papillitis) (Figure 1).

Dilated Fundus Examination of right (a) and left (b) eyes. Vitreous: Normal OU. Disc: OD: Grade I disc oedema, no pallor, no haemorrhages, no exudates, OS: Grade II disc oedema, no pallor, no haemorrhages, no exudates. Cup-to-disc: no cup noted in either optic nerve. Macula: Normal OU. Vessels: Normal OU. Periphery: Normal OU.
MRI Axial image of the brain and orbits showed heterogeneous altered signal intensity which is (a) hypointense on both T2 and (b) T2FS in the medial aspect of the right orbit involving the intraconal compartment. There is a loss of fat planes with the right medial rectus muscle. The right optic nerve appears bulky and shows fat stranding along its entire length. Heterogeneous signal intensity was seen along a left optic nerve in the region of the optic canal (Figure 2).

MRI Axial image of the brain and orbits showed heterogeneous altered signal intensity which is (a) hypointense on both T2 and (b) T2FS in the medial aspect of right orbit involving intraconal compartment. There is loss of fat planes with the right medial rectus muscle. The right optic nerve appeared bulky and shows fat stranding along its entire length. Heterogenous signal intensity seen along left optic nerve in the region of optic canal.
A diagnosis of MOG antibody-positive optic neuritis was made, and the patient was started on injections of Methylprednisolone with IVIG. Each day, the evaluation of the right eye showed remarkable improvement from finger counting to 6/6 vision.
Discussion
Isolated inflammation of the unilateral optic nerve in young patients is rare, especially with no papillitis. Optic neuritis in young patients is usually associated with systemic autoimmune or other granulomatous diseases, rather than viral illness. In our case, COVID-19 infection acts as a triggering factor for Anti-MOG antibody-positive optic neuritis confirmed by brain MRI and CSF analysis. The patient was an RT-PCR confirmed case of COVID-19 with mild clinical disease.
MOG antibody disease (MOGAD) is a neurological, immune-mediated inflammation of the optic nerve, spinal cord and/or brain. MOG protein is located on the myelin sheath surface in the CNS. While the exact function of this MOG is not known, these glycoproteins are the target of the immune system. The clinical course of MOGAD can be monophasic or relapsing and have a milder clinical course than other demyelinating disorders which does not meet the criteria for typical multiple sclerosis or other neuroinflammatory illnesses like neuromyelitis optica spectrum disorders (NMSOD). The severity and specific symptoms of MOGAD can vary from patient to patient. Patients with MOGAD have previously been diagnosed with acute disseminated encephalomyelitis (ADEM), optic neuritis (ON), longitudinally extensive transverse myelitis and transverse myelitis, based on their pattern of inflammation in the brain, spinal cord and optic nerve. 4
The two largest clinical trials were done in France and the United Kingdom in which anti-MOG antibodies were present in 44–60% of ON patients. In adult patients with late-onset disease, both optic nerves were involved and inflamed, while young patients frequently developed unilateral ON. Most patients have sudden and severe loss of vision, with eye pain at the onset but favourable vision outcomes.5,6 Similarly, it is seen in our patients.
SARS-CoV-2 is rarely found in the CSF, suggesting that the mechanism of COVID-associated neuropathology is different from direct viral toxicity. Fleischer et al. reported neurological symptoms in 60% of their (n = 102) SARS-CoV-2 patients out of which 35% were reported positive for serum autoantibodies against diverse neuronal and glial epitopes. 7 No significant correlation between the severity of SARS-Cov-2 and the presence of anti-MOG antibodies was found.
