Abstract
Background:
Thalassemia major (TM) is an inherited anaemia caused by faulty haemoglobin synthesis. Reducing serum iron levels using iron chelating agents is an important step in the treatment of TM, and the effects on the eye of both the disease and these agents can be determined by regular eye examination.
Objectives:
We evaluated macular and optic nerve vascular densities in children with TM and compared the results with healthy controls using optical coherence tomography angiography (OCTA).
Design:
This is a prospective study.
Methods:
A total of 30 children with TM and 30 healthy controls were included in the study. The area of the foveal avascular zone (FAZ) and the vascular densities (VD) of the optic nerve head (ONH), radial peripapillary capillary (RPC) and deep and superficial retinal vascular networks were measured using OCTA.
Results:
A statistically significant decrease in VD was observed in the whole image and the parafovea, superior hemi, superior and inferior parts of the superficial capillary plexus and in the whole image and the superior regions of the deep capillary plexus in the TM patient group compared with the control group (p < 0.05). A significant decrease in VD was also observed in the whole image and the inside disc, peripapillary, nasal, inferonasal and temporal regions of the ONH and in the whole image and the inside disc, peripapillary and inferonasal regions of the RPC network in patients with TM (p < 0.05). A significant positive correlation was observed between both serum ferritin levels and deferasirox dosage, on one hand, and both the superficial (p = 0.023 and p = 0.002, respectively) and deep FAZs (p = 0.015 and p = 0.045, respectively), on the other hand. A negative correlation was also found between the deferasirox dosage and the VDs of the superficial (p = 0.010) and deep (p = 0.001) foveal plexuses.
Conclusion:
Retinal VD and FAZ are affected in patients with TM. OCTA, which can noninvasively measure retinal VD in patients with TM, may be a useful tool for the early detection of retinal microvascular changes that may occur during the course of the disease.
Keywords
Introduction
Thalassemia major (TM) is an incurable hereditary anaemia that has an autosomal recessive pattern of inheritance and arises from faulty synthesis of haemoglobin, ineffective erythropoiesis and a rapid breakdown of erythrocytes. Thalassemia phenotypes are divided according to their homozygous or heterozygous genetic composition into TM or thalassemia intermedia (TI). 1 While patients with TM need close medical follow-up and regular blood transfusions from the first years of life, TI patients experience a milder disease and do not require regular treatment. 2
For TM, transfusions prevent death and reduce overall mortality, but complications due to iron accumulating from transfused red blood cells are a major problem. Iron chelation therapy, which increases iron excretion and decreases iron storage, is therefore a necessity for patients with TM, but the drugs used in iron chelation also have toxic effects.3,4 Nevertheless, the effects of the disease itself outweigh even the combined toxic effects of the drugs, especially with the new generation of iron chelation therapies that have less ocular side effects. 5
TM is a chronic disease characterised by chronic anaemia, growth retardation, bone problems, hepatosplenomegaly, diabetes, hypothyroidism and cardiac pathologies. 6 Patients with TM may present with various ocular symptoms, including dry eyes, impaired colour vision, night vision difficulties, visual field defects, lens opacity, retinal pathologies and optic nerve damage. It has been reported that agents used for iron excretion can play a role in these ocular changes, but they may also be associated with the normal progression of the disease. With close follow-up, a systematic approach and appropriate treatments, the general condition of patients improves and their life expectancy is increased, which in turn makes monitoring and management of the possible ocular complications of thalassemia more important. 7
The feasibility of using of a noninvasive and easy-to-use tool to evaluate retinal vessels and thus monitor disease progression in children with thalassemia has yet to be investigated. Optical coherence tomography angiography (OCTA) is a noninvasive multimodal imaging method that is currently used to evaluate retinal microvasculature. It uses phase or amplitude decorrelation to display ophthalmic vascular structures in three-dimensional (3D) with a resolution of 5 µm, which allows the superficial and deep capillary networks to be analysed separately. 8 For this study, we therefore used OCTA to examine TM patients for whom no pathological findings had yet been made in conventional ophthalmological examinations and compared the results with those from healthy controls.
