Abstract
Purpose:
To report the 12-month results of laser (treatment G1) versus intravitreal bevacizumab combined with laser (treatment G2) in patients with diffuse diabetic macular edema (DME).
Methods:
In this single-center randomized independent controlled trial, 32 patients were randomized to G1 (n = 15) or G2 (n = 17). In G1, laser was given at baseline and then pro re nata (PRN). In G2, three intravitreal bevacizumab (1.25 mg) injections were given once every 6 weeks, then laser and then PRN. Analysis was performed by treatment as administered. This study was registered in clinicaltrials.gov as NCT01572350 and EU Clinical Trial Registry as 2009-014654-15.
Results:
G2 was superior to G1 improving best corrected visual acuity (BCVA) with respect baseline (+8.0 vs + 3.0; p < 0.01). At month 12, a significantly greater proportion of patients had a BCVA letter score >15 and >73 in G2 (3 of 15 (20%) and 8 of 15 (53%), respectively) versus G1 (1 of 17 (6%) and 4 of 18 (23%), respectively). Health-related quality of life, assessed through National Eye Institute Visual Function Questionnaire, at 12 months was statistically indistinguishable between both groups.
Conclusion:
G2 provided superior visual acuity gains over G1 in patients with visual impairment due to center-involving diffuse DME, associated with significant gains in VFQ-25 scores.
Introduction
Anti-vascular endothelial growth factor (anti-VEGF) therapies have been tested as an alternative treatment to laser for diabetic macular edema (DME), showing such good results that they have become the first choice treatment.1–8 Bevacizumab (Avastin®; Genetech/Roche, Inc., South San Francisco, CA, USA) is a humanized full-length monoclonal antibody which encompasses all VEGF isoforms and has been used as anti-VEGF therapy for DME. Although not currently approved by the US Food and Drug Administration (FDA) for intraocular use, the injection of bevacizumab into the vitreous cavity has been performed without significant intraocular toxicity.9–11 One of the main issues of intravitreal anti-VEGF therapy is the need for repeated injections. So, it has been combined with laser looking for an additive synergistic effect and the possibility of reducing the number of intravitreal injections. It had been thought that first thinning the retina with pharmacotherapy might enable better penetration of laser light to the retinal pigment epithelium and could improve the efficacy of laser.12–14 This strategy could reduce not only the number of injections but also the atrophy and the secondary effects induced by laser coagulation. However, it has to be taken into account that the outcomes of laser treatment can be different depending on the type of DME and central involvement.
The inferiority of the laser against anti-VEGF therapy has been amply demonstrated.13–17 However, only two anti-VEFG, ranibizumab and aflibercept, have been approved by the FDA and the European Medical Agency (EMA) for use in DME. This contrasts with the fact that clinical trials on DME (T protocol) 18 and real clinical practice studies, 19 showed that the use of laser and bevacizumab are part of our therapeutic arsenal in the management of the DME. So, it is only through independent clinical trials that scientific evidence that provides data supporting the use of both therapies is being generated.
The aim of this study was to determine the efficacy and safety of the off label intravitreal anti-VEFG bevacizumab combined with grid laser photocoagulation compared with laser treatment alone in patients with diffuse DME over the course of 12 months. Diffuse DME is hard to define and characterize, with “diffuse DME” definition, we tried to clearly state that we treated DME that were non-focal and were not suitable for single laser treatment.
Materials and methods
The «alone laser versus bevacizumab plus laser for diabetic macular edema study (ALBA study)» is a single-center, prospective, 12-month, phase III, randomized, independent controlled trial conducted in patients with diffuse DME. The study was conducted in accordance with the Declaration of Helsinki and International Conference on Harmonization Good Clinical Practice guidelines. Hospital Universitario de Canarias’ Ethics Committee approval was prospectively obtained with approval number APR-DEX-2014-01. This study is registered in clinicaltrials.gov as NCT01572350 and EU Clinical Trial Registry as 2009-014654-15. Written informed consent was obtained from all individual participants included in the study. The initial protocol also included the comparison with triamcinolone. However, this comparison could not be carried out due to the difficulties to obtain such a drug in our setting, which prevented the inclusion of this comparison in the final report of the study. The size of the sample was determined by those patients who meet the inclusion criteria and were attended in our setting, Hospital Universitario de Canarias (HUC), between October 2010 and March 2013. One eye of each patient was selected and treated as the study eye. If both eyes were eligible, the eye with the worse best corrected visual acuity (BCVA) assessed at the first visit was selected as the study eye. Key inclusion criteria were (1) age above 18 years; (2) either type 1 or 2 diabetes mellitus (as per the American Diabetes Association or the World Health Organization guidelines); (3) glycosylated hemoglobin (HbA1c) ⩽11%; (4) stable medication for the management of diabetes within 3 months before randomization and expected to remain stable during the study; (5) visual impairment due to mixed or diffuse DME and no other causes; (6) BCVA letter score between 25 and 75 letters, both inclusive, based on Early Treatment Diabetic Retinopathy Study (ETDRS) visual acuity testing charts at a distance of 4 m (approximate Snellen equivalent 20/32–20/160); (7) center-involved DME, defined as central subfield thickness >300 μm in spectral domain optical coherence tomography (SD-OCT) (Carl Zeiss Meditec, Inc., Dublin, CA).