COVID-19 infection causes a dysregulated interferon response and increases the expression of several pro-inflammatory cytokines, including IL-1B, TNF and IL-6. This host response reached a critical threshold sufficient to activate MOG-IgG1 specific B-cells, leading to an increased titre of serum MOG antibodies. CD4+ and CD8+ T cell responses associated with the SARS-CoV-2 virus provide a stimulus for bystander activation and co-stimulation of autoreactive T- and B-cells. 8
These MOG antibodies in serum do not cause nervous system disease unless they cross the blood-brain barrier. In SARS-CoV-2 kinetics of the underlying mechanism of dysregulation of the blood-brain barrier is unknown. The human-induced pluripotent stem cell (hiPSC) model shows infection and multiplication of SARS-CoV-2 in various organs and transcriptional changes. This leads to the upregulation of interferon signalling pathways in the neurovascular unit. These neurovascular units maintain the physiological function of the blood-brain barrier (BBB) and comprise the brain capillary endothelial cells, pericytes, astrocytes, neurons and microglia cells. Dysregulation of BBB results in the penetration of MOG antibodies in the CNS and leads to neuro-inflammatory disorders.
MOGAD diagnosis criteria were published in 2018 based on expert consensus. According to these criteria, confirmation of MOGAD disease includes three necessary components: (1) clinical picture, (2) neuroimaging (MRI) or neurophysiological examination (in optical neurosis, optical coherence tomography or visual-evoked potentials) findings remarks demyelinating injury in the CNS, and (3) biochemical positive result of MOG-IgG antibody test performed with the modern cell-based assay. Persistently positive antibodies lead to recurrent inflammatory attacks on the CNS. 9 Patients who are positive for the MOG antibody do not test positive for the NMO antibody called aquaporin 4 (AQP-4). AQP-4 is a water channel protein and those with NMOSD produce autoantibodies against AQP-4. Patients with MOGAD seem to be less likely to have other autoimmune disorders (such as rheumatoid arthritis, Hashimoto’s thyroiditis, etc.) than those with AQP-4 positive NMOSD. 10 Similarly, it is seen in our patient.
To the best of our knowledge, a total of 17 reports identified positive connections between SARS-CoV-2 and ON, with or without spinal or cerebral demyelinating lesions; all were positive for serum antibodies against myelin oligodendrocyte glycoprotein (MOG)4,11–26 (Table 2). Out of these 17 cases, all appeared during symptomatic COVID-19 with no correlation with the severity of SARS-CoV-2 disease and 1 case appeared a few weeks after the disease. In all cases, the pre-chiasmal optic nerve was involved. MOG antibodies were measured in all cases for the confirmation of diagnosis. In all cases, positive serum MOG antibody-IgG antibodies were the diagnostic criteria related to COVID-19. MOG antibodies tested positive in three cases between 2 and 4 weeks,19,21,22 whereas in the case reported by Zorić et al. and Colantonio et al. MOG-IgG antibodies tested positive after 4 weeks and 6 months of SARS-CoV-2 RT-PCR positive test.13,22 In the remaining cases, MOG-IgG antibodies tested positive within 1 week of the SARS-CoV-2 RT-PCR positive test. Based on a finding of this database, we can suggest that the amount of time passed between the COVID-19 positive test and subsequent MOG-IgG testing should not be dismissed solely. In more than half of the cases, analysis of cerebrospinal fluid revealed pleocytosis. In contrast, only two cases by Sawalha et al. and Pinto et al. reported high protein levels in the CSF.11,24 On further analysis of the cases, two-thirds of cases showed T2 hyperintensity and post-contrast enhancement in the pre-chiasmal optic nerves with sparing of chiasma and optic tract. While Woodhall et al. was the only case to report unilateral optic nerve lesions. 19 Treatment guidelines for MOGAD have not been established, but the possible treatment for the management of acute episodes are intravenous steroids, plasma exchange (PLEX) or IVIG. A significant improvement in vision was reported in all cases after initiation of intravenous steroids. Only two cases by Sardar et al. and Woodhall et al. were treated with plasma therapy due to the partial and slow improvement in clinical symptoms.23,19
Summary of all the published studies.