Methods
The right eyes of 30 follow-up patients with TM at the haematology clinic of Dicle University (Diyarbakır, Turkey) were evaluated in the university’s ophthalmology clinic between 15 June and 15 August 2022. The right eyes of 30 healthy individuals who attended the ophthalmology outpatient clinic for other reasons were included in the study as the control group. This single-centre, prospective study was conducted in accordance with the provisions of the Declaration of Helsinki.
Those with systemic diseases, such as diabetes mellitus or hypertension, and those with corneal opacity, lens opacification, glaucoma, retinal pathology, intraocular pressure higher than 21 mmHg or a refractive error more than three dioptres were excluded from the study. All OCTA measurements were made by the same technician using an AngioVue OCTA device (Optovue, Fremont, CA, USA), and those with an OCTA image quality of 70 or higher were included in the study. While making the images, any segmentation errors, artefacts due to patient movement or blinking, flow reflections or nonvascular flow signals were removed using the device’s relevant algorithms, and more accurate measurements were then sought. Complete ophthalmologic examinations of the patients were performed, including best-corrected visual acuity, intraocular pressure, slit-lamp biomicroscopy and detailed fundus examination.
All patients with TM in this study received regular blood transfusions and took oral deferasirox (DFX; 20–40 mg/kg/day) as iron chelation therapy. Treatment with DFX was started when ferritin levels exceeded 1000 ng/ml. The number of transfusions, DFX dosage and the haematocrit (%), haemoglobin (g/dL) and ferritin (ng/ml) levels of all patients were recorded.
OCTA measurements
The superficial capillary plexus (SCP) is a 120-µm thick planar capillary network that consists of a feeding arteriole and draining venules and is located between the internal limiting membrane and the inner plexiform layer. The deep capillary plexus (DCP) has a thickness of 60 µm and is located between the inner nuclear layer and the outer plexiform layer. The vascular density (VD) was calculated from the flow produced by the erythrocytes as a percentage of the area occupied by the blood vessels (Figure 1(a) and (b)), while the nonflow assessment tool in the OCTA software (version 2017.1) was used to calculate the foveal avascular zone (FAZ) areas in the SCP and DCP (Figure 1(c) and (d)). Optic nerve scans were conducted on the optic nerve head (ONH) and radial peripapillary capillary (RPC) layers. The RPC image was a view of the VD from the internal limiting membrane (ILM) to the retinal nerve fibre layer posterior