Key exclusion criteria were (1) Focal DME; (2) tractional DME; (3) prior treatment with antiangiogenic drugs in the study eye; (4) prior vitrectomy in the study eye; (5) concomitant conditions in the study eye that could prevent the improvement in BCVA during the study according to the investigator’s opinion; (6) active intraocular inflammation or infection in either eye; (7) uncontrolled glaucoma in either eye (e.g. intraocular pressure >24 mmHg on medication, or from the investigator’s judgment); (8) previous panretinal laser photocoagulation (within 6 months) or focal/grid laser photocoagulation (within 3 months) before study entry; (9) patients with uncontrolled hypertension, uncontrolled diabetes (glycosylated hemoglobin (HbA1c) >11%), prior cerebrovascular accident, or prior myocardial infarction.
Stopping rules included: appearing of some of the exclusion criteria throughout the study, severe adverse ocular reaction (i.e. endophthalmitis, retinal detachment, macular hole, uncontrolled glaucoma, macular ischemia, atrophy, subfoveal fibrosis), severe systemic reaction, allergic reactions, being a non-compliant patient, voluntary exit, death, pregnancy.
Randomization was performed by the research unit of the HUC, using simple random sampling. Randomized tables of the type 1:1 were used using the SPSS software version 17.0 (IBM Corporation, New York, USA). Study participants were assigned randomly, using the opaque envelope method, to 1 of the 2 treatment arms:
G1. Laser photocoagulation using an ETDRS protocol at baseline. Additional laser re-treatments were applied as needed but no more frequently than every 4 months, following the ETDRS recommendations described in surgical techniques section.
G2. Three initial consecutive intravitreal bevacizumab (1.25 mg) injections were given once every 6 weeks followed by laser. Additional laser or bevacizumab re-treatments were applied according to protocol-defined re-treatment criteria described in surgical techniques section.
The primary outcome measurement was the BCVA, while secondary outcomes included central foveal thickness (CFT) and macular volume. Follow-up was planned for 1 year. During that year, follow-up visits occurred every 4 weeks (±1 week) in G2 group, while in G1 group, visits were fixed by standard protocol after the retinal laser treatment during a period of 6 months and every 4 weeks (±1 week) after 6 months. Safety evaluations, measurement of BCVA, eye examinations, and SD-OCT scans were performed at all follow-up visits. Fluorescein angiography was performed at baseline at 6, and 12 months. Measurements of glycosylated hemoglobin were obtained at baseline and at 3, 6, and 12 months. Hematology and blood chemistry tests were performed at baseline and at 6 and 12 months. Finally, health-related quality of life, assessed through the National Eye Institute Visual Function Questionnaire (NEI VFQ-25) was applied at 12 months in both groups.
All data were transcribed to a research log and registered in the SPSS software. In the middle of the study, a quality control of the data was performed by external staff attached to the research unit of the HUC, which allowed verifying that the transcribed data were adequate. Data analysis was performed at 3, 6, 9, and 12-month follow-up using SPSS software.
Surgical techniques
The intravitreal injection of bevacizumab was performed under sterile conditions. About 5% aqueous povidone iodine solution was used to clean and disinfect the eye to treat. About 1.25 mg (0.05 mL) of bevacizumab (Avastin, 100 mg/4 mL) was injected into the vitreous cavity using a sharp 27 or 30 gauge needle that was inserted into the eye 3.5–4.0 posterior from the limbus. Once the needle was removed, a sterile cotton applicator was used to prevent reflux. The postoperative medications include antibiotic drops (Exocin®, Allergan, France SA) for 1 week after the injection.
Laser photocoagulation was applied using an ETDRS protocol at baseline. Re-treatments were given in accordance with ETDRS guidelines at intervals no shorter than 4 months from the previous treatment. Only one surgeon performed macular photocoagulation for all eyes in both groups. The re-treatment criteria included BCVA worsened by ⩾5 letters associated with an increase of mean CFT ⩾10%.
Statistical analysis
Analysis was performed by treatment as administered. The non-parametric test of chi-square was used in case of qualitative variables, and data were expressed as percentage. Non-parametric Spearman test was used to analyze the correlation of two quantitative variables. To analyze independent groups, it used the Kruskal–Wallis test (more than two variables) or Mann–Whitney U-test (two variables). In dependent groups, the tests used were Friedmann (more than two variables) and Wilcoxon (two variables). Data were expressed as median and standard deviation, and median and 75 and 25 quartiles. A p value ⩽0.05 was considered statistically significant, and Bonferroni correction was applied when necessary. The statistical power of the study for the primary outcome, BCVA, was 56%, and was calculated based on the data available in previous studies.13,16,17 The data analysis was performed by SPSS software version 17.0.
