Abstract
Background:
Amiodarone is widely used for heart arrhytmia. Previous studies have suggested the possibility of optic neuropathy with the chronic use of this drug.
Objectives:
To identify structural or functional changes in the retina and optic nerve in patients on chronic amiodarone therapy without visual complaints.
Methods:
This observational study included 15 eyes of 15 patients with cardiac arrythmia on chronic amiodarone treatment and 15 healthy matched subjects as a control group. All subjects underwent electrophysiological tests [pattern visual evoked potential (PVEP), pattern electroretinogram (PERG), multifocal electroretinogram (mfERG), and optical coherence tomography (OCT) and angiography (OCTA)].
Results:
There were no statistically significant differences between the two groups regarding the PVEP, PERG, and the mfERG parameters. Macular and optic nerve head OCT and OCTA have not shown statistically significant differences except for the morphological parameters of the optic disc (p = 0.008 for the horizontal and p = 0.013 for vertical cup/disc ratio and p = 0.045 for rim area).
Conclusion:
Patients on chronic amiodarone therapy have not shown evident structural or functional changes in the retinal or optic nerve as demonstrated by electrophysiological tests, OCT, and OCTA results compared to controls.
Keywords
Introduction
An adverse drug reaction is defined by the World Health Organization as ‘a response to a medicine which is harmful and unintended, which occurs at doses normally used in man’. 1 Such adverse reactions can affect any organ or tissue, including the eye. Several toxic ocular side effects were previously reported with the use of systemic, intravitreal, and topical medications. 2 Keratopathy, cataracts, uveitis. retinopathy, optic neuropathy, and visual field defects may develop as ocular side effects. 2
Amiodarone is a potent antiarrhythmic drug that is indicated for the treatment of both atrial and ventricular arrythmias including life-threatening ventricular fibrillation and ventricular tachycardia refractory to other antiarrhythmic drugs. 3 Long-term therapy can result in a wide range of adverse effects affecting different organ systems, which can be life-threatening. Some of these can be avoided with judicious use of this medication. 3
Ocular adverse effects caused by amiodarone were first reported by Joseph and Rousselie in 1968. 4 Since then many studies have reported different amiodarone-induced ocular side effects. The most common reported symptom was colored rings around lights, whereas the commonest finding was vortex keratopathy.5,6 Lens opacities and optic neuropathy have also been reported,7,8 with few reports suggesting the possibility of retinal toxicity and other studies denying any amiodarone-induced retinal affection.9–11
To study retinal and optic nerve side effects, clinical in addition to functional and structural assessment are recommended.12,13 Objective functional assessment of the retinal and optic nerve function through different electrophysiological studies is commonly used for assessment of drug toxicity in addition to in vivo structural retinal assessment using high resolution optical coherence tomography (OCT).14,15 Optical coherence tomography angiography (OCTA) is another tool recently employed for detection of possible drug induced microvascular macular and optic disc changes. 16
Due to the sparsity of clinical studies on the effect of amiodarone on the structure and function of the retina and optic nerve with controversial results from published reports, the current study was carried out to evaluate the structure and function of the retinal and optic nerve in patients on chronic amiodarone therapy, with the aim of identifying early changes, if any. Objective functional assessment was performed using the pattern visual evoked potential (PVEP), pattern electroretinogram (PERG), and multifocal electroretinogram (mfERG). While structural assessment was done via macular and optic nerve head (ONH) OCT and OCTA.
Methods
This observational case-control study was conducted between December 2020 and September 2021.
We recruited a consecutive, non-randomized sample of 15 patients who have been diagnosed with cardiac arrhythmia and treated with amiodarone for at least 1 year (Group 2). A total of 15 healthy individuals who were sex- and age-matched to group 2 were included as a control group (Group 1).
Patients with refractive error more than ±3 diopters (D), previous ocular trauma or previous intraocular surgery, and any associated anterior segment, retinal or optic nerve pathology were excluded. Patients with any systemic disease other than cardiac arrhythmia were also excluded.
A detailed medical history was taken from each patient including duration of disease together with the dose and duration of amiodarone therapy.
Full ophthalmological examination was performed for each patient including best corrected visual acuity (BCVA) using Snellen chart, anterior segment examination using slit lamp biomicroscopy, intraocular pressure measurement using Goldmann applanation tonometry, and fundus examination. The patients were furtherly assessed using electrophysiological studies, structural OCT, and OCTA. Only the right eye of each participant was included in the study.
Optical coherence tomography
The Fourier-domain OCT system RTVue-XR Avanti (Optovue Inc, Fremont, CA, USA) was employed, running on software version 2017.1. The images were captured after pupil dilation using Mydriacyl 1% (Alcon Inc.). Only scans with good quality (>6/10) were included.