boundary. The ONH image was a view of the VD from the ILM to the retinal pigment epithelium. VD measurement of the ONH and RPC networks was made by ONH scanning, and the RPC-VD and ONH-VD were both calculated from the nasal, inferonasal, inferotemporal, superotemporal, superonasal and temporal regions. The OCTA software automatically divided the optic disc into the inside disc region and the peripapillary region, the latter being a 0.75-mm wide elliptical annulus extending from the optic disc margin (Figure 1(e) and (f)). While a 3 × 3-mm scanning protocol was used to examine the macula, the ONH was examined using a 4.5 × 4.5-mm scanning protocol. The VD values (%) of the superficial and deep parafoveal retinas, RPC network and ONH were calculated. 9
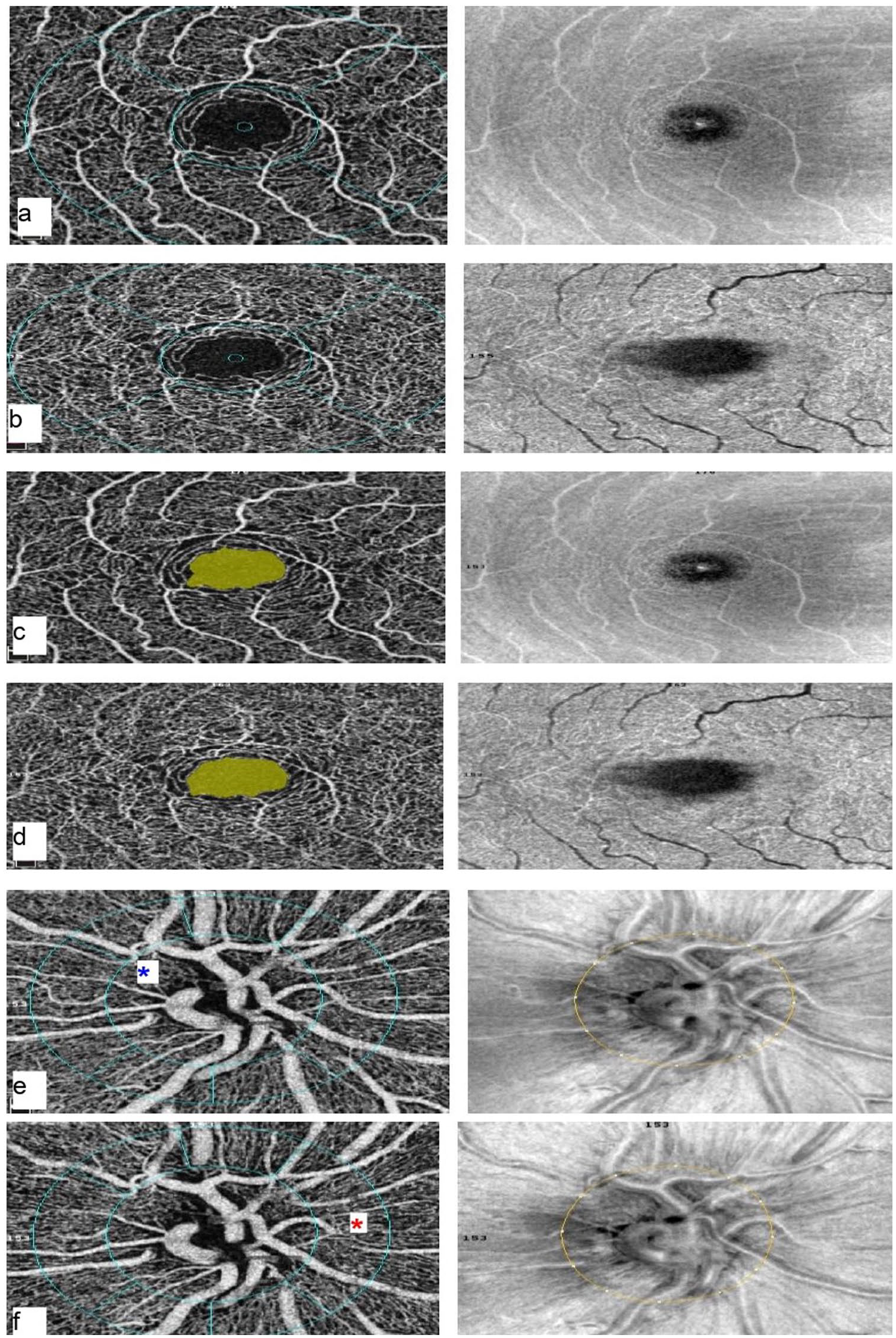
Right eye OCTA images of a 10-year-old male TM patient. (a) The superficial capillary plexus. (b) The deep capillary plexus. (c) Superficial FAZ (0.318 mm2). (d) Deep FAZ (0.336 mm2). (e) Optic nerve head, inner circle area shown with blue asterisk. (f) Radial peripapillary capillary plexus, area between outer and inner circle shown with red asterisk.
Sample size calculation
The sample size for this study was based on a previous study that compared retinal vessel density in children with TM to children without TM. 10 In this study, the sample was calculated for 80% power, a significance level of 0.05 and an effect size of 0.567.
Statistical analysis
Statistical analysis was performed using Statistical Package for the Social Sciences version 20.0 for Windows (SPSS Inc., Chicago, IL, USA) and R 3.5.1 (www.r-project.org) software. The normality of the data was assessed using a Shapiro–Wilk test, and descriptive statistics were summarised as mean ± standard deviation. Comparisons of values between the two groups were made using independent t-tests for normally distributed data and the Mann–Whitney U tests for non-normal distributions. Chi-square tests were used for between-groups comparisons of categorical variables, and the Pearson correlations were used to examine the relationships between variables. p values below 0.05 were considered statistically significant.
Results
Of the 30 patients with TM included in this study, 11 (37%) were male and 19 (63%) were female; in the control group, 18 (60%) were female and 12 (40%) were male (p = 0.491). The mean age was 11.52 ± 3.01 in the TM group and 12.43 ± 2.88 in the control group (p = 0.257). The demographic features and blood parameters, including haematocrit, haemoglobin and ferritin levels, and the DFX dosages of the TM patients are shown in Table 1. Fundus examinations of the patients revealed no pertinent symptoms or findings.