Results
Figure 1 shows the flow diagram of the study. As can be seen, 147 patients with DME were proposed to participate in the trial. A total of 90 (61%) were excluded because they did not fit the protocol (n = 89) or they did not want to participate in the study (n = 1). Consequently, 57 patients of the 147 (39%) were enrolled to the study: 32 of 57 (56%) included to the laser alone group (G1) and 25 of 57 (44%) to bevacizumab plus laser group (G2). After inclusion, 25 patients of 57 (44%) were excluded from analysis for a variety of reasons: declined to participate (n = 10), uncontrolled (n = 5), laser breakdown (n = 2), vitreous hemorrhage (n = 1), need of photocoagulation (n = 2), epiretinal membrane (ERM; n = 1), macular age degeneration (n = 1), acute myocardial infarction (n = 1), and death (n = 2). So, 15 patients in G1 and 17 patients in G2 were finally analyzed.
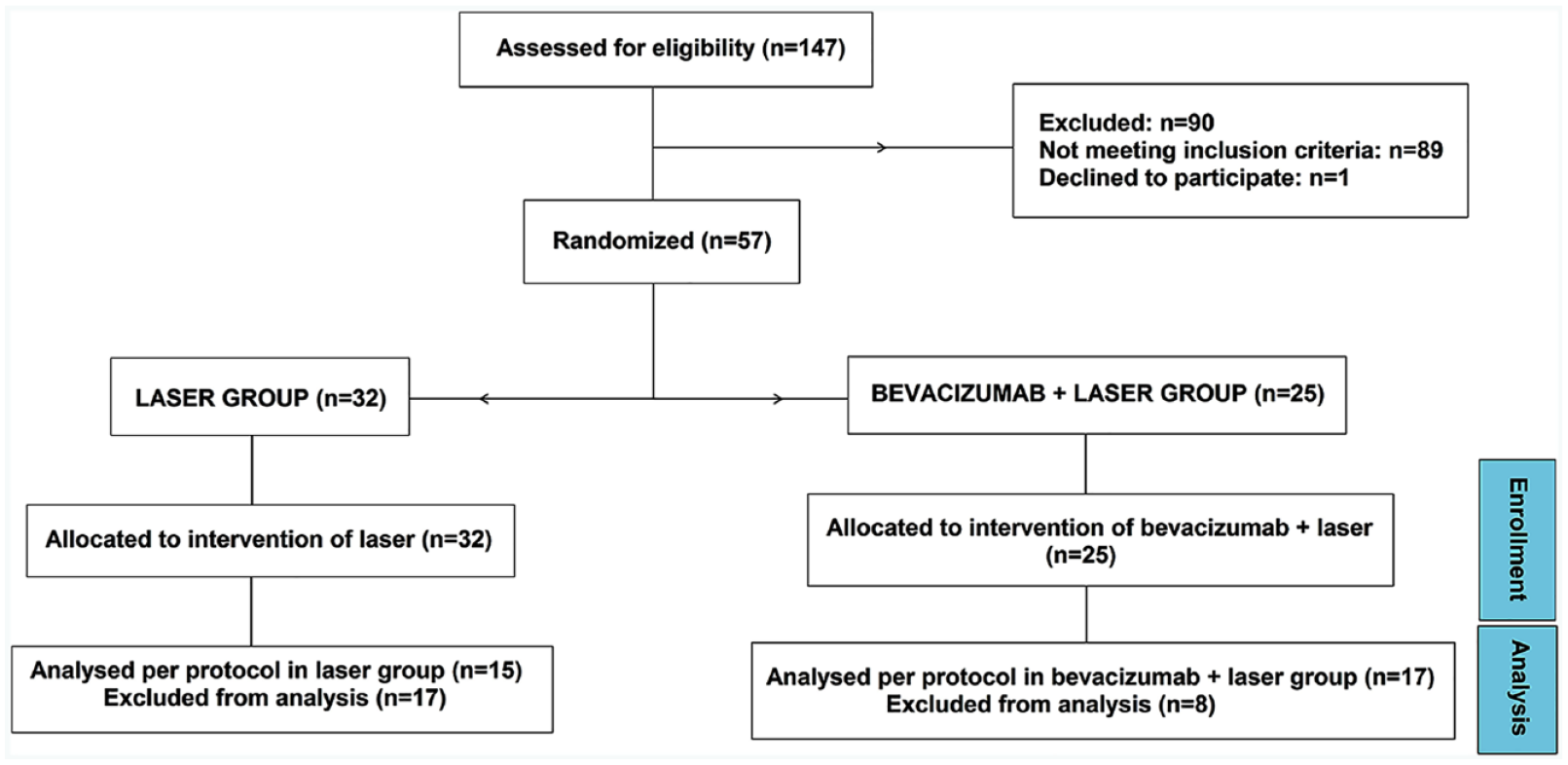
Flow diagram of the study.
Demographic data
There were no statistically significant differences in baseline characteristics of demographic data (age, sex, type of diabetes, lens status, previous treatments, laboratory measurements, vital signs, etc.) among both groups. Tables 1 and 2 show this in more detail.