We captured macula map 5 × 5 mm scans, ganglion cell complex (GCC), and ONH scans for each patient. We recorded the central subfield thickness (central 1 mm circle), parafoveal thickness (3 mm circle), and perifoveal thickness (5 mm circle) from the macula map. The average, superior and inferior GCC thickness, as the well as focal and global loss volume, were obtained from the GCC scans. The parameters extracted from ONH scans included average retinal nerve fiber layer thickness (RNFL), superior, inferior, nasal, and temporal RNFL quadrants thickness, in addition to rim area, vertical and horizontal cup/disc ratio.
Macular OCTA 6 ×6 mm scans were captured, and the vessel density (VD) of the superficial and deep capillary plexuses were documented. Foveal avascular zone (FAZ) area was also obtained from a full vascular slab of the same scan. ONH OCTA 4.5 × 4.5 mm scans were obtained for measurement of the recorded radial peripapillary capillaries vessel density.
Electrophysiological tests
Electrophysiological tests were performed with the RETI port/scan 21 (Roland Consult, Brandenburg, Germany). For each subject, PVEP, PERG, and the mfERG were recorded. The parameters of the PVEP test and the mfERG were in accordance with the International Society for Clinical Electrophysiology of Vision standard.17,18 PERG was performed according to Holder et al. 19
Statistical analysis
All data analyses were performed using the Statistical Package for Social Sciences version 15.0 (SPSS v. 15.0; SPSS Inc, Chicago, IL, USA). Quantitative data were presented as mean ± SD. Data normality assumption was tested with the (Kolmogorov–Smirnov) test. Non-parametric data sets were compared using chi square analysis. Parametric datasets were compared using the t-test for independent samples. p Values were considered statistically significant if <0.05.
Results
Demographic data and basic characteristics
The study included 15 eyes of 15 patients with chronic amiodarone usage and 15 healthy controls of the same eyes of 15 age and gender. The mean age of the study group was 41.27 ± 16.38 years while for the controls it was 41.73 ± 17.27 years (p = 0.940). The group of patients included 9 females (64%) and the control group included 10 women (67%) with no statistically significant differences (p = 0.705).
We did not find significant differences in BCVA, SE, or intraocular pressure (p = 0.072, 0.603, and 0.721, respectively). Slit lamp examination revealed that all patients on amiodarone had cornea verticillata.
The average daily dose of amiodarone was 320.00 ± 165.62 mg per day (range 200–600 mg). The mean duration of amiodarone intake was 6.73 ± 4.20 years (range 1–14 years). The mean cumulative dose was 676.4667 ± 0.045 g (range 146–2044 g).
Results of the electrophysiological tests
There were no statistically significant differences between the patients and the control group regarding the PVEP and the PERG studied parameters as shown in Table 1.
PVEP and PERG parameters in the studied groups.

µV, microvolts; ms, milliseconds; PERG, pattern electroretinogram; PVEP, pattern visual evoked potential; SD, standard deviation.
Regarding the mfERG in the five rings response density, P1 amplitude and P1 latency showed no statistically significant difference between the two groups (Table 2).
Multifocal electroretinographic five-rings RRD of the controls and patients on amiodarone.

NV/deg2, nanovolt per degree square; RRD, ring response density; SD, standard deviation.
Results of OCT and OCTA
In the studied groups, the OCT parameters did not show statistically significant difference between the study groups, including macular thickness, RNFL, and GCC, as shown in Table 3.
Structural optical coherence tomographic parameters in the studied groups.