Demographic features of participants.

BCVA, best-corrected visual acuity; IOP, intraocular pressure; TM, thalassemia major.
When the ONH parameters were compared, a statistically significant decrease in VD was observed in the whole image and the inside disc, peripapillary, nasal, inferonasal and temporal areas of patients with TM and in the whole image and the inside disc, peripapillary and inferonasal areas of the RPC network (Table 2). A statistically significant decrease in VD was observed in the whole image and the parafovea, superior hemi, superior and inferior parts of the SCP and in the whole image and the superior region of the DCP in the TM group, but the VD was high in the DCP foveal region (Table 3 and Figure 2(a) and (c)). The mean deep FAZ area was 0.38 ± 0.06 mm2 in patients with TM and 0.32 ± 0.08 mm2 in the control group (p = 0.03), and the mean superficial FAZ area was 0.30 ± 0.12 mm2 in patients with TM and 0.28 ± 0.14 mm2 in the control group (p = 0.72) (Figure 2(b) and (d)).
Comparison of optic nerve head vascular density (ONH-VD) and radial peripapillary capillary vascular density (RPC-VD) of the patients with TM and healthy controls.

TM, thalassemia major.
p<0.05 is statistically significant.
Comparison of superficial capillary plexus vessel density (SCP-VD) and deep capillary plexus vessel density (DCP-VD) values of patients with TM and healthy controls.

TM, thalassemia major.
p < 0.05 is statistically significant.

OCTA and OCT appearances of TM, and healthy control. (a1–e1) TM; 12-year-old male, left eye. (a2–e2) Healthy control; 13-year-old male, left eye. The superficial FAZs were 0.278 mm2 (b1) and 0.216 mm2 (b2). The deep FAZs were 0.171 mm2 (d1) and 0.107 mm2 (d2).
A significant positive correlation was found between both serum ferritin levels and DFX dosage, on one hand, and the areas of both the superficial (r = 0.452, p = 0.023; r = 0.604, p = 0.002, respectively) and deep (r = 0.482, p = 0.015; r = 0.413, p = 0.045, respectively) FAZs, on the other hand. Negative correlations were, however, found between DFX dosage and the VDs of both the superficial and deep foveal plexuses (r = −0.524, p = 0.010; r = −0.633, p = 0.001, respectively).
Discussion
In this study, we detected a decrease in VD in some areas of the SCP, DCP, RPC and ONH and enlargement of the deep FAZ in patients with TM. Owing to mutations in the TM globin chain, the clinical picture of the disease is characterised by an imbalance of erythrocyte precursors and chronic anaemia. 11 Increased defective globin chains cause oxidative damage and membrane abnormality in erythrocytes, and the erythropoiesis process is thus interrupted, with iron absorption from the intestines increasing as a defence mechanism. 12 An increase in the amount of free iron in the blood then leads to organ dysfunction due to excessive storage of iron in the liver, heart, lungs and endocrine organs. Iron overload in TM patients then causes free radical formation and an increase in oxidative stress, and as the disease progresses, anaemia deepens and tissue hypoxia increases.13,14
Desferrioxamine (DFO) is one of the agents currently commonly used in the treatment of transfusion-induced haemosiderosis, although ocular toxicity and systemic toxicity, such as DFO-associated cardiac toxicity, have been reported. Retinal changes have also been observed due to the use of DFO.15–17 DFX is a new oral tridentate chelator with once-daily administration that can continuously remove the toxic, nontransferrin-bound, labile plasma iron that is the main cause of tissue damage in patients with iron overload. DFX acts more rapidly than DFO, readily entering most cells and reaching the major intracellular sites of iron accumulation. 17
As DFX is a relatively new drug compared with DFO, our knowledge of its ocular toxicity is mostly in the form of case reports. Pan et al. 18 reported that a 17-year-old boy with thalassemia developed bilateral painless visual impairment, central scotoma and dyschromatopsia after treatment with DFX. They reported that the patient’s vision and visual field improved upon first discontinuing the drug and then continuing with a low dose. In another case, sudden vision loss after high-dose DFX use in a 14-year-old boy was determined to be DFX-induced optic neuritis, with improvement achieved through gradual discontinuation of the drug. 19 Poosheshdoost et al. 20 , however, stated that there was no significant relationship between DFX use and visual and auditory disorders in TM patients and that DFX was safe for both sight and hearing.