Demographic characteristics of patients at baseline: qualitative variables.
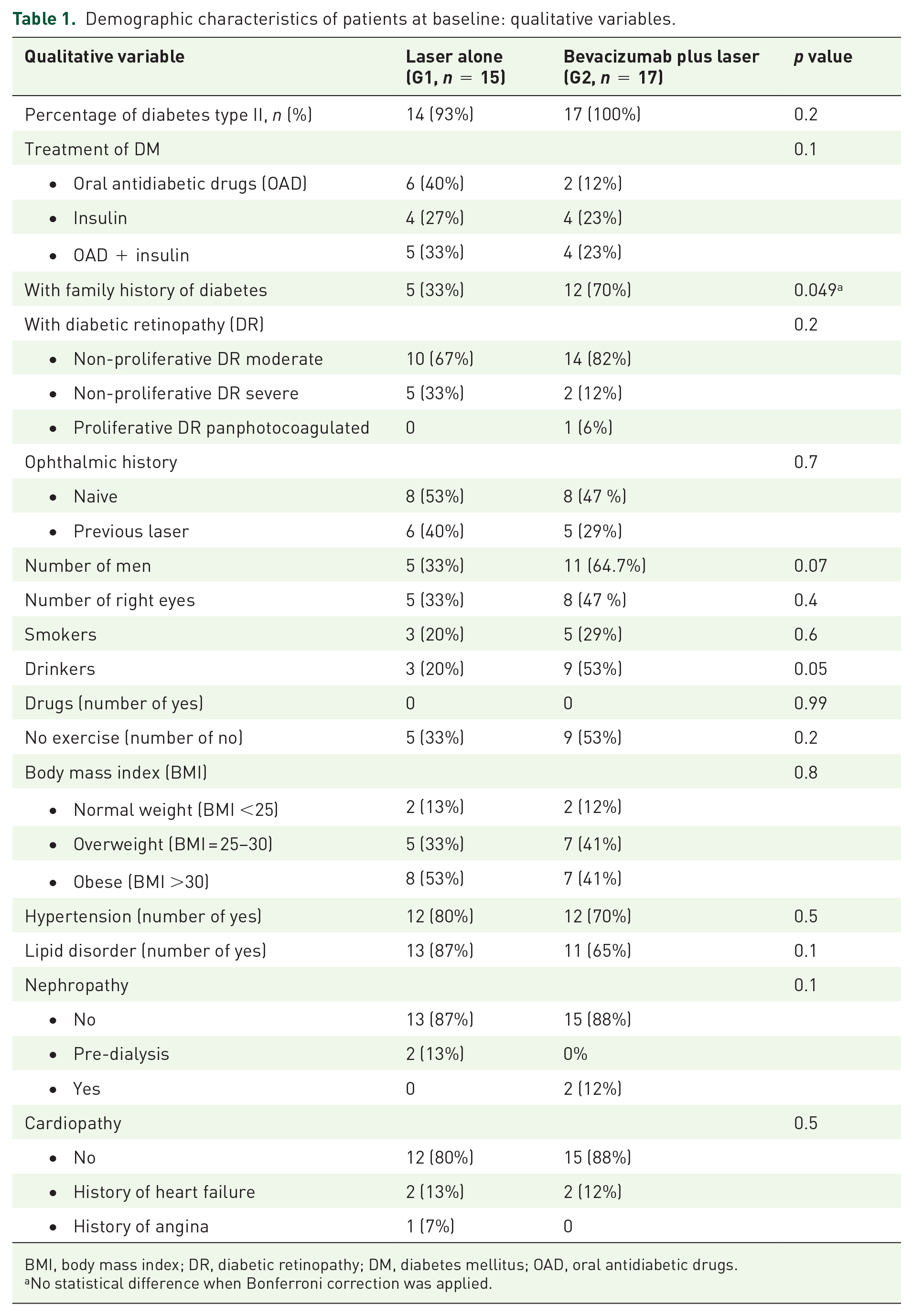
BMI, body mass index; DR, diabetic retinopathy; DM, diabetes mellitus; OAD, oral antidiabetic drugs.
No statistical difference when Bonferroni correction was applied.
Demographic characteristics of patients at baseline: quantitative variables.