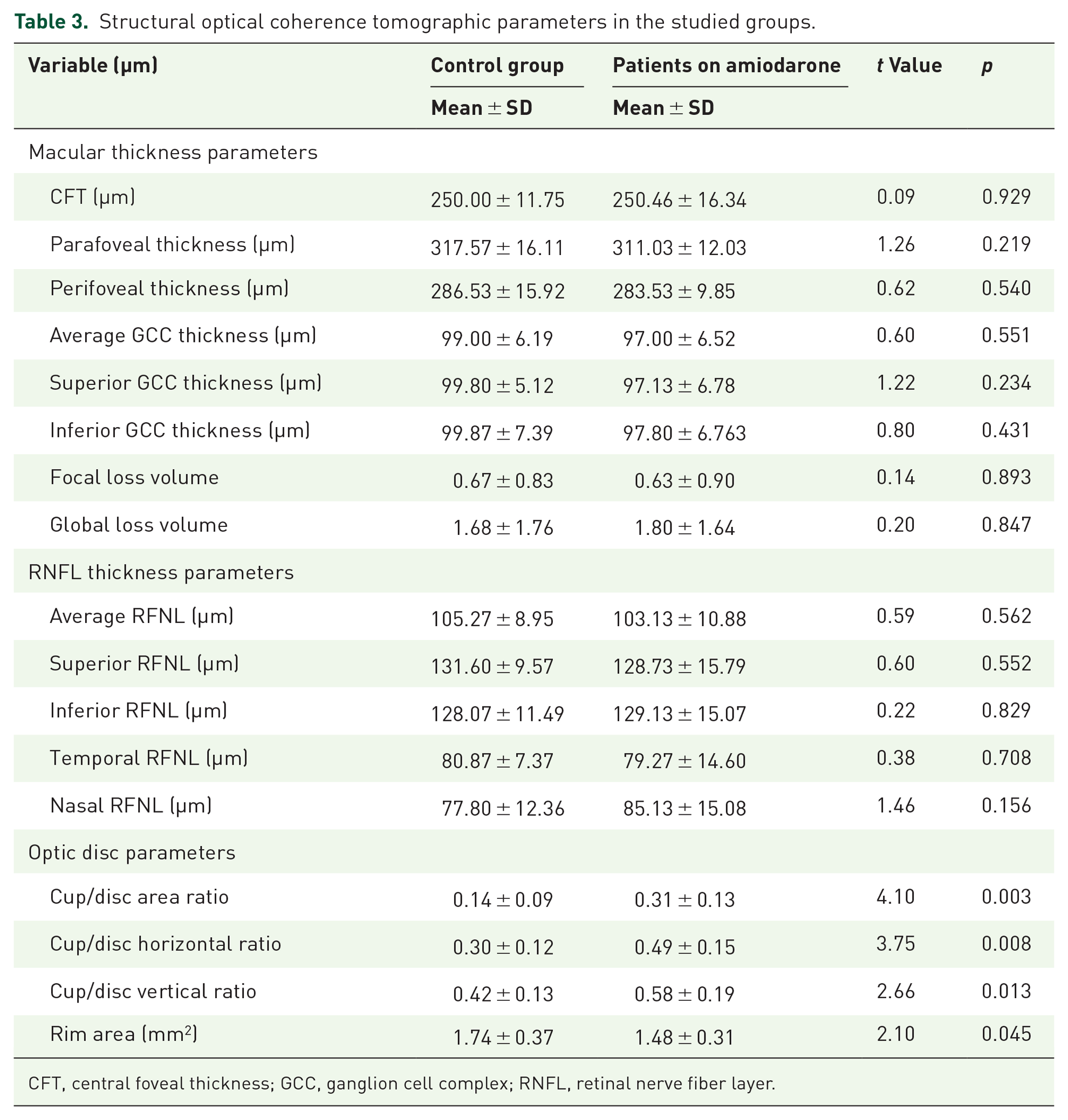
CFT, central foveal thickness; GCC, ganglion cell complex; RNFL, retinal nerve fiber layer.
Compared to controls, the results for the morphological parameters of the optic disc studied were statistically significantly higher in patients on amiodarone, with a reduced rim area (Table 3). Correlations between cup/disc ratio (horizontal and vertical) and patients’ age, dose, and duration of amiodarone were calculated. No significant correlations were found as shown in Table 4.
Correlations between cup/disc area ratio (horizontal and vertical) and age, amiodarone dose, and duration of treatment.

OCTA parameters showed no statistically significant differences as well (Table 5).
Optical coherence tomography angiography parameters in the studied groups.

DCP, deep capillary plexus; FAZ, foveal avascular zone; RPC, radial peripapillary capillary; SCP, superficial capillary plexus.
Discussion
The incidence of amiodarone induced toxic optic neuropathy incidence was estimated to be 1.76% over a 10-year period as opposed to 0.3% in an age-matched cohort not using the drug. 20 However, many later reviews have suggested that this incidence may be overestimated owing to the coexisting risk factors for non-arteritic anterior ischemic optic neuropathy among patients on chronic amiodarone therapy. To date, there are no definitive recommendations for periodic ophthalmological screening for patients on amiodarone. 21
In the current study, we evaluated structural and functional retinal and optic nerve in patients on chronic amiodarone therapy with no visual symptoms. We had a strict definition of inclusion and exclusion criteria aiming to identify any ocular toxicity related to this chronic therapy. We used different electrophysiological tests to get the benefit of functional assessment of different retinal structures in addition to using OCT and OCTA for structural and vascular evaluation of any possible changes. To our knowledge, this is the first study to employ different electrophysiological tests, as well as OCTA, in retinal and optic nerve in patients on amiodarone treatment.