In a study conducted in children with thalassemia, the mean P1 amplitude of all five rings in multifocal electroretinography was found to be significantly lower in users of both DFO and DFX compared with healthy controls, but there were no significant findings in fundus examinations. That study also found that DFO was more toxic to the macula than oral chelators like DFX. 21
Taneja et al. 22 observed ocular changes caused by the different treatment regimens recommended for patients with TM over the course of a year and reported that changes occurred in proportion to the increase in serum ferritin levels. Similarly, Cennamo et al. 23 reported, in a study that used OCTA to evaluate vascular changes in retinal and choriocapillaris perfusion, that high serum ferritin levels in TM patients may be significantly associated with choriocapillary hypoperfusion, but Kazancı et al. 10 reported that there was no relationship between ferritin levels and ocular changes when comparing TM patients with a control group in a study that also evaluated retinal vascular parameters using OCTA. In this study, a significant negative correlation was found between both serum ferritin levels and DFX doses and the VDs of the superficial and deep foveal plexuses. As the toxic effects of DFX are infrequent, VD may decrease at the more advanced stages of the disease, while DFX and ferritin levels may coincidentally increase as the disease progresses.
Cennamo et al. 23 also found a significant decrease in VD in the SCP and RPC regions of TM patients who received regular blood transfusions compared with those who did not, and they suggested that this may be due to retinal vascular damage caused by iron overload and the high oxidative environment created by increased plasmatic free iron. In this study, we found a statistically significant decrease in VD in some regions of the ONH, RPC, DCP and SCP in the TM group, which may indicate ischaemia due to disease progression; we measured high VD only in the foveal region of the DCP.
Georgalas et al. 24 reported that the SCP and DCP FAZ areas were not significantly different between their control and TM groups but that the retinal capillary plexuses – especially the DCP – presented unique morphologic changes in TM patients and that these were associated with increased urinary iron excretion. In contrast, Cennamo et al. 23 detected an enlargement in the FAZ, which was more pronounced in the DCP, in TM patients and stated that this may be due to the DCP being more sensitive to oxidative damage because it has a smaller vascular structure than the SCP. The deep FAZ area has been suggested elsewhere as a sensitive biomarker of early retinal vascular damage in various diseases,25,26 and we found a significant positive correlation between serum ferritin levels and the size of the superficial and deep FAZs and a statistically significant enlargement in the deep FAZ area in TM patients compared with the controls. The FAZ may therefore offer early indications of pathologies in the retinal capillary network, with enlargement in the FAZ reflecting deterioration in retinal vessel density and representing a possible biomarker of ischaemic damage. 26
Ophthalmologists may help to reduce ocular damage by keeping TM patients under close follow-up; using various examination methods, including OCTA; and considering possible complications caused by thalassemia itself and the ocular side effects of transfusional iron overload or iron chelators. 27 It is, however, thought that most of the ocular complications in TM patients are caused by the disease, particularly with the use of new iron chelation drugs, which are effective and have few ocular side effects. 5 Nevertheless, this still needs to be confirmed by randomised clinical trials.
The limitations of this study are that electrophysiology tests were not used; there were no comparisons with different types of disease, such as TI; there was no comparison group that had not received chelation treatment; and the FAZ could not be evaluated morphologically using the OCTA device.
In conclusion, the decrease in VD of the retinal microvascular layers and the enlargement of the FAZ may be attributed to ischaemia and to the tissue hypoxia that occurs with the disease’s progression. There was also a correlation between increased ferritin and DFX levels, which is likely due to the advanced stages of the disease and the expansion of the FAZ.
We believe that OCTA may represent a noninvasive and relatively inexpensive method for the early detection of both systemic complications and retinopathy due to the progression of TM. We think that studies with larger samples and longer follow-up periods will be beneficial in determining the most effective uses of OCTA.