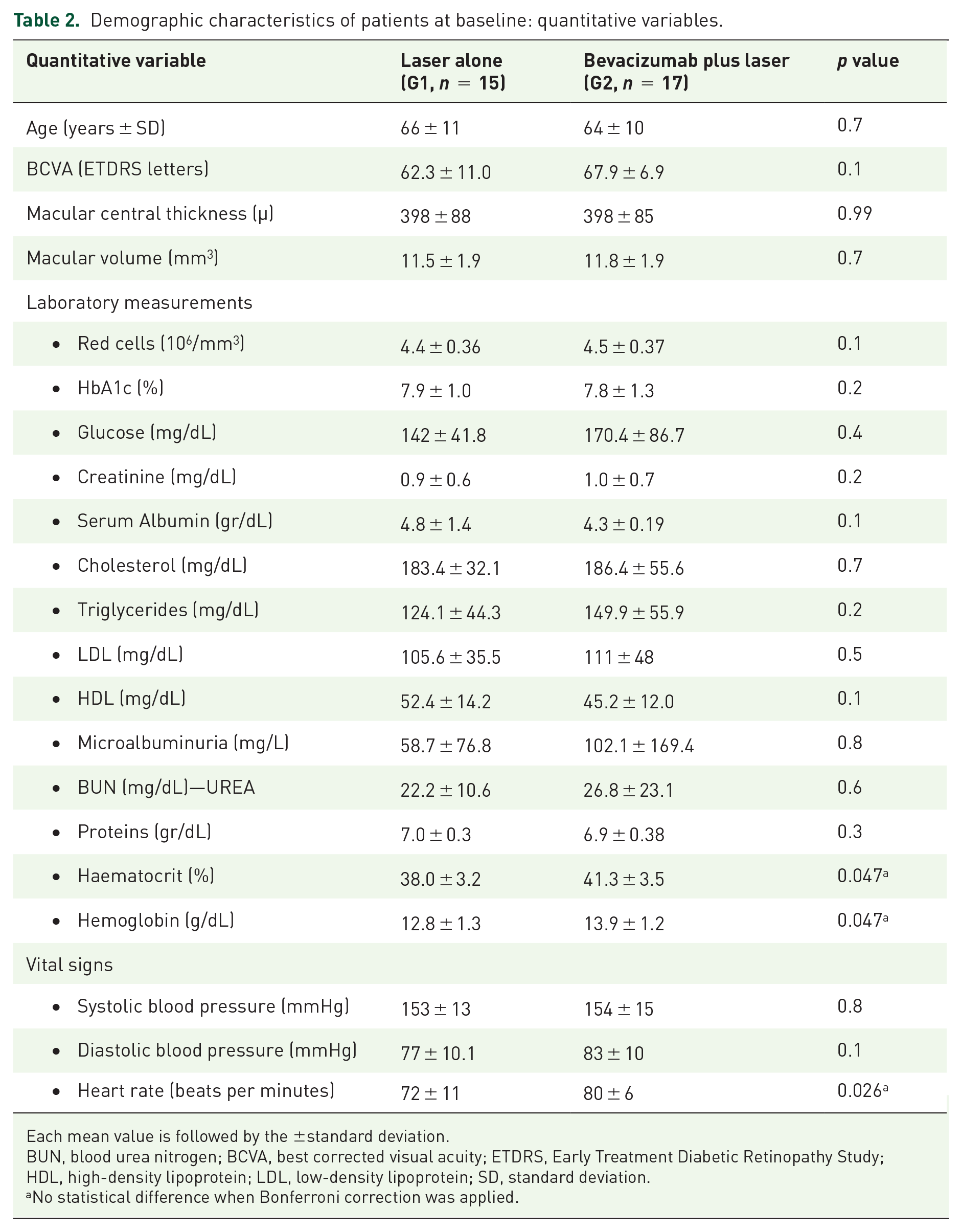
Each mean value is followed by the ±standard deviation.
BUN, blood urea nitrogen; BCVA, best corrected visual acuity; ETDRS, Early Treatment Diabetic Retinopathy Study; HDL, high-density lipoprotein; LDL, low-density lipoprotein; SD, standard deviation.
No statistical difference when Bonferroni correction was applied.
Visual acuity
As shown in Table 3, at baseline, there were no significant differences in BCVA between groups. After treatments, BCVA in G2 was statistically higher (p value ⩽ 0.01) than G1 in all follow-up visits. According to intra-group changes (statistical comparison of the value at a certain visit with respect the basal value for the same treatment), G1 provided a stable BCVA thorough the study (p value = 0.20), with a non-significant median improvement of 4 (7; –3) letters ETDRS at month 12. In contrast, G2 improved BCVA from baseline to each follow-up visit (p ⩽ 0.01) with a median improvement of +6 (11; 1.5) letters ETDRS at month 12.
Visual acuity in letters early treatment diabetic retinopathy study during the study.

BCVA, best corrected visual acuity; Q3, 75 quartile; Q1, 25 quartile; SD, standard deviation.
Statistically significant.
Regarding the percentage of visual gain (defined as an improvement >5 letters), stability (defined as a gain or loss ⩽5 letters), or loss vision (defined as a deterioration >5 letters), 29 of 32 patients (91%) gained or remained stable at month 12. Only 3 of the 32 patients (9%) lost vision (2 of 15 (13%) in G1 and 1 of 17 (6%) in G2). Regarding statistical significance, the proportion of patients with visual improvement was statistically superior in G2 compared to G1 at 3 and 6 months (p < 0.05). The proportion of patients with stable visual acuity (±5 letters) was statistically higher in G1 compared to G2 in months 3 and 6 (p < 0.05). There were no statistical differences between groups after month 6 of follow-up in the proportion of gain, stability, or loss of vision. Combined treatment achieves visual gains of ⩾10 ETDRS letters in >30% of the patients, while treatment with laser therapy does not reach this value throughout the follow-up.