Our patients received different doses of amiodarone for variable durations ranging from 1 to 14 years. The mean cumulative dose was 676.47 g, which was close to or even more than that reported by previous reports.11,22
All patients showed vortex keratopathy that manifests drug deposition in the basal epithelial layer of the cornea, where it binds to cellular lipids and becomes resistant to enzyme degradation. 23 Early histopathology reports demonstrated lysosome-like intracytoplasmic lamellar bodies observed throughout the cornea, lens, optic nerve, and in the retina. 5
Previous studies evaluating retinal drug toxicity have revealed that electrophysiological testing is considered a valuable objective tool for early detection of retinal toxicity. Electrophysiological changes may occur without or with minimal detectable structural changes. In this study, we used different electrophysiological tests to detect any changes in the function of the inner or outer retinal layers. 24 Through mfERG, simultaneous measurement of multiple responses at different retinal locations allows objective topographic mapping of retinal function in the central retinal 40°–50°. Therefore, mfERG is a valuable tool in the detection of ocular drug toxicity. 24 In our recruited cardiac patients with amiodarone, mfERG did not reveal statistically significant differences between the study groups in all the studied parameters. 11 Shaikh et al. evaluated the mf-ERG responses of 11 patients on chronic amiodarone therapy. Two patients had subnormal P1 amplitudes and mild prolongation in P1 implicit times. The authors concluded that the mildly abnormal findings with no consistent pattern could not be correlated with amiodarone exposure and were probably age related or due to testing variability. 11 In contrast, Liao et al. 9 found that rat flash electroretinogram (ERG) recordings showed that amiodarone-induced toxicity is associated with a decrease in a- and b-waves. Tajik and Shushtarian 25 also concluded that amiodarone has toxic effects on the retina, which could be detected and followed using ERG b-wave latency and amplitude.
Similarly, data obtained from PERG have not shown any statistically significant difference between waves’ implicit time, or amplitude between control group and cases group. No previous studies evaluated the effect of amiodarone therapy on PERG data.
Furthermore, there was no statistically significant difference between N75 latency (ms), P100 latency (ms), and N75-P100 amplitude (µV), in PVEP parameters between the control and cases groups. None of our patients had signs suggestive of toxic optic neuropathy at present or prior to enrollment in our study. In contrary Domingues et al. 22 found asymptomatic significant prolongation in pattern L-visual evoked potential (VEP) and a significant reduction in pattern a-VEP in patients on long-term treatment with amiodarone compared to control subjects. However, in their study, they did not exclude patients with other relevant ocular pathologies like cataract and diabetius mellitus, which might affect the proposed results. 22
All structural parameters evaluated with OCT (macular thickness and peripapillary RNFL thickness) did not show statistically significant differences. Macular OCT was not previously studied in patients on chronic amiodarone therapy apart from a case report by Joshi and Gill 10 who demonstrated interruption of the ellipsoid zone in the post-vitrectomy OCT. Accordingly, they suggested amiodarone-induced retinal phototoxicity following vitrectomy surgery. They supported their hypothesis by an earlier in vitro study which demonstrated that RPE in cases treated with amiodarone have decreased survival when exposed to UV radiation. 26
Previous studies have found that in amiodarone-related optic neuropathy, transient thickening of the RNFL occurs during the initial months of therapy, followed by subsequent axonal loss and optic nerve atrophy. 27 OCT has been proposed as a follow-up method to document edema or RNFL thinning in this setting.
When we analyzed the morphological parameters of the ONH obtained by OCT, we found statically significant differences between the study groups with more cupping and a decrease in the rim area in the cases group. However, we could not propose a causal relationship between amiodarone and optic disc cupping in absence of any significant related changes either structural (RNFL, GCC, peripapillary VD) or functional (PERG). Furthermore, we have not found significant correlation between optic disc cupping and dose or duration of amiodarone therapy.
OCTA provides objective documentation about the FAZ and vessel density characteristics. It has been suggested as a screening tool to detect early changes in the macula in some drug toxicity.28,29 Our results showed no significant changes between cases and controls in the OCTA parameters of the macular and optic disc OCTA parameters; thus denoting no variation in the FAZ area, macular vasculature, or the peripapillary vasculature related to amiodarone intake. This supports the results of the electrophysiological tests and the structural OCT to be both within normal limits. Although no previous reports in literature have used OCTA to assess amiodarone toxicity, our result – similar to previous studies – have failed to prove any retinal or optic nerve side effects of amiodarone.11,30
Owing to the strict inclusion and exclusion criteria to avoid any confounding pathology affecting our tests, only 15 patients were included in this study, which is the main limitation of the study. The variability in the amiodarone doses and duration of treatment is another limitation. Further longitudinal studies on a larger cohort of patients are warranted to support our findings.
In conclusion, our findings do not suggest retinal or optic nerve affection in patients on chronic amiodarone therapy for up to 14 years, notwithstanding meticulous assessment by multiple functional electrophysiological tests, structural OCT and OCTA in these patients. Therefore, it is plausible that the long-term use of such an indispensable drug in this group of critical patients can be considered safe on the retina and optic nerve. However, longer duration with higher doses may still need to be further investigated, and the present data are inadequate to endorse appropriate approaches for visual screening in patients on chronic amiodarone therapy.