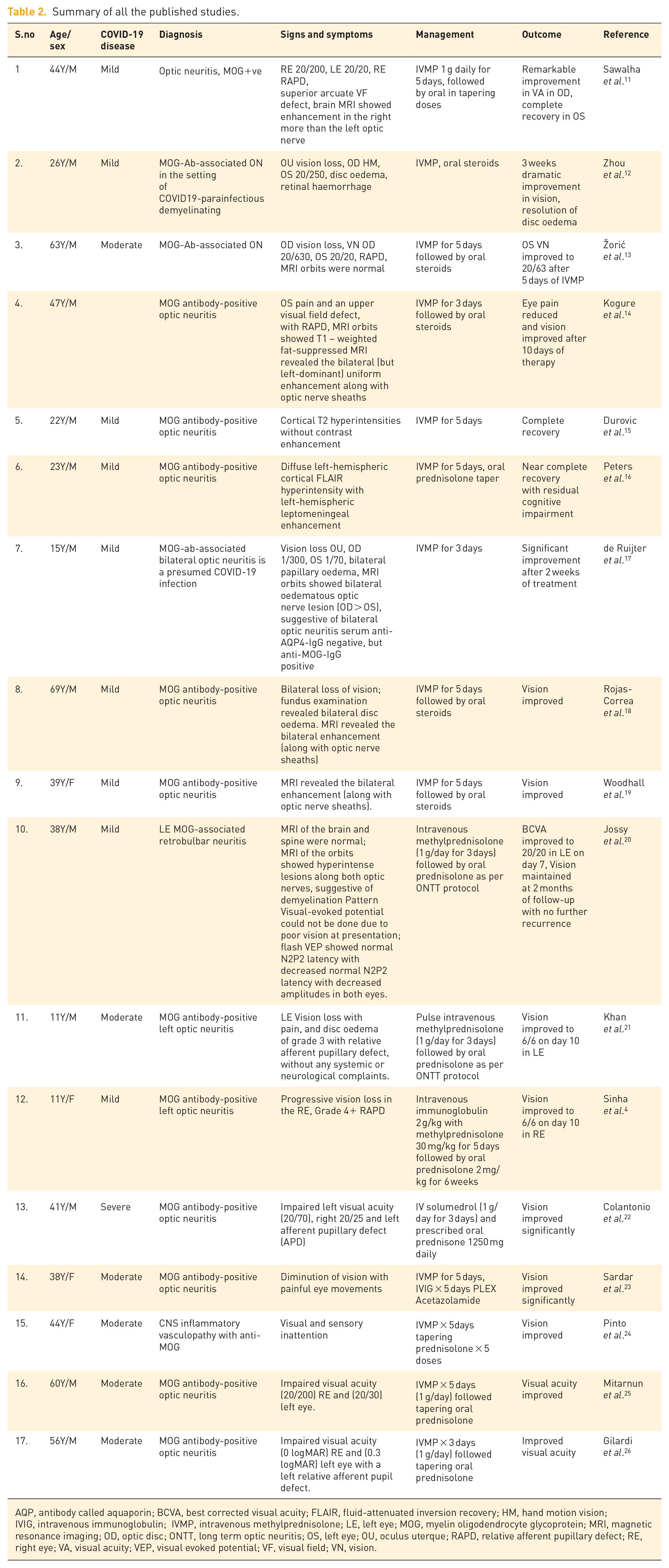
AQP, antibody called aquaporin; BCVA, best corrected visual acuity; FLAIR, fluid-attenuated inversion recovery; HM, hand motion vision; IVIG, intravenous immunoglobulin; IVMP, intravenous methylprednisolone; LE, left eye; MOG, myelin oligodendrocyte glycoprotein; MRI, magnetic resonance imaging; OD, optic disc; ONTT, long term optic neuritis; OS, left eye; OU, oculus uterque; RAPD, relative afferent pupillary defect; RE, right eye; VA, visual acuity; VEP, visual evoked potential; VF, visual field; VN, vision.
In the present case, COVID-19 infection presumably triggered an autoimmune response in the patient, resulting in the production of MOG antibodies. Though it is unclear whether the patients had a predisposition to MOG-associated disease, which relapsed during COVID-19, or whether SARS-CoV-2 set this novel process in action.
Conclusion
The present case is not able to sufficiently clarify the relationship between the two clinical conditions COVID-19 and MOGAD resulting in the form of post-infectious neuritis. It is unclear whether COVID-19 initiated this ON or was the trigger for predisposing non-recognized subclinical MOG antibodies. COVID-19 provides new challenges in both cases, shedding light on similar cases and adding more knowledge to the already present literature.