Table 4 shows the percentage of visual gain, stability, or loss vision. The proportion of patients with visual improvement was higher in G2 only at 3 and 6 months (p < 0.05), what also coincides with that the percentage of patients gaining more than 10 and 15 letters was higher in G2 at 3 and 6 months. There were no other statistical differences between groups.
Number and percentage of gain, stability, or loss vision for both groups during the study.

BCVA, best corrected visual acuity; L, letters early treatment diabetic retinopathy study.
Central foveal thickness
As showed in Table 5, the mean CFT was similar between groups at the beginning of the study (p = 0.99). Both groups did not show any significant difference in CFT at month 3, 9, and 12. However, at month 6, G2 showed statistically lower values than G1. Not every change in CFT means a real anatomic change. Only differences in absolute value above 40 µm suggest that there was a real anatomical change. This value was calculated as the 10% of the mean value (398 µm). As can be seen in the fourth column of each group, 13 of 17 patients in G2 (76%) had a decrease in CFT > 40 µm (which means an improvement) at month 12, while it occurred in 7 of 15 patients in G1 (47%). Despite the proportion of patients with CFT reduction over the year of follow-up was higher in G2 (above 65%) than in G1 (below 50%), there were no statistical differences between groups or intra-groups in proportions of patients with increase, decrease or establishment of the CFT.
Central foveal thickness (CFT) in microns for both groups during the study.

CFT, central foveal thickness; Q3, 75 quartile; Q1, 25 quartile; SD, standard deviation.
Statistically significant.
Macular volume
A normal macular volume was defined as ⩽10.62 mm3, according to the internal normative data base included in Cirrus HD SD-OCT. There was no significant difference in average macular volume between groups during the study. Any of the groups achieved a normal macular volume during the study. However, when the change in macular volume with respect to baseline was specifically analyzed, a significant reduction of macular volume in G2 was observed over time (p < 0.05), whereas this fact did not occur in G1.
Type of DME
According to the classification of Panozzo and colleagues, 20 we divided DME into three sub-groups: spongiform (E1), cyst (E2), and subretinal fluid (E3). In G1 (n = 15), there were 3 (20%), 7 (47%), and 5 (33%) patients with E1, E2, and E3, respectively. In G2 (n = 17), there were 0 (0%), 9 (53%), and 8 (47%), respectively. The classification was carried out between two ophthalmologists, each one separately, and in case there was no agreement, the case was discussed with a third party until an agreement was reached. The BCVA for each group and classification are summarized in Table 6. There were significative differences between groups in patients with DME type E2, where G2 showed better values. Analyzing intra-group differences (e.g. change with respect baseline for a certain group), we found significant improvements only for G2 with DME type E2 at all follow-up visits.
Visual acuity in letters early treatment diabetic retinopathy study depending of diabetic macular edema (DME) classification during the study.

BCVA, best corrected visual acuity; DME, diabetic macular edema; E1, diffuseME; E2, cystME; E3, subretinal fluid DME; Q3, 75 quartile; Q1, 25 quartile; SD, standard deviation.
Statistically significant.
We also analyzed the results for these three sub-groups regarding CFT and macular volume. G1 did not make any significant improvement in any of these parameters for any of the sub-groups. G2 reached a significant reduction in macular volume in DME type E2 (from a median of 11.1 (11.5–10.3) at baseline to 10.2 (10.68–10.1) at month 12) but not in CFT (p = 0.05). G2 also reached a significant reduction in CFT in E3 patients (from a median of 375 (488.75, 334.5) at baseline to 325 (355.25, 243) at month 12) with no significant changes in macular volume (p = 0.27).
Fluorescein angiography
The number of patients at baseline with macular ischemia (defined as avascular foveal area ⩾1000 µm) was 5 of 15 (33%) in G1 and 8 of 17 (47%) in G2. Progression of macular ischemia was defined as an increment in the avascular foveal area >0.1 mm2. Five of 15 patients (33%) in G1 and 6 of 17 (35%) patients in G2 suffered a progression of macular ischemia, being differences between groups statistically indistinguishable.
Correlation between variables
The correlation factor between BCVA and CFT was −0.11 (p = 0.33) for G1 and –0.32 (p < 0.01) for G2. There was not any correlation between BCVA and macular volume (p = 0.60 and p = 0.48 for G1 and G2, respectively).
Re-treatments
G1 needed a median of three laser treatments per patients while G2 needed a median of four injections (including the initial three doses) of bevacizumab plus one laser treatment. The maximum number of treatments in G1 was four lasers (in a single patient who attended the last visit 2 weeks in advance), and in G2 was six bevacizumab injections plus two laser sessions. Regarding subgroups, E3 was the type of DME that needed more re-treatments, followed by E2. A complete data sheet of re-treatments is shown in Table 7.
Treatments and re-treatments.

DME, diabetic macular edema; E1, diffuse DME; E2, cyst DME; E3, subretinal fluid DME; Q3, 75 quartile; Q1, 25 quartile; SD, standard deviation.
Safety
In relation to ocular side effects, three patients in G1 were excluded because of the need of panphotocoagulation due to the appearance of neovessels. In contrast, no progression of diabetic retinopathy was seen in G2. Also, there was ERMs development in both groups and one patient in G2 was excluded because of this cause. Although the proportion of ERMs was higher in G1, there was no statistical significance. No retinal detachment, nor increase of intraocular pressure and nor injection-related cases of infectious endophthalmitis were found. Cataract seemed to increase in G2 but not in G1, but this difference was not significant.
Two patients in G2 died, one due to meningococcal sepsis 8 months after intravitreal bevacizumab, and the other because of lung terminal tumor that was first diagnosed during the study. Also, two patients (one in each group) developed heart disease. Two hypertension episodes were found one in each group (the patient in G2 forgot to take the treatment for it). No gastrointestinal events appeared in any of the groups. Regarding systemic control, there were no differences between groups in cardiovascular risk factor control all year long (blood pressure, heart rate, lipids). Finally, differences between groups in their diabetic control (HbA1c index and microalbuminuria) were not found.
Visual function
Visual function test NEI VFQ-25 was used to determine the quality of vision after treatment. Patients were satisfied with their vision with a global score superior to 75 for both groups. The worst parameter valued by patients was «general heath» in both groups. There were no differences between groups in all parameters. The complete data for test NEI VFQ-25 can be found in Table 8.
Median values for visual function test VFQ-25 results for both groups at month 12.

Q3, 75 quartile; Q1, 25 quartile; VFQ-25, Visual Function Questionnaire.
Discussion
Bevacizumab, a monoclonal antibody approved for the treatment of colorectal cancer by the FDA and EMA, has been used, and continues to be used, out of label in clinical practice in ophthalmology. The BOLT study, a prospective randomized controlled clinical trial, found that this drug has a greater effect than macular laser treatment in patients with center-involving persistent DME. 17 In the BOLT study, at months 12 and 24, the bevacizumab group (in monotherapy) showed significant improvement in BCVA (p < 0.01) compared with the macular laser therapy group. However, therapeutic effects of intravitreal bevacizumab and other anti-VEGF are short-term and frequent injections were needed to treat DME during the BOLT study, being 13 the median number of intravitreal bevacizumab over 24 months. Other previous studies reported results of a combination treatment of intravitreal bevacizumab and triamcinolone acetonide as a treatment option for DME. However, outcomes of combined therapy with these two drugs showed no further beneficial effects than conventional treatment. 21
In this study, we tried to investigate if the response to macular laser photocoagulation combined with bevacizumab injections shows superiority compared with laser alone in patients with diffuse DME over a year of follow-up. Solaiman and colleagues 14 showed that there is an improvement in BCVA a month after intravitreal bevacizumab and, also, that only a combination with laser allows this visual improvement to last until the third month. In the meta-analysis carried out by Liu and colleagues 22 in 2014, both macular laser alone and intravitreal bevacizumab alone improved visual acuity, being bevacizumab superior only at the beginning. In our study, patients treated with laser alone achieved better visual acuity gains than the ones obtained in other studies,11,23 but similar to more current studies.13,14,19 It is important to note that in our study, laser treatment was administrated by the same surgeon in all cases. So, variability due to different surgeons was minimized.
In this work, only the bevacizumab plus laser combined treatment group (G2) achieved statistically significant visual improvement at month 12 after treatment, as can be seen in Table 3. BCVA improvements were presented at month 3 and the superiority was maintained at all time-points in G2. In laser alone group (G1), there was no significant BCVA improvement in any of the visits. This functional difference could be explained due to the higher effect of bevacizumab on retinal thickness, which allowed laser to deliver sufficient power to the retinal pigment epithelium layer without damage healthy retina surrounding it.
Besides this, in our study, the percentage of patients gaining more than 10 and 15 letters was significantly higher in G2 only at months 3 and 6. Perhaps, this was due to the low number of intravitreal injections of bevacizumab. However, there were a higher proportion of patients with a BCVA >73 letters at 6, 9, and 12 months in G2, while the number of patients losing >10 letters was higher in G1. It is in accordance with the results published by other authors.15,19,24
When comparing our results with those in the bibliography, visual improvements in most of 1-year clinical trials involving a group with a combined treatment of ranibizumab plus laser are similar to the visual improvement obtained in our work with a combined treatment of bevacizumab plus laser (+4.8 letters in READ 2 study, 3 +5.9 in RESTORE study, 19 and +5.91 in RETAIN study, 25 versus +5.0 letters in our study). On the other hand, DRCRnet study 6 evaluated the effect of three intravitreal injections of ranibizumab combined with prompt (3–10 days after injection) or deferred (⩾24 weeks after injection) laser, finding an improvement of +9 ETDRS letters in both types of treatments. Maybe, the difference with respect our study could be related with the higher number of administered anti-VEGF injections and to the lower basal BCVA of patients in their study. In their sub-group with a BCVA ⩾66 letters ETDRS (similar to the basal BCVA of our study of 70 letters ETDRS), results were similar in both studies (+6 and +5 letters ETDRS for prompt and deferred laser respectively in their study, versus +5 letters ETDRS in our study). This suggests that patients with lower BCVA have more room from improvements. It is interesting that the cited sub-group of the DRCRnet study reached those improvements with a higher number of injections (8 and 9 for prompt and deferred laser, respectively) than in our study. However, in our study, the rate of naive patients in G2 (71%) was higher than the rate of naive patients in the cited sub-group of their study (40% and 39% for prompt and deferred laser, respectively). So, it is not clear if we reached the same improvement with less number of injections due to the higher effect of bevacizumab or to the higher rate of naive patients, although we hypothesize that the latter is probably the cause. Further studies are then warranted to clarify this point.
Regarding macular volume and CFT, we did not find any statistically significant difference between the baseline and any of the follow-up visits for both groups, excepting an isolated point of statistical difference between groups for CFT at 6 months. Maybe it is required more intensive intravitreal treatment for this effect to occur. More studies are required to analyze the reason why an improvement in BCVA in G2 was observed with no corresponding significant differences in CFT and macular volume between both groups.
Some authors have investigated the predictive value of baseline characteristics in the response to intravitreal bevacizumab in patients with DME, but it although remains unclear. Al Faran and colleagues 26 found that the baseline OCT parameters were associated with visual improvement, including parameters such as the photoreceptor inner segments thickness centrally (p < 0.01) and within the central 1-mm subfield (p < 0.01), and the presence of bridging retinal processes centrally (p < 0.01). In our sample, the best response to intravitreal bevacizumab was obtained in the E2 type. Persistent macular cystoid spaces seemed to be damaging because their presence predicted a reduction in BCVA letter score. RETAIN study suggested that subretinal fluid was also a predictor of response to ranibizumab 25 and DRCR.net study reported that a low initial visual acuity was associated with better visual improvements after treatment. 15 In our study, no correlation with clinical baseline characteristics was found. It is possible that this result may be influenced with the relative small size of the sample. We could not study the relationship between vision and rupture of ellipsoid zone by OCT because there were no patients with this anatomical rupture at baseline.
Regarding side-effects, the increasing use of bevacizumab is raising concerns about safety with long-term use of these agents. This drug has the potential to inhibit all the isoforms of VEGF-A and, in consequence, all its functions such as wound healing. 9 We did not find significant differences between both treatments regarding systemic safety. However, our study has not enough patients to properly analyze the contribution of bevacizumab in the systemic side-effects that we have reported. So, future research and meta-analysis are warranted.
In the ophthalmic side-effects, no endophthalmitis, cataracts or high levels of intraocular pressure were found. However, Baek and colleagues 27 described that under long-term monitoring repeated intravitreal injections of bevacizumab, there were an association with sustained intraocular pressure elevation. In our study, patients were followed 12 months but the mean number of injections was low and maybe not high enough.
The main limitation of this study is that it does not compare the response to bevacizumab alone, along with the two other arms. So it is difficult to assess whether the improvement in BCVA is due to the combined laser and bevacizumab or the latter alone. Additionally, the G2 group inferences may be the result of confounding bias created by the fact that a standalone bevacizumab group was not used to establish the efficacy of bevacizumab combined with focal laser used as adjunct following the loading dose. However, our study also has some strengths that make it useful. It includes only diffuse DME and a high percentage of naive patients suggesting that this type of patient may require a smaller number of intravitreal treatments. Despite we found statistical signification with commonly used statistical tests, the low number of patients is the main concern of this study. The reader should bear in mind that it was such a specific type of pathology that the inclusion period had to be extended for more than 2.5 years in the context of a referral hospital that covers a population above 400 000. Nevertheless, since the statistical power was relatively low, 56%, the results should be taken with some caution, as this decreases the probability that a statistically significant result will reflect a real effect. Then, further studies are warranted to strengthen the methodology and results.
In conclusion, our study suggests that laser combined with intravitreal bevacizumab was an effective option in the management of diffuse DME with central involvement thorough the follow-up. Results suggested that after 6 months of follow-up, the combined treatment was superior to the laser, and it was maintained during the rest of the follow-up, even with the low number of injections reported in this study.
Footnotes
Author’s note
The manuscript has been read and approved by all the authors. The requirements for authorship have been met. Each author believes that the manuscript represents honest work.
The authors thank the University of La Laguna, the official medical college of Santa Crus de Tenerife, and the government of the Canary Islands for their support in carrying out this work.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Trial Registration
This study is registered in clinicaltrials.gov as NCT01572350 (https://clinicaltrials.gov/ct2/show/NCT01572350), EU Clinical Trial Registry as 2009-014654-15 (![]() ).
).
